Antidiabetic and Hypolipidemic Effects of Methanol Leaf Extract of Newbouldia laevis in Alloxan Induced Diabetic Rats
Bosha JA1*, Sule S1 and Asuzu IU2
1Department of Vet. Physiology and Pharmacology, Federal University of Agriculture Makurdi, Nigeria
2Department of Vet. Physiology and Pharmacology, University of Nigeria, Nigeria
Submission: June 06, 2019; Published: July 09, 2019
*Corresponding author:Bosha Joel Aondohulugh, Senior Lecturer, Department of Vet. Physiology and Pharmacology, Federal University of Agriculture Makurdi, PMB2373 Makurdi, Benue State, Nigeria
How to cite this article: Bosha JA, Sule S, Asuzu IU. Antidiabetic and Hypolipidemic Effects of Methanol Leaf Extract of Newbouldia laevis in Alloxan Induced Diabetic Rats. Adv Res Gastroentero Hepatol. 2019; 13(3): 555863. DOI:10.19080/ARGH.2019.13.555863.
Abstract
The antidiabetic, hypolipidemic, liver and kidney function effects of the methanol leaf extract of Newbouldia laevis (NLE) was carried out in wistar albino rats. Diabetes was induced by single intraperitoneal administration of 150 mg/kg alloxan monohydrate in overnight fasted albino rats. Newbouldia laevis extract (NLE) caused a significant (P < 0.05) time-and dose-dependent reduction in FBS especially at the dose of 250 mg/kg which caused 60.2% reduction of FBS at 24th h compared to the negative control. All doses of NLE used in the study caused significant (P < 0.05) doses-dependent decrease in all serum lipids except high density lipid level of treated rats compared to the negative control. It significantly (p < 0.05) reduced the cardiovascular risk index as represented by coronary risk and atherogenic indices, just like glibenclamide. The extract caused significant (p < 0.05) dose-dependent decrease in Alanine Aminotransferase (ALT) in the liver. Kidney function test showed significant (P < 0.05) dose-dependent decrease in serum urea and creatinine levels. The extract (NLE) has demonstrated good antidiabetic and hypolipidemic activities comparable to glibenclamide a standard antidiabetic drug and its folkloric use as an antidiabetic herb is justified and can be a good candidate as a hypolipidemic drug.
Keywords: Antidiabetic; Diabetes; Hypolipidemic; Newbouldia laevis; Methanol; Alloxan
Abbrevations: NLE: Newbouldia laevis; ALT: Alanine Aminotransferase; DM: Diabetes Mellitus; FBS: Fasting Blood Sugar; ANOVA: Analysis of Variance; SEM: Standard Error of Mean; HDL: High Density Lipoproteins; VLD: Very Low-Density Lipoproteins; LDL: Low Density Lipoproteins; AI: Atherogenic Index; CRI: Coronary Risk Index; ROS: Reactive Oxygen Species; AST: Aspartate Aminotransferase; ALP: Alkaline Phosphatase; BUN: Blood Urea Nitrogen; SUN: Serum Urea Nitrogen
Introduction
Diabetes mellitus is an abnormal metabolic condition in which there is excess glucose in the blood due to disturbance in homeostasis of carbohydrate, protein and lipid metabolism regulated by the hormone insulin Dewanjee & Rakesh et al. [1,2]. This primary defect in glucose metabolism results in wide spread multi-organ complications that encompass virtually every system of the body Wadker et al. [3]. This is so because most of the body cells utilize glucose for their energy need
Diabetes mellitus is a disease condition of both humans and other animals Frode & Medeiros [4]. The disease incidence is on the increase and has now become an epidemic that kills more than HIV/AIDS Jerald et al. [5]. The currently estimated worldwide prevalence is 8.3% and is projected to be 9.9% in the next 20 years Staykova et al. [6]. According to WHO [7] about 377 million people had diabetes worldwide in 2013 and about 3.4 million died from consequences of high blood sugar in 2004. In Africa about 10.4 million people were reported to have DM in 2008 and this figure is expected to rise to 18.7 million by 2025 Hamman [8].
In other animals, DM is more common in dogs and cats Davidson [9], Rand & Marshall [10]; Khan [11]. The disease has also been reported in horses Durham et al. [12], cattle, pigs and sheep Gould & Clark [13,14], and also in guinea pigs Lang & Mungar [15], Frode & Medeiros [4]. The trend of DM in other animals is also on the increase just as in humans especially in the companion animals (dogs and cats). The number of dogs diagnosed with diabetes mellitus has increased in the last 30 years Fleeman & Rand [16]. The disease is majorly classified into two types: type 1 and type 2 diabetes according to world health organization WHO [17]. Although, other forms of the disease do exist. For example, gestational diabetes, malnutrition related diabetes etc. ADA [18].
The ultimate goal of all diabetes treatment strategies is to lower blood glucose concentrations to levels that approximate those representing normal range and its maintenance there of Wadker et al. [3]. This has shown to lessen the risk of development and progression of the disease process and its complications. Emphasis is usually on cardiovascular risk reduction particularly hypertension control and correction of dyslipidemia Defronzo & Abdul-Ghani [19]. Since time immemorial patients with type 2 diabetes have been treated orally with a variety of plant extracts in folk medicine Ivorra et al. [20]; Andrede-cetto & Heinrich [21]. One of such plants is Newbouldia laevis Bosha et al. [22]. The present hypoglycaemic agents used in the treatment of DM have shown reduced efficacy due to drug resistance and have many serious side effects. Also, available literature did not show enough scientific validation for Newbouldia laevis’ traditional use. This study was therefore designed to evaluate the antidiabetic, hypolipidemic, liver and kidney functions as well as cardiovascular risk effects of N. laevis leaf extract in diabetic rats.
Materials and Methods
Leaves of Newbouldia laevis were collected in Makurdi, Benue State North Central Nigeria and identified by a Botanist, with the Voucher specimen UAM/FHM/205 deposited at the Hebarium of the University of Agriculture, Makurdi. It was extracted by cold maceration in 80% methanol for 72 h and was concentrated in vacuo using rotary evaporator (Rotavapor-R-215) and stored as Newbouldia laevis extract (NLE). Diabetes was induced in albino rats by single intraperitoneal injection of 150 mg/kg alloxan monohydrate (Acrose, New Jersey USA) after overnight fast Szudelski [23]. The effect of NLE on alloxan-induced diabetes was carried out using diabetic rats divided into 5 groups of 8 rats each. Any rat with fasting blood sugar ≥ 7.0 mMol/l was considered diabetic using Accu-check Advantage II® (Roche Diagnostics, New Jersey USA) Bosha et al. [24].
Rats in group 1 and 2 were given 10 ml/kg distilled water (negative control) and 2 mg/kg glibenclamide (M.P. biomedicals inc, France) (positive control), respectively. Rats in group 3-5 were given 62.5, 125 and 250 mg/kg of NLE respectively for 21 days, all by gastric gavage. The fasting blood sugar (FBS) was measured at 0, 1, 3, 6, 24 h post drug or extract administration. Blood was collected via the heart under light chloroform anesthesia on day 21 for serum biochemistry assessment. The data was presented as mean± standard error of mean (SEM) and one-way Analysis of Variance (ANOVA) was used to analyze the result. Significance was accepted at p < s 0.05. The result is presented in tables.
Results
The Effect of graded doses of NLE on FBS level of alloxaninduced diabetic rats
The effect of graded-doses of NLE on the FBS of alloxaninduced hyperglycaemic rats after 24 hours is presented in Table 1. The result indicated that there was an increase in the mean FBS of rats in the negative control group from 25.3 ± 4.12 mMol/l to 28.3 ± 2.45 mMol/l from the 0 to 24 hours respectively representing 11.9 % increase. The various doses of the extract and the reference drug (glibenclamide) produced significant (p < 0.05 – 0.01) dose and time-dependent decreases in FBS levels of treated rats when compared to the negative control especially at 6 and 24 hours. The extract doses of 62.5, 125.0 and 250.0 mg/kg produced a decrease in FBS of the rats by 24%, 25% and 33.6% at 6 hour and 27.8% 55.3% and 60.2% at 24 hour respectively as against 40% and 51.5% reduction of the FBS in rats by the reference drug at the same time frame of 6 and 24 hours respectively. The highest FBS reductions by all doses of the extract and the reference drug (glibenclamide) were obtained at 24 hours. Also, at 24 hours, there was no significant difference (p > 0.05) between the extract (125.0 and 250.0 mg/kg) and the reference drug glibenclamide
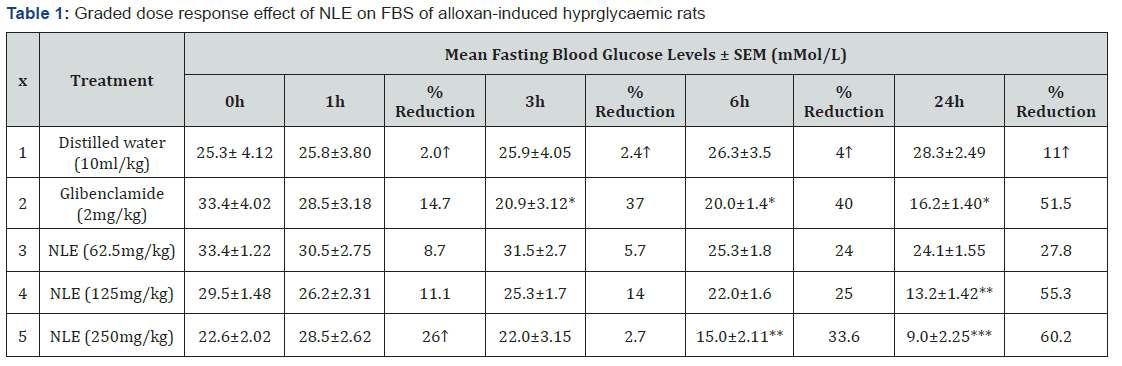
*P < 0.05, ** P < 0.01, when compared to negative control Represent increase in blood sugar level
Effect of NLE on the lipid profile of alloxan-induced diabetic rats
The mean serum level of total cholesterol, triglycerides, high density lipoproteins (HDL) low density lipoproteins (LDL) and very low-density lipoproteins (VLDL) of the controls and NLEtreated diabetic rats as well as the atherogenic index (AI) and coronary risk index (CRI) are shown in Table 2. Glibenclamide (2 mg/kg) and NLE (125 and 250 mg/kg) induced various levels of significant (p < 0.05-0.001) reduction in the serum levels of total cholesterol, triglycerides, LDL, VLDL, AI and CRI of the diabetic rats when compared with the negative control. The activity of NLE at the highest dose (250 mg/kg) was comparable to that of glibenclamide in all the parameters tested. NLE at the lowest dose (62.5 mg/kg) produced some reductions of the lipid profile and cardiovascular risk index, which were not statistically significant (p > 0.05). NLE at 62.5, 125.0 and 250.0 mg/kg produced a dosedependent significant (p < 0.05) increase of HDL in the diabetic rats when compared to the negative control.
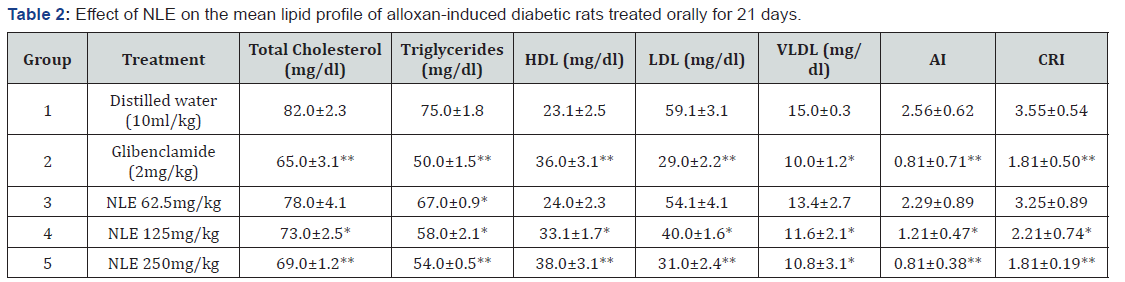
*P<0.05, **P<0.001 when compared to negative control group. HDL: High Density Lipoproteins; LDL: Low Density Lipoproteins; VLDL: Very Low-Density Lipoproteins; AI: Atherogenic Index; CRI: Coronary Risk Index
Effect of NLE on liver marker enzymes of alloxaninduced diabetic rats
The results of the effect of NLE on liver marker enzymes of alloxan-induced diabetic rats are presented in Table 3. The result indicated that there was significant (p < 0.05) decrease in mean ALT in the glibenclamide and the NLE treated groups when compared to the negative control except in the 62.5 mg/kg dose. The extract at all doses used did not cause any significant (p > 0.05) decrease in the other liver enzymes except for ALP at the 250 mg/kg dose. The total protein and bilirubin levels were not significantly (p > 0.05) changed in all treated groups when compared to the negative control group.
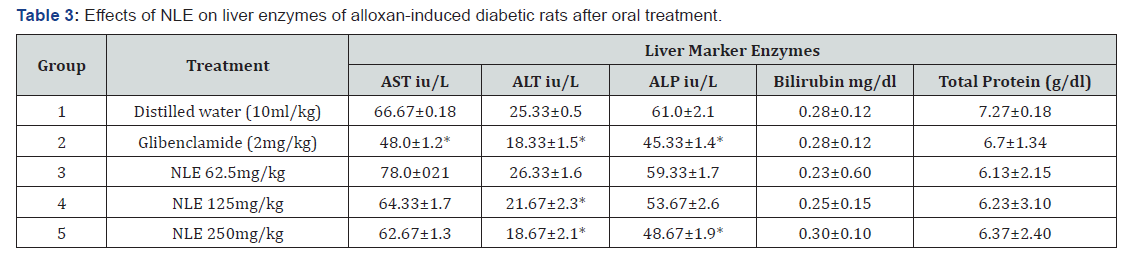
*p < 0-05 when compared to negative control group.
Effect of NLE on kidney function of alloxan-induced diabetic rats
The result of the effect of NLE on kidney function test in alloxaninduced diabetic rats is presented in Table 4. The result showed significant (p < 0.05) reduction in creatinine by the various doses (62.5, 125.0 and 250.0 mg/kg) of NLE and glibenclamide (2 mg/ kg). Creatinine was reduced from 0.40±0.16 mg/dl to 0.15±0.11 mg/dl in rats treated with the highest dose of NLE (250 mg/kg) representing 62.5% decrease. Serum urea reduced from 36.6±2.1 mg/dl in the negative control group to 25.3±2.1 mg/dl. The serum urea concentration of rats treated with 125, and 250 mg/kg were significantly (p < 0.01) different from the groups treated with glibenclamide (2 mg/kg) and NLE at 62.5 mg/kg.
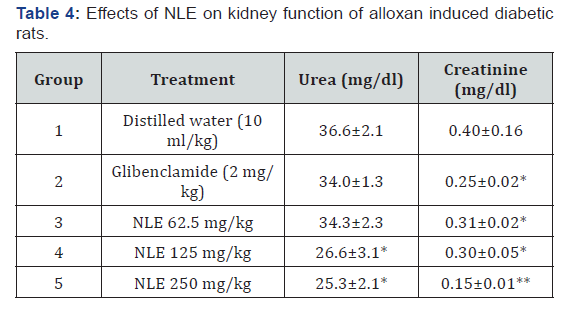
*p < 0.05, **p < 0.01 when compared to the negative control
Discussion
The ability of NLE to reduce the fasting blood sugar (FBS) of rats was tested as a means of evaluating its antidiabetic activity through a single intraperitoneal administration of 150 mg/kg of alloxan monohydrate. Fasting blood sugar (FBS) is a test of carbohydrate metabolism which measures blood glucose after a fast, which is usually 12-18 h Szudelski [23]. During this time, the body stimulates the release of the hormone glucagon which in turn releases glucose in to the blood stream through a catabolic process. Under normal circumstances, the body produces and processes insulin to counteract the rise in glucose levels, but in diabetes mellitus this process is impaired, and tested blood glucose levels remain usually high Kaufman et al. [25].
Experimental diabetes is usually induced using alloxan monohydrate or Streptozotocin Szudelski [23]; Frode & Medeiros [4]. Alloxan monohydrate (2, 4, 5, 6, - Tetraoxyprimidine, 2, 4, 5, 6, - pyrimidinetetrone) induces diabetes by the destruction of the β-cells of the islets of Langerhans of the pancreas which leads to decrease in insulin levels in the body and subsequent hyperglycaemia. Alloxan mediates its cytotoxic activity through reactive oxygen species (ROS) especially hydrogen peroxide with simultaneous massive increase in cytosolic Ca2+ concentration which causes rapid destruction of β-cells Szudelski [23]; Frode & Medeiros [4]. The pancreas is especially susceptible to the action of alloxan induced free radical-damage; subsequently, there is a decrease in endogenous insulin and this affects the utilization of glucose by the body tissues. It also leads to elevated blood glucose level, decreased protein content and increased levels of serum cholesterol, triglycerides and other bad lipids Yamamoto & Dhanabal et al. [26,27]. In this present study, diabetes was induced chemically in the rats through a single intraperitoneal injection of alloxan monohydrate at a dose of 150 mg/kg body weight as described by Szudelski [23]; Frode & Medeiros [4].
The rats were considered diabetic whenever their fasting blood sugar (FBS) was 7.0 mMol/l and more WHO, [17]; Turner et al. [28]. In the present study, methanol leaf extract of Newbouldia laevis (NLE) at doses of 62.5, 125.0 and 250.0 mg/kg body weight caused significant (p < 0.05 – 0.01) dose and time-dependent decreases in the FBS levels of the alloxan-induced diabetic rats. The time of maximum effect was at the 24th h in the dose response study. The dose and time-dependent reductions in the FBS levels also manifested in the subacute study of 21 days oral administration of NLE Bosha et al. [29]. The NLE at the highest dose (250.0 mg/kg) caused 60.2% decreases in FBS at 24 h, when compared to the negative control rats as against the reference drug glibenclamide (2 mg/kg) which caused 51.5% decrease under the same condition (Table 1). The dose and time-dependent decrease of FBS in diabetic rats by Newbouldia laevis has been reported by other workers also Tanko et al. [30]; Owolabi et al. [31]; Kolawole et al. [32]; Bosha et al. [22]; Bosha et al. [24]; Bosha et al. [33]
Diabetes mellitus is also associated with increase in serum lipid which usually leads to coronary heart disease complications. The lipids that are normally elevated in diabetes mellitus include total cholesterol, triglycerides, low density lipids, (LDL), very low-density lipids (VLDL) but a decrease in high density lipids (HDL) ADA [18,34]. The increase in the concentration of body lipids especially the visceral lipids results in the liberation of some lysosomes and other pro-inflammatory substances like some free fatty acids and cytokinins that trigger inflammation and cell degeneration, which are capable of causing other complications Tajudin & Nasiruddin [35]; Umesh et al. [36]. In the present study, NLE significantly (p 0.05) reduced the elevated serum levels of total cholesterol, triglycerides, LDL and VLDL of the treated rats dose-dependently when compared to the negative control group (Table 2).
NLE also reduced the cardiovascular risk index dosedependently as shown by the reduction of the Coronary Risk Index (CRI) from 3.55 in the control group to 1.81 and Atherogenic Index (AI) from 2.56 to 0.81, just like glibenclamide. These reductions are likely beneficial in preventing the development of the disease and its complications Cho et al. [37]. These findings also point to the fact that NLE has some cardiovascular protective property and can positively intervene in cardiovascular complications associated with the natural history of diabetes. Another good finding about NLE is that its dose-dependently increased the concentration of HDL which is also needed to ameliorate the cardiac risk associated with diabetes mellitus
The liver is the major organ of metabolism and biotransformation as well as gluconeogenesis and glycogenolysis. Glucose homeostasis is physiologically maintained by the balance between glucose production by the liver and glucose utilization by the peripheral tissues Duncan & Bond [38]; Navarro et al. [39]. The effect of substances on the liver depends on the duration of exposure and the chemical nature of the substance.
Alloxan monohydrate is toxic to the liver apart from the pancreas such that the induction of diabetes with alloxan also causes damage to the liver and affects its function and integrity El-Demerdash et al. [40]. This damage is manifested by leakages of liver enzymes into the blood and this leads to increased enzyme levels Navarro et al. [39]. NLE caused dose-dependent decrease in elevated serum level of these enzymes (AST, ALT and ALP) but only ALT was significant (p < 0.05) when compared to the untreated control (Table 3). Increase beyond the normal level of some or all of these enzymes (AST, ALT and ALP) in the serum are indicative of toxicity of the liver by drugs or other hepatic toxins Ramaiah [41].
Among all the enzymes, ALT is the most specific to the liver and a much better parameter to detect liver injury. Aspartate aminotransferase (AST) apart from being observed in liver injury is also associated with the diseases of other organs like the heart and skeletal muscles Ozer et al. [42]. Alkaline phosphatase (ALP) is mostly present in the cells linning the billiary duct system of the liver and is used to diagnose obstruction to the billiary system such that its elevation in the blood is associated with cholestatic diseases such as gallstone or tumour blocking the bile duct Burtis & Ashwood [43]; Kolawole et al. [32]. In this present study, there was no significant change in the values of AST, ALT and ALP in the duration of the study. Kolawole et al. [32] also reported nonsignificant changes in these liver enzymes in rats. This also suggests a hepatoprotective effect of NLE in diabetes which may be due to cell membrane stabilization and repair of damaged tissues as mentioned by Argawal et al. [44]. Bosha et al. [33] reported the erythrocytes and membrane protective effects of NLE in diabetic rats.
The protein level of the NLE-treated rats was slightly lower than the untreated diabetic group though the difference was not statistically significant. This does not agree with the findings of Duncan & Bond [38] and Ezeja et al. [45]. They reported that the protein levels of the treated rats were slightly reduced after 3 weeks of diabetic induction but higher than the control. In this study, the protein levels of treated rats were slightly reduced but lower than the untreated control. Among the NLE-treated groups there was a dose-dependent difference in the protein level. The glibenclamide-treated group also showed a similar decrease in protein level. The serum bilirubin of the control and the treated groups of rats did not show any significant difference. This lack of difference in the serum bilirubin level may indicate that there was no hepatic injury. Bilirubin is a product of haemoglobin breakdown and is associated with hepatic diseases like jaundice and ineffective erythropoiesis. Increase in the level of bilirubin indicates the depth of jaundice in the individual Thapa & Walia [46].
Kidneys are major organs of excretion and play very important roles in glucose metabolism. Their role in gluconeogenesis and glucose excretion helps in the development, maintenance and resolution of hyperglycaemia Marseric [47]. It takes up glucose from circulation and reabsorbs glucose from glomerular filtrate. The kidney responds to noxious substances or injuries in various ways ranging from decreased waste elimination to cell death. The major biochemical parameters that are measured to access the integrity and normal function of the kidney include increased blood urea nitrogen (BUN) or increased plasma creatinine levels Gerich [48]; Braide & Anika [49].
In this study, untreated diabetic rats showed higher levels of serum urea and creatinine when compared to the treated groups. Kolawole et al. [32] also reported a similar finding in rats. NLE showed a dose-dependent decrease in serum urea nitrogen (SUN). At the highest dose (250 mg/kg), it performed significantly (p 0.05) better than the reference drug glibenclamide. This may be possible due to improved waste product clearance and adequate regulation of blood pH which are affected in diabetes mellitus due to disturbance of tubular integrity Frode & Medeiros [4]. This significant reduction in both serum urea and ceratinine suggest that NLE may improve nephropathy and its associated complications in diabetes (Table 4). Serum urea and creatinine are important markers of kidney dysfunction Mukinda & Eagles [50].
Conclusion
In conclusion, Newbouldia laevis leaf extract (NLE) has demostrated significant antidiabetic activity which is comparable to glibenclamide a standard sulfanylurea, in this study. It also demonstrated dyslipidemic effects by lowering the bad lipids and increasing the good lipids thereby reducing the cardiovascular risk index as much as glibenclamide. It also protects the liver and the kidney by lowering the index enzymes. This establishes the basis for its pharmacological use as an antidiabetic plant in Nigerian folk medicine and can be a good candidate for hypolipidemic drug.
Acknowledgements
Dr Bosha acknowledged the support of University of Agriculture, Makurdi for the study leave to carry out the work. He also acknowledges TET Fund through the same University for partly sponsoring the work.
References
- Dewanjee S, Bose SK, Sahu R, Mandal SC (2008) Anti-diabetic effect of matured fruits of Dispyros peregrina in alloxan-induced diabetic rats. International Journal of Green Pharmacy 2: 95-99.
- Rakesh B, Sanjay J, Deep O, Armit J, Girraj ST, et al. (2010) Antidiabetic activity of aqueous root extract of Inchnocarpus fruitescens in STZ and Nicotinamide-induced Type 2 diabetes in rats. Indian J Pharmacol 40(1): 19-22.
- Wadkar KA, Magdum CS, Patil SS, Naikwade NS (2008) Antidiabetic potential of Indian medicinal plants. Journal of Herbal and Medical Toxicology 2: 45-50.
- Frode TS, Medeiros A (2008) Animal Models to test drugs with potential antidiabetic activity. J Ethnopharmacol 115(2): 173-183.
- Jerald EE, Joshi SB, Jain DC (2008) Antidiabetic activity of flower buds of Michehia champaca (Linn). Indian J Pharmacol 40(6): 510-571.
- Staykova J, Manologa A, Tzolova G (2014) Diabetes Mellitus Type 2 Prevalence, Risk Factors and Complications in the Region of Kardzhali, Bulgaria. Journal of Pharmacy and Pharmacology 2: 145-151.
- (2013) WHO. Factsheet No 312.
- Hamman RF (2010) Mortality risk in long-standing Type 1 Diabetes: Hope and concerns. Diabetes 59(12): 2997-2998.
- Catchpole B, Ristic JM, Fleeman LM, Davidson LJ (2005) Canine diabetes mellitus: Can old dogs teach us new tricks. Diabetologica 48(10): 1948-1956.
- Rand J, Marshall R (2005) Understanding feline diabetes mellitus.
- Khan MC (2010) Diabetes mellitus. In: The merck Veterinary Manual. (10th), pp. 439-440.
- Durham AE, Huges KJ, Cottle HJ, Rendle DI, Boston RC (2009) Type 2 diabetes mellitus with pancreatic β cell dysfunction in 3 horses confirmed with minimal model analysis. Equine Vet J 41(9): 924-929.
- Gould AC (1981) Diabetes mellitus in Cattle. Vet Rec 109(24): 539.
- Clark Z (2003) Diabetes mellitus in a 6-month old Charolias heifer Can Vet J 44(11): 921-922.
- Lang CM, Mungar BL (1976) Diabetes mellitus in the Guinea pig. Diabetes 25(5): 434-443.
- Fleeman LM, Rand J (2005) Beyond Insulin Therapy: Achieving optimal control in diabetic dogs. Centre for companion animal, the University of Queensland Australia. WALTHAM focus 15(3): 12-18.
- (1980) World Health Organization study group 1980: Technical report series No. 727.
- (2012) American Diabetes Association. Periodic release.
- Defronzo RA, Abdul-Ghani M (2011) Type 2 diabetes can be prevented with Early Pharmacological Intervention. Diabetes care 34(2): 5202-5209.
- Ivorra MD, Paya M, Villar A (1989) A review of Natural products and plants as potential antidiabetic drugs. J Ethnopharmacol 27(3): 243-275.
- Andrede-Cetto A, Heinrich M (2005) Mexican Plants with hypoglycaemic effect used in the treatment of Diabetes. J Ethnopharmacol 99(3): 325-348.
- Bosha,JA, Asuzu IU, Anaga AO (2013) Antidiabetic effects of Newbouldia laevis methanol leaf-extract on alloxan-induced diabetic rats. 37th Annual Conference of The West African Society for Pharmacology (WASP) held at Sheraton Hotel Lagos, Nigeria, p. 45.
- Szudelski T (2001) The mechanism of Alloxan and Streptozotocin actions in β- cell of the rats’ pancreas. Physiol Res 50(6): 536-546.
- Bosha JA, Anaga AO, Asuzu IU (2015) Bioassay-Guided Isolation and Structural Elucidation of Antidiabetic Principle of Methanol Leaf Extract of Newbouldia laevis (P Beauv). Journal of Pharmacy and Pharmacology 3: 515-526.
- Kaufman FR, Gallivan JM, Warren-Boulton E (2009) Overview of diabetes in Children and teens. American Journal of Health Education 40: 259-263.
- Yamamoto H, Uchigata Y, Okamoto H (1981) Streptozotocin and Alloxan induced DNA strand breaks and poly (ADP)-ribose sythetase in pancreatic islets. Nature 294(5838): 284-286.
- Dhanabal SP, Raja MM, Ramathan M, Suresh B (2007) Hypoglycemic activity of Nymphaea stellata leaves ethanolic extract in alloxan induced diabetic rats. Fitoterapia 78(4): 288-291.
- Turner R, Hansson P, Lancet S (2000) Issues of diabetes. British Medicinal Journal 351: 1755-1762.
- Bosha JA, Anaga AO, Asuzu IU (2016a) Antidiabetic Effects of Newboldia laevis (P. beauv) Methanol Leaf Extract on Alloxan- induced Diabetic Rats. Nigerian Society for Animal Production (NSAP). Proceedings of 41st Annual Conference at Federal University of Abeokuta, Nigeria, pp. 848-850.
- Tanko Y, Okasha MA, Saleh MIA, Mohammed A, Yerima M, et al. (2008) Antidiabetic effects of the ethanolic flower-extracts of Newbouldia laevis on blood glucose level in Streptozotocin- induced diabetic Wistar rats. Medwell Research Journal of medical Science 2(2): 62-65.
- Owolabi JO, Amechina CF, Okoro M (2011) Effect of Ethanol leaf-extracts of Newbouldia laevis on blood glucose levels in diabetic rats. Tropical Journal of Pharmaceutical Research 10(3): 249-254.
- Kolawole OT, Akanji MA, Akiibinu MO (2013) Toxicological Assessment of Ethanolic extract of Leaves of Newbouldia laevis (P. Beauv). American Journal of Medicine and Medical Sciences 3(4): 74-80.
- Bosha JA, Adenkola AY, Asuzu IU (2016b) Erythrocyte Stability, Membrane Protective and Haematological activities of Newbouldia laevis in Alloxan-induced diabetic Rats. Sokoto Journal of Veterinary Sciences 14(1).
- (2003) American Diabetes Association. Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Follow up report on the Diagnosis of Diabetes Mellitus. Diabetes Care 26: 3160-3167.
- Tajudin M, Nasiruddin WA (2006) Effect of Unani formulation on lipid profile in rats. Indian J pharmacol 38(1): 56-57.
- Umesh K, Mishara M, Lal SS, Tayyab F (2012) Medicinal plants and its impact on diabetes. World Journal of Pharmaceutical Research 1(4): 1019-1046.
- Cho SY, Park TY, Park IM, Choi MS, Lee MU, et al. (2002) Alteration of Hepatic Antoxidant Enzymes activities and lipid profile in STZ-induced diabetic rats by supplementation of dandetion water extract. Clin Chem Alta 317(1-2): 109-117.
- Duncan WE, Bond JS (1981) Decrease turnover of soluble liver proteins in mice with alloxan- induced diabetes. Am J Physiol 241(2): E1151-E1159.
- Navarro CM, Montilla PM, Martin A, Jimenez J, Utrilla PM (1993) Free radicals scavenger and antihepatotoxic activity of Rosmarinus. Plant Medicine 59(4): 312-314.
- El-Dermerdash FM, Yousy MV, El-waga NIA (2005) Biochemical study on the hypoglycaemic effects of onion and garlic in alloxan-induced diabetic rats. Food Chem Toxicol 43(1): 57-63.
- Ramaiah SK (2011) Preclinical safety assessment. Current gaps, Challenges and approaches in identifying translatable biomarkers of drug-induced liver damage. Clin. Laboratory Med 31(1): 161-172.
- Ozer J, Ratweb M, Shawe M, Baileya W, Schomaker S (2008) The current state of serum biomarker of hepatoxicity. Toxicity 245(3): 194-205.
- Burtis CA, Ashwood ER (2001) Enzymes In: Tietz Fundamentals of Clinical Chemistry. (5th), New York USA, W.B Saunder company, pp. 352-369.
- Argawal V, Shama AK, Upadhyay A, Singh G, Gupta R (2012) Hypoglycaemic effects of Citullus colocynthis Acta Poloniae Pharmaceutica-drug research 9(1): 75-79.
- Ezeja MI, Anaga AO, Asuzu IU (2014) Acute and Sub-chronic Toxicity profile of methanol leaf extract of Gouania longipetala in rats. J Ethnopharmacol 151(3): 1155-1164.
- Thapa BR, Walia A (2007) Liver function test and their interpretation. India J Pediatr 74: 663-671.
- Marseric O (2009) Glucose control by the kidney: an emerging target in diabetes. Am J kidney Dis 53(5): 875-883.
- Gerich JE (2010) Role of kidney in normal glucose homeostasis and in the hyperglycaemia of diabetes mellitus: Therapeutic implication. Diabetes medicine 27(2): 136-142.
- Braide VB, Anika SM. Environmental Toxicology. (1st ), SNAAP press Ltd, Enugu. p. 89-99.
- Mukinda JT, Eagles FK (2010) Acute and Sub-chronic oral toxicity profile of the aqueous extract of Pohygala fruticosa in female mice and rats. J Ethnopharmacol 128: 236-240.






























