Burden of HCV in Bangladesh: Warrants the Screening for Blood Donors
Salahuddin Mahmud1, Manzoor Hussain2, Syed Shafi Ahmed3, Manjuma Rahman4, Farhana Tasneem5 and Mahenaz Afroz6
1Assistant Professor, Pediatric Gastroenterology, Hepatology & Nutrition, Bangladesh Institute of Child Health, Dhaka Shishu (Children) Hospital, Bangladesh
2 Professor & Head, Pediatric Medicine & Pediatric Cardiology, Bangladesh Institute of Child Health, Dhaka Shishu (Children) Hospital, Bangladesh
3 Professor & Head, Pediatric Gastroenterology, Hepatology & Nutrition, Bangladesh Institute of Child Health, Dhaka Shishu (Children) Hospital, Bangladesh
4 Consultant, Blood Transfusion Department, Square Hospital Limited (SHL), Dhaka
5 Registrar, Neonatology, Apollo Hospitals, Bangladesh
6Assistant Professor of Gynaecology & Obstetrics, National Institute for Cancer Research Hospital (NICRH), Bangladesh
Submission: April 05, 2017; Published: June 07, 2017
*Corresponding author: Dr. Salahuddin Mahmud, Assistant Professor, Dhaka Shishu (Children) Hospital, Dhaka, Bangladesh.
How to cite this article: Salahuddin M, Manzoor H, Syed S A, Manjuma R, Farhana T, et al. Burden of HCV in Bangladesh: Warrants the Screening for Blood Donors. Acad J Ped Neonatol. 2017; 4(5): 555706. DOI: 10.19080/AJPN.2017.04.555706
Abstract
Globally, the morbidity and mortality attributable to hepatitis C virus (HCV) infection continues to increase. Approximately 7,00,000 persons die each year from HCV related complications, which include cirrhosis, hepatocellular carcinoma (HCC) and liver failure. It is estimated about 180 million people having hepatitis C virus infection but most are unaware of their infection. Highest number of transmission of HCV through unscreened or improperly screened blood transfusion (BT). In every case of BT in Bangladesh, so called proper donor screening done every day. But After reviewing lots of national & international guidelines and papers of blood transfusion, proved that blood donor screening tests and method of testing are not adequate & inappropriate. Lots of multi-transfused thalassemic and hemophiliac patients getting hepatitis C virus undoubtedly. Transfusions should be planned judiciously and side by side, efforts should be made to minimize the risk of transfusion transmissible infections (TTI) through adopting the international guidelines for safe blood transfusion. Awareness and knowledge would be the key to prevent the transfusion of transmissible diseases. Implementations of strict donor selection criteria and use of sensitive laboratory screening tests reduces the incidence of HCV transfusion.
Keywords: Blood transfusion; Blood donor screening; Transfusion transmissible infections
Introduction
Transfusion of blood components (TBC) continues to be an important therapeutic resource into the 21st century [1]. It is a specialized modality of patient management saves million of lives but many patients requiring transfusion do not have timely access to safe blood [2]. The safety of the transfusion lies not only in the correct selection, preparation and administration of blood products, but also in the ability to correctly interpret when such intervention is appropriate [1]. Providing safe and adequate blood should be an integral part of every country’s national health care policy and infrastructure [2].
The microbiological safety of blood donations may be affected by donor’s exposure to HIV, hepatitis B, hepatitis C, syphilis and other transfusion-transmissible infections (TTI). Through unsafe blood transfusion these microbial agents may transmit to the recipient blood and can cause morbidity and mortality [3]. The infectious agents may present in the blood for long periods, sometimes in high titers, stability in blood stored at 4 ̊C or lower, long incubation period before the appearance of clinical signs, asymptomatic phase or only mild symptoms in the blood donor, hence not identifiable during the blood donor selection process [4].
The primary responsibility of a blood transfusion service (BTS) is to provide a safe, sufficient and timely supply of blood and blood products. In fulfilling this responsibility, the BTS should ensure that the act of blood donation is safe and causes no harm to the donor [3]. However, in developing countries transmission of infectious agents through blood transfusion are continuing [5]. This is mostly due to inability of the test to detect the disease or the diagnostic window during which an acutely infected blood donor may harbor large amounts of highly infectious viruses without developing symptoms or detectable antigen and antibody concentration or laboratory errors [6].
Hepatitis C virus (HCV) is serious threat for South East Asia [7]. Bangladesh, a developing country of South-East Asia, has a population of 160 million [8]. HCV is emerging as one of the major health problem in Bangladesh [7]. It is encountered sporadically in this country [9]. It is one of the main causes of chronic liver disease, cirrhosis and hepatocellular carcinoma (HCC) worldwide and also in Bangladesh [10,11]. It is imperative to screen and diagnose HCV infection in high risk population specially blood donors so that those at risk of progressive liver disease may benefit from anti-viral therapy and counseling [12]. The introduction of highly sensitive second-generation & third-generation screening assays for HCV antibodies (anti-HCV) everywhere, where as Bangladesh usually practiced rapid strip method which is less sensitive & specific with lots of chance of viral missing [6,13]. This review aims to aware all health care practitioners about present dangerous situation of donor screening and wake up the policy makers regarding safe blood transfusion.
Hepatitis C Virus (HCV)
Hepatitis C virus (HCV) is a hepatotropic RNA virus of the genus Hepacivirus in the Flaviviridae family [14]. It was discovered in 1989, and since been identified as the major cause of transfusion associated non-A, non-B hepatitis [6].
Epidemiology & Burden
The number of deaths per year due to HCV-related diseases continues to increase. According to estimates from the Global Burden of Disease study, the number of deaths due to hepatitis C was 3,33,000 in 1990, 4,99,000 in 2010 and 7,04,000 in 2013.A more recent systematic review estimated that 115 million persons are anti-HCV (antibody to HCV) positive and 80 million have chronic infection. The increase in number of deaths reflects the high incidence of hepatitis C [15]. There is lack of representative population study in Bangladesh regarding prevalence of HCV infection [9]. In the past, it was 2.4% (WHO, 1999) [7]. In a recent study from Mahtab et al. [9] that was 0.88%. HCV poses a huge burden on the health of Bangladeshis, being a leading cause of all forms of chronic liver diseases next only to HBV [16,17]. This is similar to the experience in India [18,19], Pakistan [20] and Nepal [21,22]. HCV also ranks to be a leading cause of HCC in Bangladesh [23] as well as in the region including India [18] and Pakistan [24]. There are also published data from Bangladesh identifying HCV to be the etiological agent in 24.1% of patients with chronic liver diseases [25]. In another study in Bangladesh, anti-HCV was positive in 1.7% in acute viral hepatitis, 5.5% in sub-acute hepatic failure, 6.8% in post transfusion hepatitis, 24.1% of chronic liver disease, 9.6% cases hepatocellular carcinoma [26].
Hepatitis C Virus (HCV) Infection
HCV infection can be cured by antiviral treatment; however, due to the asymptomatic nature of the disease, many infected persons are unaware of their infection and for those who are diagnosed, access to treatment remains poor in many settings Figure 1 [15].
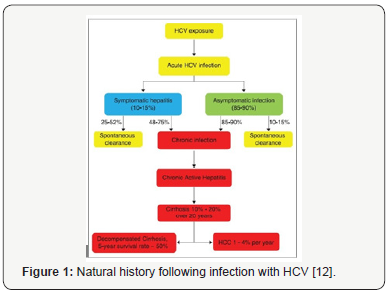
Acute Hepatitis C Virus (HCV) Infection: Most of the cases of acute hepatitis C are asymptomatic (85-90%). Symptomatic acute hepatitis with jaundice is seen in 10-15% cases and asymptomatic infection in 85-90% cases [12]. Diagnosis of acute HCV infection is based on detection of antibodies to HCV (Anti-HCV) by enzyme immunoassay. Presence of anti-HCV indicating history of exposure to HCV but not indicate the resolved infection or chronicity and persist throughout the life [15].
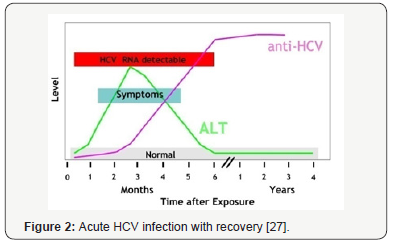
Resolved infection: In 15-45% cases a person clear the HCV within 6 months of exposure without any chronicity. A negative test result for hepatitis C virus RNA in the presence of a positive antibody indicates a resolved infection. The persons who already resolved infection, anti-HCV may persist life-long but are no longer infected with HCV (Figure 2) [15].
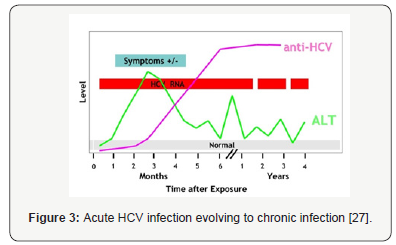
Chronic Hepatitis C Virus (HCV) infection: Continued presence of HCV RNA in the blood six months or more after acquiring infection is called chronicity. Chronic infection with HCV is usually clinically silent, and is only very rarely associated with life-threatening disease. Left untreated, chronic HCV infection can cause chronic liver disease (70-80%), cirrhosis (10-20%) and hepatocellular carcinoma (HCC) (1-4%) (Figure 3) [12,15].
Incubation period
The time between exposure of HCV & appearance of symptoms. It is about 2 weeks to 6 months commonly 6-9 weeks [12,15].
Window Period
This is about 4-6 weeks before developing anti-bodies against HCV. As there is no antibody detection, the ELISA will come up negative [12,15].
Transfusion Transmissible Infections (TTI)
Many viruses [mostly hepatitis B virus, hepatitis C virus, HIV and less commonly cytomegalovirus (CMV) and Epstein Barr Virus (EBV)], bacteria (treponema pallidum) and protozoa (malarial parasite) can be transmitted by transfusion [3,28]. Like other developing countries, blood banking in Bangladesh does not get enough attention for development from authorities. Many blood recipients remained at risk of TTI transmission as a result of poor blood donor recruitment and the use of low-quality testing in TTI screening [29].
Transmission of HCV through transfusion
HCV is spread predominantly by percutaneous or mucosal exposure to infected blood [15]. Several studies from different countries including Bangladesh indicate that, blood transfusion is the main source for transmission of HCV (Table 1 & 2).
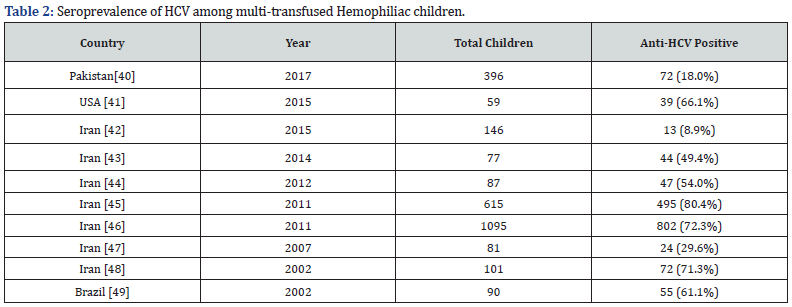
Donor screening tests for HCV in Bangladesh & Abroad
Nowadays in Bangladesh, routine screening of blood donors for HCV is only anti-HBC (antibody to HCV) [50]. Along this test, International organizations added some recent tests for prevention of HCV transmission. With anti-HCV, American red cross [51], Australian red cross[52], U.S. food & drug administration (FDA) [53], United Kingdom (UK) [54], Singapore health science authority [55], recommends Nuclieic acid testing (NAT) [56] (It is a molecular technique for screening of HCV which providing an additional layer of blood safety). Centers for disease control & prevention (CDC) [57] recommends the cost effective approach. If anti-HCV positive then they advice HCV RNA. World Health Organization (WHO) [3] recommends HCV antibody immunoassay or a combination HCV antigen-antibody immunoassay. American association for the study of liver diseases (AASLD) [58] and American family physician (AFP) [59] recommends like CDC. But when history of exposure to HCV or having suspected liver disease or HCV infection should do HCV RNA. European association for the study of liver (EASL) [10] is little bit different. With positive anti- HCV, HCV RNA is recommended. If HCV RNA not possible then HCV core antigen (HCV Ag) is the answer. I think western world doing the great job with no chance of missing HCV.
Limitations of antibody to Hepatitis C Virus (Anti-HCV) test
Anti-HCV usually appear in the blood after 6-10 weeks of infection by enzyme immunoassay. Due to large gap between infection & anti-body appearance, lots of chance of false positive & false negative results.
False Positive results of Anti-HCV [60]
I. Antibodies that the immune system has produced to combat infections other than hepatitis C (known as “cross-reactive”). The ELISA winds up picking up on these antibodies’ presence and incorrectly coming up positive.
II. If individuals suffering from autoimmune disorders like lupus.
False Negative results of Anti-HCV [61]
I. Incubation period (It is about 2 weeks to 6 months commonly 6-9 weeks).
II. Window period (around 4-6 weeks before developing antibodies against HCV).
III. Initial period of infection (before appearing antibody).
IV. If patient is immuno-compromised
V. Low viremia
Advantages of HCV Core Antigen (HCV Ag)
During the past decade, several assays for the detection of the core antigen of HCV by ELISA (Enzyme immunoassays) or CLIA (Chemiluminescent immunoassays) have been developed. These assays were envisioned as alternatives to NAT to be used in resource-limited settings, where molecular laboratory services are either not available or not widely utilized owing to cost issues. Since these assays are either ELISA or CLIA based, they are user friendly, require less technical expertise and are less expensive compared to molecular techniques [12]. HCV core antigen assays are less sensitive than HCV RNA assays (lower limit of detection equivalent to approximately 500 to 3000 HCV RNAIU/ml) [10]. But it is more sensitive than anti-HCV. It appears just after appearing of HCV RNA (about 7-8 weeks earlier than the anti-HCV) with small window period and it’s follow the HCV RNA dynamics [12].
Advantages of HCV RNA
It is the beautiful test which developed world already adopted. It diagnosed HCV infection in incubation period (after 1 week of infection), window period and with low viremia (as low as 30 copies/ml). Nucleic acid testing (NAT) is considered the ‘gold standard’ for detecting active HCV replication. It is extremely useful in establishing the diagnosis of acute HCV infection, since RNA is detectable as early as 1 week after exposure via needle-stick or blood transfusion, and at least 4-6 weeks prior to seroconversion as demonstrated in a number of transmission settings. The diagnosis of HCV infection is established with antibody screening followed by NAT for HCV RNA (Table 3) [12].
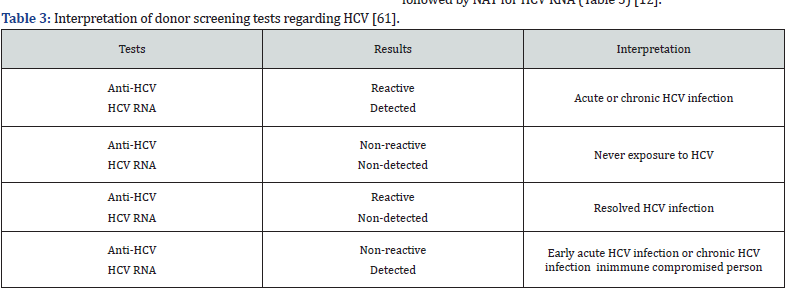
Tests Methods
According to WHO & National guideline, the main types of assay used for blood screening of HBV are [50]:
• Immunoassays (lAs):
• Enzyme immunoassays (EIAs)
• Chemiluminescent immunoassays (CLIAs)
• Rapid/simple single‐use assays (rapid tests)
• Nucleic acid amplification technology (NAT) assays.
8. Universal Choice
The blood transfusion department of medical colleges, institutes and specialized hospitals should perform EIAs and CLIAs for blood screening in addition to the current system of rapid testing [50].
In Special Situation
Only rapid tests may be considered in case emergency screening (when blood is needed urgently) or in remote areas with low workloads or limited number of tests are performed daily and limited facilities, when equipment is lacking or where there may be no electricity e.g. district blood centers and Upazila health complexes [50].
Why EIAs and CLIAs Rather than Strip
In case of rapid strip test, lots of false positive/negative results may occur. It is most commonly used, only 30 taka/strip, less sensitive & specific, manual entry of test results and not recommended by WHO as a universal screening of blood donors in Bangladesh. When false positive results occur, prospective blood donors are unnecessarily excluded from blood donation. On the other hand, when false negatives occur, this poses a great challenge to the quality and reliability of blood screening and to patient safety. Errors may also be produced if samples for retesting are improperly stored and/or transported. On the other side, quite unlikely to occur when screening is done by EIA, Chemiluminoassay, or PCR technology [50].
Present Situation in Bangladesh
In Bangladesh, Transfusion Transmissible Infections (TTI) screening is done mainly by rapid assay. Only at private sector centers (Square Hospital Limited [62] & another is Apollo Hospital, Dhaka [63]) are screening done by EIA/ CLIA (Chemiluminescent Assay). The blood donation system in Bangladesh is decentralized; all centers collect, and process and distribute blood. A beautiful study was done in 2013, which was organized by WHO, DGHS (Director General Health Services) & IEDCR (Institute of Epidemiology, Disease Control and Research) to assess the donor screening quality of different blood transfusion centers of Bangladesh. A total of 12 centers from Dhaka city (Apollo hospital, Square hospital, United hospital, Lab Aid hospital, Armed forces institutes of pathology, Bangladesh Medical College hospital, Cancer and rehabilitation hospital, Chest diseases hospital, Kidney and urology hospital, Institutes of child and maternal health etc) and 15 centers from outside the Dhaka (Rajshahi medical college hospital, Khulna medical college hospital, Faridpur medical college hospital, Comilla medical college hospital & lots of district hospitals) were included in this study. A total 915 blood samples were received from testing centers and all samples were accepted forre‐testing in IEDCR. Out of 27 testing centers, results from 18 centers were correct (66.67%) and those from 9 centers were incorrect (33.33%) when compared with the results obtained at IEDCR. Among incorrect results, HBV (53.3%) is the dominant one followed by HCV (40%) and malaria (6.7%). No results of disparity observed among HIV and syphilis cases [13].
Conclusion
Although so called proper donor screening prior to blood transfusion every time, they were unable to prevent HCV transmission. Lots of national & international studies proved that, multi-transfused thalassemic & hemophiliac patients gradually positive with HCV after few times of blood transfusion. Not only the strip method but also only antibody (anti-HBC) screening may missing the HCV anytime. The risk of disease transmission increases many fold if blood donor selection is inappropriate and method of testing is inadequate. The hazards of blood transfusion, specially the risk of transfusion transmissible infections especially HCV are now the burning issue. The policy makers, academicians, physicians and hepatologists are equally unaware of the seriousness, loop holes and magnitudes of ongoing blood transfusion program of this country. Percentage of anti-HCV negative (with strip method) healthy donors without knowing the HCV RNA status warrants the decision makers for rethinking about silent HCV infection.
Recommendations
• Anibody (anti-HCV) screening should be on Enzyme immunoassays (EIAs) and Chemiluminescent immunoassays (CLIAs) in place of strip method.
• Nucleic acid testing (NAT) for HCV should be included to avoid of missing initial period of infection/window period and for safe blood transfusion in Bangladesh.
• Core antigen (HCV Ag) may include in national donor screening program if nucleic acid testing (NAT) for HCV are not available or not affordable.
Future Prospect
Establishment of a nationally coordinated blood transfusion service, collection of blood only from voluntary donors, testing of all blood for compatibility and transfusion transmissible infections (TTIs) with appropriate method of testing and reduction of unnecessary transfusion will be the key factors for excellent blood transfusion service (BTS) in future.
References
- Diaz MQ, Borobia AM, Erce JAG, Eid CM, Fabra S, et al. (2017) Appropriate use of red blood cell transfusion in emergency departments: a study in five Emergency departments. Blood Transfus 15(3): 199-206.
- World Health Organization (2016) Blood safety & availability. WHO fact sheet, WHO, Geneva, Switzerland.
- World Health Organization (2012) Blood donor selection: Guideline on assessing donor stability for blood donation. WHO, Geneva, Switzerland, pp. 1-128
- World Health Organization (2009) Screening donated blood for transfusion transmissible infections: Recommendations. Geneva, Switzerland, pp. 1-72.
- Saha SK, Banik RK, Saha MR, Habibullah MM, Mahtab MA (2011) Prevalence of transfusion transmitted infection in healthy blood donors in Sir Salimullah Medical College Dhaka, Bangladesh. Euroasian J Hepato-Gastroenterol 1(2): 68-70.
- Forcic D, Zgorelec R, Branovic K, Gulija TK, Santak M, et al. (2001) Incidence of hepatitis C virus RNA in anti-HCV negative plasma pools in Croatia. Transfusion of Apheresis Science 24(3): 269-278.
- Ahad MA (2008) Current challenges in Hepatitis C. 21(1): 93-96.
- Jahan M, Islam MA, Akbar SMF, Takahashi K, Tabassum S, et al. (2016) Anti-HBc Screening of Blood Donors in Bangladesh: Relevance to Containment of HBV Propagation. J Clin Exp Hepatol 6(2): 115-118.
- Mahtab MA, Rahman S, Karim F, Foster G, Solaiman S (2009) Epidemiology of hepatitis C virus in Bangladeshi general population. Bangabandhu Sheikh Mujib Med Univ 2(1): 14-17.
- European association for the study of the liver guidelines (2016) EASL recommendations on treatment of hepatitis C. Journal of Hepatology, pp. 1-33.
- Islam MS, Miah MR, Roy PK, Rahman O, Siddique AB, et al. (2015) Genotypes of hepatitis C virus infection in Bangladeshi population. Mymensingh Med J 24(1): 143-151.
- Gupta E, Bajpai M, Choudhary A (2014) Hepatitis C virus: Screening, diagnosis, and interpretation of laboratory assays. Asian J Transfus Sci 8(1): 19-25.
- National Guidelines (2013) Transfusion Transmissible Infections: Testing at Blood Transfusion Centers. External Quality Assessment Report. WHO, Bangladesh.
- Kim CW, Chang KM (2013) Hepatitis C virus: Virology and life cycle. Clin Mol Hepatol 19(1):17-25.
- World Health Organization (2016) Guidelines for the screening, care and treatment of persons with chronic hepatitis C infection. WHO, Geneva, Switzerland, pp. 1-140
- Mahtab MA, Rahman S, Khan M, Kamal M, Karim MF, et al. (2007) Aetiology of chronic hepatitis: Experience from a tertiary centre in Bangladesh. Indian J Gastroenterol 26(2): 142
- Afroz S, Mahtab MA, Rahman S, Khan M. (2007) Hepatitis B virus is the leading cause of cirrhosis of liver in Bangladesh. Hepatol Int 1(1): 120.
- Sarin SK, Chari S, Sundaram KR, Ahuja RK, Anand BS, et al. (1988) Young v adult cirrhotics: a prospective comparative analysis of the clinical profile, natural course and survival. Gut 29(1): 101-107.
- Acharya SK, Panda SK, Duphare H, Dasarathy S, Ramesh R, et al. (1993) Chronic hepatitis in a large Indian hospital. Nat Med J India 6(5): 202- 206.
- Zuberi SJ (1996) Seroepidemiology of HBV/HCV in Pakistan. Int Hepatol Comm 5(1): 19-26.
- Shreatha SM, Tsuda F, Okamoto H, Tokita H, Horikita M, et al (1994) Hepatitis B virus subtypes and hepatitis C virus genotypes in patients with chronic liver disease in Nepal. Hepatology 19(4): 805-809.
- Shreatha SM (1987) Incidence of HBsAg carrier rate in pregnant women in Kathmandu. J Inst Med 1987: 71-76.
- Khan M, Zaki KMJ, Ahmed KU (1991) Clinical profile: Prognostic index in hepatocellular carcinoma. Bangladesh Med Res Council Bull XVII: 49-62.
- Abdul Mujeeb S, Jamal Q, Khanani R, Iqbal N, Kaher S (1997) Prevalence of hepatitis B virus Surface antigen and HCV antibodies in hepatocellular carcinoma cases in Karachi, Pakistan. Trop Doct 27: 45- 46.
- Khan M, Ahmed N, Rabtuan S, Zaki KMJ, Matin MA (1995) Interferon therapy in chronic viral hepatitis in Bangladesh: A preliminary report. Int Hepatol Common 3(Suppl): 104.
- Khan M, Kiyosawa K, Yano M et al. (1994) HCV seroprevalance in liver disease in Bangladesh. IXth biennial scientific meeting, Asia Pacific Association for the study of the Liver, Kualalampur, Malaysia.
- Guichard R (2012) Hepattitis C: Health from trusted sources.
- Centers for disease control & prevention (2013) Donor screening and testing. CDC, Atlanta, USA.
- Islam MB (2009) Blood transfusion services in Bangladesh. Asian J Transfus Sci 3(2): 1-3.
- Mittal K, Abrol P, Yadav J (2017) Prevalence of transfusion transmitted infections amongst multiple blood transfused patients of β-thalassemia major in a tertiary care hospital. Int J Res Med Sci 5(1): 181-185.
- Mahmoud RA, El-mazary AAM, Khodeary A (2016) Seroprevalence of Hepatitis C, Hepatitis B, Cytomegalovirus, and Human Immunodeficiency Viruses in Multitransfused Thalassemic Children in Upper Egypt. Adv Hematol 16(1): 1-7
- Bazi A, Mirimoghaddam E, Rostami D, Dabirzadeh M (2016) Characteristics of Seropositive Hepatitis B and C Thalassemia Major Patients in South-East of Iran. Biotech Health Sci 3(2): 1-6.
- Sheikh MA, Rabbani MW, Ali Z, Babar Z, Iqbal I (2015) Prevalence of Hepatitis B and Hepatitis C in Thalassemia Major Patients receiving Multiple Transfusions at the Children’s Hospital Multan. Pak Pediatr J 39(2): 69-72.
- Haque ATME, Latiff HZBA (2015) Prevalence of Hepatitis B and Hepatitis C Infections among Multi-transfused Thalassaemic Patients. PTB Reports 1(2): 69-72.
- Chakrabarty P, Rudra S, Hossain MA (2014) Prevalence of HBV and HCV among the multi-transfused beta thalassemic major patients in a day care centre of blood transfusion department of Mymensingh Medical College Hospital. Mymensingh Med J 23(2): 235-241.
- Karim AKMR, Islam A, Jamal CY, Matin A, Hossain M, et al. (2013) Seroprevalence of Hepatitis B, Hepatitis C and Human Immunodeficiency Virus Among Multitransfuse Thalassaemic Children in Dhaka, Bangladesh. Bangladesh J Child Health 37(3): 146-153.
- Mankad GP, Singh SP (2013) Incidence of viral hepatitis in thalassemic patients as a consequence of multiple blood transfusions. Int J Sci Res 3(8): 1-4.
- Shekhar HU, Kabir Y, Hossain M, Uddin M, Jannat KK, et al. (2007) Blood transfusion-mediated viral infections in thalassemic children in Bangladesh. J Med Sci 7(1): 131-135.
- Mollah AH, Nahar N, Siddique A, Anwar KS, Hassan T, et al. (2003) Common transfusion-transmitted infectious agents among thalassaemic children in Bangladesh. J Health Popul Nutr 21(1): 67-71.
- Junaid M, Siddique AN, Khan MTM, Alam I, Waqas M, et al. (2017) Detection and prevalence of hepatitis B, C and HIV viral infections amonghemophilia patients in Peshawar, Pakistan. J Entomol Zool Stu 5(2): 180-184.
- Cisak KI, Pan J, Rai SN, Ashby P, Sharma VR (2015) Prevalence of Hepatitis C Infection and Virus Clearance in Patients with Hemophilia. Blood 126(23): 4706-4713.
- Shamsdin SA, Sepehrimanesh M, Pezeshki B, Nejabat M (2015) Seroprevalence of Hepatitis B and C in patients with Hemophilia: A Single-Centre Descriptive Study. Shiraz E-Med J 16(7): 1-3.
- Esfahani H, Bazmamoun H (2014) The prevalence of blood-borne viral infection (HBV, HCV, HIV) among hemophilia patients in Hamedan Province of Iran. Int J Biodivers 6(4): 209-211.
- Assarehzadegan MA, Boroujerdnia MG, Zandian K. (2012) Prevalence of hepatitis B and C infections and HCV Genotypes among Haemophilia patients in Ahvaz, Southwest Iran. Iran Red Crescent Med J 14(8): 470- 474.
- Kalantari H, Mirzabaghi A, Akbari M, Shahshahan Z. (2001) Prevalence of hepatitisC virus, hepatitis B virus, human immunodeficiency virus and related risk factors among hemophilia and thalassemia patients In Iran. Iran J Clin Infect Dis 6(2): 82-84.
- Mousavian SA, Mansouri F, Saraei A, Sadeghei A, Merat S (2011) Seroprevalence of Hepatitis C in Hemophilia Patients Refering to Iran Hemophilia Society Center in Tehran. GOVARESH 16(3): 169-174.
- Sharifi-Mood B, Eshghi P, Sanei-Moghaddam E, Hashemi M (2007) Hepatitis Band C virus infections in patients with hemophilia in Zahedan, southeast Iran. Saudi Med J 28(10): 1516-1519.
- Ghanaei FM, Fallah MS, Shafaghi A. Mashhoor MY, Ramezani N, et al. (2002) Prevalence of hepatitis B and C seromarkers and abnormal liver function tests among hemophiliacs in Guilan (northern province of Iran). Med Sci Monit 8(12): CR797-CR800.
- Barbosa AP, Martins RMB, Teles RS, Silva SA, Oliveira JM, Clara FT et al. (2002) Prevalence of hepatitis C virus infection among hemophiliacs in Central Brazil. Short Communication 97(5): 643-644.
- National Guidelines (2013) Screening donated blood for transfusion transmissible infections in Bangladesh.
- American red cross (2017) Blood testing.
- Australian red cross (2016) Australian red cross blood service
- U.S. Food & Drug administration (2016) Complete list of donor screening assays for infectious agents and HIV diagnostic assays. FDA.
- UK NHS Blood & transplant (2015) Tests we carry out
- Singapore Health Science Authority (2017) Blood donor testing.
- Hans R, Marwaha N (2014) Nucleic acid testing benefits and constraints. Asin J Transfus Sci 8(1): 2-3.
- Centers for disease control & prevention (2015) Testing recommendations of HCV infection. CDC, Atlanta, USA.
- AASLD/IDSA HCV Guidance Panel (2015) Hepatitis C guidance: AASLDIDSA recommendations for testing, managing and treating adults infected with hepatitis C virus. Hepatology 62(3): 932-954.
- Wilkins T, Akhtar M, Gititu E, Jalluri C, Ramirez J (2015) Diagnosis and management of hepatitis C. American family physician 91(12): 835- 842.
- Ryan B (2014) Why hepatitis C tests may give false results. Patient Assistance for Lab Services.
- Centers for disease control & prevention (2013) Testing for HCV infection: An update of guidance for clinicians and laboratorians. MMWR Morb Mortal Wkly Rep 62(18): 362-365.
- https://www.squarehospital.com/?q=content/pathology-lab-centre
- http://www.apollodhaka.com/blood-bank-2/#about






























