Exogenous Biotin May Be Leading You Astray: A Novel Cause Of Factitious Neonatal Graves’ Disease
Nora E Renthal MD PhD1, Yachana Kataria PhD2, Rosalind S Brown MD1 and Jessica R Smith MD1
1 Division of Endocrinology, Department of Medicine, Boston Children’s Hospital, USA
2Department of Laboratory Medicine, Boston Children’s Hospital, USA
Submission: May 01, 2017; Published: June 08, 2017
*Corresponding author: Jessica R Smith, MD, Division of Endocrinology, Department of Medicine, Boston Children’s Hospital, USA.
How to cite this article: Renthal NE, Kataria YP, Brown RS, Smith JR. Exogenous Biotin May Be Leading You Astray: A Novel Cause Of Factitious Neonatal Graves’ Disease. Acad J Ped Neonatol. 2017; 5(1): 555710. DOI: 10.19080/AJPN.2017.05.555710
Abstract
Normal thyroid function is critical for brain development from infancy through early childhood. Accurate assays to measure thyroid function are essential to properly diagnose thyroid disease in this high-risk population. We report an infant who was misdiagnosed with neonatal Graves’ disease and treated with antithyroid drug (methimazole) due to a biotin-streptavidin laboratory artifact.
Keywords: Thyroid; Graves’ disease; Methimazole; Biotin-streptavidin
Abbrevations: TSH: Thyrotropin Stimulating Hormone; FT4: Free Thyroxine; T4: Thyroxine; T3: Triiodothyronine; TRAbs: Thyrotropin Receptor Antibodies; TBII: Thyrotropin Receptor Binding Inhibitory Immunoglobulins; TSHR: Thyrotropin Receptor; DOL: Day of Life
Introduction
Neonatal Graves’ disease is a rare form of pediatric hyperthyroidism that occurs in 1-5% of infants born to mothers with Graves’ disease [1,2]. The risk of neonatal Graves’ disease is highest in those infants born to mothers with active or refractory Graves’ disease during pregnancy. However, infants whose mothers have undergone definitive therapy with either radioiodine ablation or surgical thyroidectomy can still be affected.
Neonatal Graves’ disease results from the transplacental passage of maternal thyrotropin receptor antibodies (TRAbs) which stimulate the fetal thyrotropin receptors (TSHR) on the neonatal thyroid gland. Hyperthyroidism can develop in utero, “fetal thyrotoxicosis,” or in the postpartum period, “neonatal thyrotoxicosis.” Hyperthyroidism that develops in utero can lead to prolonged fetal tachycardia and an increased risk of fetal demise or miscarriage [2,3]. After birth, neonatal Graves’ disease is a self-limiting condition as maternal TSH receptor antibodies classically remit within 12 weeks of life. However, if not immediately identified and treated, neonatal thyrotoxicosis can be life threatening.
The diagnosis of neonatal Graves’ disease is made in coordination with a pediatric endocrinologist. Infants with neonatal Graves typically demonstrate suppressed serum TSH and elevated free T4 and T3 levels. TSH receptor antibodies are also detectable in both the infant and the mother.
Once the diagnosis is confirmed, treatment should be promptly initiated. Therapy includes both hemodynamic stabilization with beta-adrenergic blockade (propranolol/atenolol) and anti-thyroid drug (methimazole) with the goal of achieving rapid euthyroidism. In acutely ill infants, inorganic iodine and glucocorticoids may be required. With clinical stability, frequent laboratory assessments are necessary in order to titrate antithyroid drug, monitor thyroid function and prevent the development of iatrogenic hypothyroidism [4].
The proper diagnosis of neonatal Graves’ disease is highly reliant on the ability to accurately assess thyroid function. Laboratory interference by heterophile antibodies to both thyroxine and thyrotropin is well reported; however, artifacts that utilize the biotin-streptavidin system are not widely appreciated in pediatrics. We report a case of a 1-week-old neonate treated with high doses of biotin for neurologic stabilization that was misdiagnosed with neonatal Graves’ disease due to biotin-streptavidin laboratory interference.
Case
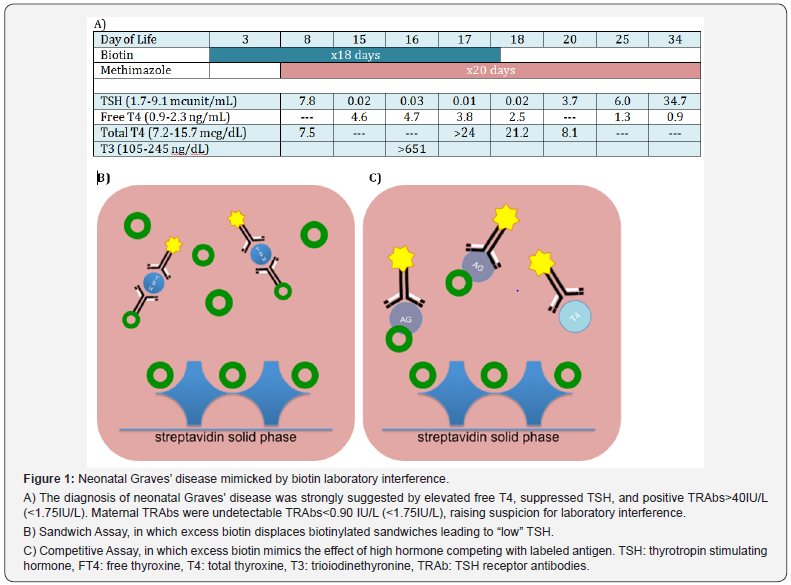
The patient was delivered by emergency cesarean section at 31 weeks due to decreased fetal movement. At birth, he had marked hydrops and required urgent intubation and ventilation due to severe respiratory depression and lactic acidosis. He developed seizures, jaundice, tachycardia, and hypertension, and subsequently, he was administered therapeutic doses of thiamine (10mg/kg/day) and biotin (8mg/kg/day) for the presumed diagnosis of pyruvate carboxylase deficiency. Physical examination was remarkable for upper extremity tremors, but did not identify orbitopathy or goiter. Thyroid function tests were notable for a suppressed serum TSH 0.02 mcunit/mL (1.7-9.1), and elevated serum free T44. 6ng/mL (0.9-2.3 ng/mL) and total T3 >651 ng/ dL (105-245ng/dL) levels, consistent with neonatal thyrotoxicosis (Figure 1). The diagnosis of neonatal Graves’ disease was strongly suggested by the infant’s positive TSH receptor antibody titer, TRAbs >40 IU/L (<1.75).
Of importance, the patient’s mother was treated with thyroid hormone replacement therapy for chronic lymphocytic thyroiditis diagnosed four years prior to the pregnancy. It was uncertain as to whether TRAbs were measured at the time of her diagnosis; however, maternal TRAbs, obtained during the patient’s evaluation, were undetectable TRAbs <0.90 IU/L (<1.75 IU/L).
Anti-thyroid medication (methimazole 0.5mg/kg/day) was initiated on DOL15, and there was a gradual reduction in serum free T4 and T3 levels (Figure 1). Biotin therapy was discontinued on DOL19 when a therapeutic effect was not clinically observed. The patient’s newborn screen (DELFIA Perkin-Elmer), performed on DOL20, was discordant with serum thyroid function tests obtained one day prior. On DOL21, the patient was clinically stable and transferred to another facility. Thyroid function tests, obtained on DOL 34, were notable for an elevated TSH 34.7 mcunit/mL (1.7-9.1) and normal free T4 0.9 ng/mL (0.9-2.3), and methimazole was discontinued.
Methods
Assays performed at Boston Children’s Hospital (Boston, MA) and the New England Newborn State Screening Laboratory (Jamaica Plain, MA). At Boston Children’s Hospital, serum T4, free T4, T3, and TSH were measured by an electrochemiluminescence immunoassay on the Cobas Roche analyzer (Indianapolis, IN). TSH receptor antibody (TBII) was measured at ARUP laboratories (Salt Lake City, Utah). TSH receptor antibody (TRAb) was measured by a quantitative electrochemiluminescent immunoassay on the Cobas Roche analyzer (Salt Lake City, Utah).
Discussion
Biotin is a water-soluble B-vitamin found naturally in foods, which acts as a coenzyme in the carboxylation, transcarboxylation, and decarboxylation reactions of gluconeogenesis, lipogenesis, fatty acid synthesis, propionate metabolism, and catabolism of leucine [5]. Biotin is highly concentrated in liver (100mcg per 100g), whereas most other meats contain trace amounts (1mcg per 100g). Leafy green vegetables, peanuts, and raw egg yolk also contain large amounts of biotin. Egg whites contain the protein avidin, which binds strongly to biotin, thus reducing its bioavailability. A typical Western diet contains adequate amounts of biotin with a recommended daily intake of 30mcg/day.
Biotin deficiency is rare. Older clinical studies with subjective measures indicated a potential role for biotin in the improvement of hair growth, skin radiance, and fingernails [6]. Quality clinical studies supporting this application are lacking. In the pediatric population, biotinidase deficiency or partial biotinidase deficiency can be difficult to diagnose, particularly in the neonatal period. Presently, high dose biotin (1-6 months: 5mcg/day or ~0.7mcg/ kg/day) is more commonly being used in a neonatal intensive care unit [7]. In rare cases of refractory juvenile onset myoclonic dystrophies, a trial of high dose biotin therapy is attempted. In addition, many adolescents self-administer high dose of biotin (5- 10 grams per day) in an effort to improve hair loss, dry skin and brittle nails.
In-vitro diagnostic platforms take advantage of the biotinstreptavidin interaction due to its analytical sensitivity, specificity and ease of performance [8]. Manufacturers add streptavidin to the assay reagents to scavenge the free biotin and reduce interference upto a manufactured specified level. However, these assays are subjected to interference from large doses of biotin and can lead to aberrant laboratory results. This has recently been reported in the literature [9-12] and is illustrated in this case of transient subclinical hypothyroidism resulting from the use of anti-thyroid medication for a presumed diagnosis of neonatal Graves’ disease.
Several commercially available TSH assays utilize a sandwich immunoassay, in which biotinylated-capture and labeleddetection antibodies bind to different epitopes on TSH to form a sandwich complex. This complex becomes bound to the solid phase due to the interaction of biotin and streptavidin. Unbound substances are removed, and bound substances are detected via chemiluminescent technology. Therefore, the biotinylated labeled sandwich immunocomplex is directly proportional to the amount of analyte present in the serum. In this case, the excess serum biotin present in our patient’s sample resulted in a reduced sandwich complexes and a falsely low TSH level.
Many of the TBII, free and total thyroxine assays also utilize a competitive immunochemical assay design. These assays result in competition between antibody binding of endogenous hormone and a detectable, biotinylated labeled analyte analogue. The immunocomplex of biotinylated analogue and antibody is formed and is inversely proportional to the amount of hormone present in the sample. Streptavidin on the solid phase immobilizes the labeled biotinylated analogues. The unbound substances are removed, and the bound substances are measured via chemiluminescent technology. Conversely, the excess serum biotin in our patient’s sample resulted in falsely elevated T4, T3 and TBII concentrations.
Biotin interference also occurs in the measurement of other competitive assays including testosterone, estrogen and DHEAS. Therefore, the combination of assay interference with orally ingested biotin can be misleading and potentially result in the misdiagnosis of thyrotoxicosis and other endocrinopathies. In addition to the treatment of children with metabolic and neurologic disease, biotin is widely available over-the-counter as a natural supplement. Thus, biotin laboratory interference must be considered in all patients undergoing any endocrine evaluation, but particularly thyroid dysfunction, especially when there is a discrepancy between the clinical scenario and laboratory results.
In this case, the absence of maternal TRAbs was inconsistent with the diagnosis of neonatal Graves’ disease, leading to the postulation of biotin interference. In these circumstances, we recommend evaluating the serum on an alternative platform (such as the Abbott Architect i1000 platform, which does not have streptavidin-biotin as part of the system). Alternatively, biotin interference can be eliminated from an assay by withholding the supplement for at least 72 hours prior to retesting. Evaluating for biotin inference leads to an improvement in the interpretation of thyroid function, patient diagnosis and clinical care. A misdiagnosis of neonatal Graves’ disease is not benign, as antithyroid medications can result in iatrogenic hypothyroidism with potential adverse effects on neonatal growth and neurocognitive development. Thus, increased awareness of biotin interference is critical in this age group.
Established Facts
• Neonatal Graves’ disease is a potential serious complication occurring in 1-5% of neonates born to mothers with a history of Graves’ disease.
• The diagnosis of neonatal Graves’ disease is confirmed with laboratory studies demonstrating a suppressed serum TSH, elevated FT4 and T3 and the presence of TRAbs.
Novel Insights
• Many laboratory assays assessing thyroid function utilize a competitive immunochemical design, which can be affected by elevated plasma biotin in a patient’s serum.
• Exogenous biotin administration can mimic the laboratory profile of neonatal Graves’ disease.
• An increased awareness of this phenomenon is critical to prevent iatrogenic hypothyroidism in infants being evaluated for hyperthyroidism.
Conflict of Interest
The authors declare no conflicts of interest.
References
- McKenzie JM, Zakarija M (1992) Fetal and neonatal hyperthyroidism and hypothyroidism due to maternal TSH receptor antibodies. Thyroid 2(2): 155-159.
- De Groot L, Abalovich M, Alexander EK, Amino N, Barbour L, et al. (2012) Management of thyroid dysfunction during pregnancy and postpartum: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 97(8): 2543-2565.
- Batra CM, Gupta V, Gupta N, Menon PS (2015) Fetal Hyperthyroidism: Intrauterine Treatment with Carbimazole in Two Siblings. Indian J Pediatr 82(10): 962-964.
- van der Kaay DC, Wasserman JD, Palmert MR (2016) Management of Neonates Born to Mothers With Graves’ Disease. Pediatrics 137(4). doi: 10.1542/peds.2015-1878.
- (1998) Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. National Academies Press, Washington (DC), USA.
- Hochman LG, Scher RK, Meyerson MS (1993) Brittle nails: response to daily biotin supplementation. Cutis, 1993. 51(4): 303-305.
- McVoy JR, Levy HL, Lawler M, Schmidt MA, Ebers DD, et al. (1990) Partial biotinidase deficiency: clinical and biochemical features. J Pediatr 116(1): 78-83.
- Spencer CA (2000) Assay of thyroid hormones and related substances. South Dartmouth, MA, USA.
- Kwok JS, Chan IH, Chan MH (2012) Biotin interference on TSH and free thyroid hormone measurement. Pathology 44(3): 278-280.
- Wijeratne NG, Doery JC, Lu ZX (2012) Positive and negative interference in immunoassays following biotin ingestion: a pharmacokinetic study. Pathology 44(7): 674-675.
- Kummer S, Hermsen D, Distelmaier F (2016) Biotin Treatment Mimicking Graves’ Disease. N Engl J Med 375(7): 704-706.
- Barbesino G (2016) Misdiagnosis of Graves’ disease with apparent severe hyperthyroidism in a patient taking biotin megadoses. Thyroid 26(6): 860-863.






























