Bubble CPAP versus Variable Flow CPAP in Preterm Newborns with Respiratory Distress: A Randomized Controlled Trial
Romero-Maldonado S1, Hernández-Ruíz M2, Pérez-Valdez MT3, López-Pérez MR3, Delgado Franco D2, Maida-Claros R2 and Echániz Avilés MOL2*
1Head of the Neonatal Intermediate Care Unit, Instituto Nacional de Perinatología, México
2Neonatology Department, Instituto Nacional de Perinatología, México
3Respiratory Support Group, México
Submission: March 10, 2017; Published: June 01, 2017
*Corresponding author: Echániz Avilés MOL, Neonatology Department, Instituto Nacional de Perinatología, Calle Montes Urales 800, Miguel Hidalgo, Lomas Virreyes, 11000 Ciudad de México, CDMX, Mexico.
How to cite this article: Romero-M S, Hernández-R M, Pérez-V M, López-P M. Bubble CPAP versus Variable Flow CPAP in Preterm Newborns with Respiratory Distress: A Randomized Controlled Trial . Acad J Ped Neonatol. 2017; 4(5): 555704. DOI: 10.19080/AJPN.2017.04.555704
Abstract
Objective: To assess the efficacy and safety of continuous positive airway pressure using variable flow (VF) against nasal bubble continuous positive airway pressure (nCPAP).
Methods: Between August 2011 and November 2012, one hundred and one preterm newborns, less than 34 weeks gestational age with RDS requiring CPAP were randomly assigned to two study groups: 47 to the VF and 54to the nCPAP group. CPAP failure, presence of air leaks, hyperemia of nasal mucosa and need for mechanical ventilation were recorded, of, as well as associated morbidities such as Intraventricular hemorrhage (IVH), Necrotizing Enterocolitis (NEC), pneumonia, Bronchopulmonary dysplasia (BPD) and Patent Ductus Arteriosus (PDA). Categorical outcome variables were tested using the chi-square test or Fisher’s exact test. Continuous variables were analyzed using Student’s t-test or the Mann-Whitney test. Level of significance was set at p<0.05.
Results:CPAP failure was reported in 23% of VF and 17% for nCPAP (p = 0.39). Air leak syndrome occurred in 3.7% for nCPAP and 4.3% for VF (p=0.88), and hyperemia of the nasal mucosa was 5.6% for nCPAPvs 12.8% for VF (p=0.20).
Conclusion: In newborns <34 weeks’ gestational age and moderate RDS, the use of continuous flow nCPAP showed the same benefits as variable flow nCPAP.
Keywords: Continuous positive airway pressure (CPAP); Nasal CPAP; Variable flow CPAP; Respiratory distress
Abbrevations: WHO: World Health Organization; RDS: Respiratory Distress Syndrome; CPAP: Continuous Positive Airway Pressure; FRC: Functional Residual Capacity; WOB: Work Of Breathing; IFD: Infant Flow Driver
Introduction
The World Health Organization (WHO) reports 15 million preterm newborns per year [1,2]. Respiratory distress is one of the main causes of perinatal morbidity and mortality. The incidence of respiratory distress syndrome (RDS) is 10% among preterm newborns in our hospital’s Intermediate Care Unit. The Instituto Nacional de Perinatología is a tertiary-level hospital, where 300 preterm infants with <1500g birth weight are taken care of and more than 500 nCPAP devices are used each year [3].
Continuous positive airway pressure (CPAP) has been widely used to manage RDS in newborn infants [4,5]. The clinical goals of CPAP are to maintain the functional residual capacity (FRC) of the lungs and support gas exchange to reduce apnea, work of breathing (WOB) and lung injury [6,7]. The Infant Flow Driver (IFD) device consists of a flow driver which provides a continuous blended gas source and an airway pressure monitoring system. Experimental data have shown that IFD is capable of providing a consistent airway pressure and has also been shown to impose a lower WOB than other devices that use fluidic control to maintain CPAP [8,9].
In our Institution we started to use IFD in 2011 and had few clinical data to evaluate its superiority over nasal bubble CPAP. Therefore, to compare both types of respiratory devices, a randomized clinical trial was conducted to assess their efficacy and safety in our Institution.
Methods
Enrollment
One hundred and twenty newborns less than 34 weeks’ gestational age who were admitted to the intermediate or intensive care units with moderate respiratory distress during the period between August 2011 and November 2012 were recruited on their date of birth. Ninety of them were excluded (Figure 1) All infants less than 12 hours old who were diagnosed with moderate RDS (clinical respiratory distress, pCO2<65mmHg, oxygen requirement greater than 30%, radiological findings of poor lung expansion) were considered for the study.

Respiratory distress was defined as sternal retraction, intercostal and sub costal recession, grunting, tachypnea. Exclusion criteria were major congenital malformations, neuromuscular disease, severe birth asphyxia (Apgar score at 5 minutes of less than 4, serum bicarbonate <12mmol/l in the first hour), sepsis, pneumothorax, cleft palate and hydrops fetalis [10]. The study protocol was approved by our Ethics Institutional Review Board and informed consent was obtained from the children’s parents in every case.
Delivery of continuous distending pressure (CDP)
We have routinely delivered nCPAP with a hybrid system: binasal Hudson prongs combined with the Fisher & Paykel pressure delivery device (Fisher & Paykel Healthcare Limited, Auckland, New Zealand) [11,12]. This system was utilized in every case assigned to nCPAP. As for the Infant Flow Driver (VF) group, we used the Viasys SiPaP system (Viasys Healthcare, Yorba Linda, CA, USA) with nasal prongs. Both devices were calibrated against and independent oxygen analyzer to determine any differences in oxygen delivery.
Randomization and allocation concealment
After parental consent was obtained in every case, the infants were prospectively and randomly assigned (by drawing a sealed, numbered envelope) to either VF or nCPAP. This was done by the inhalotherapy group, using sequentially numbered containers. According to a preset protocol, continuous distending pressure was initially set at 5cm H2O in both groups. Similarly, CDP was increased if a fraction of inspired O2(FiO2)>0.6 was required to keep saturated O2 in the range of 90-95%, depending on gestational age.
Only in those who developed apnea, theophylline was started with a loading dose of 5mg/kg given intravenously followed by a daily maintenance dose of 1mg/kg given intravenously every 8h, as we didn’t have caffeine in México. Oxygen requirements, respiratory rate, heart rate and saturated O2 by pulse oximetry were monitored continuously. Blood gases were determined on capillary blood every 6h during the first 24 hours of treatment and every 8h thereafter or at the discretion of the health care team.
Non-invasive blood pressure was obtained by oscillometry every 8h during the first 24h of treatment and every 12h thereafter. All infants underwent a brain ultrasound at enrollment and at least three times subsequently; the prongs were fixed with the help of a technician in order to get the cerebral ultrasound.IVH was classified as described by Papile et al
Any adverse clinical events (abdominal distension, pulmonary air leaks, nasal injury and damage to septal mucosa) were also prospectively recorded. Demographic data, antenatal steroids, surfactant use, vital signs, arterial gases and ventilator parameters were recorded, as well as CPAP failure and total CPAP and oxygen time. We also documented associated morbidities such as NEC, PDA and BPD.
Outcome measures
Success was defined as the ability to remain CPAP-free (medically stable with pCO2<60mmHg, saturated O2>95% without supplementary O2 requirement). Failure to wean was defined by an increase in pCO2 above 65mmHg and/or pH<7.25 on two consecutive occasions, O2 requirement >60% at CDP of 6cm H2O to keep saturated O2>90% for more than 4 consecutive hours, more than 2apneic episodes/hour requiring stimulation or bag ventilation. Adverse events such as air leaks and hyperemia of the nasal mucosa are defined in our Neonatal Guidelines. In such cases, further management was at the discretion of the health care team.
Statistical design and analysis
The sample size was estimated from a previous study that reported 20% failure with the conventional nCPAP system in our Institution [13] using a power of 85% and a significance level of 0.05. We used the difference in proportions formula [14] with p=90, p2=70, Δ=20, β=0.20and α=0.05, which yielded a sample size of 50 patients per group. Differences between the two groups were assessed using the chi-square test or Fisher’s exact test for categorical variables, whereas continuous variables were analyzed using Student’s t-test or the Mann-Whitney test. Data were processed using the SPSS software package (Chicago, Illinois, USA).
Results
A total of 101 infants were enrolled in our study and randomly assigned to one of the two treatment groups (Figure 1). The baseline characteristics of infants are summarized in Table 1 & 2. There were no significant differences in relevant clinical data at the time of enrollment. Gestational age, birth weight and gender at the beginning of the study, were similar among treatment groups. Physiological variables including respiratory rate and FiO2 requirement at entry didn’t show significant differences. History of antenatal steroids was present in almost half of the infants in both groups. Exogenous surfactant administration was similar between both groups as well as xanthine management for apneas. As for relevant morbidities during hospitalization, there were also no significant differences between the two treatment groups.
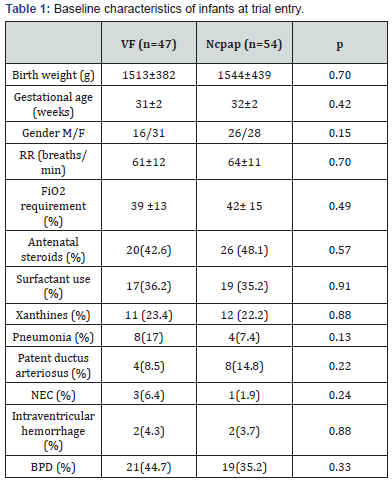
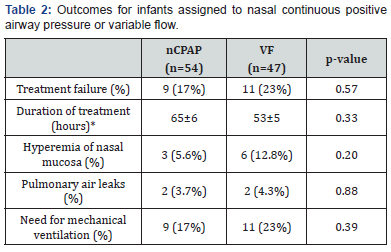
In this study we found a similar proportion of treatment failure between both modalities of CPAP. The duration of treatment in hours differed slightly and it was longer in the nCPAP group but without reaching statistical significance. With regard to adverse events, we found a higher proportion of nasal mucosa hyperemia in the VF group, although the difference was not statistically significant. We found the same number of pulmonary air leaks in both groups.
Discussion
The main purpose of this study was to compare the safety and efficacy of nCPAP vs. VF in the management of moderate RDS in our Institution. In a randomized controlled trial done in Rio J et al. [15]. evaluated efficacy and safety of nCPAP using devices with variable flow or bubble continuous positive airway pressure. CPAP failure, presence of air leaks, total CPAP and oxygen time in neonates with moderate respiratory distress and birth weight >1500g was evaluated and found no differences between the two groups. They concluded, as we did that nCPAP showed the same benefits as the use of variable flow NCPAP [15]. In a previous RCT by Mazzella et al. [9] comparing the effectiveness of the infant flow driver (IFD) against single prong nasal continuous positive airway pressure (nCPAP), IFD-treated patients were shown to have lower oxygen requirement, a decreased respiratory rate and higher rate of successful weaning and shorter duration of treatment, although the difference in these two last outcomes failed to reach statistical significance..
In our study we found a slightly longer duration of treatment in the nCPAP group but without statistical significance as well as a similar proportion of treatment failure for both modalities. In 2010 Lista et al. [16] compared synchronized bi-level CPAP and standard nCPAP in infants 28-34 weeks gestation with respiratory distress. They found no differences in short-term markers of inflammation between treatments. They found that infants treated with bi-level CPAP had a significantly shorter duration of respiratory support and supplemental oxygen than the group randomized to standard CPAP [16]. We were not able to find such differences. For the past 15 years, the medical staff, residents and nurses have acquired expertise and are used to handle nCPAP, and this could be an important issue associated with these outcomes, since the IFD system is relatively new compared with nasal bubble CPAP.
In 2012 O’ Brien et al. [17] performed a randomized controlled trial of bi-level CPAP versus standard nasal CPAP to facilitate sustained extubation in babies <1250g and found that the use of bi-level CPAP was as effective as standard CPAP [17]. As for BPD incidence in this study, it is reported high in both groups. México City is located at 2420 m above sea level and for many years it has been noted that oxygen dependency is greater at higher altitudes, and this leads to an increase in the diagnosis of BPD. Despite this, the standard for the diagnosis of BPD is the requirement of and oxygen concentration over 21% as it is for patients at sea level. At present, there are no references for oxygen dependency at higher altitudes according to barometric pressures for the purpose of BPD classification. This could be the reason for the variation in oxygen dependency and the apparent increase in BPD incidence in México City [18].
Conclusion
In newborns with <34 weeks of gestational age and moderate RDS, the use of continuous flow nCPAP showed the same benefits as the use of variable flow nCPAP in our Institution.
Acknowledgement
The authors thank Dr Carlos Alfonso Fajardo (Director of the SIBEN Neonatal Network) for the revision of this article and useful input and the nursing staff of the InstitutoNacional de Perinatología for their invaluable help in this research.
Contributors
Dra Romero-Maldonado and DraEchániz-Avilés contributed equally in the design and analysis of the results. Hernández-Ruiz and Delgado Franco participated in the recruitment and follow-up of the patients. Pérez-Valdez as well as López-Pérez was invaluable in assisting in the patient’s management.
Ethics Approval
This study was conducted with the approval of the InstitutoNacional de Perinatología Investigation Comittee with the registration number 212250-30011.
References
- Kinney MV, Howson CP, McDougall L, Lawn JE (2010) Nacido demasiado pronto: Informe de Acción Global sobre Nacimientos Prematuros. WHO, pp.1-2.
- Angus DR, Linde-Zwirble WT, Clermont G, Griffin MF, Clark RH (2001) Epidemiology of neonatal respiratory failure in the United States. Am J RespirCrit Care Med 164(7): 1154-1160.
- Anuario Estadístico del Instituto Nacional de Perinatología (2013) Isidro Espinosa de los Reyes.
- Gregory GA, Kitterman JA, Phibbs RH, Tooley WH, Hamilton WK (1971) Treatment of the idiopathic respiratory-distress syndrome with continuous positive airway pressure. N Engl J Med 284(24): 1333- 1340.
- Wiswell TH, Srinivasan P (2003) Continuous Positive Airway Pressure. In: Goldsmith JP, et al. (eds.), Assisted ventilation of the neonate. (3rd edn), Saunders, Philadelphia, USA, pp. 127-143.
- Morley C (1999) Continuous distending pressure. Arch Dis Child Fetal Neonatal Ed 81(2): 152F-F156.
- Yu VYH, Rolfe P (1977) Effect of continuous positive airway pressure on cardiorespiratory function in infants with respiratory distress syndrome. Acta Paediatr Scand 66(1): 59-64.
- Kavvadia V, Greenough A, Dimitriou G (2000) Effect on lung function of continuous positive airway pressure administered either by infant flow driver or a single nasal prong. Eur J Pediatr 159(4): 289-292.
- Mazzella M, Bellini C, Calevo MG, Campone F, Massocco D, et al. (2001) A randomized control study comparing the Infant Flow Driver with nasal continuous positive airway pressure in preterm infants. Arch Dis Child Fetal Neonatal Ed 85(2): F86-F90.
- InstitutoNacional de Perinatología México (2009) Normas y procedimientos de neonatología. Marketing y Publicidad de México, México.
- Wung JT, Driscoll JM, Epstein RA, Hyman AI (1975) A new device for CPAP by nasal route. Crit Care Med 3(2): 76-78.
- Davis P, Davies M, Faber B (2001) A randomized controlled trial of two methods of delivering nasal continuous positive airway pressure after extubation to infants weighing less than 1000g: binasal (Hudson) vs single nasal prongs. Arch Dis Child Fetal Neonatal Ed 85(2): F82-F85.
- Méndez-Fuentes AP (2013) Apego al uso de CPAP nasal de burbuja en el INPer “Isidro Espinosa de los Reyes”. México Tesis neonatología, México.
- Hulley DB, Cummings SM, Browner WG, Newman JB (2008) Cálculo de la potencia y el tamaño de la muestra: aplicaciones y ejemplos. In: Hulley SB et al. (eds.), Diseño de investigacionesclínicas. (3rd edn), Lippincott Wilkins Williams, Philadelphia, USA, pp. 98-99.
- Yagui AC, Vale LA, Haddad LB, Prado C, Rossi FS, et al. (2011) Bubble CPAP versus CPAP with variable flow in newborns with respiratory distress: a randomized controlled trial. J Pediatr Rio J 87(6): 499-504.
- Lista G, Castoldi F, Fontana P, Daniele I, Cavigioli F, et al. (2010) Nasal continuous positive airway pressure (CPAP) versus bi-level nasal CPAP in preterm babies with respiratory distress syndrome: a randomized control trial. Arch Dis Child Fetal Neoantal Ed 95(2): F85-F89.
- O’Brien K, Campbell C, Brown L, Wenger L, Shah V (2012) Infant flow biphasic nasal continuous psotive airway pressure (BP-NCPAP) vs. infant flow NCPAP for the facilitation of extubation in infants <1250 grams: a randomized controlled trial. BMC Pediatr 12: 43.
- Fernández CL, Fajardo CA, Favareto MV, Hoyos A, Jijón- Letort FX, et al. (2014) Oxygen dependency as equivalent to bronchopulmonarydisplasia at different altitudes in newborns <1500g at birth from the SIBEN network. J Perinatol 34(7): 538-542.






























