When Should Thiopurine Therapy During Sustained Remission in Inflammatory Bowel Disease be Stopped?
Julia Herreras and Marisa Iborra*
Department of gastroenterology, University Hospital La Fe, Spain
Submission: March 01, 2017; Published: April 24, 2017
*Corresponding author: Marisa Iborra, Department of Gastroenterology, University Hospital La Fe, Spain, Fax: +34 961245859/+34 961246257; Email: marisaiborra@hotmail.com
How to cite this article: Julia H, Marisa I. When Should Thiopurine Therapy During Sustained Remission in Inflammatory Bowel Disease be Stopped?. Adv Res Gastroentero Hepatol 2017; 4(5): 555647. DOI: 10.19080/ARGH.2017.04.555647
Abstract
Azathioprine is effective for maintenance of remission in inflammatory bowel disease, nonetheless, duration of efficacy and the dose response relationship has not been fully evaluated. Currently, there are no general recommendations yet that can help us selecting patients who would benefit from the discontinuation of TP without an increased relapse risk.
Keywords: Immuno suppression; Thiopurines; Azathioprine; Inflammatory bowel disease
Abbreviations: AZA: Azathioprine; IBD:Inflammatory Bowel Disease; CD: Crohn's Disease; UC: Ulcerative Colitis; anti-TNF: Anti-Tumour Necrosis Factor
Introduction
Azathioprine (AZA) have been used in clinical practice for more than 50 years and remain the mainstay of maintenance treatment for inflammatory bowel disease (IBD), either as alone and/or in combination with anti-tumour necrosis factor (anti-TNF) drugs [1]. Today, Thiopurine have demonstrated their capacity maintaining remission in the long-term in both Crohn's disease (CD) [2,3] and ulcerative colitis (UC) [4,5], and it seems that the effect does not disappear after for up to 5 years [6]. Moreover, during follow-up responders had a significantly reduced risk of intestinal surgery and perianal surgery [7].
Discussion
Despite increasing evidence of safety from several studies, a percentage of responders developed cancers, including nonmelanoma skin cancers [7,8]. A rare and usually fatal lymphoma, hepatosplenic T-cell lymphoma, has been related with younger IBD patients who received long-term therapy (at least 2 years) with Thiopurine [9]. Thiopurine use in IBD appears to be strongly related with an increased risk of Epstein-BARGH virus-positive lymphoma [10,11]. Recently, it has been linked immunosuppressive treatment with opportunistic infections during severe lymphopenia in IBD patients [12]. Regular monitoring of blood counts and liver test is required in order to early detection of bone mARGHow and liver toxicities [13,14].
Treatment strategies have changed accordingly. Presently, the early introduction of Immuno modulators and anti- TNF therapy targeting a window of opportunity before the development of potentially intestinal complications and they are capable of change the disease evolution. However, the clinicians unknown the best moment of therapy stopping once remission is achieved. Identifying IBD patients with increased risk of relapse after Thiopurine withdrawal during sustained clinical remission is essential for appropriate management.
From a clinical point of view, our patients are young and have a long life expectancy. Physicians should consider maintaining thiopurinesonly in cases in which a clear benefit is expected. On the other hand, whether or not AZA can be stopped is an important question and factors involved in the decision to removal the drug in patients with IBD are necessary. Previous retrospective study suggested that the risk of relapse appeared to be similar if we withdrawal or maintain AZA after 4 years of remission in CD patients [15]. In Table 1 you can see the most relevant studies assessing relapse rate in IBD patients under immuno suppressive therapy in case of discontinuation as well as the predictive factors.
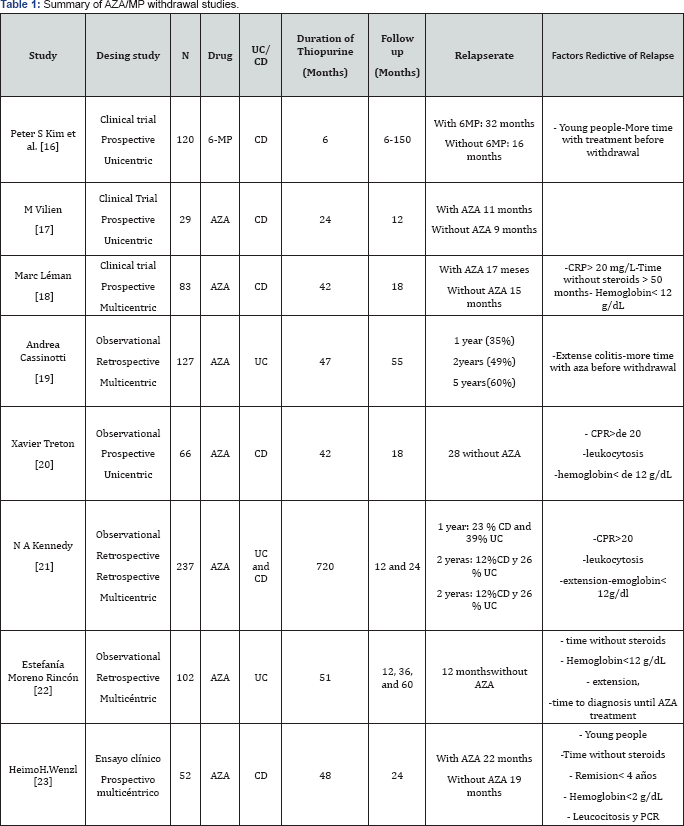
AZA (Azathioprine), 6-MP (6 mercaptopurine), CPR ( C reactive protein), UC (ulcerative colitis), CD (Crohn's disease)
Conclusion
In conclusion, Thiopurine withdrawal in the context of sustained remission is associated with a high risk of relapse. Currently discontinuation of AZA may be considered after 4 years in IBD patients in sustained remission and steroid free [24,25]. Further investigations are necessary in order to identify risk factors of relapse after stopping immunosuppressive therapy The safety and actual risk/benefit ratio of therapy withdrawal needs to be studied in prospective controlled trials, given the need to optimize the use and duration of potentially risky and costly therapies.
Conflict of Interest
No financial support was received for the preparation of this study. The authors declare that no conflict of interest exists.
References
- Brooke BN, Hoffmann DC, Swarbrick ET (1969) Azathioprine for Crohn's disease. Lancet 2(7621): 612-614.
- Candy S, Wright J, Gerber M, Adams G, Gerig M, et al. (1995) A controlled double blind study of azathioprine in the management of Crohn's disease. Gut 37(5): 674-678.
- Chande N, Patton PH, Tsoulis DJ, Thomas BS, MacDonald JK et al. (2015) Azathioprine or 6-mercaptopurine for maintenance of remission in Crohn's disease. The Cochrane database of systematic reviews (10),CD000067.
- Gisbert JP, Linares PM, McNicholl AG, Mate J, Gomollon F (2009) Meta-analysis: the efficacy of azathioprine and mercaptopurine in ulcerative colitis. Aliment Pharmacol Ther 30(2): 126-137.
- Timmer A, Patton PH, Chande N, McDonald JW, MacDonald JK (2016) Azathioprine and 6-mercaptopurine for maintenance of remission in ulcerative colitis. Cochrane Database Syst Rev. (5): CD000478.
- Fraser AG, Orchard TR, Jewell DP (2002) The efficacy of azathioprine for the treatment of inflammatory bowel disease: a 30 year review. Gut 50(4): 485-489.
- Camus M, Seksik P, Bourrier A, Nion-Larmurier I, Sokol H, et al. (2013) Long-term outcome of patients with Crohn's disease who respond to azathioprine. Clinical gastroenterology and hepatology: the official clinical practice journal of the American Gastroenterological Association 11(4): 389-394.
- Kopylov U, Vutcovici M, Kezouh A, Seidman E, Bitton A, et al. (2015) Risk of Lymphoma, Colorectal and Skin Cancer in Patients with IBD Treated with Immuno modulators and Biologics: A Quebec Claims Database Study. Inflamm Bowel Dis 21(8): 1847-1853.
- Kotlyar DS, Osterman MT, Diamond RH, Porter D, Blonski WC, et al. (2011) A systematic review of factors that contribute to hepatosplenic T-cell lymphoma in patients with inflammatory bowel disease. Clinical gastroenterology and hepatology: the official clinical practice journal of the American Gastroenterological Association 9(1): 36-41.e1.
- Dayharsh GA, Loftus EV, Sandborn WJ, Tremaine WJ, Zinsmeister AR, et al. (2002) Epstein-BARGH virus-positive lymphoma in patients with inflammatory bowel disease treated with azathioprine or 6-mercaptopurine. Gastroenterology 122(1): 72-77.
- Vos AC, Bakkal N, Minnee RC, Casparie MK, de Jong DJ, et al. (2011) Risk of malignant lymphoma in patients with inflammatory bowel diseases: a Dutch nationwide study. Inflamm Bowel Dis 17(9): 1837-1845.
- Vogelin M, Biedermann L, Frei P, Vavricka SR, Scharl S, et al. (2016) The Impact of Azathioprine-Associated Lymphopenia on the Onset of Opportunistic Infections in Patients with Inflammatory Bowel Disease. PloS one 11(5): e0155218.
- Gisbert JP, Gomollon F (2008) Thiopurine-induced myelotoxicity in patients with inflammatory bowel disease: a review. Am J Gastroenterol 103(7): 1783-1800.
- Goldberg R, Irving PM (2015) Toxicity and response to thiopurines in patients with inflammatory bowel disease. Expert Rev Gastroenterol Hepatol 9(7): 891-900.
- Bouhnik Y, Lemann M, Mary JY, Scemama G, Tai R, et al. (1996) Long-term follow-up of patients with Crohn's disease treated with azathioprine or 6-mercaptopurine. Lancet 347(8996): 215-219.
- Kim PS, Zlatanic J, Korelitz BI, Gleim GW (1999) Optimum duration of treatment with 6-mercaptopurine for Crohn's disease. Am J Gastroenterol 94(11): 3254-3257.
- Vilien M, Dahlerup JF, Munck LK, Norregaard P, Gronbaek K, et al. (2004) Randomized controlled azathioprine withdrawal after more than two years treatment in Crohn's disease: increased relapse rate the following year. Aliment Pharmacol The 19(11): 1147-1152.
- Lemann M, Mary JY, Colombel JF, Duclos B, Soule JC, et al. (2005) Therapeutique des Affections Inflammatoires du Tube D: A randomized, double-blind, controlled withdrawal trial in Crohn's disease patients in long-term remission on azathioprine. Gastroenterology 128(7): 18121818.
- Cassinotti A, Actis GC, Duca P, Massari A, Colombo E, et al. (2009) Maintenance treatment with azathioprine in ulcerative colitis: outcome and predictive factors after drug withdrawal. The Am J Gastroenterol 104(11): 2760-2767.
- Treton X, Bouhnik Y, Mary JY, Colombel JF, Duclos B, et al. (2009) Azathioprine withdrawal in patients with Crohn's disease maintained on prolonged remission: a high risk of relapse. Clin Gastroenterol Hepatol 7(1): 80-85.
- Kennedy NA, Kalla R, Warner B, Gambles CJ, Musy R, et al. (2014) Thiopurine withdrawal during sustained clinical remission in inflammatory bowel disease: relapse and recapture rates, with predictive factors in 237 patients. Aliment Pharmacol Ther 40(11-12): 1313-1323.
- Moreno-Rincon E, Benitez JM, Serrano-Ruiz FJ, Vazquez-Moron JM, Pallares MH, et al. (2015) Prognosis of Patients with Ulcerative Colitis in Sustained Remission After Thiopurines Withdrawal. Inflammatory bowel diseases 21(7): 1564-1571.
- Wenzl HH, Primas C, Novacek G, Teml A, Offerlbauer-EA, et al. (2015) Withdrawal of long-term maintenance treatment with azathioprine tends to increase relapse risk in patients with Crohn's disease. Dig Dis Sci 60(5): 1414-1423.
- Dignass A, Van Assche G, Lindsay JO, Lemann M, Soderholm J, et al. (2010) The second European evidence-based Consensus on the diagnosis and management of Crohn's disease: Current management. J Crohns Colitis 4(1): 28-62.
- Pittet V, Froehlich F, Maillard MH, Mottet C, Gonvers JJ, et al. (2013) When do we dare to stop biological or immunomodulatory therapy for Crohn's disease? Results of a multidisciplinary European expert panel. J Crohns Colitis 7(10): 820-826.






























