Therapeutic Potential of Lab-Grown Autologous BioSphincters for the Treatment of Fecal Incontinence in a Long-Term Pre-Clinical Study
Elie Zakhem1*, Jaime L Bohl2*, Riccardo Tamburrini2, Prabhash Dadhich1, Christie Scott1, Erin L Mitchell3, J Mark Cline4 and Khalil N Bitar1,5,6*
1Wake Forest Institute for Regenerative Medicine, USA
2Department of Surgery, Wake Forest School of Medicine, USA
3Animal Resources Program, Wake Forest School of Medicine, USA
4Department of Comparative Medicine, Wake Forest School of Medicine, USA
5Section on Gastroenterology, Wake Forest School of Medicine, USA
6Virginia Tech-Wake Forest School of Biomedical Engineering Sciences, USA
Submission: January 24, 2019; Published: March 12, 2019
*Corresponding author:Khalil N Bitar, Wake Forest Institute for Regenerative Medicine, Section on Gastroenterology, Virginia Tech-Wake Forest School of Biomedical Engineering Sciences, Wake Forest School of Medicine, 391 Technology Way, Winston-Salem NC, 27101, USA
How to cite this article:Khalil N B, Elie Z, Jaime L. B, Riccardo T, Prabhash D,et al. Therapeutic Potential of Lab-Grown Autologous BioSphincters for the Treatment of Fecal Incontinence in a Long-Term Pre-Clinical Study. Adv Res Gastroentero Hepatol. 2019; 12(4): 555845. DOI: 10.19080/ARGH.2019.12.555845.
Abstract
Passive fecal incontinence (FI) is a clinical condition when patients lose the ability to control the voluntary passage of fecal material. The internal anal sphincter is responsible to maintain the closure of the sphincter and its intrinsic innervation is responsible to control the relaxation of the sphincter. Loss of this coordinated interplay leads to fecal incontinence. We previously created a model of passive FI in rabbits and treated it with autologous engineered BioSphincters. Rabbits were followed for 3 months. The objective of this study was to extend the follow up on the animals for 12 months to demonstrate safety and efficacy of the BioSphincters in rabbits. Seventeen female rabbits underwent sphincterotomy to induce fecal incontinence. This was demonstrated by reduction in anal basal pressure and a reduction in rectoanal inhibitory reflex (RAIR). Rabbits were divided into three groups; non-treated group, treated group with BioSphincters and sham group. Anorectal manometry was performed on the rabbits in all groups regularly to assess their anorectal physiology incontinence. Rabbits in the non-treated group maintained a reduced anal pressure and reduced RAIR for 12 months. Rabbits in the treated group had their anal pressure and RAIR restored, compared to baseline (pre-sphincterectomy) and maintained the restored anorectal physiology for 12 months. Rabbits in the sham group behaved similarly to rabbits in the non-treated group. Restoration of smooth muscle continuity was observed in rabbits in the treated group compared with the non-treated group. Pathological findings in this study were minor and rabbits maintained good health in all groups. The results obtained in this study support the long-term safety and efficacy of using BioSphincters to treat FI in preclinical large animal models of FI. BioSphincters provide a potential therapeutic approach to treat and manage fecal incontinence.
Keywords: BioSphincters; Sphincterotomy; Rabbits; Fecal incontinence; Fecal material; Infections; Malfunction; Therapeutic potential
Abbrevations: FI: Fecal Incontinence; IAS: Internal Anal Sphincter; EAS: External Anal Sphincter; RAIR: Recto Anal Inhibitory Reflex; NO: Nitric Oxide
Introduction
Fecal incontinence (FI) is a common devastating clinical condition. Patients lose the ability to control the passage of fecal material. Additionally, the quality of life of patients becomes impaired, limiting their social activities leading to social embarrassment and loss of jobs [1]. Given that fecal incontinence is a multifactorial disease, it is hard to estimate the prevalence of fecal incontinence in the total population, rather the prevalence was subdivided into categories of patients [2-4]. Additionally, people with fecal incontinence are embarrassed to report it which leads to inaccuracy in the published numbers. In a recent study by Menees S et al. [5]based on a large population in the United States, it was reported that 1 out 7 had FI.
At the pathophysiological level, one or more components of the anal sphincter complex become impaired and lead to loss of function. Normal anal sphincter complex is composed of a smooth muscle component (internal anal sphincter, IAS), a striated muscle (external anal sphincter, EAS), and enteric neurons [6]. A coordinated interplay between these components and the anorectum result in normal anal sphincter function. Loss of integrity of any of those 3 major players leads to fecal incontinence [7,8]. The IAS is predominantly (70-85%) responsible to maintain closure of the anal sphincter and therefore maintain basal tone. The EAS supports the IAS tone by providing squeeze pressure.
Fecal incontinence resulting from IAS damage is considered as passive fecal incontinence and the one resulting from EAS damage is considered as urge-related incontinence. Rectal distension due to fecal material in the anal canal induces the IAS to relax and allow the passage of fecal material. This local reflex is referred to as Recto Anal Inhibitory Reflex (RAIR) and is mediated by the enteric neurons, specifically nitric oxide (NO) neurons [9]. Inhibition of the NO-mediated relaxation of the smooth muscle can be achieved by N(ω)-nitro-L-arginine methyl ester (L-NAME), which is a compound used in organ bath studies to test the integrity of the neural circuitry.
Patients presenting with fecal incontinence currently have limited options with unsatisfactory outcomes. Injection of inert bulking agents, sacral nerve stimulation, implantation of artificial anal sphincters and colostomies are common treatments [10-12]. However, they are associated with short-term efficacy, infections and malfunction. Given the complex structure of the anal sphincter, strategies to treat fecal incontinence must take into account the reason of the failure to maintain continence. In the case of passive fecal incontinence, restoration of the IAS smooth muscle integrity and its enteric neural circuitry is essential to restore the basal tone, and therefore the anal closure and continence.
Current animal studies focus on injuries to the EAS or to the IAS and EAS combined without the distinction between passive and urge-related fecal incontinence [13,14]. Our lab developed a large animal model (rabbit) of passive fecal incontinence by surgical damage of the IAS integrity to cause fecal incontinence that mimicks passive fecal incontinence seen in human patients. The rabbits were also treated with autologous IAS BioSphincters engineered in the lab using the rabbit’s own smooth muscle cells and enteric neurons. Results have shown that over a follow up of 3 months, rabbits that received BioSphincter implantation restored fecal continence [15]. In this study, we followed the rabbits for a longer term (12 months) to validate the safety and efficacy of Bio- Sphincters. The results of this study demonstrated that lab-grown autologous BioSphincters have a therapeutic potential in treating fecal incontinence in human patients.
Methods
Study design
The design of this study is summarized in Table 1. Twenty-six female New Zealand white rabbits were included in the whole study. All the rabbits were six months old at the beginning of enrolment. The average weight of the rabbits was 2.9±0.2 kg, at the beginning of enrolment. All animals underwent IAS hemi-sphincterectomy to induce FI. Rabbits were randomly divided into 3 experimental groups: 1) Non-treated group (incontinent control), 2) Treated group (rabbits received surgical implantation of BioSphincters 6-8 weeks following sphincterectomy through a surgical opening of the anal verge), and 3) Sham surgery group (surgical opening of the anal verge was performed followed by immediate closure without implantation of BioSphincters). Rabbits were followed for 12 months.
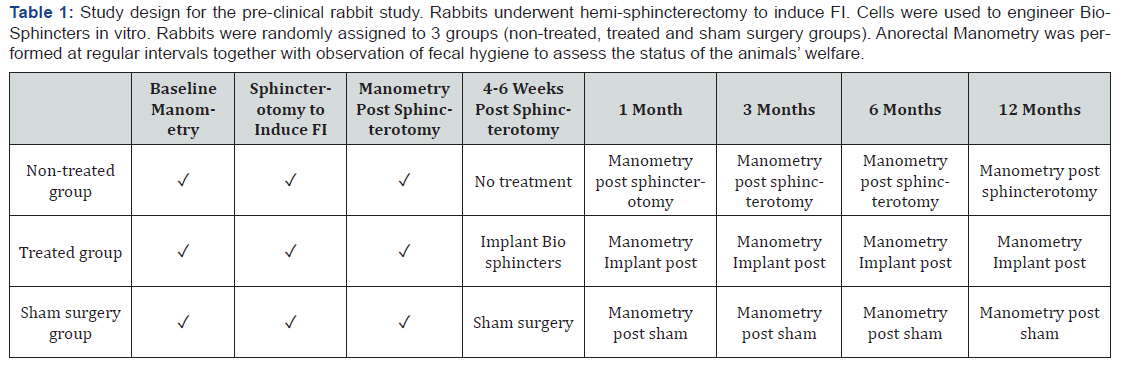
Measures for fecal incontinence
All rabbits underwent IAS hemi-sphincterectomy to induce FI. FI was assessed in each animal after sphincterectomy and compared to before sphincterectomy by: (1) regular observation of fecal hygiene and (2) regular anorectal manometry
Anorectal manometry was performed as follows: A catheter with 4 circumferential sensors attached to an inflatable balloon was used to measure 2 parameters: anal basal pressure and Recto Anal Inhibitory Reflex (RAIR). These 2 parameters were used as a measure for normal anorectum physiological functioning and FI.
Anal basal pressure is the highest identified pressure zone generated by the IAS. RAIR is the ability of the IAS to relax following inflation of the balloon. Baseline readings were obtained on all rabbits before any surgeries. Following hemi-sphincterectomy, anorectal manometry was performed on all rabbits to confirm FI, which was identified by lack of fecal hygiene and by significant decrease in anal basal pressure and RAIR in all rabbits. All rabbits in the 3 groups were followed by anorectal manometry regularly until they reached their 12-month time point.
Collection of biopsies for cell isolation and expansion in vitro:
All procedures were performed following the guidelines established by the Institutional Animal Care and Use Committee (IACUC) at Wake Forest School of Medicine. General anesthesia was induced using isoflurane via masking. During the surgical preparation, animals were intubated, eye lube applied, blood collected, the surgical area shaved and prepped for aseptic surgery, and a catheter was inserted intravenously. Blood pressure, heart rate, respiration status (CO2, SPO2) and temperature were monitored throughout the procedures. Procedures were performed to collect small intestine biopsy and IAS biopsies as previously described [15].
Briefly, before IAS hemi-sphincterectomy surgery, on the same day, rabbits underwent small intestinal biopsy through laparotomy. Two full thickness intestinal biopsies were obtained. Following the small intestine biopsy, the IAS area was prepped and draped in the standard surgical fashion. A ventral hemi-circumferential, curvilinear incision was made through the anocutaneous tissue. The IAS was then dissected from the submucosal plane and the overlying EAS for 1 cm. The IAS fibers were then sharply amputated. IAS tissue was then immediately placed in 2 - 8oC Hank’s balanced salt solution (HBSS) transport medium. Antibiotics (Enrofloxcin) were given at 10 mg/kg intramuscular for 5 days post operatively. Analgesics were given orally for 3 days post operatively.
Autologous cell isolation
Autologous cell isolation was performed as previously described [15].
Neural progenitor cells: The intestinal biopsies were washed extensively with HBSS containing 2X antibiotic/antimycotic. The tissue was minced, washed, and subjected to digestion twice in a mixture containing type II Collagenase and Dispase II. The cells were recovered by centrifugation and washed before being passed through a 70 μm nylon cell strainer. Cells were washed and re-suspended in neural growth medium before being filtered through a 40 μm nylon cell strainer. NPC were plated in neural growth media consisting of Neurobasal media (Invitrogen) supplemented with 1% N2 supplement, 20 ng/ml bFGF, 20 ng/ml EGF, 1 mM L-Glutamine and 1x antibiotic/antimycotic. Under these culture conditions, single cells aggregated, replicated, and formed enteric NPC at 37°C under 7% CO2
Smooth muscle cells: IAS tissue excised from the rabbit was rapidly cleaned in ice-cold carbonated HBSS containing 2X antibiotic/ antimycotic (200U/ml Penicillin G, 200 μg/ml Streptomycin and 0.5 μg/ml Amphotericin B) and 50 μg/ml Gentamicin. The tissue was finely minced and digested twice using Collagenase type II for 1 hour each digest. The cells were collected, washed, and re-suspended in growth medium before plating onto tissue culture flasks at 37°C under 5% CO2. Smooth muscle cells media consisted of DMEM high glucose (Invitrogen) supplemented with 10% FBS, 2.5 mM L-glutamine and 1x antibiotic/antimycotic.
Both cell types were expanded for 4 weeks to obtain the required number of cells to engineer the BioSphincters.
Autologous cells characterization
Isolated autologous IAS smooth muscle cells and small intestine neural progenitor cells were characterized for identification before engineering the BioSphincters.
Smooth muscle cells: Autologous smooth muscle cells isolated from rabbit IAS biopsy were allowed to grow in culture. Microscopic evaluation of the morphology of the smooth muscle cells was performed. Phenotype of smooth muscle cells was evaluated by immunofluorescence. Cells grown in culture were fixed in 4% formaldehyde, followed by blocking and permeabilization. Smooth muscle cells were stained with primary antibodies against smooth muscle markers; α-smooth muscle actin and smoothelin. Appropriately conjugated secondary antibodies were used to visualize the positive stains.
Additionally, the phenotype of the isolated smooth muscle cells was characterized by flow cytometry. Cultured IAS smooth muscle cells were incubated with primary α-smooth muscle actin and smoothelin antibodies followed by incubation with appropriately conjugated fluorescent secondary antibody. Unstained cells and cells stained with secondary antibody only were used as control.
Neural progenitor cells: Autologous neural progenitor cells isolated from rabbit small intestine were visualized under the microscope for clusters formation. Cells were then characterized by immunofluorescence. Cells were fixed followed by blocking. Cells were incubated in primary antibody solution to stain for p75NTR.
Engineered bio sphincters for implantation
Six BioSphincters were engineered per rabbit. Two BioSphincters were randomly selected for characterization studies. The remaining 4 BioSphincters were used for implantation into the rabbits.
Neural progenitor cells were counted using a hemocytometer and collected at a density of 1.2 million cells/6 BioSphincters (200,000 NPC/BioSphincter). The cells were then suspended in a 6-mL collagen/laminin hydrogel mixture. One milliliter of hydrogel with suspended NPC was pipetted around a sterilized cylindrical post placed in the center of pre-coated Sylgard plates. The solution was spread uniformly on the plates and allowed to gel at 37°C for 15 minutes.
In the meantime, confluent smooth muscle cells were trypsinized and counted using a hemocytometer. A density of 3 million cells/6 BioSphincters (500,000 SMC/BioSphincter) was collected. The cells were then suspended in a 6-mL collagen hydrogel mixture. 1 mL of the smooth muscle hydrogel mixture was pipetted on top of gelled NPC layer in each plate and allowed to gel for an additional 15 minutes at 37°C. Differentiation media (Neurobasal- A media supplemented with B27 supplement, 1% FBS and 1x antibiotic/antimycotic) was then added. Media was changed every other day.
On the 12th day, 2 of the engineered BioSphincters were used for characterization and the remaining 4 BioSphincters were transported to the surgery suite for implantation.
Characterization of the bio sphincters
Immunofluorescence of bio sphincters: BioSphincters were fixed and processed for histological evaluation. Processed BioSphincters were embedded in paraffin and sectioned. Sections were stained with primary antibodies against the smooth muscle marker; smooth muscle actin and neural marker; βIII tubulin.
Protein expression using qPCR: BioSphincters were obtained at days 7, 10, 11, 12, 13, and 14 post-engineering to assess (1) the maturity of the smooth muscle component and (2) the differentiation of the neural progenitor cells into mature neurons in the BioSphincters prior to implantation
Physiological testing of bio sphincters
BioSphincters were tested for functionality using an isometric force transducer. BioSphincters were placed between a stationary central pin and the measuring arm of the organ bath transducer (Harvard Apparatus, Holliston MA). BioSphincters were immersed in 4 mL of DMEM buffered with 25 mM HEPES which was changed at the end of every experiment. BioSphincters were maintained at 37°C
BioSphincters were tested for their ability to generate spontaneous basal tone. Functionality was quantified following pharmacological or electrical stimuli. Muscle functionality was tested by smooth muscle membrane depolarization using 60 mM potassium chloride (KCl). Functionality of the neurons in the BioSphincters was evaluated by electrical field stimulation (EFS, 5 Hz, 0.5 ms) using parallel platinum plate electrodes in the organ bath. Bio- Sphincters were stimulated in the absence and presence of neurotoxin (tetrodotoxin, TTX).
Implantation of bio sphincters
Engineered autologous BioSphincters were implanted approximately 6-8 weeks post biopsy procedure. Animals in the non-treated group did not go through any additional surgeries. During implantation surgery, general anesthesia was rendered using isoflurane via mask in animals in both the treated and shamsurgery groups. Similar pre-operative procedures were performed as described for the biopsy procedure
The surgical site was appropriately prepared. A circumferential curvilinear incision through a cutaneous tissue was made. A space between the rabbit’s own IAS and external anal sphincter (EAS) was created by dissecting a plane between the two anal sphincters. In the treated group, 4 BioSphincters (day 12 post-engineering) were implanted in this created space. Incisions were closed using simple-interrupted 5-0 prolene sutures. In the sham group, the space between the rabbit’s IAS and EAS was created but no BioSphincters were implanted in the space. Incisions were immediately closed using simple-interrupted 5-0 prolene sutures
Anorectal manometry
Anorectal manometry is a technique used to measure contractility in the anus and rectum. Anorectal manometry was performed initially at baseline prior to any surgery. These measurements reflected the control state for all animals in this study. Anorectal manometry was performed prior to any surgery (before animals went for any procedure) to record the baseline, and one month following IAS hemi-sphincterectomy (biopsy), then at 3, 6, and 12 months in each experimental group. Light anesthesia was induced using isoflurane via mask. Following sedation, the rabbits were laid on their right side and the catheter was inserted into the rectum 6 cm deep.
The catheter has four air-charged pressure transducers arranged at the same level circumferentially and 90° apart were used. The catheter was then withdrawn in 1 cm increments and the area of maximum resting pressure (anal basal pressure) was identified. Anal basal pressure was recorded. At this identified anal basal pressure, the s balloon attached to the distal aspect of the catheter was inflated to a volume of 20mL and then deflated. This technique is used to evaluate Recto Anal Inhibitory Reflex (RAIR), which is expressed as the percentage decrease in anal basal pressure in response to rectal balloon inflation. Data acquisition and analysis was performed using BioVIEW software (Sandhill Scientific, Littleton, CO, http://www.diversatekhealthcare.com/).
Histological evaluation
Sections of the surgical site were obtained at the time of euthanasia and fixed in formaldehyde. Tissues were then processed, embedded in paraffin and sectioned for histological analysis. H&E and Masson’s trichrome were performed on the obtained sections
Clinical evaluation of the animals
The rabbits’ weights were recorded at baseline (when rabbits arrived at the facility) before any surgery was performed and then regularly every month post surgeries. Additionally, venous blood samples were taken from the jugular vein of rabbits at baseline (prior to any surgeries as part of their physical) and at the defined time-points. The samples were placed on wet ice and delivered to a contract laboratory (IDEXX, North Grafton, MA) the same day. Blood was analyzed and compared among different groups.
Histopathological evaluation
Following euthanasia of the rabbits (when reached their defined time-points), the surgical site was accessed, the implantation site was harvested and immediately fixed in formaldehyde for histological analysis. In addition to the surgical site, biopsies from other tissues and organs were obtained for histological analysis. Tissues were then processed, embedded and sectioned. The tissue sections were stained with Hematoxylin and Eosin (H&E) and Masson’s trichrome. The sections were coded (blinded) and sent to a board-certified veterinary pathologist, for analysis of any pathology that might exist. Once analyzed, the results were un-blinded, and the pathologist prepared a report with the findings separated as to treatment conditions.
Statistical analysis
One-way ANOVA was performed with Bonferroni post-hoc analysis to compare anal basal pressure and RAIR among different groups.
Results
Cell characterization
Smooth muscle cells: Autologous smooth muscle cells isolated from rabbit IAS biopsy were allowed to grow in culture. Identity of smooth muscle cells was first confirmed microscopically (Figure 1). Smooth muscle cells exhibited spindlelike morphology (Figure 1A). Smooth muscle cells stained positive with smooth muscle markers; α-smooth muscle actin (Figure 1B) and smoothelin (Figure 1C). Positive stains confirmed contractile phenotype of isolated smooth muscle cells.
Autologous smooth muscle cells were also characterized by flow cytometry. IAS smooth muscle cells were incubated with primary α-smooth muscle actin antibody followed by incubation with appropriately conjugated fluorescent secondary antibody. Unstained cells and cells stained with secondary antibody only were used as control. Flow cytometry data analysis showed that over 94.5 ± 4.5% (n=3) of the isolated IAS smooth muscle cells expressed α-smooth muscle actin. Graphical representation is shown in Figure 1D.
Neural progenitor cells: Autologous neural progenitor cells isolated from rabbit small intestine were visualized under the microscope. Cells clustered into small floating bodies called neurospheres. Cells were then characterized by immunofluorescence and stained positive for p75NTR (Figure 1E).
Characterization of engineered bio sphincters
BioSphincters were characterized for the presence of contractile smooth muscle and mature neurons by immunohistochemistry and quantified by qPCR, followed by functionality testing
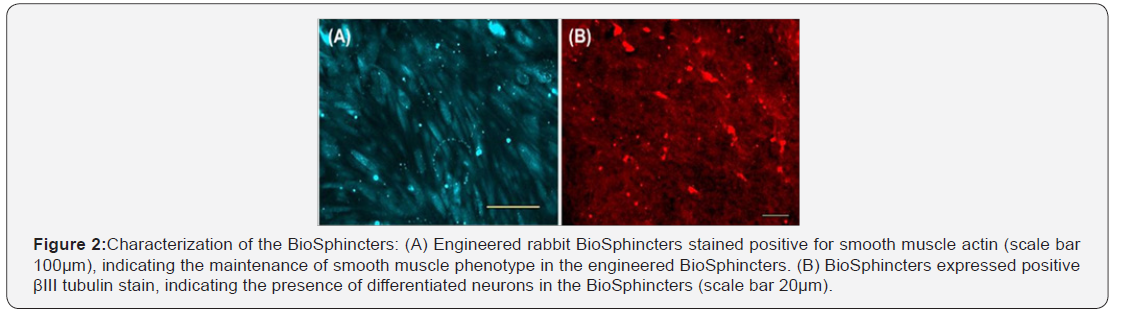
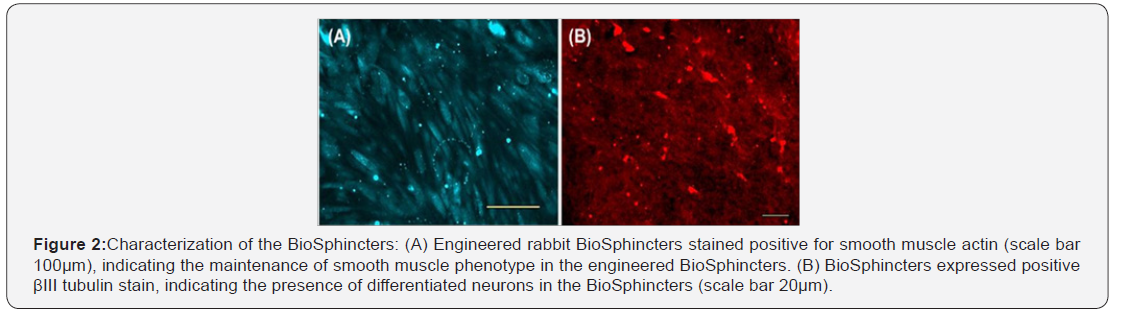
BioSphincters stained positive for α-smooth muscle actin and βIII tubulin, confirming the presence of mature contractile smooth muscle and differentiated neurons, respectively (Figure 2).
qPCR was conducted to quantify genetic expression of smoothelin (smooth muscle) and βIII tubulin (differentiated mature neurons) (Figure 3). BioSphincters were harvested starting at day 7 and followed up to day 14. Smoothelin in the BioSphincters was similar from day 7 to day 14 (without significant difference in the level of expression) indicating that the contractile phenotype of the smooth muscle component in the BioSphincters was maintained during the culture time. βIII tubulin expression increased significantly from day 7 to day 10 and reached plateau starting at day 12 through day 14, indicating that full differentiation of neural progenitor cells into neurons in the engineered BioSphincters was reached by day 12 post-engineering.

BioSphincters were tested for functionality using an isometric force transducer. Constructs were placed between a stationary central pin and the measuring arm of the organ bath transducer (Harvard Apparatus, Holliston MA). BioSphincters were immersed in 4 mL of DMEM buffered with 25 mM HEPES which was changed at the end of every experiment. BioSphincters were maintained at 37°C.
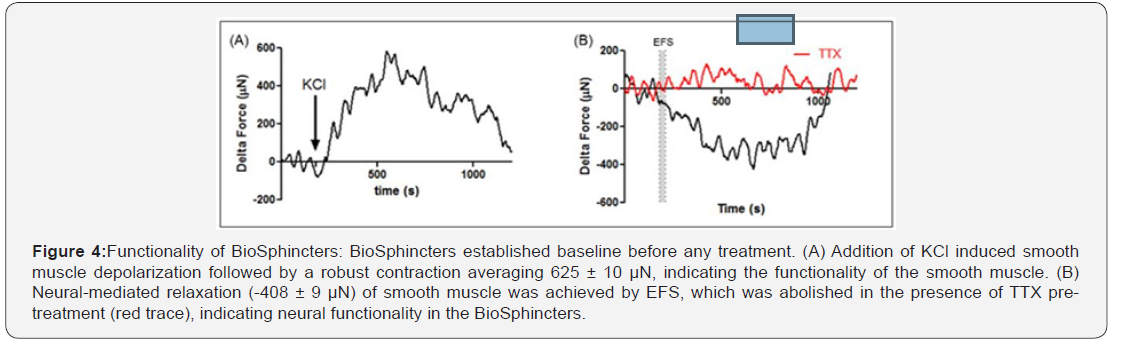
BioSphincters were able to generate spontaneous basal tone in the absence of any external stimulation that averaged 541 ± 13 μN (n=10). Generation of spontaneous basal tone is a characteristic of sphincter tissue. Functionality was quantified following pharmacological or electrical stimuli. Muscle functionality was tested using addition 30 mM potassium chloride (KCl). When BioSphincters established a baseline, addition of KCl caused a contraction above established basal tone, with maximal average contraction of 625 ± 10 μN (Figure 4).
To test the neural functionality, neuronally evoked relaxation was achieved by electrical field stimulation (EFS) (5 Hz, 0.5 ms) between parallel platinum plate electrodes in the organ bath. Maximal relaxation averaged -408 ± 9 μN (Figure 4B). EFS-mediated relaxation of the BioSphincter was totally inhibited in the presence of neurotoxin tetrodotoxin (TTX), indicating that the relaxation was mediated by functional neurons in the BioSphincters EFS-induced relaxation was also partially inhibited by nitric oxide neuron inhibitor (LNAME), indicating that the EFS-induced relaxation was partially mediated by functional nitric oxide neurons in the BioSphincters.
Anorectal manometry
Anorectal manometry was performed on all rabbits before any surgery at baseline. Manometry was then performed on all rabbits 1 month post-sphincterectomy to confirm fecal incontinence. Manometry was then performed regularly on rabbits in their respective groups.
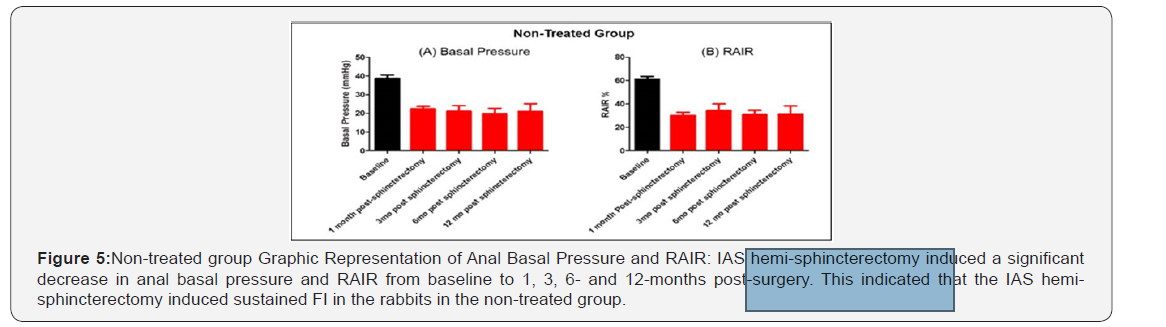
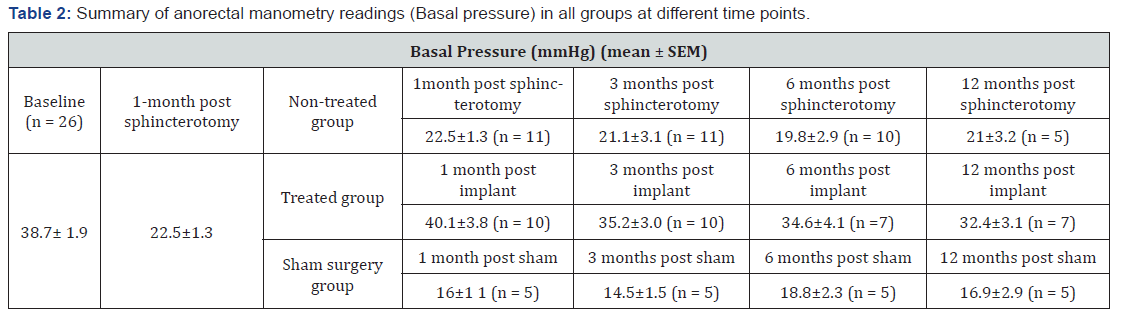
Basal Pressure: Hemi-circumferential internal anal sphincterectomy resulted in significant decrease in basal tone in all groups at 1-month post-sphincterectomy. In the non-treated group (Figure 5A), basal pressure remained low over the study period. Basal pressure decreased from 38.7 ± 9 mmHg to 22.5 ± 1.3 mmHg at 1-month, 21.1 ± 3.1 mmHg at 3 months, 19.8 ± 2.9 mmHg at 6 months and 21 ± 4.2 mmHg at 12 months post sphincterectomy (p < 0.0001). Basal pressures at 1, 3, 6, and 12 months post-sphincterectomy were not significantly different from each other (p > 0.05).
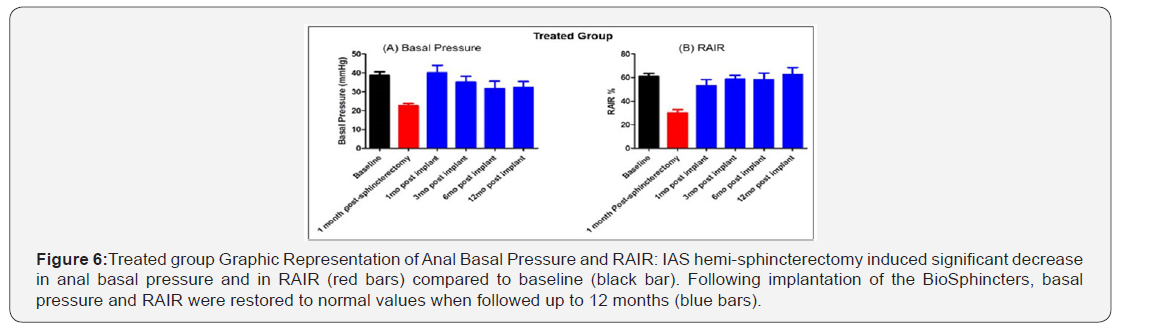
In the treated group prior to implantation, anorectal readings were performed on the rabbits following sphincterectomy to confirm their incontinence (Figure 6A – red bar). Anorectal manometry readings following implantation of 4 BioSphincters were conducted on the rabbits (Figure 6A – blue bars). Basal pressure significantly increased following implantation of the bioengineered BioSphincters (blue bars, p < 0.0001) compared to basal pressure following sphincterectomy (22.5 ± 1.3 mmHg). Anorectal manometry post-implantation indicated that basal pressure at 3 months (35.2 ± 3 mmHg), 6 months (37.0 ± 3.4 mmHg), and 12 months (32.4 ± 3.1 mmHg) was restored back to normal compared to baseline (p > 0.05).
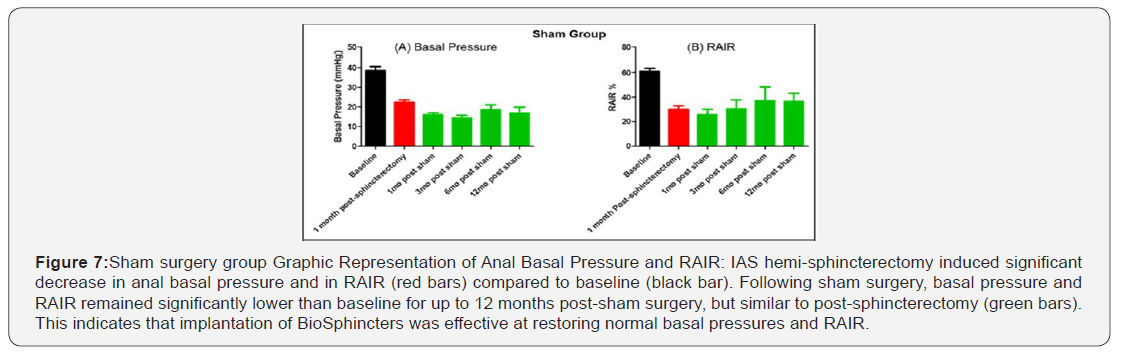
In the sham surgery group, basal pressure was significantly reduced from 38.7 ± 9 mmHg (baseline) to 16 ± 1.1 mmHg at 1-month time point, 14.5 ± 1.5 mmHg at 3-month time point, 18.8 ± 2.3 mmHg at 6-month time point, and 16.9 ± 2.9 mmHg at 12-month time point post-sham (p < 0.0001) (Figure 7A). Basal pressure at 1, 3, 6, and 12 months post sham was not significantly different from basal pressure at 1, 3, 6, and 12 months post-sphincterectomy (p > 0.05), however it was significantly lower than basal pressure post-implantation at 1, 3, 6, and 12 months (p < 0.05).
Recto anal inhibitory reflex or Balloon expulsion test RAIR: Sphincterectomy induced a significant decrease in RAIR in all rabbits. In the non-treated group, RAIR significantly decreased from 61.1 ± 2.4% (baseline) to 30 ± 2.7% at 1 month and remained significantly low (31 ± 7.3%) at 12 months post-sphincterectomy (p < 0.0001) (Figure 5B). RAIR at 1 and 12 months post-sphincterectomy were not significantly different from each other (p > 0.05).
In the treated group, implantation of 4 engineered BioSphincters restored RAIR to baseline (Figure 6B – blue bars). RAIR in the treated group was significantly higher than RAIR in the non-treated group at 12-month time point (p < 0.0001). RAIR in the treated group increased to 62.5 ± 5.9% at 12 months.
In the sham surgery group, RAIR was also significantly reduced from 61.1 ± 2.4% (baseline) to 36.6 ± 6.6% at 12-month time point (p < 0.05) (Figure 7B – green bars). RAIR at 12 months in the sham group was not significantly different from RAIR at 12 months in the non-treated group (p > 0.05); however, it was significantly lower than RAIR at 12 months in the treated group (p < 0.05).Histological findings
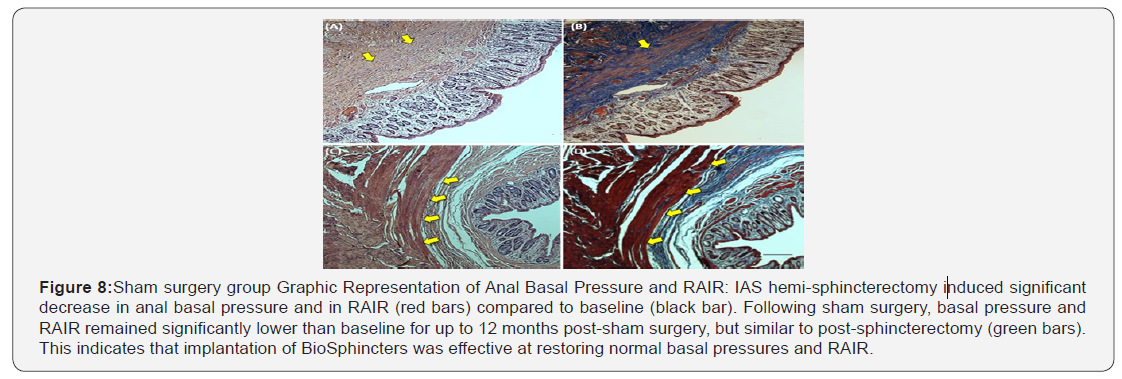
H&E and Masson’s trichrome (Figure 8A&B) of the non-treated group (sphincterectomy only without treatment) confirmed the discontinuity of the smooth muscle at the surgical site. The H&E stained sections of the implanted site confirmed the appearance of engineered BioSphincters (Figure 8C). The thick continuous sheet of IAS tissues validated the manometry outcomes. There was absence of any fibrosis or avascular collagen around the implant, indicating no foreign-body reaction with the implants. Sections stained with Masson’s trichrome showed abundant red stained smooth muscle cells interspersed with the blue stained collagen (Figure 8D).
Clinical assessment
Rabbits maintained healthy weights and gained weights throughout the study period. There were adverse events in four rabbits. These events were attributed to the physiology and behavior of the rabbit species and are not associated with the Bio- Sphincters or related procedures. Complete blood count (CBC) and blood chemistry in rabbits in all groups showed values within the normal range with no significant changes in the levels (Table 2).
Histopathological assessment
All sectioned tissues were examined by a board-certified veterinary pathologist, and recorded, in a blinded fashion with respect to treatment group. All lesions were identified by major process, location, distribution, and if possible, time course, and graded as minimal, mild, moderate or marked. No animals showed any evidence of neoplasia. Pathologic findings in this study were generally minor and consisted primarily of a low incidence of background changes and minor changes attributable to surgery. No histologic changes were seen in the collected tissues and organs (other thanthe surgical site). One animal in the treated group had ulcerative lesion in the rectum which may have been the result of a surgical complication or an infection, or an injury or infection unrelated to the procedure.
Discussion
As stated earlier, passive fecal incontinence is the result of the loss of integrity of the internal anal sphincter and its intrinsic enteric innervation. Cell-based therapy for passive fecal incontinence must take into account the restoration of the IAS-enteric neurons interaction. In our previous study, we created and validated a large animal model of passive fecal incontinence in rabbits by causing surgical injury to the IAS. Rabbits were then treated with autologous engineered BioSphincters. In that study, rabbits were followed over a short period of time (3 months) [15]. In this study, we performed a long term follow up on the rabbits (12 months) to determine the efficacy of the BioSphincters.
In previous animal studies, creation of theincontinence model was a challenge. In rats, resting pressure was restored after 14 days of the injury caused by sphincterotomy [16]. In dog and rabbit models, even though anal resting pressures were decreased, there was no distinction in the injury model between IAS and EAS [14,17]. In our model, we specifically target the IAS to mimick passive fecal incontinence seen in humans. Over a period of 12 months, rabbits in the non-treated group maintained a significant and sustained decrease in their anal basal pressure and RAIR compared to baseline. This was comparable to rabbits in the sham group. Histologically, the site of injury showed a discontinuity in the smooth muscle component of the sphincter.
Our lab developed a technique to engineer BioSphincters using IAS smooth muscle cells and neural progenitor cells. During the sphincterectomy surgery, the IAS obtained from the injury was used to isolate smooth muscle cells in the lab. During the same surgery, small intestinal biopsy was obtained to isolate neural progenitor cells. Following cell expansion, autologous BioSphincters were engineered. During the culture period, neural progenitor cells differentiated into mature functional excitatory and inhibitory motor neurons. Rabbits in the treated group received implantation of the engineered BioSphincters. Rabbits in this group showed an increase in the anal basal pressure and restoration of RAIR compared to baseline. This effect was sustained over the 12 months follow up. This indicates that the engineered innervated smooth muscle integrated with the host IAS tissue and helped in restoring functionality of the sphincter. RAIR is an important measure of fecal incontinence (Table 3).

RAIR is a coordinated reflex in the anal sphincter mediated by the enteric neurons of the myenteric plexus that sense the distension in the rectum. During manometry, after inflation of the balloon to cause distension of the rectum, rabbits exhibited restoration of RAIR values similar to baseline. This reflex was not seen in rabbits in the non-treated group. Rabbits in the sham group behaved similarly to rabbits in the non-treated group. Those rabbits had their IAS site surgically opened without implantation of Bio- Sphincters. This indicated that the restoration of basal pressure and RAIR in the treated group was not due to fibrosis or scar tissue as a result of the second surgery.
There has been no universal method to create a model of fecal incontinence. Models of fecal incontinence differ in the location and extent of caused injury. Injury to the anal sphincter (sphincterotomy) in rats has been reported to decrease the anal pressure however, this effect has been shown to be reversed within 4 weeks [16]. This was attributed to the low extent of injury caused to the sphincter. In a recent rat study, Kuismanen et al. [18] induced fecalincontinence in rats by causing an injury to external and internal anal muscles and to the anal mucosa
.Their injury caused a sustained decrease in anal pressure for up to 4 weeks. Most animal models don’t distinguish between the different muscles of the sphincter. Pudendal nerve injury is another way to create a model of fecal incontinence without any focus on the IAS component. In our model, the injury was specific to the internal anal sphincter. The incision performed at the anal sphincter separated the internal anal sphincter from the mucosa and the external sphincter, leaving them intact. A consistent injury was caused to all rabbits. The follow-up was extended to 12 months. Anal pressures in the non-treated rabbits were lower than those in the treated rabbits throughout the study period.
Previous attempts to treat fecal incontinence in animal models involve the use of stem cell therapy [19-21]. While some studies showed positive results, others have shown mixed outcomes. Kuismanen et al. [18] used adipose derived stem cells to treat incontinence in rats. Higher pressures were observed compared to untreated rats. The challenge with stem cell injection is finding an optimal route and a carrier for the cells for a spatiotemporal delivery of the injected cells. The study showed that injecting cells in saline induced inflammation compared to hydrogel carrier. In another model of fecal incontinence in rats, a group compared the delivery of cells via bio sutures or injection. While results were positive in terms of safety, the long-term fate, survival and effect of the cells is still in question. Our model has the advantage of providing autologous components necessary to remedy passive fecal incontinence. Smooth muscle cells in the BioSphincters contribute to the restoration of basal tone in the rabbits. The neural progenitor cells in the BioSphincters were allowed to differentiate into functional neurons in vitro prior to implantation of the Bio- Sphincters. The differentiated neurons integrated with the native myenteric plexus as demonstrated by restoration of RAIR. Specifically, nNOS-mediated inhibitory reflex was maintained for the 12 months follow up, which shows the long-term efficacy of the Bio- Sphincters.
Our large animal pre-clinical study showed the long-term safety and efficacy of the BioSphincters. The results of this study are promising as the BioSphincter approach provides an autologous solution for patients with passive fecal incontinence. Results have shown that the basal pressure and RAIR were restored, 2 important features of fecal continence
Acknowledgment
This work was supported by Army, Navy, NIH, Air Force, VA and Health Affairs to support the AFIRM II effort, under Award No.W81XWH-13-2-0052; GU 7 and NIH/NIDDK R01DK071614 and R42DK105593 to CELL- BIO LLC.
References
- Bartlett L, Nowak M, Ho YH (2009) Impact of fecal incontinence on quality of life. World J Gastroenterol 15(26): 3276-3282.
- Whitehead WE, Borrud L, Goode PS, Meikle S, Mueller ER, et al. (2009) Fecal incontinence in US adults: epidemiology and risk factors. Gastroenterology 137(2): 512-517.
- Varma MG, Brown JS, Creasman JM, Thom DH, Van Den Eeden SK, et al. (2006) Fecal incontinence in females older than aged 40 years: who is at risk? Dis Colon Rectum 49(6): 841-851.
- Jerez Roig J, Souza DL, Amaral FL, Lima KC (2015) Prevalence of fecal incontinence (FI) and associated factors in institutionalized older adults. Arch Gerontol Geriatr 60(3): 425-430.
- Menees SB, Almario CV, Spiegel BM, Chey WD (2018) Prevalence of and Factors Associated With Fecal Incontinence: Results From a Population-Based Survey. Gastroenterology 154(6): 1672-1681.
- Rao SS (2004) Pathophysiology of adult fecal incontinence. Gastroenterology 126: S14-S22.
- Huebner M, Margulies RU, Fenner DE, Ashton Miller JA, Bitar KN, et al. (2007) Age effects on internal anal sphincter thickness and diameter in nulliparous females. Dis Colon Rectum 50(9): 1405-1411.
- Lindsey I, Jones OM, Smilgin Humphreys M, Cunningham C, Mortensen NJ (2004) Patterns of fecal incontinence after anal surgery. Dis Colon Rectum 47(10): 1643-1649.
- Remes Troche JM, Rao SS (2008) Neurophysiological testing in anorectal disorders. Expert Rev Gastroenterol Hepatol 2(3): 323-335.
- Maslekar S, Smith K, Harji D, Griffiths B, Sagar P (2013) Injectable collagen for the treatment of fecal incontinence: long-term results. Dis Colon Rectum 56(3): 354-359.
- Hull T, Giese C, Wexner SD, Mellgren A, Devroede G, et al. (2013) Long-term durability of sacral nerve stimulation therapy for chronic fecal incontinence. Dis Colon Rectum 56(2): 234-245.
- Darnis B, Faucheron JL, Damon H, Barth X (2013) Technical and functional results of the artificial bowel sphincter for treatment of severe fecal incontinence: is there any benefit for the patient? Dis Colon Rectum 56(4): 505-510.
- Healy C, Herlihy C, Brien C, Connell P, Jones J (2008) Experimental models of neuropathic fecal incontinence: an animal model of childbirth injury to the pudendal nerve and external anal sphincter. Dis Colon Rectum 51(11): 1619-1626.
- Kang SB, Lee HS, Lim JY, Oh SH, Kim SJ (2013) Injection of porous polycaprolactone beads containing autologous myoblasts in a dog model of fecal incontinence. J Korean Surg Soc 84(4): 216-224.
- Bohl JL, Zakhem E, Bitar KN (2017) Successful Treatment of Passive Fecal Incontinence in an Animal Model Using Engineered Biosphincters: A 3‐Month Follow‐Up Study. Stem Cells Transl Med 6(9): 1795-1802.
- Salcedo L, Damaser M, Butler R, Jiang HH, Hull T, et al. (2010) Long-term effects on pressure and electromyography in a rat model of anal sphincter injury. Dis Colon Rectum 53(8): 1209-1217.
- Kajbafzadeh AM, Elmi A, Talab SS, Esfahani SA, Tourchi A (2010) Functional external anal sphincter reconstruction for treatment of anal incontinence using muscle progenitor cell auto grafting. Dis Colon Rectum 53(10): 1415-1421.
- Kuismanen K, Juntunen M, Narra Girish N, Tuominen H, Huhtala H, et al. (2018) Functional outcome of human adipose stem cell injections in rat anal sphincter acute injury model. Stem Cells Transl Med 7(3): 295-304.
- Pathi SD, Acevedo JF, Keller PW, Kishore AH, Miller RT, et al. (2012) Recovery of the injured external anal sphincter after injection of local or intravenous mesenchymal stem cells. Obstet Gynecol 119(1): 134-144.
- Salcedo L, Mayorga M, Damaser M, Balog B, Butler R, et al. (2013) Mesenchymal stem cells can improve anal pressures after anal sphincter injury. Stem Cell Res 10(1): 95-102.
- Salcedo L, Penn M, Damaser M, Balog B, Zutshi M (2014) Functional outcome after anal sphincter injury and treatment with mesenchymal stem cells. Stem Cells Transl Med 3(6): 760-767.






























