Is Calcium Supplementation Required in Osteoarthritis Patients?
Vikram Khanna1*, Harish Chandra Khanna2 and Ajit Saigal3
1Department of Orthopaedics, National Institute of Medical Sciences, India
2Department of Internal Medicine, Ranjana Hospital, India
3Department of Orthopaedics, Mangalam Hospital, India
Submission: August 19, 2016; Published: September 01, 2016
*Corresponding author: Vikram Khanna, Department of Orthopaedics, National Institute of Medical Sciences, Jaipur, India.
How to cite this article: Vikram K, Harish C K, Ajit S. Is Calcium Supplementation Required in Osteoarthritis Patients?. Adv Res Gastroentero Hepatol. 2016; 1(5): 555575. DOI: 10.19080/ARGH.2016.01.555575
Abstract
Introduction: Osteoporosis is a condition in which the bone mineral density is decreased. Osteoarthritis consists of a group of mechanical abnormalities causing degradation of articular cartilage and subchondral bone. Physicians tend to prescribe calcium supplementation in cases of osteoarthritis, as it was thought to be helpful in this case. This study was done to find out if there was any correlation between osteoarthritis and osteoporosis and inturn to assess the usefulness of calcium supp! Lementation in osteoarthritis.
Materials and Methods: A multicentric study of 1500 patients ageing 45 years and above was done in Lucknow, Allahabad and Varanasi. After an informed consent the patients were assessed using Kellegren Lawrence grading and Singh and Maini Index. All the patients with any history predisposing them to secondary arthritis were excluded along with patients on long term use of corticosteroids. Both the KL grading and the Singh and Maini index were compared using the Pearsons coefficient of correlation.
Results: On comparing 1500 patients it was seen that Osteoporosis was most commonly seen in post-menopausal females. The urban population was affected with osteoporosis more than rural population and vice versa in osteoarthritis. There was a slight positive correlation between Kellegren Lawrence grading and Singh and Maini Index. This indicated that osteoarthritis and osteoporosis were slightly inversely proportional to each other.
Conclusion: This study shows that there is a slight negative correlation between osteoarthritis and osteoporosis. Hence, this puts into question the use of calcium supplementation in cases of osteoarthritis.
Keywords: Osteoarthritis; Calcium supplementation
Introduction
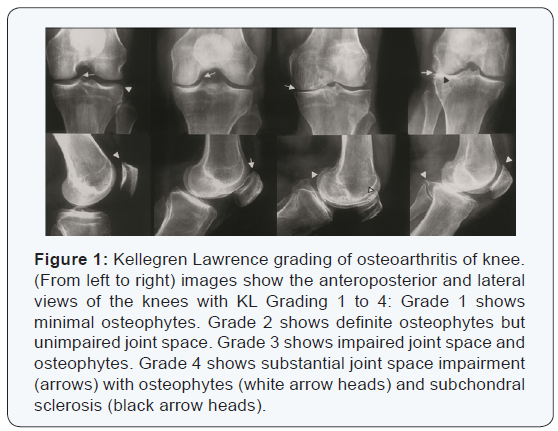
Osteoporosis (OP) is a condition with decreased bone density leading to fragility fractures. Osteoarthritis (OA) is a group of mechanical abnormalities causing degradation of articular cartilage and subchondral bone. The prevalance of OA is on the rise due to increased life expectancy and increasing incidences of trauma and obesity [1]. The coexistence of OA and OP is uncommon. In fact, it is seen that in cases operated for osteoarthritis of hip the bone density is higher than normal. This may be attributed to subchondral sclerosis which causes local rise in the bone density. Studies have shown that OP and OA may not be present in the same patient [2-4]. However, this association has not been emphatically proven. Kellegren Lawrence grading system (Figure 1) uses radiological findings of the joint to divide osteoarthritis into 4 grades [5,6]. The KL grade increases with increase in the severity of OA.
The direct measurement of bone mineral density can be calculated either by quantitative ultrasonographic study or dual energy X-ray absorptiometry. However, these modalities are not easily available & are expensive hence, indirect techniques for the assessment of OP may be used for screening like the Singh and Maini index (Figure 2) [7,8]. This index is based on the presence of compressive and tension trabeculae pattern of the proximal femur. The degree of osteoporosis decreases with an increase in the index from 1 to 6.
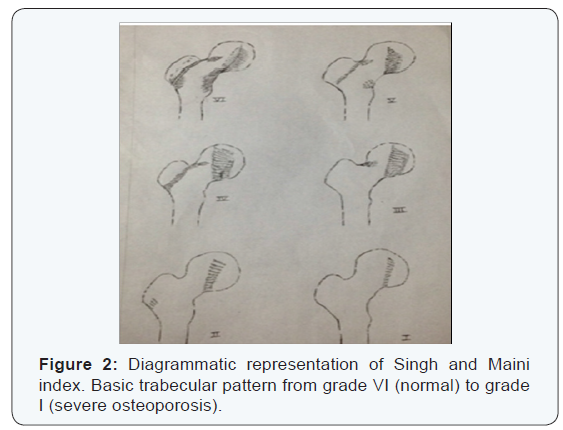
In this study the Kellegren Lawrence (KL) grading and the Singh and Maini (SM) index has been compared to find out the correlation between osteoarthritis and osteoporosis.
Materials & Methods
This multicentric study was conducted on 1500 patients of 45 years or more coming to hospital in 3 major cities of north India: Lucknow, Allahabad and Varanasi. After an informed consent, blood workup was done to rule out the causes of secondary osteoarthritis of knee. This included erythrocyte sedimentation rate, C- reactive protein and RA Factor. All cases of previous trauma and known cases of inflammatory arthritis were excluded from this study. This was followed by a plain radiograph of pelvis with both hips anteroposterior view and standing radiographs of the knee, anteroposterior and lateral view. These radiographs were then assessed by a single investigator and osteoporosis was assessed using the pelvis with both hips anteroposterior view with the Singh and Maini index.
The osteoarthritis was then assessed using the Kellegren Lawrence grading on the knee X-rays. In case if both the knees had different grades of osteoarthritis then the higher value was taken for calculation for that particular patient. Both the grading was compared and tabulated. A correlation was calculated using the Pearsons coefficient of correlation. The patients were then given symptomatic treatment for the complain which they were having along with physiotherapy and asked to follow-up. The calculation was done using the SPSS software version 20.
Results
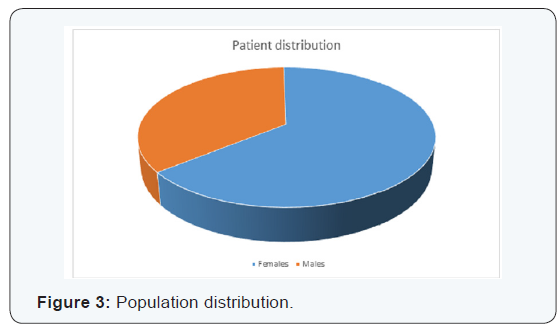
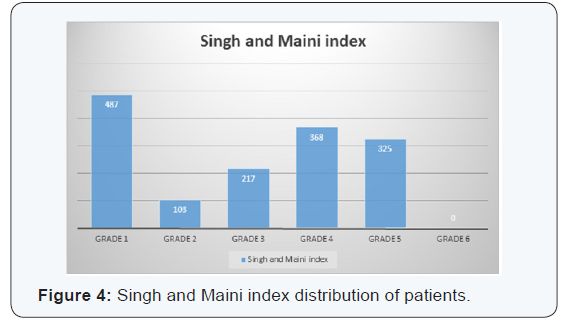
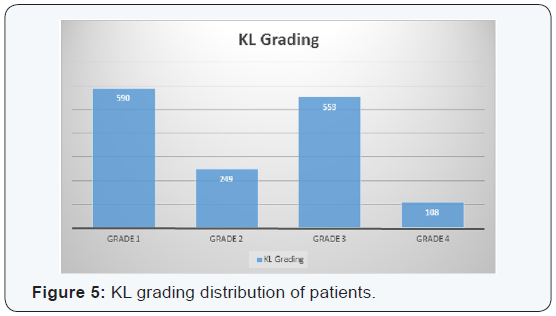
A total of 1500 patients were included in this study. Out of 1500, 967 (64.5%) patients were females and the remaining 533 (35.5%) patients were males (Figure 3). The average age of the patients was calculated to be 61.3 years with a minimum of 45 years and a maximum of 78 years of age. On tabulating the scores from the Singh and Maini index it was found that the maximum number of cases were seen in the Singh and Maini index grade 6 with 487 cases (32.5%) and the minimum no cases in Singh and Maini index grade 1 which didn’t have even a single case (Figure 4). On tabulating the scores from the Kellegren Lawrence grading the maximum numbers of cases were seen of grade 3 which constituted of 553 cases (36.87%) and the minimum number of cases was seen in grade 4 which constituted of 108 cases (7.2%) (Figure 5).
There was an increase in the grade of osteoarthritis with the increase in age group seen for both the sexes and this was found to be statistically significant (p<0.05). Whereas, in the case of osteoporosis there was a marked decrease in the Singh and Maini index seen in postmenopausal females. Moreover, this change in the osteoporosis between the premenopausal and postmenopausal females was statistically significant (p<0.05). On calculating the correlation between the Singh and Maini Index and the Kellegren Lawrence grading it was found that there was a weak positive correlation (+0.453 by Pearson’s coefficient of correlation) between both the grading systems. Since, Singh and Maini index is inversely proportional to the severity of the osteoporosis, which indicates that there is a weak negative correlation between osteoporosis and osteoarthritis.
Discussion
Osteoarthritis and osteoporosis are common conditions of the elderly population. In our extensive search of the literature there appears to be negative correlation between osteoarthritis and osteoporosis [2,3,9-11]. Whereas, some studies have proven that both the diseases exist in the same individuals [12,13]. This study was conducted in 3 cities which are within 200 kilometers of each other. This was done to avoid any major ecological changes which maybe a confounding factor. Osteoporosis and osteoarthritis have been observed on separate joints to avoid any false reading in osteoporosis due to sclerosis and other bony changes associated with osteoarthritis.
Other grading systems like DEXA scanning and QUS scanning for OP and WORMS grading for osteoarthritis are more accurate but very expensive. On a large sample size like the one in this study there were affordability issues for expensive investigations like DEXA and MRI hence; scoring systems based on radiographs was used in this study. The Os’ des Femmes de Lyon (OFELY) study showed that the fracture risk did not decrease even with the BMD higher than normal. It was also seen that there was decreased in correlation between BMD and joint space nARGHowing. It was seen that the incidence of fractures may be increased in OA due to instability. Hence, studies using vertebral fractures as an indicator of osteoporosis were faulty [14,15]. In this study 2 grading systems have been used to quantify and compare the severity of OA and OP.
The relationship between OA and OP has been explained with the help of many theories. Osteoarthritis is primarily a disease of the subchondral bone. If the BMD is high, then the hard bone causes increase in the stress on the cartilage. On the other hand, osteoporotic bone being soft absorbs little of the stress and hence there is lesser extent of cartilage damage than what is seen in OA [16]. Radin and Rose [17] came up with the theory that the loss of elasticity of the subchondral bone was responsible for OA. Subsequently, due to secondary vascular changes there is further necrosis and secondary cartilage damage. OA develops due to healed micro fractures. OP bones are more elastic and hence the cartilage damage is lesser than that seen in OA. Greavers et al. [18] also supported this theory and said that primary OA was primarily a bone disease and the cartilage disorder followed later.
Pogrund et al. [11] found that the hypothesis by Radin and Rose was contrary to the findings present in the bone. In OP the number of trabeculae were decreased, a condition which was not conducive for bone elasticity. Todd et al. [19] found out that isolated trabeculae fracture was associated with osteoporotic proximal femur fracture. These micro fractures caused incompatibility of the femoral articular margin and which lead to OA. Hordon et al. [20] compared the BMD of postmenopausal females with generalized OA with that in normal individuals. They found out that the BMD was more in OA patients as compared to the control group. Whereas, Reid et al. [21] and Price et al. [22] found that there was no difference in the BMD between the normal and the OA patients.
Dequeker et al. [23] said that high BMD in case of OA was due to low bone turnover. They also found that the levels of IGF I-II and TGF were higher in the region of OA. Dai [24] assessed the relationship between OA and fractures of the spine. It was seen that the BMD was more in cases of spinal OA as compared to normal cases. The search of the literature seems to be divided and in order to get some clarity this study was done. In this study the grading of knee X-ray was assessed for the severity of osteoarthritis and a pelvis X-ray was assessed for the severity of osteoporosis. There was a weak negative correlation seen. The association is very weak but as all causes of secondary osteoarthritis have been assessed and patients having any predisposing cause have been excluded it gives a much better association between primary OA and OP.
The negative correlation between osteoarthritis and osteoporosis was first observed by Foss and Byers [25]. This was seen when they observed that only 3 out of 140 patients of osteoporotic hip fracture had osteoarthritis. They concluded that it was indirectly indicative towards the protective influence of osteoarthritis on osteoporosis and vice versa. In a recent study done by Abdin-Mohamed et al. [26] in 2009 found out that the radiologic changes of OA were associated with the increase in the strength of the bone. However, on quantitative analysis of the same it was seen that there was no increase in the bone mineral density in OA cases.
However, Bağış et al. [27] did not detect any relationship between the BMD and the radiological evaluation of the hand. Hence, they concluded that both osteoarthritis and osteoporosis were separate entities. This study had several limitations. Both the osteoarthritis and osteoporosis have been calculated using indirect means and that they are not very specific and may be associated with intra observer variations but the large sample size may be helpful in providing adequate data for the association between OA and OP and eventually the use of calcium supplementation in OA patients.
Conclusion
This study of 1500 patients concludes that in patients coming with osteoarthritis there is little chance of them having osteoporosis. Hence, this study question the need of calcium supplementation in osteoarthritis patients and in the long run may help to decrease the financial burden on the patient and also on the healthcare system.
References
- Grainger R, Cicuttini FM (2004) Medical management of osteoarthritis of the knee and hip joints. Med J Aust 180(5): 232-236.
- Cooper C, Cook PL, Osmond C, Fisher L, Cawley MI (1991) Osteoarthritis of the hip and osteoporosis of the proximal femur. Ann Rheum Dis 50(8): 540-542.
- Sowers M, Lachance L, Jamadar D, Hochberg MC, Hollis B, et al. (1999) the associations of bone mineral density and bone turnover markers with osteoarthritis of the hand and knee in pre- and premenopausal women. Arthritis Rheum 42(3): 483-489.
- Burger H, van Daele PL, Odding E, Valkenburg HA, Hofman A, et al. (1996) Association of radio graphically evident osteoarthritis with higher bone mineral density and increased bone loss with age. The Rotterdam Study. Arthritis Rheum 39(1): 81-86.
- Kellgren JH, Lawrence JS (1957) Radiological assessment of osteoarthrosis. Ann Rheum Dis 16(4): 494-502.
- Emrani PS, Katz JN, Kessler CL, Reichmann WM, Wright EA, et al. (2008) Joint space nARGHowing and Kellgren-Lawrence progression in knee osteoarthritis: An analytic literature synthesis. Osteoarthritis Cartilage 16(8): 873-882.
- Singh M, Nagrath AR, Maini PS (1970) Changes in trabecular pattern of the upper end of the femur as an index of osteoporosis. J Bone Joint Surg Am 52(3): 457-467.
- Koot VCM, Kesselaer SMMJ, Clevers GJ, de Hooge P, Weits T, et al. (1996) Evaluation of the Singh index for measuring osteoporosis. J Bone Joint Surg Br 78(5): 831-834.
- Zhang Y, Hannan MT, Chaisson CE, McAlindon TE, Evans SR, et al. (2000) Bone mineral density and risk of incident and progressive radiographic knee osteoarthritis in women: the Framingham Study. J Rheumatol 27(4): 1032-1037.
- Weintroub S, Papo J, Ashkenazi M, Tardiman R, Weissman SL, et al. (1982) Osteoarthritis of the hip and fracture of the proximal end of the femur. Acta Orthop Scand 53(2): 261-264.
- Pogrund H, Rutenberg M, Makin M, Robin G, Menczel J, et al. (1982) Osteoarthritis of the hip joint and osteoporosis: a radiological study in a random population sample in Jerusalem. Clin Orthop Relat Res (164): 130-135.
- Dequeker J (1999) The inverse relationship between osteoporosis and osteoarthritis. Adv Exp Med Biol 455: 419-422.
- Solomon L, Schnitzler CM, Browett JP (1982) Osteoarthritis of the hip: the patient behind the disease. Ann Rheum Dis 41(2): 118-125.
- Arden NK, Nevitt MC, Lane NE, Gore LR, Hochberg MC, et al. (1999) Osteoarthritis and risk of falls, rates of bone loss, and osteoporotic fractures. Study of Osteoporotic Fractures Research Group. Arthritis Rheum 42(7): 1378-1385.
- Sornay-Rendu E, Munoz F, Duboeuf F, Delmas PD, OFELY Study, et al. (2004) Disc space nARGHowing is associated with an increased vertebral fracture risk in postmenopausal women: the OFELY Study. J Bone Miner Res 19(12): 1994-1999.
- Buckland-Wright C (2004) Subchondral bone changes in hand and knee osteoarthritis detected by radiography. Osteoarthritis Cartilage 12(Suppl A): S10-19.
- Radin EL, Rose RM (1986) Role of subchondral bone in the initiation and progression of cartilage damage. Clin Orthop Relat Res (213): 34- 40.
- Gevers G, Dequeker J, Martens M, Van Audekercke R, Nyssen-Behets C, et al. (1989) Biomechanical characteristics of iliac crest bone in elderly women according to osteoarthritis grade at the hand joints. J Rheumatol 16(5): 660-663.
- Todd RC, Freeman MA, Pirie CJ (1972) Isolated trabecular fatigue fractures in the femoral head. J Bone Joint Surg Br 54(4): 723-728.
- Hordon LD, Stewart SP, Troughton PR, Wright V, Horsman A, et al (1993) Primary generalized osteoarthritis and bone mass. Br J Rheumatol 32(12): 1059-1061.
- Reid DM, Kennedy NS, Smith MA, et al. (1984) Bone mass in nodal primary generalised osteoarthrosis. Ann Rheum Dis 43(2): 240-242.
- Price T, Hesp R, Mitchell R (1987) Bone density in generalized osteoarthritis. J Rheumatol 14(3): 560-562.
- Dequeker J, Mohan S, Finkelman RD, Aerssens J, Baylink DJ (1993) Generalized osteoarthritis associated with increased insulin-like growth factor types I and II and transforming growth factor beta in cortical bone from the iliac crest. Possible mechanism of increased bone density and protection against osteoporosis. Arthritis Rheum 36(12): 1702-1708.
- Dai LY (1998) The relationship between osteoarthritis and osteoporosis in the spine. Clin Rheumatol 17(1): 44-46.
- Foss MV, Byers PD (1972) Bone density, osteo-arthrosis of the hip, and fracture of the upper end of the femur. Ann Rheum Dis 31(4): 259-264.
- Abdin-Mohamed M, Jameson K, Dennison EM, Cooper C, Arden NK, et al. (2009) Volumetric bone mineral density of the tibia is not increased in subjects with radiographic knee osteoarthritis. Osteoarthritis Cartilage 17(2): 174-177.
- Bağış S, Şahin G, Özışık S (2003) The relationship between hand osteoarthritis and osteoporosis in postmenopausal women. T Klin J PM&R 3: 1-5.






























