New Frontiers in Nephrotic Syndrome in Children
Bharat J Parmar*
Department of Pediatrics, BJ Medical College, Civil Hospital Ahmedabad, Gujarat, India
Submission: November 25, 2016; Published: August 30, 2017
*Corresponding author: Bharat JParmar, Department of Pediatrics, BJ Medical College, Civil Hospital G-103 Shilalekh Opp. Police Stadium Shahibaug Ahmedabad 380004, Gujarat, India,.
How to cite this article: Bharat J P. New Frontiers in Nephrotic Syndrome in Children. Acad J Ped Neonatol. 2017; 5(4): 555726. DOI: 10.19080/AJPN.2017.05.555726
Abstract
The commonest type of Nephrotic Syndrome in Children is Idiopathic Nephrotic Syndrome (90%) & are called “Minimal Change Nephotic Syndrome“. Minimal Change Nephritic Syndrome (MCNS) is unique among other verities in that despite dramatic clinical expression the kidney shows either minimal or no change on light or immune fluorescent microscopy. Hallmark of MCNS are generlised anasarca, massive protenuria, (>2mg/day or >40mg/24hr) hypoalbunemia S. Albumin (<2.5mg/Dl), hypercholesterolemia S. Cholesterol>200mg/dL. It is auto immune disease with frequent remission & relapse.
Keywords: Minimal change nephrotic syndrome (MCNS); Steroid dependant syndrome (SDNS); Frequently relapsing nephritic syndrome (FRNS); Comprehensive management
Introduction
Clinical expressions
Gradual onset of oedema on face more in the morning then spread to whole body in next few day. Development of generlised anasarca, ascites, marked oedema of genitals. No fever and blood pressure is normal. Nephrotic range of protenuria is documented with early morning urine protein 3 + or 4 +ondipstick or turbidometry method, spot urine protein/ceatenine ratio>2mg/mg and urine albumin excretion >40mg/m2/hr on a timed-sample. As per Indian Society of Pediatric Nephrology (ISPN) guidelines, precise quantitative assessment of protenuria is seldom necessary.
Idiopathic nephritic syndrome is histologically characterized by minimal change disease, Focal and Segmental Glomerulo Sclerosis (FSGS) & diffuse mesenngeal proliferation. Idiopathic nephrotic syndrome is most common (90%) various other causes associated with NS. Infections like malaria, hepatitis B, Hepatitis C, Systemic diseases SLE & HSP one of the hematological disease Thalasemia major diabetes mellitus.
Pathogenesis
Idiopathic nephrotic syndrome occur as a result of immunological dysfunction. Recentally according to new theory protenuria is as result of molecular mechanism of glomerular filtration barrier. One of the essential component of glomerolar basement membrane i.e. podocyte plays a main role in filtration mechanism. The multiprotein complex between adjacent podocyte process, the slit diaphragm, is essential for the control of the act in cytoskeleton and cell morphology of podocyte. Genetic mutation encoding podocyte and slit diaphragm proteins nephrin, podocin, transient receptor potential-6 channel, Wilms’ tumor 1 gene and alpha-actinin-4 have identified by genetic studies of inherited nephrotic syndrome. Circulating permeability factors like IL-13 and Nuclear Factor Kappa b (NF-Kb) which alters the glomerular filtration barrier can cause protenuria. Finally the response to rituximab in nephrotic syndromre, suggest role for B cell involvement and/or B cell-T Cell cross talk in causation of nephrotic syndrome.
Oedema in nephrotic Syndrome
Oedema is a classical expression of nephrotic syndrome.The undrfill hypothesis state thatdecrease in oncotic pressure leads to excess filrerate of the fluid from intravascular space to the intersticial space causing hypovulemia, renal hypoperfusion activate rennin angiotensin aldosterone systean and secondary renal sodium retension.
Overfill hypothesis nephrotic protenuria causes primary renal sodium retension lead in to oedema. Recentally proposed mechanism of sodium retention regardless of intravascular volume status and it is due to the activation of epithelial sidium channel by serine proteases in the glomerular filterate of nephrotic patient. Plasminogen filtered by the nephrotic glomerulli is converted to plasm in by urokinase-type plasminogen activator in the cortical collecting duct cells. Plasmin then proteolytically removes the inhibitory domain from epithelial sodium channel (ENaC), resulting in near full activation of ENaC. Other proposed mechanism of oedema formation include (a) increased angiotensin ii independent afferent and efferent arteriolar tone because of the increased efferent sympathetic nervous activity, (b) tubular resistance to atrial natriuritic peptide and (c) increased number and activity of cortical collecting duct Na/K ATP as e-channel.
Assesment of Neprotic Syndrome
a. Initial: Height, weight and Blood pressure
b. Physical examination: Severity of oedema, focus of sepsis, signs of systemic illness, hepatospleenomegaly, pallor, skin rash, arthritis.
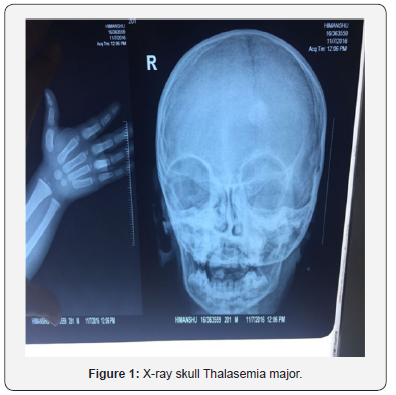
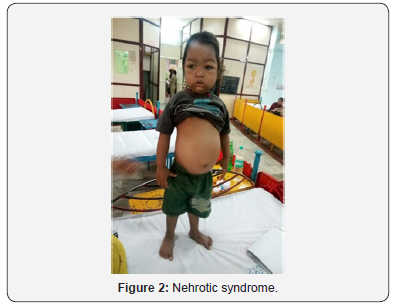
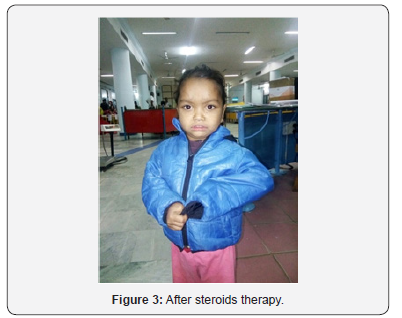
c. Investigation: Urine, CBC, Renal function tests, S. Albumin, S. Cholesterol, Manutox test, ESR, X- ray chest, USG Abdomen, S. Complement C3, C4, During October 2016 Two patients of nephotic syndrome admitted in my institute I.e. Neprotic syndrome & Thalasemia major, Nehrotic syndrome & Diabetes mellitus as Steroid toxicity. First Patient presented as Nephrotic syndrome with Thasasemia major. Patient named Himanshu aged 3 year old with clinical expressions: Generlised anasarca, anorexia, pallor Urine protein 3+ 4+, S. albumin 1.5g/Dl, S. chlolesterol 394 mg/ Dl, Blood urea & S. creatinine are normal. O/E B P normal, Anemia, Hepatospeenomegaly, haemolytic face X-ray Skull widen deploic space thinner outer table skull (Figure 1-3).
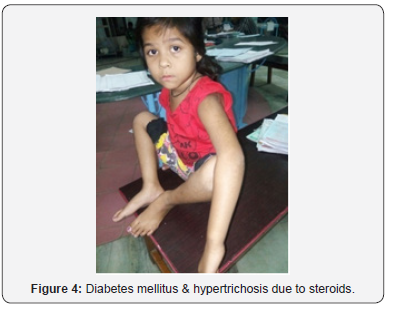
Second patient named Sakhi Vikrambhai Parmar aged 5.5 year presented as Diabetes mellitus due to steroid toxicity with clinical expressions: Polyuria, breathlessness, vomiting Diabetic ketoacidosis, Blood sugar 450mg/Dl, ketonuria and ketonemia. Past history Neprotic syndrome diagnosied at age of 1 year and treated with steroids and immunomodulator for 4 time In next 1 year. Steroids stopped since 8 months (Figure 4).
Comprehensive management
Supportive management: Adequet protein (1.5-2mg/kg) and calories Salt restriction (1-2gm/day) with persistent oedema, Fluid restriction if necessary in massive oedema. Supplement of calcium and vitamin D Immunization Pneumococcal vaccine do not give Live Vaccines Measles, MMR, Chicken pox chil receving high doses of steroids. Growth monitoring weight and height Parents should be told about importance of early treatment of inter current infection and indication of admission. Ensure compliance regarding medication encourage physical activity and play activity.
Diuretics: In nephrotic syndromre induction of Na, K- ATP as e-channel there increase in sodium reabsorption there by reducing natriuretic effect of furosemide. This can be corrected by co- administration of amiloride which inhibits the distal sodium reabsorption by blocking the ENaC channels and there by overcome furosemide resistance. Use combination furosemide and thiazide or furosemide and amiloride. It is also considered in resistant oedema. Continuous infusion of the loop diuretics has greater diuretic potency then intermittent therapy. In refractory oedema, 20% albumin infusion (1gm/kg) can br tried. It is ideal to give furocemideat the end of infusion for to prevent pulmonary oedema thus plasma volume expansion with IV Albumin might play an adjunctive role in diuretic management.
Specific management:, The ultimate goal of our long term management is to induce the remission; maintain it and at the same time minimize side effects of medications. Neprotic Syndrome should be treated adequately with corticosteroids, both in terms of dose and duration, which is an important determinant of long term course of the disease. Intial dose of nephrotic syndrome should be treated with predenisolone 2mg/kg per day (maximum 60 mg /day)for 6 weeks, followed by 1.5 mg/kg ( maximum 40 mg /day) as single morning dose on alternate days for the next 6 weeks followed by tapering ove 2 to 4 weeks. Child dose not go to remission with 4 weeks daily steroid therapy designated as steroid resistant nephrotic syndrome.
Relapse of nephrotic syndrome always look for precipitating factor, which most often is an infection. Adequate treatment of infection may result in spontaneous remission. Relapse is treated with prednisolone 2mg/kg/day until urine protein is trace or nil for three consecutive days. Subsequently prednisolone is given in single morning dose of 1.5mg/kg on alternate day for 4weeks and then discontinued.
Role of steroid sparing drugs in nephrotic syndrome
a. Levamisole: Frequent relapse NS & Steroid dependant NS where the steroid threshold is high ideal treatment is Levamisole. It is immune modulator. Dose 2 to 2.5mg/kg on alternate days 12 to 24 months along with Prednisolone 1.5mg/kg on alternate day to start with, gradually tapering it to a maintenance dose 0.20 to 0.25mg/kg continued for 6 to 12months. Levamisole is used to achieve a steroid sparing effect, ideally to started after inducing remission. Monitor WBC every 12 to 16 weeks.
b. Cyclophosphamide: Indication of use of this cytotoxic alkylating agent for FRNS & SDNS. 2 to 2.5mg/kg/day for 12 weeks orally or IV monthly pulses of 6 doses ensuring that cumulative dosage does not exceed 168 mg/kg/per course. It should be start preferably after remissin of proteinuria in FRNS & SDNS Prednisolone is co- administered for 6 to 12 months. Monitoring WBC for leucopenia every 2 weeks.
c. Mycophenolate mofetil (MMF): It is an antiproliferative agent that inhibits T & B lymphocyte proliferation by inhibition of inosin monophosphate dehydrogenase which is key enzyme in Perine biosynthesis. Dose 800 to 1200 mg/kg/m in 2 divided doses along with tapering dose of prednisolone for 12 to 24 months. Monitor WBC for leucopenia every 1-2 months.
d. Calcineurine inhibitors (CNIs): 5mg/kg for 12-24 months. Tacrolimus .1 to .2mg/kg daily for 12-24months. Prednisolone is co- administered and tapered to a loe maintenance .25 -.5mg/kg continued for 6 or more months. It is indicated when no response of Levamisole, cyclophosphemide and MMF in SRNS AND difficult SDNS. Tacrolimus is preferred over cyclosporine in view of lack of cosmetic side effects like hirsutism, ginivalhyperplasia which is seen in cyclosporine.
e. Rituximab: It is novel genetically engineered anti-CD20 monoclonal antibody that selectively targets CD20-Positive B cells Mechanism of this drug in NS are restoration of T-Regulatory (T REG) cell population and/or up regulation of their function and decreasing proteinuria. It inhibit the production of cytokines, surpasses the circulating permeability factor. Rituximab is very effective in difficult SDNS and SRNS not responding to CNI’s. Dose is 375 mg /week for 2 or more doses to achieve CD19 level below 1 % of leucocyte. Side effects of drug are hypotension or hypertension, flu like symptoms and risk of infection.
i. Complications
a. Infections: Prone to develop primary peritonitis and systemic infection due to urinary loss & inefficient production of immune globulins, defective cell mediated immunity and use of immune suppressive drugs.
b. Hyperlipidemia: It may be due to increases hepatic synthesis of lipoproteins secondary to hypoalbunemia. Hyperlipidemia may lead to platelet aggregates and risk of renal vein thrombosis.
c. Electrolyte disturbances: Hyponatrimia, hyperkalemia and hypocalcemia. Hypocalcemia as result of reduction of protein bound calcium secondary to hypoalbunemia, urinary loss of D-binding globulin, 25-hydroxyvitamin D3 and further aggravated by prolonged corticosteroids therapy.
d. Acute renal failure: It is due to intrinsic glomerular damage in atypical nephrotic syndrome. These patient are very sensitive to reduction of blood volume. As result of diarrhea, vomiting or rapid dieresis they cal develop hypovolemic shocl and pre renal azotemia promptly managed by administration of albumin or plasma to prevent renal dysfunction.
ii. Key massages: Most common causen nephrotic syndrome in children is idiopathic neprotic syndrome. Though it is called idiopathic, various pathogenetic mechanisms for proteinuria include deregulation of T cells, genetic mutation, circulating permeability factors and aberrant cross talk between B & T cell. Overfill hypothesis of oedema formation is supported by activation of tubular ENaC. Nephrotic oedema should be treated cautiously with appropriate diuretics either alone or combination with serial monitoring of electrolytes and other adverse effects. Neprotic syndrome should be treated adequetaly with corticosteroids both in terms of dosage and duration. In case of relapse, adequate treatment of infection may result in spontaneous remission. Loe dose of steroids is always co-administered with steroids sparing drugs in the initial period of treatment of FRNS and SDNS. All steroids sparing drugs have their own benefits and adverse effects. Hence serial monitoring to look for adverse effects should be stressed. Rituximab, a novel genetically engineered anti CD20 monoclonal antibody which selectively targets CD20- positive B cells is useful in difficult SDNS and SRNS. Patients of nephrotic syndrome should be counselled regarding the need for pnemococcal vaccination when the children in remission. In children with risk of suppression hypothalamopitutory adrenal axixs should get stress dose of steroids during period of stress if they have received steroids in the past one year. Complications due to disease and drugs per say should be addressed as early as possible in children with neprotic syndrome to prevent the adverse consequences.
Acknowledgement
The Author wish to thank BJ. Medical College, Civil Hospital Ahmadabad for support this work.
References
- Niaudet P, BOYER O (2009) Idiopathic Nephrotic Syndrome in Children: Clinical Aspects. In: Avner ED, Harmon WE, Niaudet P, Yoshika N (Eds.) Pediatric Nephrology, 6th edn. Springer-Verlag Berlin Heidelberg. Pp 667-702.
- Indian pediatric nephrology group, Indian academy of pediatrics (2008) Management of steroid sensitive nephrotic Syndrome: Revised Guidelines. Indian Pediatrics 45: 203-214.
- Sangeetha G, Amish Udani, Vijaykumar M (2013) Steroid-sensitive nephrotic syndrome. In: Vijaykumar M, Nammalwar BR (Eds). Principles and practice of Pediatric Neprology, 2nd edn. Jaypee Brothers Medical Publisher Pvt Ltd; New Delhi pp. 324-343.
- Ding WY, Saleem MA (2012) Current concepts of the podocyte in neprotic syndrome. Kidney Res Clin Pract 31(2): 87-93.
- Prajnya R (2013) Genetic Basis of Podocytopathies. In: Vijaykumar M, Nammalwar BR (Eds ). Principles and practice of pediatric Nephrology, 2nd edn. Jaypee Brothers Medical Publishers Pvt Ltd; New Delhi, pp 309-316.
- Greenbaum LA, Bendrof R, Smoyer WE (2012) Childhood nephrotic syndrome--current and future therapies. Nat Rev Nephrol 8(8): 445- 458.
- Gbadegesin R, Smoyer WE (2008) Nephrotic syndrome. In: Geary DF, Schaefer F (Eds.). Comprehensive Pediatric Nephrology 1st edn. Mosby Elsevier Philadelphia pp. 204-218.
- EC Siddall, J Radhakrishnan (2012) The Pathophysiology of Edema Formation in the Nephrotic Syndrome. Kidney Int 82: 635-642
- Doucet A, Favre G, Deschenes G (2007) Molecular mechanism of edema formation in nephrotic syndrome: therapeutic implications. Pediatr Nephrol 22(12): 1983-1990.
- Floge J, Feehally j (2010) Introduction to Glomerular Disease: Clinical Presentations. In: Floege J, Jihnson RJ, Feehally J ( eds). Comprehensive Clinical Nephrology, 4th edn. Missouri: Elsevier Saunders pp. 193-207.
- Sinha A, Bagga A (2013) Rituximab therapy in nephrotic syndrome: implications for patients’ management. Nat Rev Nephrol 9(3): 154- 169.






























