Association between Lower Folic Acid Level and Unbalanced Neuron Excitability in Chinese Pediatric Patients
Yiwei Liu, Xuan Zhang, Yuncui Yu, Xiaolu Nie and Xiaoling Wang*
Pharmacy Department, Beijing Children’s Hospital, Beijing, China
Submission: June 12, 2017; Published: August 10, 2017
*Corresponding author: Al Daghamin S, Riyadh Colleges of Dentistry and Pharmacy, Riyadh, Kingdom of Saudi Arabia.
How to cite this article: Yiwei L, Xuan Z, Yuncui Y, Xiaolu N, Xiaoling W. Association between Lower Folic Acid Level and Unbalanced Neuron Excitability in Chinese Pediatric Patients. Acad J Ped Neonatol. 2017; 5(4): 555723. DOI: 10.19080/AJPN.2017.05.555723
Abstract
Introduction: Folic acid (FA or VB9) is an essential factor for many biochemical reactions. However, human cannot synthesis it on their own. The main source for folic acid is from one’s diet. The deficiency of foliate intake would result in multiple neurological and hematological diseases. To confirm its association with different neuro-etiology, we were conducted an analysis of nine types of vitamin monitoring data from 4367Chinese pediatric patients that visited our hospital.
Method and patient:We collected data on nine types of vitamins (VA, VC, VD, VE, VB1, VB2, VB6, VB9, VB12) in our monitoring data from 4367 pediatric patients that visited our hospital since August 2016 to April 2017, and extracted a diagnosis related to neuropsychiatry, including ‘seizure’,‘epilepsy’, ‘convulsion’, ‘mental/language deterioration’, ‘gait disturbances’, etc. to evaluate if there is any association between phenotype and one’s vitamin level. The monitoring method was an electrode detection.
Results:Patients (n=475) with unbalanced neuron excitability (seizure, epilepsy, convulsion)symptoms are significantly (P<0.001) associated with a lower FAlevel[95%CI: -8.54 to -4.09, nmol/L] when compared to healthy controls (n=882) or the remaining patients (n=2903), but no difference was found in mental/language deterioration, gait disturbance, ASD or autism symptoms (n=107).
conclusion The association between neuropsychiatry and FA deficiency was established several decades ago. However, our study reflects a slightly different result via a clinical outcome, which divides these neuropsychiatry into two groups: association with all cause unbalanced neuron excitability and no association with mental developmental disorder. The intrinsic cause of this result is still under investigation.
Background
Vitamin B9, or folic acid (FA), is a water-soluble vitamin. FA is the substrate for Tetrahydrofolate (THF). They are involved in many biochemical reactions including DNA methylation, DNA maintenance, etc. Their metabolism pathway is shown in Figure 1 [1].
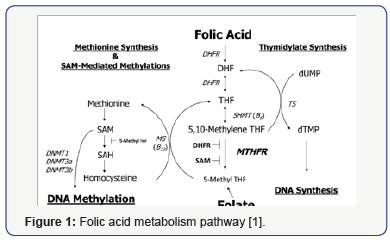

The FA deficiency could lead to a higher risk of cardiovascular disease, multiple cancers, anemia, and neural tube defects. The neurological phenotype for pediatric folic acid deficiency including cognitive change, affective disorder, peripheral neuropathy, seizure, behavior disturbance, spastic paresis, etc. Those symptoms vary depending on the age of symptom presentation (Figure 2). The VB12 deficiency usually have similar symptoms compare to FA deficiency. Folate disorder may be caused by multiple etiologies such as absorption, transport, metabolism and diet disorder [2-6]. Although we have monitored 9 vitamins’ levels for all patients, this article focuses on the FA level in patients with different neurological symptoms. We were trying to separate those symptoms into different groups, to validate if they are related with FA level.
Patients and Method
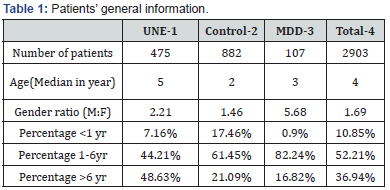
We collected 9 types whole blood vitamin level (VA, VC, VD, VE, VB1, VB2, VB6, VB9, VB12) from 4,367 patients since Aug 2016 to Apr 2017. We extracted a diagnosis and labeled the patients as having either: ‘seizure, epilepsy, convulsions, twitch or tremor’. All of those patients were group 1-Unbalanced Neuron Excitability (UNE), (n=475); patients with ‘physical examination’ but no other disease would be labeled as group 2-Control (n=882); patients with a ‘mental development disorder, language development stunting, autism, mental deterioration, attention disorder’ would be labeled as group 3-Mental Development Disorder (MDD) (n=107); and the rest of all patients visiting our hospital would be labeled as group 4-Total (n=2903). Patients’ general information (age, gender, age distribution) are shown in Table 1. A significant difference was observed between age, gender ratio and age distribution in group 1, 2, 3. The monitoring method was electrode chemical monitoring (LK3000V, from Tianjin blue label electronic Ltd.). Patients’ blood sample (whole blood) was collected with heparin tube. All monitoring experiments were performed by the same technician.
Statistics
All calculations were performed with SPSS-22.0 software. Statistical analysis includes ANOVA and Kruskal Wallis test. P<0.05 will be considered statistically significant.
Results
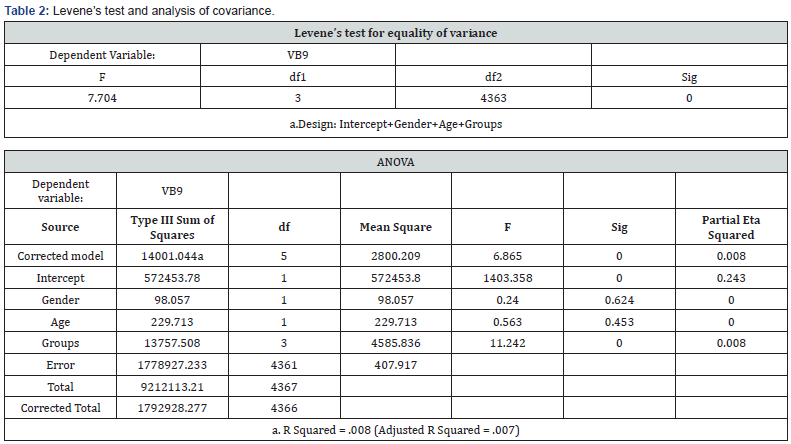
There is no significant vitamin level difference between the control, MDD and total group (data no shown). We only found a significant difference (P<0.001) in the FA level between the UNE group and the other 3 groups (Figure 3), but notin other vitamins (data no shown). There was a significant difference in age and gender between the UNE group and the control or total group (data no shown). To avoid the influence of age and gender for FA, we used a Levene’s test for equality of variance and analysis of covariance, the result is shown in Table 2. A Kruskal Wallis test as nonparametric showed the same result compare to ANOVA (data not shown).
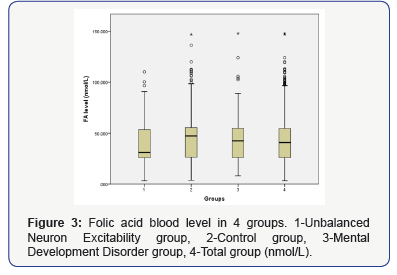
Conclusion and Discussion
Our study found an association between lower FA levels and UNE patients; no other vitamin level difference was found. We were unable to find any association between Vitamin B12 level and all causes of neuropsychiatry. Due to the phenotype similarity of VB12 deficiency and FA deficiency that was demonstrated by Shorvon et al. [7] this situation still remains unknown. The association between neuropsychiatry and FA deficiency was already established decades ago6. However, our study reflects a slightly different result from Ogier De Baulny H, et al.’s study on clinical outcome, which divides these neuropsychiatries into two groups: association with unbalanced neuron excitability and no association with mental developmental disorder. The subject number of Ogier De Baulny H et al.’s retrospective study was limited (n=141) and had no control group. However, that study was based on a Caucasian population, our study has a different ethnic background. There might be some intrinsic causes for that issue, such as gene polymorphism on FA metabolism pathway [7] like Dihydrofolate reductase (DHFR), C1-THF synthetase, Thymidylate synthase (TS), 5,10-Methylenetetrahydrofolate reductase (MTHFR), Methionine synthase (MTR), Cystathionine β-synthase (CBS) and cystathionine γ-lyase (CTH), etc. In fact, the association between MTHFR polymorphism and homocysteine (Hcy) has already been found in anxiety and depression patients [8,9]. Furthermore, FA level has a reverse association with Hcy level. So MTHFR polymorphism might influence the FA level as well. We will continue this in a subsequent study.
Conflict of Interest
There is no conflict of interest for all authors.
References
- Reynolds EH (2014) The neurology of folic acid deficiency. Handb Clin Neurol 120: 927-943.
- Rosenblatt DS, Fenton WA (2001) Inherited disorders of folate and cobalamin transport and metabolism. In: Scriver CS, Beaudet AL, Sly WS, Valle D, Childs B, et al. (Eds.), The Metabolic Basis of Inherited Disease. (8th edn), McGraw-Hill, New York, pp. 3897-3933.
- Surtees R (2001) Cobalamin and folate responsive disorders. In: Baxter P (Ed.), Vitamin Responsive Conditions in Paediatric Neurology. MacKeith Press, London, UK, pp. 96-109.
- Whitehead VM (2006) Acquired and inherited disorders of cobalamin and folate in children. Br J Haematol 134(2): 125-136.
- Crider K S, Yang T P, Berry R J, Bailey LB (2012) Folate and DNA methylation: a review of molecular mechanisms and the evidence for folate’s role. Adv Nutr 3(1): 21-38.
- Ogier de Baulny H, Gérard M, Saudubray JM, Zittoun J (1998) Remethylation defects: guidelines for clinical diagnosis and treatment. Eur J Pediatr 157(Suppl 2): S77-S83.
- Shorvon SD, Carney MW, Chanarin I, et al. The neuropsychiatry of megaloblastic anaemia. Br Med J 281(6247): 1036-1038.
- Nazki FH, Sameer AS, Ganaie BA (2014) Folate: metabolism, genes, polymorphisms and the associated diseases. Gene 533(1): 11-20
- Bjelland I, Tell GS, Vollset SE, Refsum H, Ueland PM (2003) Folate, Vitamin B12, Homocysteine, and the MTHFR 677C→T Polymorphism in Anxiety and Depression: The Hordaland Homocysteine Study. Arch Gen Psychiatry 60(6): 618-626.






























