Association between Oxidative Stress, Systemic Inflammation and Renal Function in Type 2 Diabetic Nephropathy
Mohammed H Saiem Al-Dahr* and Essam H Jiffri
Department of Medical Laboratory Technology, King Abdulaziz University, Jeddah, Saudi Arabia
Submission: May 08, 2017; Published: August 11, 2017
*Corresponding author: Mohammed H Saiem Al-Dahr, Department of Medical Laboratory Technology, Faculty of Applied Medical Sciences, King Abdulaziz University, P.O. Box 80324, Jeddah, 21589, Saudi Arabia, Tel: +966505608510; Email: mdahr@kau.edu.sa
How to cite this article: Mohammed H S, Essam H J.Association between Oxidative Stress, Systemic Inflammation and Renal Function in Type 2 Diabetic Nephropathy. Adv Res Gastroentero Hepatol 2017; 6(4): 555695. DOI: 10.19080/ARGH.2017.06.555695
Abstract
Background: Diabetic nephropathy (DN) is the common microvascular diabetic complication worldwide. However, there is still insufficient understanding of the full mechanism related to progressive diabetic renal disease. Objective: This study aimed to detect the association between oxidative stress, systemic inflammation and kidney function in patients with type 2 diabetic nephropathy.
Material and Methods: One hundred-twenty obese T2DM patients (72 males and 48 females), their body mass index (BMI) was 31-36 Kg/ m2 and the mean of diabetes chronicity was 12.65±2.49 years enrolled in the present study in addition to sixty apparently healthy individuals were recruited to serve as non-diabetic control. Participants were included three groups; seventy-five subjects with diabetic nephropathy (group I), 45 diabetic subjects without nephropathy (group II) and 60 controls (group III).
Results: The mean values of the biochemical parameters of the three groups proved that both group (I) and group (II) had a significant higher serum creatinine, TNF-α, IL-6, CRP and MDA levels than group (III) in addition to a significant lower serum GPX, GSH and SOD levels in group (III) than both group (I) and group (II). However, there were significant differences in all biochemical characteristics between group (I) and group (II). Moreover, the values of IL-6, TNF-α, CRP and MDA showed a strong direct relationship with value of creatinine in the three groups. However, the values of GPX, GSH and SOD showed a strong inverse relationship with value of creatinine in the three groups (P<0.05).
Conclusion: There is an evidence that oxidative stress and inflammatory cytokines appear to be significantly correlated with renal function among patients with diabetic nephropathy.
Keywords: Inflammatory cytokines; Diabetic nephropathy; Oxidative stress
Introduction
Diabetic nephropathy (DN) is a metabolic disorder with high rate of morbidity and mortality. Global pattern of diabetic nephropathy incidence is not entirely explored, but its incidence has already occurred widely and is estimated to be increased in prevalence [1]. However, DN is one of the main microvascular diabetic complications that leads to renal failure worldwide [2-5]. While, DN occur in 20-40% of type 2 diabetes mellitus (T2DM) patients and the principal etiology of renal failure [6,7].
Patients with DN suffer from high rate of morbidity and mortality. In fact, a rapid kidney function decline is a predictor for both cardiovascular disorders as well as all-cause mortality [8-10]. Risk factors of DN include poor metabolic control, diabetes duration, race, heredity, life style, diet composition, aging and hypertension. On the other hand, oxidative stress and inflammatory factors are 2 serious elements in promoting DN [11,12].
Hyperglycemia induces oxidative stress and inflammation [13]. In addition, poor glycemic control induce abnormal level of oxidative stress markers [14,15]. In the other hand, oxidative stress induce dysfunction of β-cell that lead to insulin resistance development, diabetes and its associated microvascular complications [16-21], so that patients with T2DM are under oxidative stress because of prolonged exposure to hyperglycemia [22].
Researches proved that hyperglycemia that induced oxidative stress and inflammation may play vital role in the pathogenesis of DN [23,24]. Hyperglycemia in diabetic patients leads to mitochondrial dysfunction, advanced glycation end processes and other factors, and generate the reactive free radicals, then triggers the DNA fragmentation that lead to cell death [25]. Hyperglycemia also causes oxidative stress, decreases the regeneration of glutathione (GSH) from oxidized GSH and reduces the availability of nicotinamide adenine dinucleotide phosphate [26,27]. This study aimed to detect the association between oxidative stress, systemic inflammation and kidney function in patients with type 2 diabetic nephropathy.
Materials and Methods
Subjects
One hundred-twenty obese T2DM patients (72 males and 48 females), their body mass index (BMI) was 30 to 36 Kg/m2 and the mean of diabetes chronicity was 12.65±2.49 years were selected from the out-patient diabetic clinic of the King Abdalziz Teaching Hospital. Exclusion criteria included smokers, kidney insufficiency, congestive heart failure, pregnant female patients, hepatitis and respiratory failure. In addition, sixty apparently healthy, medically free, and treatment naive individuals were recruited to serve as non-diabetic control. Participants were included three groups; seventy-five diabetic nephropathy patients (group I), 45 diabetic patients without nephropathy (group II) and 60 controls (group III). The Ethics Committee of the Faculty of Applied Medical Sciences, King Abdulaziz University, approved this study.
Measurements
The baseline and anthropometric data were collected for all participants at the time of enrollment. Independent assessors who were blinded to group assignment and not involved in the routine treatment of the patients performed clinical evaluations and laboratory analysis. Body mass index (BMI) was calculated on the basis of weight (kilograms) and height (meters), and subjects were classified as normal weight (BMI 18.5-24.9 kg/ m2), overweight (BMI 25-29.9 kg/m2), and obese (BMI ≥30 kg/ m2). In addition, between 07:30 and 09:00, after an overnight fast of 12 h fasting blood sample was drawn. Triglycerides, high-density lipoprotein cholesterol (HDL-c) and plasma glucose concentration were determined (Roche Diagnostics GmbH, Mannheim, Germany) using commercially available assay kits. Glycated hemoglobin (HbA1c) was measured using a kit obtained from BioSystems (Spain). Serum creatinine was measured with a kit obtained from Stanbio Laboratory (USA).
A. Measurement of oxidative stress markers and anti-oxidant status: For all participants serum (from 10 ml blood in plain vial) and plasma (from 5 ml blood in EDTA vial) were separated from the sample within 30 min of collection and was stored in pyrogen free polypropylene cryo-tubes at (-80°C) until analysis. Assessment of lipid markers for peroxidation as malondialdehyde (MDA) were determined according to Buege and Aust. [28]. However, Anti-oxidant status, glutathione (GSH) that was determined by the method of Beutler and colleagues [29], in the other hand, glutathione peroxidase (GPx) and superoxide dismutase (SOD) were measured by the method of Nishikimi and colleagues [30].
B. Measurement of inflammatory cytokines: Venous blood samples after a 12-hours fasting were centrifuged at 4 °C (1000Xg for 10 min). "Immulite 2000" immune-assay analyzer (Siemens Healthcare Diagnostics, Deerfield, USA) analyzed IL-6 level. However, TNF-a and CRP levels were measured by ELISA kits (R&D, USA) by using ELISA technique (ELX 808; Bio-Tek Instruments, USA).
Statistical Analysis
SPSS (Chicago, IL, USA) version 21 was used for statistical analysis of data. Quantitative variables were described as mean±SD. An independent t-test was used to compare mean values of each parameter among the groups. To observe possible relationships between creatinine and TNF-α, IL-6, CRP, MDA, GPX, GSH and SOD, Pearson's correlation coefficient (r) was used. All assumptions were carefully appreciated in each model we followed. All variables with p- value less than 0.05 were considered as statistical significance.
Results
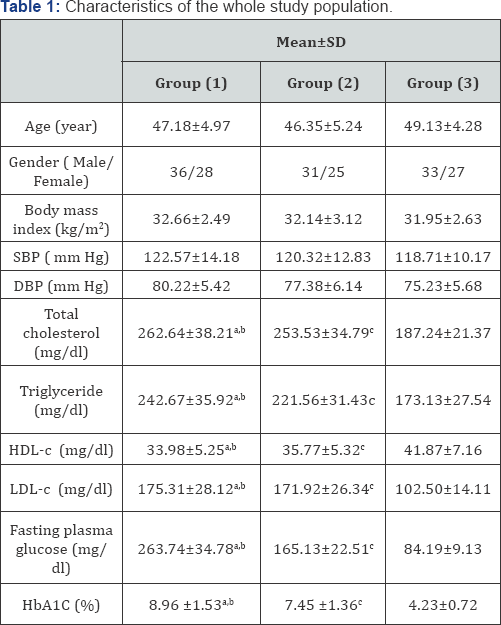
SBP: Systolic blood pressure; DBP: Diastolic blood pressure; TG: Triglyceride; HDL-c: High-density lipoprotein cholesterol; LDL-c: Low- density lipoprotein cholesterol; HBA1c: glycosylated hemoglobin; a P <0.05 : group (1) compared with group (2); b P<0.05 : group (1) compared with group (3); c P < 0.05 : group (2) compared with group (3); *Significant level (p<0.05).
Detailed baseline characteristics of the participants of the three groups presented in Table 1. There was a significant difference for all characteristics of type 2 diabetic patients with nephropathy (group 1) and both type 2 diabetic patient without nephropathy (group 2) and the non-diabetic patients (group 3), except in the age, gender, body mass index, systolic blood pressure and diastolic blood pressure (Table 1).
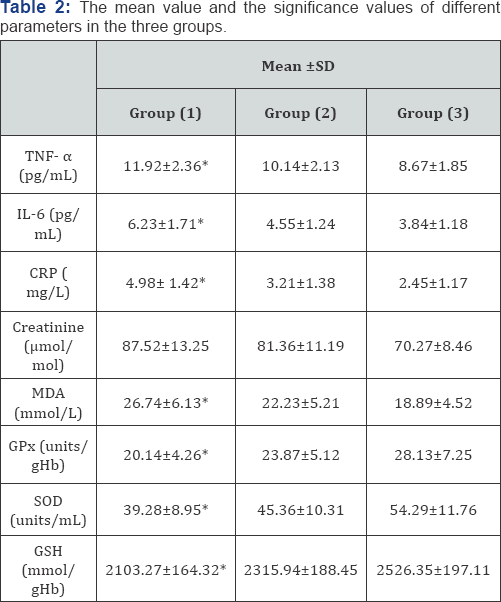
TNF-α tumor necrosis factor - alpha; IL-6: Interleukin-6; CRP: C-Reactive Protein; CD: Conjugated Dienes; MDA: Malondialdehyde; Gpx: Glutathione Peroxidase; SOD: Superoxide Dismutase; GSH: Glutathione; A P< 0.05 : Group (1) Compared With Group (2); B P <0.05 : Group (1) Compared With Group (3); C P<0.05 : Group (2) Compared With Group (3); *Significant Level (P<0.05).
Regarding the biochemical characteristics of the three groups, Table 2 shows that both group (1) and group (2) had a significant higher serum creatinine, TNF-α, IL-6, CRP and MDA levels than group (3) in addition to a significant lower serum GPX, GSH and SOD levels in group (3) than both group (1) and group (2). However, there were significant differences in all biochemical characteristics between group (1) and group (2) (P<0.05).
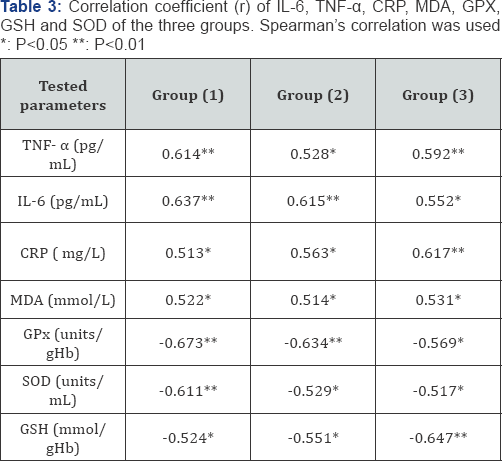
The values of IL-6, TNF-α CRP and MDA showed a strong direct relationship with value of creatinine in the three groups. However, the values of GPX, GSH and SOD showed a strong inverse relationship with value of creatinine in the three groups (Table 3) (P<0.05).
Discussion
Diabetic nephropathy (DN) considered as the most serious diabetic complication; while renal replacement is required for the majority of subjects with chronic renal disease among patients with T2DM [31,32], where poor glycemic control [33] is related to abnormal oxidative stress and systemic inflammation that induce progressive diabetic renal lesion [34-36]. Therefore, the purpose of this study was to investigate the association between oxidative stress, inflammatory cytokines and kidney function in patients with type 2 diabetic nephropathy
The principal finding of our study indicated that creatinine, inflammatory cytokines and oxidative stress markers were significantly higher in T2DM with DN and T2DM without DN than the non-diabetic control subjects. These results agreed with researchers conducted by Sekizuka et al., Suzuki et al. & Choudhary et al. [37-39] showed that serum IL-6 levels increased in type 2 diabetic nephropathy patients compared with diabetic patients without nephropathy. Similar result also found by Shelbaya et al. involved 50 subjects, consisted of 40 patients with type I diabetes and 10 normal subjects [40]. The research found that serum IL-6 levels and CRP were significantly higher in type 1 diabetics. However, NavARGHo et al. found an increase in the gene expression for pro-inflammatory cytokine in patients with DN [41]. Several studies reported that TNF-α levels increased along with progressivity of DN. It indicates that there is an association between the increase of TNF-α levels and the incidence & the course of renal lesion among diabetics [42-44]. Moreover, El Mesallamy et al. [45] reported that among 65 type 2 diabetic subjects and 17 control, there was a significant elevation in inflammatory cytokines in T2DM with DN as compared with control and normo-albuminuric subjects [45]. However, NavARGHo et al. & Moriwaki et al. [46,47] reported an association between progression of DN and urinary TNF-α excretion.
Regarding the oxidative stress markers, several reports stated that there was reduced level of GSH in diabetes associated with systemic inflammation [40-50]. In addition, in p-cell dysfunction may be related to abnormal GSH level which has a role in the incidence of long-term diabetic complications [51]. Moreover, low GSH is related to DNA oxidative damage in T2DM [52]. Many studies reported decline in the level of SOD in diabetic tissue and blood [53-56]. While, study performed by Lucchesi and colleagues to observe the oxidative balance of diabetic rats reported diminished activity of SOD and other antioxidative enzymes in the liver tissue [57]. In the other hand, several studies reported an increased MDA level in patients with T2DM [58,59]. In addition, Baynes & Ramesh et al. [60,61] reported that lipid peroxidation in diabetes induced many secondary chronic complications including atherosclerosis and neural disorders. While, Yang et al. [62] found greater serum lipid peroxidation evaluated in terms of MDA in hyperglycemic mice and proposed that the increase in lipid peroxidation exacerbated the occurrence of myocardial infraction through NADPH oxidase activation.
Finally, in our study, there was a strong direct relationship between values of inflammatory cytokines and oxidative stress markers with value of creatinine in the three groups. However, the values of anti-oxidative stress markers showed a strong inverse relationship with value of creatinine in the three groups. Our results consistent with the studies of Chen et al., Shikano et al. & Kafle et al. [63-65]who confirmed the possible role of IL-6 and TNF-α and Gpx in diabetic renal damage progress. While, Xu et al. [66] conducted a cohort study on 176 patients with chronic kidney disease and 67 healthy controls and reported increased level of CRP, IL-6 and MDA in addition to decreased levels of SOD and GSH-PX (glutathione peroxidase) along with inverse relationship between estimated glomerular filtration rate (eGFR) and MDA associated with positive relationship with SOD and GSH-PX among patients with chronic kidney disease (CKD). Moreover, Aslan et al. [67] reported significant correlations between oxidative stress and microalbuminuria levels in patients with DS. However, Sreeram et al. [68] reported that among 108 CKD patients, as the renal damage progressed the values of MDA & CRP increased while the values of GPx and SOD decreased.
Conclusion
The current study provides evidence that oxidative stress and inflammatory cytokines appear to be significantly correlated with renal function among patients with diabetic nephropathy.
References
- Whiting DR, Guariguta L, Weil C, Shaw J (2011) IDF diabetes atlas: global estimates of the prevalence of diabetes for 2011 and 2030. Diabetes Res Clin Pract 94(3): 311-321.
- Moradi M, Rahimi Z, Amiri S, Rahimi Z, Vessal M, et al. (2015) AT1R A1166C variants in patients with type 2 diabetes mellitus and diabetic nephropathy. J Nephropathol 4(3): 69-76.
- Gheith O, Farouk N, Nampoory N, Halim MA, Al Otaibi T (2016) Diabetic kidney disease: worldwide difference of prevalence and risk factors. J Nephropharmacol 5(1): 49-56.
- Akbari F, Shahinfard N, Mirhoseini M, Shirzad H, Heidarian E, et al. (2016) Impacts of Hibiscus esculentus extract on glucose and lipid profile of diabetic rats. J Nephropharmacol 5(2): 80-85.
- Adeshara KA, Diwan AG, Tupe RS (2016) Diabetes and Complications: Cellular Signaling Pathways, Current Understanding and Targeted Therapies. Curr Drug Targets 17(11): 1309-1328.
- Tuttle KR, Bakris GL, Bilous RW, Chiang JL, de Boer IH, et al. (2014) Diabetic kidney disease: a report from an ADA Consensus Conference. Diabetes Care 37(10): 2864-2883.
- Ahn JH, Yu JH, Ko SH, Kwon HS, Kim DJ, et al. (2014) Prevalence and determinants of diabetic nephropathy in Korea: Korea National Health and Nutrition Examination Survey. Diabetes Metab J 38(2): 109-119.
- Mahmoodi BK, Matsushita K, Woodward M, Blankestijn PJ, Cirillo M, et al. (2012) Associations of kidney disease measures with mortality and end-stage renal disease in individuals with and without diabetes: a meta-analysis. Lancet 380(9854): 1649-1661.
- Levey AS, Becker C, Inker LA (2015) Glomerular filtration rate and albuminuria for detection and staging of acute and chronic kidney disease in adults: A systematic review. JAMA 313(8): 837-846.
- Matsushita K, van der Velde M, Astor BC, Woodward M, Levey AS, et al. (2010) Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: A collaborative meta-analysis. Lancet 375(9731): 2073-2081.
- Badal SS, Danesh FR (2015) Diabetic Nephropathy: Emerging Biomarkers for Risk Assessment. Diabetes 64(9): 3063-3065.
- Rahimi Z, Mansouri Zaveleh O, Rahimi Z, Abbasi A (2013) AT2R-1332 G: A polymorphism and diabetic nephropathy in type 2 diabetes mellitus patients. J Renal Inj Prev 2(3): 97-101.
- Dandona P, Mohanty P, Chaudhuri A, Garg R, Aljada A (2005) Insulin infusion in acute illness. J Clin Invest 115(8): 2069-2072.
- Monnier L, Mas E, Ginet C, Michel F, Villon L, et al. (2006) Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA 295(14): 1681-1687.
- Paravicini TM, Touyz RM (2008) Nadph oxidases, reactive oxygen species, and hypertension: clinical implications and therapeutic possibilities. Diabetes Care 31(Suppl 2): S170-S180.
- Ceriello A, Motz E (2004) Is oxidative stress the pathogenic mechanism underlying insulin resistance, diabetes, and cardiovascular disease? The common soil hypothesis revisited. Arterioscler Thromb Vasc Biol 24(5): 816-823.
- Robertson RP, Harmon J, Tran PO, Tanaka Y, Takahashi H (2003) Glucose toxicity in b-cells: type 2 diabetes, good radicals gone bad, and the glutathione connection. Diabetes 52(3): 581-587.
- Pan HZ, Zhang L, Guo MY, Sui H, Li H, et al. (2010) The oxidative stress status in diabetes mellitus and diabetes nephropathy. Acta Diabetol 47(1): 71-76.
- Odum EP, Ejilemele AA, Wakwe VC (2012) Antioxidant status of type 2 diabetic patients in Port Harcourt, Nigeria. Niger J Clin Pract 15(1): 55-58.
- Shi YC, Pan TM (2012) Red mold, diabetes, and oxidative stress: A review. Appl Microbiol Biotechnol 94(1): 47-55.
- Lima V, Sampaio F, Bezerra D, Neto J, MARGHeiro D (2011) Parameters of glycemic control and their relationship with zinc concentrations in blood and with superoxide dismutase enzyme activity in type 2 diabetes patients. Arq Bras Endocrinol Metab 55(9): 701-707.
- Likidlilid A, Patchanans N, Peerapatdit T, Sriratanasathavorn C (2010) Lipid peroxidation and antioxidant enzyme activities in erythrocytes of type 2 diabetes patients. J Med Assoc Thai 93(6): 682-693.
- Bhattacharya S, Manna P, Gachhui R, Sil PC (2013) D-saccharic acid 1,4-lactone protects diabetic rat kidney by ameliorating hyperglycemia- mediated oxidative stress and renal inflammatory cytokines via NF- kappaB and PKC signaling. Toxicol Appl Pharmacol 267(1): 16-29.
- Chow F, Ozols E, Nikolic Paterson DJ, Atkins RC, Tesch GH (2004) Macrophages in mouse type 2 diabetic nephropathy: correlation with diabetic state and progressive renal injury. Kidney Int 65(1): 116-128.
- Maritim AC, Sanders RA, Watkins III JB (2003) Diabetes, oxidative stress, and antioxidants: a review. J Biochem Mol Toxicol 17(1): 24-38.
- Vander Jagt DL, Hassebrook RK, Hunsaker LA, Brown WM, Royer RE (2001) Metabolism of the 2-oxoaldehyde methylglyoxal by aldose reductase and by glyoxalase-I: roles for glutathione in both enzymes and implications for diabetic complications. Chem Biol Interact 30: 549-562.
- Ayalasomayajula SP, Kompella UB (2005) Subconjunctivally administered celecoxib-PLGA microparticles sustain retinal drug levels and alleviate diabetes-induced oxidative stress in a rat model. Eur J Pharmacol 511(2-3): 191-198.
- Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52: 302-310.
- Beutler E, Duron O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Lab Clin Med 61: 882-888.
- Nishikimi M, Appaji Rao N, Yagi K (1972) The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem Biophys Res Commun 46(2): 849-854.
- Bukhari SA, Shamshari WA, Ur Rahman M, Zia Ul Haq M, Jaafar HZ (2014) Computer aided screening of secreted frizzled related protein 4 (SFRP4): a potential control for diabetes mellitus. Molecules 19(7): 10129-10136.
- Grace BS, Clayton P, McDonald SP (2012) Increases in renal replacement therapy in Australia and New Zealand: understanding trends in diabetic nephropathy. Nephrology (Carlton) 17(1): 76-84.
- Schaffer SW, Jong CJ, Mozaffari M (2012) Role of oxidative stress in diabetes mediated vascular dysfunction: Unifying hypothesis of diabetes revisited. Vascul Pharmacol 57(5-6): 139-149.
- Remuzzi G, Benigni A, Remuzzi A (2006) Mechanisms of progression and regression of renal lesions of chronic nephropathies and diabetes. J Clin Invest 116(2): 288-296.
- Kotur Stevuljevic J, Simic Ogrizovic S, Dopsaj V, Vujovic A, Stefanovic A, et al. (2012) A hazardous link between malnutrition, inflammation and oxidative stress in renal patients. Clin Biochem 45(15): 1202-1205.
- Massy ZA, Stenvinkel P, Drueke TB (2009) The role of oxidative stress in chronic kidney disease. Semin Dial 22(4): 405-408.
- Sekizuka K, Tomino Y, Sei C, Kurusu A, Tashiro K, et al. (1994) Detection of serum IL-6 in patients with diabetic nephropathy. Nephron 68(2): 284-285.
- Suzuki D, Miyazaki M, Naka R, Koji T, Yagame M, et al. (1995) In situ hybridization of interleukin 6 in diabetic nephropathy. Diabetes 44(10): 1233-1238.
- Choudhary N, Ahlawat RS (2008) Interleukin-6 and C-reactive protein in pathogenesis of diabetic nephropathy: new evidence linking inflammation, glycemic control, and microalbuminuria. Iran J Kidney Dis 2(2): 72-79.
- Shelbaya S, Amer H, Seddik S, Allah AA, Sabry IM, et al. (2012) Study of the role of interleukin-6 and highly sensitive C-reactive protein in diabetic nephropathy in type 1 diabetic patients. Eur Rev Med Pharmacol Sci 16(2): 176-182.
- NavARGHo JF, Milena FJ, Mora C, Leon C, Garci'a J (2006) Renal pro- inflammatory cytokine gene expression in diabetic nephropathy: Effect of angiotensin-converting enzyme inhibition and pentoxifylline administration. Am J Nephrol 26(6): 562-570.
- Moriwaki Y, Yamamoto T, Shibutani Y, Aoki E, Tsutsumi Z, et al. (2003) Elevated levels of interleukin-18 and tumor necrosis factor-alpha in serum of patients with type 2 diabetes mellitus: relationship with diabetic nephropathy. Metabolism 52(5): 605-608.
- NavARGHo JF, Mora C, Maca M, Garca J (2003) Inflammatory parameters are independently associated with urinary albumin in type 2 diabetes mellitus. Am J Kidney Dis 42(1): 53-61.
- NavARGHo JF, Mora C, Muros M, Garc'ia J (2006) Urinary tumour necrosis factor-alpha excretion independently correlates with clinical markers of glomerular and tubu-lointerstitial injury in type 2 diabetic patients. Nephrology Dialysis Transplantation 21(12): 3428-3434.
- El Mesallamy HO, Ahmed HH, Bassyouni AA, Ahmed AS (2012) Clinical significance of inflammatory and fibrogenic cytokines in diabetic nephropathy. Clin Biochem 45(9): 646-650.
- NavARGHo JF, Mora C, Rivero A, Gallego E, Chahin J, et al. (1999) Urinary protein excretion and serum tumor necrosis factor in diabetic patients with advanced renal failure: Effects of pentoxifylline administration. Am J Kidney Dis 33(3): 458-463.
- Moriwaki Y, Yamamoto T, Shibutani Y, Aoki E, Tsutsumi Z, et al. (2003) Elevated levels of interleukin-18 and tumor necrosis factor-a in serum of patients with type 2 diabetes mellitus: Relationship with diabetic nephropathy. Metabolism 52(5): 605-608.
- Rahigude A, Bhutada P, Kaulaskar S, Aswar M, Otari K (2012) Participation of antioxidant and cholinergic system in protective effect of naringenin against type-2 diabetes-induced memory dysfunction in rats. Neuroscience 226: 62-72.
- Calabrese V, Cornelius C, Leso V, Trovato Salinaro A, Ventimiglia B, et al. (2012) Oxidative stress, glutathione status, sirtuin and cellular stress response in type 2 diabetes. Biochim Biophys Acta 1822(5): 729-736.
- Das J, Vasan V, Sil PC (2012) Taurine exerts hypoglycemic effect in alloxan-induced diabetic rats, improves insulin-mediated glucose transport signaling pathway in heart and ameliorates cardiac oxidative stress and apoptosis. Toxicol Appl Pharmacol 258(2): 296-308.
- Callum Livingstone, James Davis (2007) Targeting therapeutics against glutathione depletion in diabetes and its complications. British Journal of Diabetes and Vascular Disease 7(6): 258-265.
- Yildiz Dinner, Tulay Akfay, Zeynep Alademir, Hasan Ilkova (2002) Assessment of DNA base oxidation and glutathione level in patients with type 2 diabetes. Mutation Research 505(1-2): 75-81.
- Giugliano D, Ceriello A, Paolisso G (1995) Diabetes mellitus, hypertension, and cardiovascular disease: which role for oxidative stress. Metabolism 44(3): 363-368.
- He K, Li X, Chen X, Ye X, Huang J, et al. (2011) Evaluation of antidiabetic potential of selected traditional Chinese medicines in STZ-induced diabetic mice. J Ethnopharmacol 137(3): 1135-1142.
- Shukla K, Dikshit P, Tyagi MK, Shukla R, Gambhir JK (2012) Ameliorative effect ofWithania coagulans on dyslipidemia and oxidative stress in nicotinamide streptozotocin induced diabetes mellitus. Food Chem Toxicol 50(10): 3595-3599.
- Kim CH (2013) Expression of extracellular superoxide dismutase protein indiabetes. Arch Plast Surg 40(5): 517-521.
- Lucchesi AN, Freitas NT, Cassettari LL, Marques SF, Spadella CT (2013) Diabetes mellitus triggers oxidative stress in the liver of alloxan- treated rats: a mechanism for diabetic chronic liver disease. Acta Cir Bras 28(7): 502-508.
- Moussa SA (2008) Oxidative stress in diabetes mellitus. Romanian Journal of Biophysics 18: 225-236.
- Bandeira Sde M, Guedes Gda S, da Fonseca LJ, Pires AS, Gelain DP, et al. (2012) Characterization of blood oxidative stress in type 2 diabetes mellitus patients: increase in lipid peroxidation and SOD activity. Oxid Med Cell Longev 2012: 819310.
- Baynes JW (1991) Role of oxidative stress in development of complications in diabetes. Diabetes 40(4): 405-412.
- Ramesh B, Karuna R, Sreenivasa RS, Haritha K, Sai MD, et al. (2012) Effect of Commiphora mukul gum resin on hepatic marker enzymes, lipid peroxidation and antioxidants status in pancreas and heart of streptozotocin induced diabetic rats. Asian Pac J Trop Biomed 2(11): 895-900.
- Yang Z, Laubach VE, French BA, Kron IL (2009) Acute hyperglycemia enhances oxidative stress and exacerbates myocardial infarction by This work is licensed under Creative Commons Attribution 4.0 License DOI: 10.19080/ARGH.2017.06.555695 activating nicotinamide adenine dinucleotide phosphate oxidase during reperfusion. J Thorac Cardiovasc Surg 137(3): 723-729.
- Chen S, Cohen MP, Lautenslager GT, Shearman CW, Ziyadeh FN (2001) Glycated albumin stimulates TGF-beta 1 production and protein kinase C activity in glomerular endothelial cells. Kidney International 59(2): 673-668.
- Shikano M, sobajima H, Yoshikawa H, Toba T, Kushimots H, et al. (2000) Usefulness of highly sensitive urinary and serum IL-6 assay in patients with diabetic nephropathy. Nephron 85(1): 81-85.
- Kafle D, Singh N, Singh S, Mandal F , Rimal B (2012) Relationship between Hyperglycemia, Inflammation and Oxidative Stress in Type-2 Diabetic Nephropathy Subjects. International Journal of Pharmaceutical & Biological Archive 3(5): 1204-1206.
- Xu G, Luo K, Liu H, Huang T, Fang X, et al. (2015) The progress of inflammation and oxidative stress in patients with chronic kidney disease. Ren Fail 37(1): 45-49.
- Aslan M, Sabuncu T, Kocyigit A, Celik H, Selek S (2007) Relationship between total oxidant status and severity of diabetic nephropathy in type 2 diabetic patients. Nutr Metab Cardiovasc Dis 17(10): 734-740.
- Sreeram M, Suryakar A, Kulhalli P (2013) A Study of Bio-Markers of Oxidative Stress and Inflammation in Chronic Kidney Disease. Journal of Dental and Medical Sciences 11(2): 6-10.






























