Malignant Degeneration of a Giant Hepatic Adenoma during Pregnancy
Nicolas Duchateau*, Ben Gys, Arno Talboom, Glenn de Weerdt and Bart Bracke
Hepatobiliary surgery, University Hospital Antwerp, Belgium
Submission: January 28, 2019; Published: March 11, 2019
*Corresponding author:Nicolas Duchateau, Hepatobiliary surgery, University Hospital Antwerp, Doornelei 22, B-2018 Antwerpen, Belgium
How to cite this article:Nicolas D, Ben G Arno T, Glenn d W, Bart B. Malignant Degeneration of a Giant Hepatic Adenoma during Pregnancy. Adv Res Gastroentero Hepatol. 2019; 12(4): 555843. DOI: 10.19080/ARGH.2019.12.555843.
Keywords: Hepatocytes; Liver tumors; Vacuolated cytoplasm; Giant hepatocellular adenoma; Malignant transformation; Vaginal delivery; Gestational age; Hemorrhage; Intrahepatic hemorrhage
Abbrevations: OC: Oral Contraceptive; EASL: European Association for the Study of the Liver; BMI: Body Mass Index; HCA Hepatocellular Adenoma; NGS: Next Generation Sequencing; NSE: Non-Standard Exception; BeLIAC: Belgian Liver & Intestine Advisory Committee; ICU: Intensive Care Unit; FFP: Fresh Frozen Plasma
Introduction
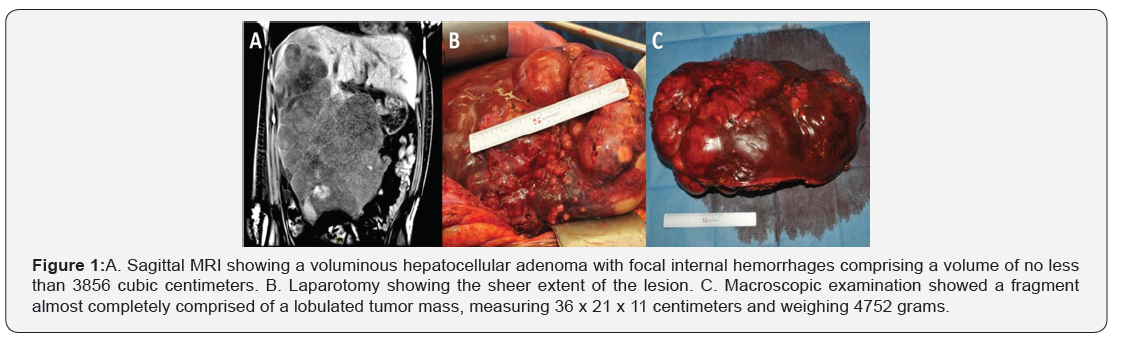
Hepatocellular adenomas are rare, benign, hormonally inactive liver tumors composed of well-circumscribed nodules that contain sheets of hepatocytes with a vacuolated cytoplasm. Most diagnosed tumors are discovered incidentally because of modern imaging. Hepatocellular adenomas are usually asymptomatic, but symptoms such as pain, dyspepsia and abdominal mass are described as the result of tumor bulk or hemorrhage. They are commonly found in people with elevated systemic levels of estrogen. Malignant transformation is rare. We report the case of a giant hepatocellular adenoma in a 30-year-old female who acutely presented with abdominal pain three days after uncomplicated vaginal delivery. The resected tumor measured 36 x 21 x 11 centimeters
Case Report
A 30-year-old pregnant woman presented to the emergency department with preterm premature rupture of the membranes at 35 weeks and 5 days gestational age. Shortly thereafter, she developed spontaneous contractions leading to an uncomplicated vaginal delivery. Three days postpartum, she experienced complaints of a generally swollen and tender abdomen accompanied by a stabbing pain in the right upper quadrant, prompting readmission to the emergency department. Urgent abdominal computed tomography scanning revealed an extremely enlarged right liver lobe with multiple heterogeneous hypodense nodules together with intrahepatic bleeding components, clustering into one sizable mass. Diagnosis of a giant hepatocellular adenoma with contained intrahepatic hemorrhage was made. Subsequently, regarding further diagnostic work-up, magnetic resonance
imaging was conducted confirming a voluminous hepatocellular adenoma with focal internal hemorrhages, comprising a volume of no less than 3856 cubic centimeters (Figure 1, panel A). Remarkably, prenatal ultrasounds did not withhold any abnormalities. Apart from one event two weeks before going into labor, the patient was completely asymptomatic. She experienced vague abdominal pain with nausea and vomiting, common symptoms during pregnancy and therefore believed to be pregnancy-related. Liver function tests during the first trimester of pregnancy were normal, afterwards no liver function tests were taken.
Surgical Technique
Given the sheer extent of the lesion, the bleeding diathesis, and the abdominal discomfort, an open right hemi hepatectomy was performed via Mercedes incision (Figure 1, panel B). An anterior approach was used with minimal mobilization of the right liver. After careful dissection of the liver hilum, the right portal vein was ligated, followed by the right hepatic artery. To further minimize intraoperative bleeding, an intermittent Pringle maneuver was applied at the time of liver transection which consisted of cross-clamping the hepatoduodenal ligament for 15 minutes and releasing the clamp for 5 minutes until the liver transection was completed. The inferior vena cava was not clamped but porto-venous extracorporeal bypass was stand-by. Total procedure time was 5 hours. Intraoperative blood loss was 10 liters of which 4 liters was recovered using a Cell Saver® with leukocyte depletion filter. An additional 6 units of packed cells, 9 units of fresh frozen plasma (FFP) and 2 units of platelet concentrate were given. Postoperative stay in the intensive care unit (ICU) was characterized by transient right-sided heart decompensation and pleural effusion after extensive fluid resuscitation, which was managed by the administration of diuretics. After 5 days, she was transferred to the ward and further recovery was uneventful. She was discharged 11 days after surgery.
Liver transplantation was considered, however when liver adenomas are requested for listing via the Belgian Liver & Intestine Advisory Committee (BeLIAC) they only receive a Non-Standard Exception (NSE) in Belgium. If the indication was approved, the patient would receive a MELD score of 22 points that increased 3 points every 90 days, giving her a long waiting time. In addition, the liver adenoma was respectable
Results
The resected tissue specimen was examined anatomopathological. Macroscopic examination showed a fragment almost completely comprised of a lobulated tumor mass, measuring 36 cm x 21 cm x 11 cm and weighing 4752 grams (Figure 1, panel C). Cross section revealed lobules containing whitish-pink tissue and hemorrhagic caverns. Microscopic examination showed a sharply demarcated tumor without obvious capsulation. The tumor itself had double cell-layered trabecular architecture with isolated blood vessels and absence of accompanying bile ducts. Nodules within the tumor showed trabecular architecture with more than three cell layers and nuclear pleomorphism matching a well differentiated, grade 2 hepatocellular carcinoma (HCC) originating from a hepatocellular adenoma, with no evidence for blood vessel invasion. Molecular studies using Next Generation Sequencing (NGS) revealed no mutations in BRAF, CTNNB1, RET, TP53 or PIK3CA. There were no mutations found in β-catenin. Surrounding liver parenchyma showed normal architecture apart from reactive changes secondary to tumor mass effect. Pathologic staging was pT3 G1, and complete resection was obtained.
Close follow-up was advised after multidisciplinary discussion. Magnetic resonance imaging was planned one month postoperatively together with alpha-fetoprotein testing. Based on these results it will be decided whether or not to consider adjuvant therapy.
Discussion
A hepatocellular adenoma (HCA) is a rare benign liver tumor, making up 2 percent of all liver neoplasms with an estimated incidence of 3 per million per year in the general population. They are predominantly found in young women in their reproductive years, with an incidence of 3-4 per 100.000 women [1]. These types of adenomas usually vary in size between 1 and 30 cm diameter, HCAs exceeding a diameter of 30cm are extremely rare [2].
Typically, patients with HCAs are asymptomatic. With increasing size, they can cause symptoms of vague abdominal pain, gastro-intestinal discomfort and in extreme cases, jaundice because of mass effect. However, abdominal distension, vague abdominal pain and gastro-intestinal discomfort are also very common symptoms during pregnancy and can thus be attributed to pregnancy, delaying diagnosis [3]. The most significant complications of hepatocellular adenomas are spontaneous hemorrhage and malignant transformation [4].
Risk factors for hemorrhage in liver adenomas are the presence of more than ten lesions, a BMI (Body Mass Index) of more than 25kg/m2, tumors located in segments II-III, an exophytic localization in relation to the liver capsule, the presence of central or peripheral arteries and lesion size greater than 35 mm [5]. Spontaneous rupture or spontaneous hemorrhage can be life-threatening [6,7].
Of all HCAs, approximately 4.2 percent will eventually transform into malignant hepatocellular carcinoma [8].
Guidelines of the European Association for the Study of the Liver (EASL) stated that resection is indicated for HCAs greater than 5 cm, which do not regress by 6 months after conservative measures or those continuing to grow [9]. Very seldom does an HCA smaller than 5 cm progress to an HCC.
Risk factors for malignant degeneration include a history of anabolic androgen intake, male gender, increasing size of the adenoma, patients with type I or type III glycogen storage disease and adenomas containing a β-catenin mutation [10]. The only risk factor present in our patient was increasing size of the adenoma
Oral contraceptive (OC) drug use could also play a potential role in malignant degeneration; however, this is not yet clearly established. OCs do stimulate the growth of HCAs and an increase in adenoma size raises concern for malignant degeneration. Given the influence of estrogens on tumor growth, all women diagnosed with HCA should initially be treated conservatively through cessation of oral contraceptives and weight reduction
Little is known about the behavior of HCAs during pregnancy. The build-up of estrogen levels, the hyperdynamic circulation and the increase in liver vascularization during pregnancy, especially in the third trimester can lead to growth of the adenoma [11]. We hypothesize that this physiologic elevation of pregnancy hormones contributed to the malignant degeneration of the HCA.
Conclusion
Although abdominal discomfort, nausea and vomiting are frequent symptoms experienced during pregnancy, one should always thoroughly investigate these complaints. Liver tumors such as hepatocellular adenomas are rare but should be kept in mind. Malignant transformation is even more infrequent. We believe that increased endogenous levels of steroid hormones during pregnancy can lead to rapid, hormone-induced growth of hepatocellular adenomas, promoting rupture and effectuating malignant degeneration.
References
- Thomeer MG, Broker M, Verheij J, Doukas M, Terkivatan T, et al. (2016) Hepatocellular adenoma: when and how to treat? Update of current evidence. Therap Adv Gastroenterol 9(6): 898-912.
- Vijay A, Elaffandi A, Khalaf H (2015) Hepatocellular adenoma: An update. World J Hepatol 7(25): 2603-2609.
- van Aalten SM, de Man RA, JN IJ, Terkivatan T (2012) Systematic review of haemorrhage and rupture of hepatocellular adenomas. Br J Surg. 99(7): 911-916.
- Bieze M, Phoa SS, Verheij J, van Lienden KP, van Gulik TM (2014) Risk factors for bleeding in hepatocellular adenoma. The British journal of surgery 101(7): 847-855.
- Klompenhouwer AJ, de Man RA, Thomeer MG, Ijzermans JN (2017) Management and outcome of hepatocellular adenoma with massive bleeding at presentation. World J Gastroenterol 23(25): 4579-4586.
- Mota C, Carvalho AM, Fonseca V, Silva MT, Victorino RMM (2017) Exuberant liver adenomatosis presenting with iron deficiency anemia. Clin Case Rep 5(5): 574-577.
- Stoot JH, Coelen RJ, De Jong MC, Dejong CH (2010) Malignant transformation of hepatocellular adenomas into hepatocellular carcinomas: a systematic review including more than 1600 adenoma cases. HPB (Oxford) 12(8): 509-522
- (2016) EASL Clinical Practice Guidelines on the management of benign liver tumors. J Hepato 65(2): 386-398.
- Farges O, Ferreira N, Dokmak S, Belghiti J, Bedossa P, et al. (2011) Changing trends in malignant transformation of hepatocellular adenoma. Gut 60(1): 85-89.
- van Aalten SM, Broker ME, Busschbach JJ, de Koning HJ, de Man RA, et al. (2012) Pregnancy and liver adenoma management: PALM-study. BMC Gastroenterol 12: 82.






























