Can Treatment Lead to Chronic Diarrhea in a Young Adult?
Sridhar Sundaram1* and Vaneet Jearth2
1 Department of Gastroenterology, Seth GS Medical College and KEM Hospital, India
2Department of Digestive Diseases and Clinical Nutrition, Tata Memorial Centre, India
Submission: September 01, 2018; Published: October 09, 2018
*Corresponding author: Sridhar Sundaram MD (Medicine), DM (Gastroenterology), Department of Gastroenterology, Seth GS Medical College and KEM Hospital, Dr. E Borges Road, Parel, Mumbai, India, Pin Code: 400012, Tel: +919860096846; Email: drsridharsundaram@gmail.com
How to cite this article: Sridhar S, Vaneet J. Can Treatment Lead to Chronic Diarrhea in a Young Adult?. Adv Res Gastroentero Hepatol. 2018; 11(2): 555807. DOI: 10.19080/ARGH.2018.11.555807.
Abstract
Immunodeficiency disorders can present primarily with gastrointestinal manifestations as seen in Combined Variable Immuno-deficiency (CVID) and Selective IgA deficiency. Rarely, treatment with immunosuppressants like rituximab can lead to persistent hypogammaglobulinemia which may have consequences like recurrent gastrointestinal infections. We present a rare case of chronic diarrhea resulting from rituximab induced hypogammaglobulinemia, presenting with ileal giardiasis.
Keywords: Immunodeficiency disorders; Gastrointestinal manifestations; IgA deficiency; Immunosuppressants; Ileal giardiasis; Hypogammaglobulinemia; Immunoglobulins therapy
Abbrevations: NHL: Non-Hodgkin Lymphoma; CVID: Combined Variable Immuno-Deficiency; IBD: Inflammatory Bowel Disease; Ig: immunoglobulin
Introduction
Rituximab may cause hypogammaglobulinemia in a subset of patients, especially if given in multiple cycles. Early clinical trial data suggested that hypogammaglobulinemia is transient, however subsequent reports have described hypogammaglobulinemia that is both persistent and clinically significant, necessitating immunoglobulins therapy in some cases [1-5]. Gastrointestinal disease is identified in approximately 10 to 20 percent of these patients and may be the presenting symptom in some [6]. Many of these disorders mimic classic forms of disease (in the absence of immunodeficiency) such as celiac sprue, inflammatory bowel disease (IBD), and pernicious anemia but differ in pathogenesis and are often unresponsive to conventional therapies. Gastroenterologists therefore must be able to diagnose and treat patients with immunodeficiency..
Case
A 30 years old male was referred to our hospital with chief complains of low backache from last 2 months with referred pain to right lower limb. On evaluation outside, CECT Abdomen and pelvis was suggestive of 7.5 X 7.5cm ill-defined homogenous lesion in upper portion of right psoas muscle extending along aortocaval and prevertebral region. Right paravertebral lesion biopsy was suggestive of High-grade Non-Hodgkin Lymphoma (NHL). Patient received 6 cycles of chemotherapy containing Rituximab as part of NHL BFM (Berlin-Frankfurt-Münster) 90 Protocol followed by radiotherapy to Right paravertebral lesion to a dose of 45 Gy/25 cycles. Post 4 cycles of chemotherapy PET CT showed complete metabolic response. Patient tolerated chemotherapy well. Patient was on regular follow up every 3 monthly.
After 9 months of treatment completion, patient presented with complaints of increased frequency of stools, 7 to 8 episodes per day, large volume, not associated with blood or pain abdomen from last 1 month. There was also history of weight loss of approximately 12kg in 4months. Clinically patient was pale, anicteric, cachexic with no pedal edema. Differentials for small bowel diarrhea included Radiation enteropathy, Crohn’s disease, recurrence of the lymphoma and possibility of celiac disease. On investigations, total protein was 6.2 g/l with albumin of 4.0 g/l. Haemoglobin was 10.2 g% with normal total leucocyte counts and platelets. Kidney and liver function tests were unremarkable. Stool examination was unremarkable. ESR was 45mm/hr while CRP was 1.2 mg%. IgA for tissue transglutaminase was negative.
CT Enterography showed thickened large bowel with normal small bowel loops. UGIE showed scalloping of duodenal folds with edematous mucosa. Biopsy from D1, D2 and jejunum showed show similar histomorphological features of mild increase in lymphocytic infiltrate along with neutrophils and few eosinophilis in the lamina propria with mild blunting of the villi. Ileocolonoscopy was done which showed normal appearing mucosa. Mapping biopsies were taken which was suggestive of Giardiasis in Ileum along with nonspecific inflammation in colon and small bowel. Patient then complained of pus discharge in the perianal region with pain. Local examination suggested hard tender perianal swelling. MRI pelvis showed small 5 x 2 mm sized pocket of air in the Intersphincteric plane at 1-2o clock position, with surrounding T2/ FLAIR hyperintensity showing post contrast enhancement, suggestive of infective collection. He was treated for the likely pilonidal abscess with ciprofloxacin and metronidazole and recovered over 3 weeks.
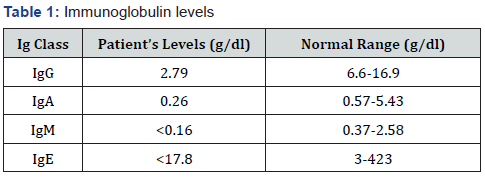
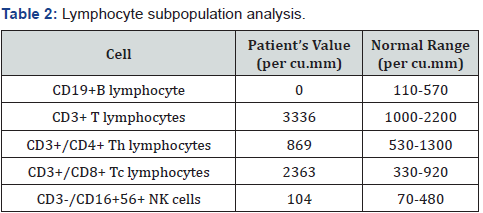
As for the diarrhea, patient had intermittent relief in symptoms on antibiotics, but symptoms relapsed again after 1 month. Biopsy was again reviewed for any evidence of IBD but histologic findings were nonspecific. In view of past history of receiving Rituximab, on and off symptoms, improvement with antibiotics, and ileal giardiasis and perianal infection, we sent his immunoglobulin profile which showed low levels of all immunoglobulins (Table 1). Lymphocyte subpopulation analysis revealed absence of CD19 and CD20 B lymphocytes, likely long-term adverse effect to Rituximab (Table 2). Patient was subsequently treated as Rituximab induced hypogammaglobulinemia with gastrointestinal manifestations. He was started on IV Immunoglobulins (IVIg) monthly and eventually his symptoms improved over next 3 months.
Discussion
Rituximab is a chimeric immunoglobulin (Ig)G1 CD20-specific monoclonal antibody. The primary use of rituximab is in the treatment of B cell malignancies and in selected autoimmune disorders, including rheumatoid arthritis, autoimmune cytopenias, autoimmune pancreatitis, Sjögren syndrome, and some forms of vasculitis. In a review of 211 patients treated with rituximab for B cell lymphoma, 38 percent of patients with initially normal serum IgG levels developed hypogammaglobulinemia [7]. Immune globulin therapy was needed in 6.6 percent to control infections.
Iatrogenic hypogammaglobulinemia is an acquired hypogammaglobulinemia caused by various drugs but clinically, it may be difficult to distinguish it from common variable immune deficiency (CVID), which is a congenital disorder that involves low levels of most or all of immunoglobulins, lack of B-lymphocytes or plasma cells that are capable of producing antibodies, and repeated bacterial infections.
It is difficult to differentiate whether the hypogammaglobulinemia is caused by rituximab or the preexisting CVID has been aggravated by rituximab treatment [5], especially if there are no previous immunology and biochemistry tests available as in our case. Chronic diarrhea is also one of the most common clinical presentation of CVID. This patient reported no previous history of any significant infection prior to rituximab therapy. The diagnosis of immunodeficiency, in this case, severity of infection, response to antibiotic, and a previous history of rituximab therapy led to our speculation of an acquired hypogammaglobulinemia likely as consequence of rituximab therapy.
In our case, the first clue regarding underlying immunodeficiency came from diagnosis of ileal giardiasis. Our patient had on and off symptoms with response to certain antibiotics. Presence of perianal abscess with background inflammatory changes in colonic biopsies and scalloping of duodenal folds with non specific inflammation in our case again is in concurrence with fact that hypogammaglobinemia can mimic as IBD and celiac disease respectively. Giardiasis is known to involve the proximal small intestine, however in presence of significant inflammation in the duodenum, ileal biopsies may support a diagnosis by demonstration of trophozoites [8]. Our patient also had giardiasis seen in the terminal ileum without any trophozoites seen in the duodenal biopsies with mild villous blunting seen.
Because GI disease may be the first presentation of an underlying immunodeficiency, it is imperative to consider immunodeficiency in any patient with intractable diarrhea, malabsorption, and failure to thrive that is resistant to conventional treatments with the increasing use of rituximab in non-malignant settings, it is important for clinicians treating these patients to be aware of hypogammaglobulinemia and serious infections occurring even years after completion of treatment.
References
- Thiel J, Rizzi M, Engesser M, Dufner AK, Troilo A, et al. (2017) B cell repopulation kinetics after rituximab treatment in ANCA-associated vasculitides compared to rheumatoid arthritis, and connective tissue diseases: a longitudinal observational study on 120 patients. Arthritis Res Ther 19(1): 101.
- Adeli MM, Eichner BH, Thornburg C, Williams L (2009) Persistent antibody depletion after rituximab in three children with autoimmune cytopenias. Pediatr Hematol Oncol 26(8): 566-572.
- Irie E, Shirota Y, Suzuki C, Tajima Y, Ishizawa K, et al. (2010) Severe hypogammaglobulinemia persisting for 6 years after treatment with rituximab combined chemotherapy due to arrest of B lymphocyte differentiation together with alteration of T lymphocyte homeostasis. Int J Hematol 91(3): 501-508.
- Cooper N, Davies EG, Thrasher AJ (2009) Repeated courses of rituximab for autoimmune cytopenias may precipitate profound hypogammaglobulinaemia requiring replacement intravenous immunoglobulin. Br J Haematol 146(1): 120-122.
- Diwakar L, Gorrie S, Richter A, Chapman O, Dhillon P, et al. (2010) Does rituximab aggravate pre-existing hypogammaglobulinaemia? J Clin Pathol 63(3): 275-277.
- Agarwal S, Mayer L (2009) Pathogenesis and treatment of gastrointestinal disease in antibody deficiency syndromes. J Allergy Clin Immunol 124(4): 658-664.
- Casulo C, Maragulia J, Zelenetz AD (2013) Incidence of hypogammaglobulinemia in patients receiving rituximab and the use of intravenous immunoglobulin for recurrent infections. Clin Lymphoma Myeloma Leuk 13(2): 106-111
- Oberhuber G, Mesteri I, Kopf W, Müller H (2016) Demonstration of Trophozoites of G. Lamblia in Ileal Mucosal Biopsy Specimens May Reveal Giardiasis in Patients with Significantly Inflamed Parasite-free Duodenal Mucosa. Am J Surg Pathol 40(9): 1280-1285






























