Abstract
Microbial fermentation represents a sustainable strategy for producing novel polysaccharides with desirable properties for biomaterial applications, particularly those synthesized by food grade bacteria. Lactic acid bacteria, including Lactobacillus, Leuconostoc, Weissella, Lactococcus, Streptococcus, Pediococcus, and Bifidobacterium spp., are known to produce extracellular polysaccharides that can be classified according to their chemical composition and structural features. In this context, the present study provides a comprehensive chemical and physicochemical characterization of the exopolysaccharide (EPS) synthesized by Lactiplantibacillus plantarum LH04, a strain isolated from human milk. The EPS was identified as a heteropolysaccharide composed primarily of glucose, galactose, fructose, N acetylglucosamine, and succinic acid. The extraction and purification process yielded 1.09 g·L⁻¹ of EPS, containing 86.38% (w/w) total carbohydrates, 0.3% (w/w) protein, and less than 0.001% (w/w) reducing sugars. The EPS exhibited notable thermal stability, with decomposition occurring above 259 °C, and displayed a predominantly amorphous structure, as confirmed by X-ray diffraction analysis, with a crystallinity index of 1.8%. Additionally, it showed an uncommon solubility profile, being insoluble in water and organic solvents but fully soluble in NaOH and KOH. Overall, the compositional and physicochemical characteristics of this HePS underscore its potential for diverse applications in food, biomedical, and environmental technologies.
This is ultimately followed by the convergence of finely, tuned epigenetic switches and the synthetic biology, leading to a safer, more precise, and diverse process of disease modeling as well as the therapeutic intervention.
Keywords:Lactiplantibacillus plantarum LH04; Exopolysaccharides; Characterization, Fermentation; Physical-chemical characterization; Ecofriendly process
Introduction
Exopolysaccharides (EPS) are macromolecules made up of linear or branched long chains of sugar molecules which have high molecular weight and are obtained from different sources such as bacteria, algae, plants, and animals [1,2]. Interest in exploring emerging biopolymers, such as bacterial EPS, has opened a growing research field due to the unique physicochemical properties that each biopolymer exhibits [3]. The EPS has proven to have applications in the pharmaceutical, food, biomedical, and bioremediation sectors, including emulsification, stabilization, bacterial proliferation, structural support in biofilm formulation, preservative, additive, and others [4,5]. In the context of polysaccharides and EPS as microbial metabolites, the former has been associated with the promotion of pathogenic bacteria, while the latter are produced to cope with environmental stress.
These sugar complexes adhere to the cell wall structure acting as a water reserve, and a carbon source. Likewise, once secreted into the extracellular space, EPS can interact with other biomolecules such as acids, proteins, and lipids in the culture medium to form an extracellular matrix [6,7]. The EPS can be classified into two main groups: homopolysaccharides (HoPS), which are conformed to only one kind of monosaccharide, such as D-glucose, D-galactose, D-fructose, D-mannose, L-rhamnose, and so on. The synthesis of HoPS is typically carried out by a single sucrase enzyme that can polymerize subunits of glucose in the cell wall. For example, dextran is made up of subunits of glucose through glycosidic link α-1-6; this EPS is achieved by the dextransucrase enzyme [8]. On the other hand, heteropolysaccharides (HePSs) have two or more types of monosaccharides in the sugar polymer chain, such as D-galactose, L-rhamnose, D-glucose, D-fructose, D-mannose, glucuronic acid, among others [9]. On the other hand, techniques such as colorimetric tests using Congo Red Agar and Calcofluor White, precipitation with organic solvents, visual detection of viscosity in the culture medium, and carbohydrates analysis (e.g. uronic acid via m-hydroxydiphenyl colorimetry) are used to demonstrate whether a bacterium can produce EPS [10,11].
Among a wide variety of microorganisms, lactic acid bacteria (LAB) have demonstrated to produce EPS with a peculiar physicochemical profile. This is due not only to their characteristics as additives, flocculants, and emulsifiers, but also as a source used in the formulation of anti-tumor drugs and cholesterol-lowering agents [12,13]. LABs isolated from human milk have demonstrated a unique metabolic profile, producing specific metabolites such as butyrate, polyamines, bioactive peptides, oligosaccharides, and exopolysaccharides in contrast to LAB isolated from other sources [14,15]. Despite the considerable interest in these microorganisms, the research on the EPS produced by Lactiplantibacillus plantarum isolated from human breast milk remains relatively limited, particularly in terms of their physicochemical characterization. According to the extant literature, the majority of EPS reported by LAB exhibit characteristics such as complete water solubility and low thermal resistance. These characteristics serve as the primary parameters for determining applications.
Nevertheless, studies demonstrate that the properties of each EPS are contingent on the strain and the culture medium, potentially resulting in inconsistencies with typical EPS profiles depending on the microbial source. In particular, the discovery of EPS with atypical characteristics, such as limited water solubility, incorporation of nitrogenous carbohydrates, organic acid, or greater thermal stability, is rarely described in the case of LAB isolation from breast milk. These atypical characteristics have the potential to expand their application beyond the conventional, thereby facilitating exploration in the domains of biomaterials, environmental processes, and separation technologies [16,17].
In this context, the main objective in the present study is the production and comprehensive characterization of an exopolysaccharide (EPS) synthesized by L. plantarum LH04 (isolated from breast milk). The EPS was analyzed in terms of its monosaccharide composition, purity, solubility profile, crystallinity, thermal behavior, functional groups, and elemental composition, that represent the basis of biopolymer characterization (either a homopolysaccharide or a heteropolysaccharide) for its future applications. Considering the composition and physicochemical features, this work aims to provide the structure and properties of EPS with uncommon characteristics, contributing to the variability of EPS within each strain and supporting its use as an additive in food formulation, a prebiotic for the proliferation of beneficial bacteria for health (Bifidobacteria and Lactobacillus), or in the development of drug-loaded gels, where EPS may serve as a structural matrix or membrane scaffold.
Materials and methods
Materials
Sodium dodecyl sulfate (SDS, 99.0%), sugar standards (maltose, lactose, lactulose, galacturonic acid, glucose, galactose, fructose, arabinose, N-acetylglucosamine), citric acid, and succinic acid (99.0%) were purchased from Sigma-Aldrich (USA). Trichloroacetic acid (TCA, 99.0%) was procured from Golden Bell-Reactivos (Mexico). Sulfuric acid (H2SO4, 97.0%), barium hydroxide octahydrate (Ba(OH)₂·8H2O, 99.0%), manganese sulfate monohydrate (MnSO4·H2O, 99.0%) -all of analytical gradewere purchased from Fermont (Mexico). Lactiplantibacillus plantarum LH04 strain was isolated from human milk and identified by molecular biology at the human milk laboratory, Centro Universitario de Ciencias Exactas e Ingenierías (CUCEI). The strain was grown on De Man, Rogosa, and Sharpe agar (MRS, BD Difco, USA).
L. plantarum fermentation for EPS production
Culture medium composition: The culture medium used for EPS production by L. plantarum LH04 was a modified MRS broth, prepared according to the formulation described by Wang et al. [18]. The composition of the medium (g·L-1) was as follows: proteose peptone No. 3, 10.0; beef extract, 10.0; yeast extract, 5.0; dextrose, 20.0; polysorbate 80, 1.0; ammonium citrate, 2.0; sodium acetate, 5.0; magnesium sulfate, 0.1; dipotassium phosphate, 2.0; manganese sulfate, 0.6; pH 6.4. The medium was sterilized at 121°C for 15 min.
Inoculum and fermentation medium conditions: The preinoculum was prepared by inoculating 80 mL of modified MRS medium to a final concentration of approximately 1x108 CFU/mL and incubated at 37°C for 24 h under static conditions [19]. For the main fermentation, the pre-inoculum was aseptically transferred into 720 mL of fresh modified MRS medium (final volume: 800 mL) and incubated at 37°C for 72 h without agitation.
Extraction and purification of EPS: After 72 h incubation period, the fermented culture medium was stopped by adding trichloroacetic acid (TCA) to final concentration of 5% (w/v) to precipitate proteins, and sodium dodecyl sulfate was added to a final concentration of 0.1% (w/v) to facilitate the EPS release from cell surfaces. Then, bacterial cells and proteins were separated by centrifugation at 4,664 x g for 20 min at 4°C in a refrigerated centrifuge (Sigma Zentrifugen 8K10). The resulting cell-free supernatant was treated with 1:3 (v/v) culture: chilled isopropyl alcohol and left to rest at 4°C overnight to allow EPS agglomeration. After this period, the agglomerate was recovered by centrifugation at 4,664 x g for 20 min at 4°C. The supernatant was discarded, and the EPS pellet was resuspended in deionized water and dialyzed for 72 h using a 12-14 kDa molecular weight cut-off cellulose membrane (Spectra/Por Dialysis membrane). Water was used as the dialysis buffer and changed twice per day. Finally, the dialyzed EPS was lyophilized to obtain a dry powder.
Physicochemical characterization of EPS
Determination of total sugars using the phenol-sulfuric acid method: The phenol-sulfuric acid method was used to determine the total carbohydrate content in the EPS, with glucose as the standard compound [20]. To perform the assay, approximately 6 mg of EPS was dissolved in 2 mL of 0.2 M NaOH. After dissolving the sample, 8 mL of distilled water was added and stirred. Then, 0.1 mL of the sample was diluted to 1.0 mL with distilled water to obtain 0.06 mg·mL-1 as a final concentration. The reaction was carried out by transferring 0.25 mL of diluted sample to the test tube, followed by the addition of 0.15 mL of phenol reagent (5.0% w/v in water) and 1.0 mL of concentrated sulfuric acid. The mixture was stirred and incubated until it reached room temperature. The absorbance of the samples (in triplicate) was measured at 490 nm using Thermo Scientific Genesys 20 spectrophotometer. The total carbohydrate content of the EPS was estimated from the linear regression of absorbance values from a five-point glucose standard curve (10, 20, 40, 80, and 100 μg·mL-1). Distilled water was used as the blank for all measurements.
Determination of reducing sugars by 3,5-Dinitrosalicilic Acid (DNS): To detect the presence of free carbonyl groups (C=O) as reducing sugar, the EPS samples were analyzed using the 3,5-dinitrosalicylic acid (DNS) method at 540 nm with a Thermo Scientific Genesys 20 spectrophotometer. A standard curve (300, 600, 900, 1,200, and 1,500 μg·mL-1) was used to estimate the concentration of free carbonyl groups [21].
Total protein: To estimate total protein content, an EPS sample was prepared at a final concentration of 2000 μg·mL-1 and measured using the Bradford assay at 595 nm with a Thermo Scientific Genesys 20 spectrophotometer [22]. A standard curve was prepared using a linear regression of bovine serum albumin (BSA) (0, 10, 25, 50, 75, and 100 μg·mL-1).
Thin-layer chromatography (TLC): Thin-layer chromatography was performed by adding 5 μL of both hydrolyzed and non-hydrolyzed EPS samples on the silica gel (Sigma-Aldrich TLC plates silica gel matrix), as well as glucose, galactose, fructose, mannose, chicory inulin, and pectin as standard compounds. The plate was developed with a solvent mixture of water, 1-butanol, and glacial acetic acid (10:25:15, v/v) in a total volume of 50 mL. After drying the plate, a 9:1 (v/v) ethanol:sulfuric acid solution was sprayed onto the TLC plate and heated at 80°C for 30 min to visualize hydrolyzed products and reference standards [23].
Monosaccharide composition: To determine the monosaccharide composition, 10 mg of purified EPS was hydrolyzed with 2.0 M H2SO4 at 120°C for 30 min. The hydrolyzed mixture was neutralized by the addition of 2.0 M Ba(OH)₂. The mixture was stirred and centrifuged at 5,750 x g for 10 min at 4°C (using a Labnet Hermle Z300 K MicroCentrifuge) to precipitate the barium sulfate, the reaction product. After centrifugation, 1.0 mL of supernatant was collected, and the monosaccharide composition was analyzed by HPLC (Agilent Series 1200) using a MetaCarb H-Plus column with a refractive index detector (RID). The mobile phase consisted of 0.0125 M H2SO4 at a flow rate of 0.5 mL·min-1 at 50°C.
Elemental composition: Approximately 2.5 mg of the EPS sample were wrapped in aluminum foil and compressed to determine the elemental composition of carbon (C), hydrogen (H), nitrogen (N), and sulfur (S) using an elemental analyzer (TrueSpec® Micro Series, UK).
FTIR - EPS analysis: The EPS functional groups were determined by Fourier-transform infrared spectroscopy (FTIR) using an Agilent Cary 630 FTIR spectrophotometer. Approximately 10 mg of the EPS sample (previously oven-dried at 60°C for 24 h) were placed in the diamond-window and scanned from 500 to 4000 cm-1 at scan rate of 32 scans and a spectral resolution of 2 cm-1.
Thermogravimetric analysis (TGA): EPS thermal behaviour was analyzed by thermogravimetric analysis (TGA) using a Discovery Series, TA Instruments, USA. The sample was evaluated over a temperature range from 30 to 600°C under nitrogen atmosphere at a heating rate of 10°C·min-1.
Differential Scanning Calorimeter (DSC): The thermal properties of the EPS were determined by differential scanning calorimetry (DSC, Discovery Series, TA Instruments, USA). Approximately 5 mg of EPS was analyzed over a temperature range from 30 to 350°C, at a heating rate of 5°C·min-1 under nitrogen atmosphere.
X-Ray Diffraction (XRD): X-ray diffraction (XRD) measurements of the purified EPS were performed using a Malvern PANalytical EMPYREAN diffractometer. Data were collected over the 2θ range from 5 to 90°, with a step size of 0.02°, and a scanning speed of 30°·min-1, using a Cu anode as the X-ray source.
Qualitative solubility test
The qualitative solubility of the EPS was determined using different solvents. Acetonitrile, methanol, acetone, ethyl acetate, ethanol, dimethyl sulfoxide and hexane as organic solvents and sodium chloride (NaCl), calcium chloride (CaCl2), potassium chloride (KCl), ammonium acetate (NH4CH3CO2), ammonium nitrate (NH4NO3), ammonium chloride (NH4Cl), ammonium sulfate ((NH4)2SO4) and sodium hydroxide (NaOH) as alkaline solutions were tested at concentration of 0.1, 0.25 and 0.5 M. Each of the solutions was assessed according to the U.S. Pharmacopoeia to determine the degree of solubility of the EPS [24].
Statistical analysis
Data were analyzed using JMP Pro software for Windows (version 18.0.2, NC, SAS Institute Inc., Cary, NC, USA), and the Student’s t-test was considered to determine statistical inference at a level of α = 0.05.
Results and Discussion
Chemical composition analysis of EPS
Crude exopolysaccharide (EPS) was obtained from L. plantarum fermentation by precipitation with chilled isopropyl alcohol (4 °C), yielding approximately 1.09 g·L⁻¹ of a beige, powder-like material. The resulting precipitate was subsequently dialyzed to further purify the sample (Figure 1).

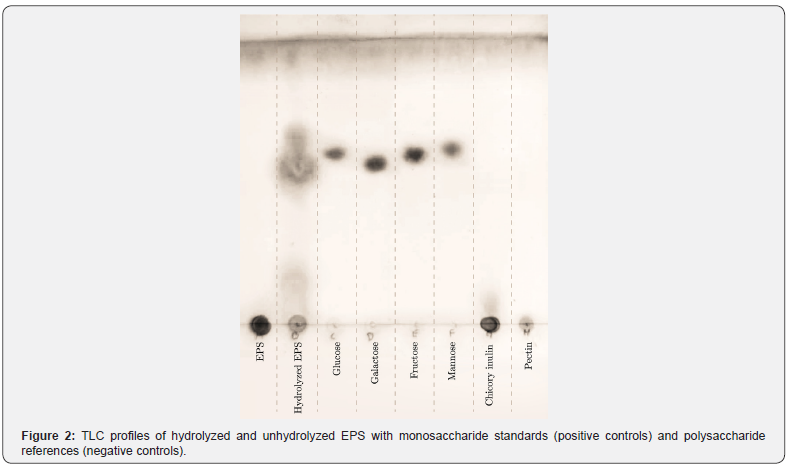
Chemical composition analysis revealed that the purified EPS contained 86.38% (w/w) total carbohydrates, 0.300% (w/w) protein, and less than 0.001% (w/w) reducing sugars, as determined by the DNS assay. Thin-layer chromatography of the unhydrolyzed EPS (Figure 2) showed no detectable migration on the silica plate, a behavior consistent with the negative controls (chicory inulin and pectin). No residual monosaccharides were observed in this sample. In contrast, the positive controls (glucose, galactose, fructose, and mannose) exhibited clear migration. Notably, the lane corresponding to the hydrolyzed EPS displayed brown spots that migrated almost identically to the monosaccharide standards. In this sense, the TLC results indicate that the EPS produced by L. plantarum LH04 probably consists of multiple monosaccharide units. Moreover, the absence of free glucose in the unhydrolyzed sample confirms that there is no residual sugar from MRS medium in the EPS after purification. This finding aligns with previous studies showing that lactic acid bacteria are capable to synthesize both homopolysaccharides and heteropolysaccharides with diverse monosaccharides composition [25].
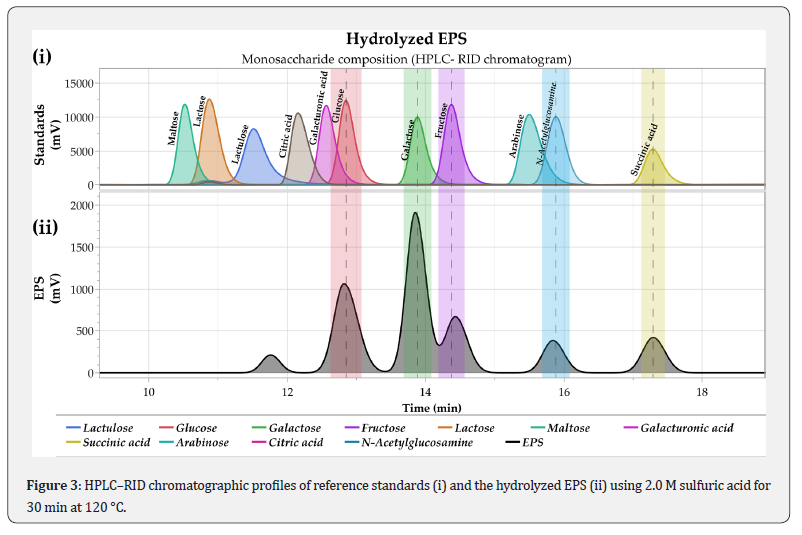
The HPLC–RID chromatogram (Figure 3) confirms that the EPS produced by L. plantarum LH04 is a heteropolysaccharide. Chromatographic analysis of the hydrolyzed sample revealed the presence of glucose, galactose, fructose, N acetylglucosamine, and succinic acid. Among these components, glucose, galactose, and fructose were detected at higher concentrations compared with N acetylglucosamine and succinic acid. Notably, galactose was identified as the most abundant monosaccharide in the EPS structure. The monosaccharide profile of the EPS produced by L. plantarum LH04 aligns with previous reports indicating that EPS synthesized by lactic acid bacteria may contain a wide range of monosaccharides, including glucose, galactose, mannose, fructose, N acetylglucosamine, N acetylgalactosamine, and glucuronic acid [26-30] reported that L. plantarum PC715 produced a heteropolysaccharide composed of glucose, mannose, and rhamnose, while Chin Feng et al. [31] found that the EPS from L. plantarum NTU 102 contained fructose, arabinose, galactose, glucose, mannose, and maltose. Collectively, these findings support the ability of L. plantarum LH04 to synthesize an EPS with a diverse monosaccharide composition.
The elemental composition of the EPS produced by Lactiplantibacillus plantarum LH04 was determined by quantifying the percentages of carbon (C), hydrogen (H), nitrogen (N), and sulfur (S), yielding values of 39.15%, 5.30%, 3.84%, and 0.0%, respectively. The measurable nitrogen content, together with the absence of sulfur, initially suggested the possible presence of protein residues; however, this interpretation is inconsistent with the low protein concentration obtained by the Bradford assay (0.3% w/w). Instead, the nitrogen detected is more plausibly attributed to amino sugars-particularly N acetylglucosaminewhich contributes nitrogen through its amide group. This explanation aligns with previous reports of nitrogen containing polysaccharides, such as chitin, which contains approximately 7% nitrogen [32,33]. The presence of nitrogen in the EPS from L. plantarum LH04 is further supported by the functional groups identified in the FTIR spectrum and the monosaccharide composition determined by HPLC, indicating that amide linked residues are incorporated into the polymer structure.
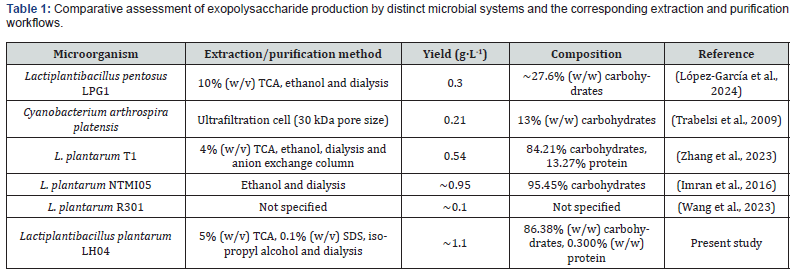
The EPS yield obtained from Lactiplantibacillus plantarum LH04 in this study was higher than the values previously reported for other Lactiplantibacillus strains (Table 1) [34-37]. This result underscores the strong capacity of L. plantarum to synthesize EPS efficiently, producing a polymer with a predominantly carbohydrate rich composition (86%). The enhanced EPS yield observed here may be associated with methodological optimizations introduced during both the cultivation and recovery stages. In particular, the use of isopropyl alcohol-whose lower dielectric constant relative to ethanol, likely promotes polymer aggregation-appears to have facilitated more efficient precipitation [38]. Moreover, the modified MRS medium, supplemented with an increased concentration of MnSO₄, may have improved the availability of essential cofactors involved in EPS biosynthesis. Finally, the remaining 13.62% of non-carbohydrate material suggests the presence of additional components such as succinic acid, which are commonly found in bacterial EPS matrices and may contribute to their structural or functional properties.
Physicochemical Characterization of the EPS
The functional groups of the purified EPS were characterized using FTIR spectroscopy (Figure 4). The spectrum exhibited a broad absorption band at 3550–3100 cm⁻¹, confirming the presence of hydroxyl groups (–OH) typically associated with polysaccharide structures. Weak peaks at 2938 cm⁻¹ and 1421 cm⁻¹ corresponded to C–H stretching vibrations of aliphatic sugar chains [39]. A distinct signal at 1640 cm⁻¹ was attributed to the C=O stretching vibration of free carboxylic acid groups (– COOH). This feature supports the presence of succinic acid in its monoester form, as no strong ester carbonyl band was observed in the 1730–1750 cm⁻¹ region, which would indicate complete diesterification [40].
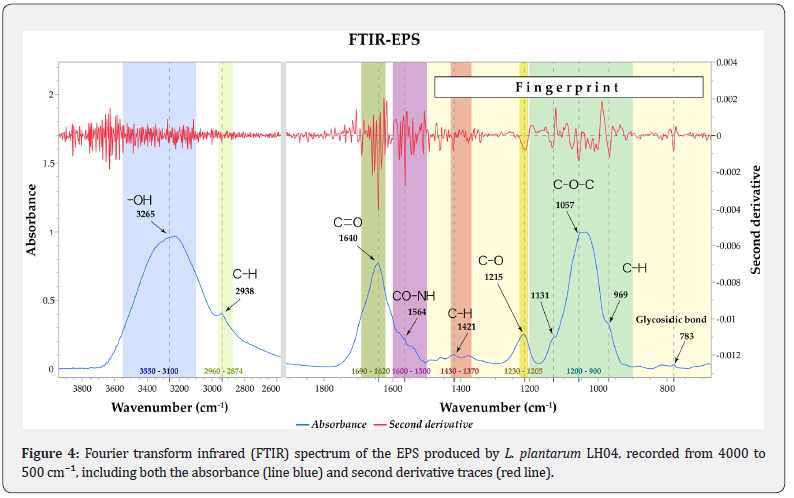
Second derivative analysis revealed an additional overlapping peak at 1564 cm⁻¹ within the 1500–1600 cm⁻¹ region, corresponding to amide II vibrations (N -H bending and C–N stretching), thereby confirming the presence of nitrogen containing functional groups such as N acetylglucosamine [41]. An absorption peak at 1215 cm⁻¹, within the characteristic 1230– 1205 cm⁻¹ range, indicated ester (C–O) stretching vibrations, further supporting the incorporation of succinic acid into the EPS structure [42]. The broad region between 1200 and 900 cm⁻¹ displayed several absorption bands at 1131, 1057, and 969 cm⁻¹, corresponding to the fingerprint region of exopolysaccharides and indicative of pyranose ring structures (β glucans) [43]. Finally, the peak at 783 cm⁻¹ confirmed the presence of β glycosidic linkages, while the absence of absorption bands at 766, 844, and 917 cm⁻¹ indicated that α glycosidic linkages were not present [44].
The thermal behavior of the EPS was assessed using thermogravimetric analysis (TGA) over a temperature range of 30–600 °C. The thermogram and its first derivative revealed five distinct stages of weight loss (Figure 5 i). The first stage (50–100 °C) corresponded to moisture evaporation, resulting in a 10.6% mass loss. A slight decrease was also observed around 60 °C, likely due to the evaporation of residual water and alcohol retained within the EPS matrix, a phenomenon attributed to strong interactions between these molecules and carboxyl groups [45]. The second stage (100–215 °C) showed no significant mass loss, indicating that the polymer remained thermally stable in this region. The third stage (215–303 °C) exhibited major degradation, with a 24.4% mass loss and an inflection point at 259.6 °C, consistent with previous findings for EPS produced by L. plantarum MTCC 9510, which also showed substantial decomposition near this temperature [46]. The fourth stage displayed a decomposition temperature (Td) at 345.8 °C, accompanied by a 16.5% mass loss. The final stage (380–600 °C) showed a gradual decline, accounting for an additional 22.7% mass loss. The residual mass at 600 °C was 7.97%, indicating that the EPS from L. plantarum LH04 exhibits considerable thermal stability across a broad temperature range. Overall, the multi-step degradation pattern suggests the presence of heterogeneous structural domains with varying thermal resistance.
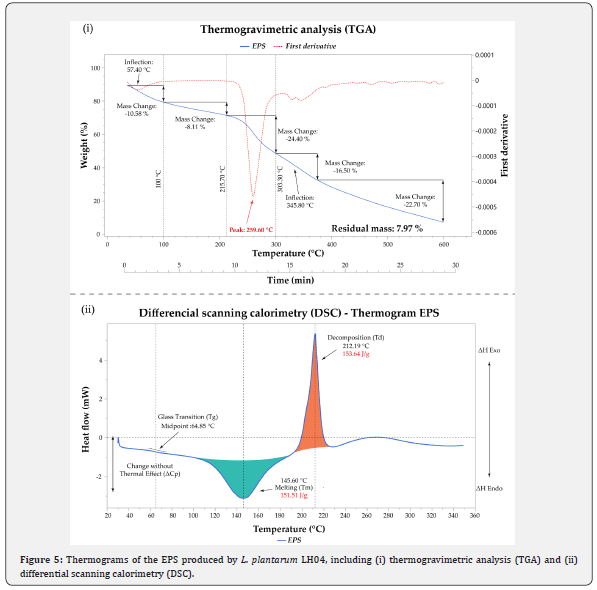
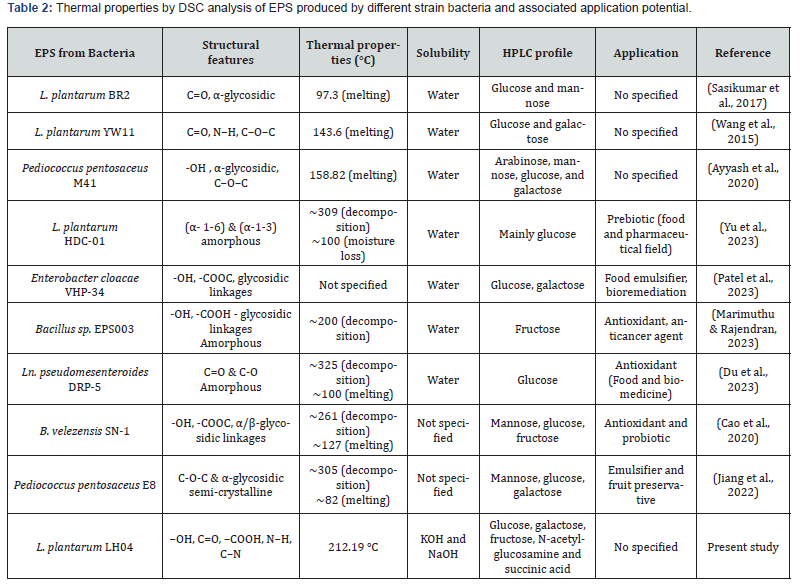
The thermal properties of the EPS were further examined using differential scanning calorimetry (DSC), a complementary thermogravimetric technique that provides insight into energy transitions and enthalpy changes (ΔH). Understanding these thermal events is essential for the development of biomaterials, films, gels, and coatings, as they influence stability during processing and application. The DSC scan performed from 30 to 350 °C (Figure 5 ii) revealed a glass transition temperature (Tg) at 64.85 °C, marking the onset of molecular mobility within the amorphous regions of the polymer. This was followed by an endothermic melting peak (Tm) at 145.60 °C, associated with the disruption of crystalline or ordered domains, and accompanied by an enthalpy change (ΔH) of 151.51 J·g⁻¹. An exothermic decomposition peak (Td) was subsequently observed at 212.19 °C, with an enthalpy change of 153.64 J·g⁻¹. The melting temperature obtained in this study aligns with previously reported values (Table 2), being higher than those of EPS produced by L. plantarum BR2 (97.3 °C) [47] and L. plantarum YW11 (143.6 °C) [48], but lower than that of Pediococcus pentosaceus M41 (158.82 °C) [49]. It is important to note that typical processing conditions in the food and pharmaceutical industries do not reach such elevated temperatures, indicating that the EPS maintains structural integrity under conventional application environments. However, the thermal stability of EPS produced by L. plantarum LH04 is an important indicator of its structural robustness. The thermal properties suggest that the EPS can support thermal treatments such as pasteurization, sterilization, or drying processes (e.g., spray-drying) without significant degradation.
Understanding the molecular organization of an EPS, particularly the distribution of amorphous and crystalline phases, provides valuable insight into its potential industrial applications. Amorphous or semi amorphous structures are generally associated with enhanced water holding capacity and viscosity, whereas a higher degree of crystallinity often reduces dispersibility. The X ray diffraction (XRD) pattern of the EPS (Figure 6) revealed a crystallinity index of 1.80%, confirming that the material is predominantly amorphous. The use of the second derivative function facilitated the identification and deconvolution of overlapping peaks. Broad diffraction signals at approximately 10° and 25° (2θ) are characteristic of amorphous regions, while a small but distinct peak near 45° (2θ) indicates the presence of a minor crystalline domain within the polymer matrix.
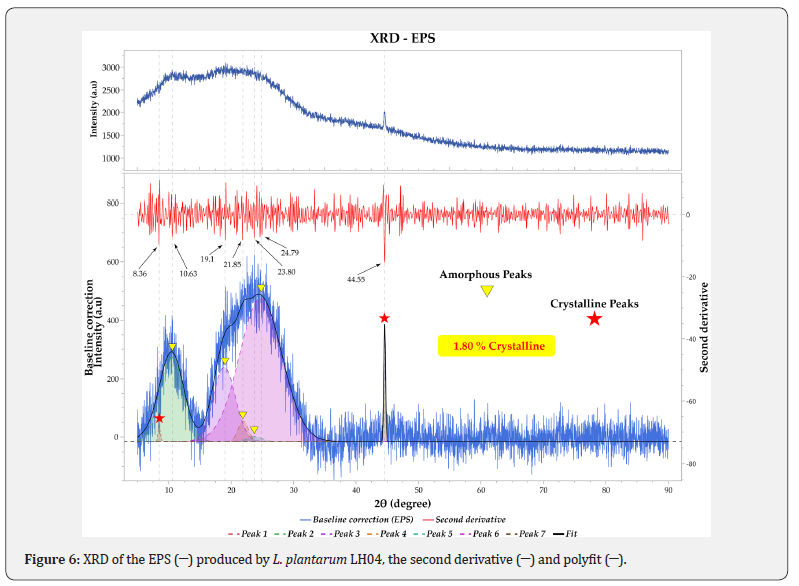
This predominance of broad peaks is consistent with previous reports for EPS produced by L. plantarum HDC 01 and B. cereus KMS3 1, which also exhibited amorphous halos around 10° and 25° (2θ) [50,51]. The weak crystalline peak at ~45° (2θ) similarly aligns with observations in EPS from I. obliquus, Fusarium sp., L. lactis KC117496, and L. delbrueckii GRIPUMSK [52-55], all of which display partial ordering within an otherwise amorphous structure. The limited crystallinity observed in the EPS from L. plantarum LH04 may be influenced by the presence of Mn²⁺ ions in the modified MRS medium, as this strain appears capable of utilizing metal ions as cofactors during EPS biosynthesis, thereby affecting polymer conformation and molecular organization. Additionally, the EPS may interact with metal ions in the medium through chelation, contributing to the coexistence of amorphous and crystalline phases. This interpretation is supported by previous findings showing Mn(II) biosorption by an EPS produced by Rhizobium etli [56].
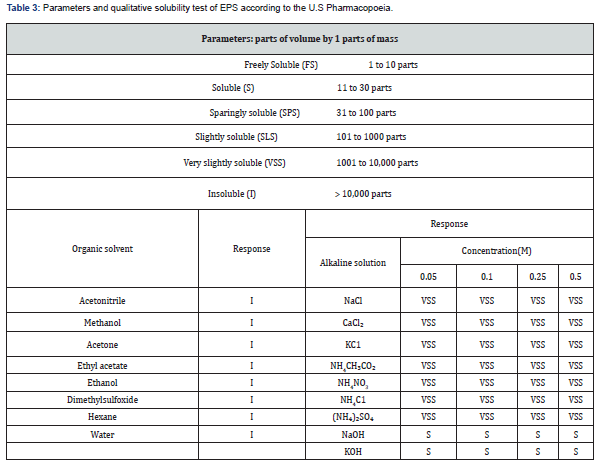
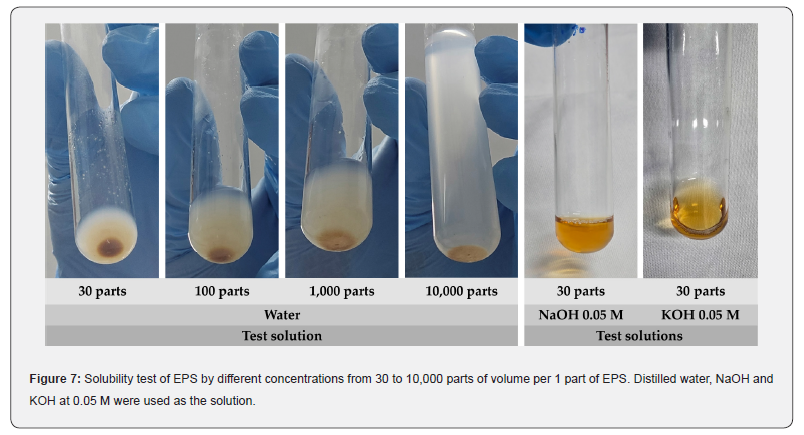
The qualitative solubility assessment of the EPS in different solutions provides insight into its potential applicability in processes involving organic solvents or salt based systems. The EPS produced by Lactiplantibacillus plantarum LH04 was insoluble in all organic solvents tested (Table 3). Similarly, water solubility tests at ratios of 30, 100, 1,000, and 10,000 parts of water per 1 part of EPS showed no dissolution after 30 min of resting (Figure 7). In contrast, the EPS exhibited limited solubility in alkaline solutions (0.05, 0.1, 0.25, and 0.5 M), yet it was fully soluble in both NaOH and KOH at all concentrations evaluated (Table 3). The pronounced insolubility in water suggests a structural organization influenced by crystalline regions and the presence of ester functional groups, both of which restrict interactions with water molecules. The semi crystalline nature of the polymer may also account for its solubility in alkaline environments, where hydroxide ions can hydrolyze ester linkages and promote polymer dispersion. Similar behavior was reported by Ahmed et al., who described an EPS produced by L. plantarum isolated from apple and cultivated in a modified Hestrin and Schramm medium (MHS), which was likewise water insoluble and identified as bacterial cellulose. These observations underscore the significant influence of culture medium composition on the structural and functional properties of microbial EPS [57-62].
Conclusion
In the present study, the characterization of an EPS produced by L. plantarum LH04 isolated from human milk, was performed through a modified MRS broth and characterized using chemical, spectroscopic, thermal and structural approaches. The EPS was classified as a heteropolysaccharide based on its composition, which includes more than two types of monosaccharides in its structure, including glucose, galactose, fructose, N-acetylglucosamine and succinic acid. The physicochemical analysis revealed several unusual characteristics for an EPS produced by L. plantarum LH04. The polymer exhibited crystalline components in its structure, along with remarkable thermal stability, as well as unusual solubility behavior, being insoluble in water and organic solvents but completely soluble in alkaline media. Importantly, the distinctive characteristics of the EPS produced by L. plantarum LH04 differentiate it from previously reported LAB-derived EPS, which are generally water-soluble and less thermal resistance. These properties suggest a range of applications, including heat sterilization, alkaline environments, prolonged thermal exposure, food encapsulation, as a packing base for chromatographic columns, or in formulation of biofilms, and as a metal scavenger in water treatment.
Acknowledgments
The authors thank the National Council for Humanities, Sciences and Technologies (SECIHTI) for financial support for the first author’s doctoral studies. They also gratefully acknowledge the support of L’Oréal-UNESCO-AMC for this publication.
References
- Dilna SV, Surya H, Aswathy RG, Varsha KK, Sakthikumar DN, et al. (2015) Characterization of an exopolysaccharide with potential health-benefit properties from a probiotic Lactobacillus plantarum RJF4. LWT - Food Science and Technology 64(2): 1179-1186.
- Wang J, Zhang J, Guo H, Cheng Q, Abbas Z, et al. (2023 Optimization of Exopolysaccharide Produced by Lactobacillus plantarum R301 and Its Antioxidant and Anti-Inflammatory Activities. Foods 12(13): 2481
- Netrusov AI, Liyaskina EV, Kurgaeva IV, Liyaskina AU, Yang G, et al. (2023) Exopolysaccharides Producing Bacteria: A Review. Microorganisms 11(6): 1541.
- Jivkova D, Sathiyanarayanan G, Hari M, Hertkorn N, Schmitt-Kopplin P, et al. (2022) Production and Characterization of a Novel Exopolysaccharide from Ramlibacter tataouinensis. Molecules 27(21): 7172
- Paikra SK, Panda J, Sahoo G, Mishra M (2022) Characterization of exopolysaccharide derived from Enterobacter ludwigii and its possible role as an emulsifier. 3 Biotech 12(9): 212.
- Khan R, Shah MD, Shah L, Lee PC, Khan I, et al. (2022) Bacterial polysaccharides—A big source for prebiotics and therapeutics. Frontiers in Nutrition.
- Prasad S, Purohit SR (2023) Microbial exopolysaccharide: Sources, stress conditions, properties and application in food and environment: A comprehensive review. International Journal of Biological Macromolecules 242(Pt 3): 124925
- Schmid J, Sieber V, Rehm B (2015) Bacterial exopolysaccharides: Biosynthesis pathways and engineering strategies. Frontiers in Microbiology 26:6:496.
- Lynch KM, Zannini E, Coffey A, Arendt EK (2018) Lactic Acid Bacteria Exopolysaccharides in Foods and Beverages: Isolation, Properties, Characterization, and Health Benefits. Review of Food Science and Technology 9(1): 383-410.
- Alan Y, Keskin AO, Sönmez M (2025) Probiotic and functional characterization of newly isolated Lactiplantibacillus plantarum strains from human breast milk and proliferative inhibition potential of metabolites. Enzyme and Microbial Technology: 182.
- Rühmann B, Schmid J, Sieber V (2015) Methods to identify the unexplored diversity of microbial exopolysaccharides. Frontiers in Microbiology 6: 565.
- Di W, Zhang L, Wang S, Yi H, Han X, et al. (2017) Physicochemical characterization and antitumour activity of exopolysaccharides produced by Lactobacillus casei SB27 from yak milk. Carbohydrate Polymers 171: 307-315.
- Srinivash M, Krishnamoorthi R, Mahalingam PU, Malaikozhundan B (2023) Exopolysaccharide from Lactococcus hircilactis CH4 and Lactobacillus delbrueckii GRIPUMSK as new therapeutics to treat biofilm pathogens, oxidative stress and human colon adenocarcinoma. International Journal of Biological Macromolecules 250: 126171
- Łubiech K, Twarużek M (2020) Lactobacillus bacteria in breast milk. Nutrients 12(12): 1-13.
- Toscano M, Grandi R, De Grossi E, Drago L (2017) Role of the human breast milk-associated microbiota on the newborns’ immune system: A mini review. Frontiers in Microbiology 8:2100
- Hernández-Figueroa RH, López-Malo A, Mani-López E (2025) Lactic Acid Bacteria-Derived Exopolysaccharides: Dual Roles as Functional Ingredients and Fermentation Agents in Food Applications. In Fermentation Multidisciplinary Digital Publishing Institute (MDPI) 11(9): 538.
- Ouarabi Lylia, Ouarabi Liza, Hamma-Faradji S, Mohedano ML, López P, et al. (2025) Exopolysaccharides from lactic acid bacteria: Structure, biosynthesis, and health benefits. In Microbes and Infection 27(8): 105581.
- Wang J Zhang J, Guo H, Cheng Q, Abbas Z, et al. (2023) Optimization of Exopolysaccharide Produced by Lactobacillus plantarum R301 and Its Antioxidant and Anti-Inflammatory Activities. Foods 12(13): 2481.
- Üçok G, Sert D (2020) Growth kinetics and biomass characteristics of Lactobacillus plantarum L14 isolated from sourdough: Effect of fermentation time on dough machinability LWT 129.
- Kurzyna-Szklarek M, Cybulska J, Zdunek A (2022) Analysis of the chemical composition of natural carbohydrates – An overview of methods. Food Chemistry 394: 133466.
- McCleary BV, McGeough P (2015) A Comparison of Polysaccharide Substrates and Reducing Sugar Methods for the Measurement of endo-1,4-β-Xylanase. Applied Biochemistry and Biotechnology 177(5): 1152-1163.
- Kielkopf CL, Bauer W, Urbatsch IL (2020) Bradford assay for determining protein concentration. Cold Spring Harbor Protocols (4) :136-138.
- Zhou Z, He N, Han Q, Liu S, Xue R, Hao, et al. (2021) Characterization and Application of a New β-Galactosidase Gal42 From Marine Bacterium Bacillus sp. BY02. Frontiers in Microbiology 12: 742300.
- Censi R, Di Martino P (2015) Polymorph impact on the bioavailability and stability of poorly soluble drugs. Molecules 20(10): 18759-18776.
- Toschkova S (2022) Structural characterization of an exopolysaccharide produced by Lactobacillus plantarum Ts . Acta Scientifica Naturalis 9(3): 71-83.
- Mao Y, Wang W, Mo W, Yang B, Han Y, et al. (2024) Purification, characterization, and hypoglycemic activity of exopolysaccharides from Lactiplantibacillus plantarum MY04. International Journal of Biological Macromolecules 282: 137008.
- Tallon R, Bressollier P, Urdaci MC (2003) Isolation and characterization of two exopolysaccharides produced by Lactobacillus plantarum EP56. Research in Microbiology 154(10): 705-712.
- Zaghloul EH, Ibrahim MIA (2022) Production and Characterization of Exopolysaccharide From Newly Isolated Marine Probiotic Lactiplantibacillus plantarum EI6 With in vitro Wound Healing Activity. Frontiers in Microbiology 13: 903363.
- Zhou X, Hong T, Yu Q, Nie S, Gong D, et al. (2017) Exopolysaccharides from Lactobacillus plantarum NCU116 induce c-Jun dependent Fas/Fasl-mediated apoptosis via TLR2 in mouse intestinal epithelial cancer cells. Scientific Reports 7(1): 14247.
- Tan X, Ma B, Wang X, Cui F, Li X, et al. (2024) Characterization of Exopolysaccharides from Lactiplantibacillus plantarum PC715 and Their Antibiofilm Activity Against Hafnia alvei. Microorganisms 12(11): 2229.
- Liu CF, Tseng KC, Chiang SS, Lee BH, Hsu WH, et al. (2011) Immunomodulatory and antioxidant potential of Lactobacillus exopolysaccharides. Journal of the Science of Food and Agriculture 91(12): 2284-2291.
- Banerjee A, Rudra SG, Mazumder K, Nigam V, Bandopadhyay R (2018) Structural and Functional Properties of Exopolysaccharide Excreted by a Novel Bacillus anthracis (Strain PFAB2) of Hot Spring Origin. Indian Journal of Microbiology 58(1): 39-50.
- Rani RP, Anandharaj M, Sabhapathy P, Ravindran AD (2017) Physiochemical and biological characterization of novel exopolysaccharide produced by Bacillus tequilensis FR9 isolated from chicken. International Journal of Biological Macromolecules 96: 1-10.
- Imran MYM, Reehana N, Jayaraj KA, Ahamed AAP, Dhanasekaran D, et al. (2016) Statistical optimization of exopolysaccharide production by Lactobacillus plantarum NTMI05 and NTMI20. International Journal of Biological Macromolecules 93: 731-745.
- López-García E, Marín-Gordillo A, Sánchez-Hidalgo M, Ávila-Román J, Romero-Gil V, et al. (2024) Functional features of the exopolysaccharide extracts produced by Lactiplantibacillus strains isolated from table olives. Food and Function 15(4): 1938-1947.
- Trabelsi L, M’sakni NH, Ouada H Ben, Bacha H, Roudesli S, et al. (2009) Partial characterization of extracellular polysaccharides produced by cyanobacterium Arthrospira platensis. Biotechnology and Bioprocess Engineering 14(1): 27-31.
- Zhang X, Liu Y, Zhao X, Ma K, Zhang C, et al. (2023) A novel viscous hydrophilic colloidal polysaccharide produced by Lactiplantibacillus plantarum T1: Structural characterization, rheological behavior and biological activity. Process Biochemistry 131: 101-113.
- Bahl MA, Schultheis E, Hempel DC, Nörtemann B, Franco-Lara E, et al. (2010) Recovery and purification of the exopolysaccharide PS-EDIV from Sphingomonas pituitosa DSM 13101. Carbohydrate Polymers 80(4): 1037-1041.
- Derdak R, Sakoui S, Pop OL, Cristian Vodna D, Addoum B, et al. (2022) Screening, optimization and characterization of exopolysaccharides produced by novel strains isolated from Moroccan raw donkey milk. Food Chemistry X 14: 100305.
- Bhowmik B, Afrin S, Jui AH, Bhuiyan RH, Rashid MM, et al. (2025) Exploring the purification, characterization, and industrial applications of Exopolysaccharide (EPS) from Bacillus amyloliquefaciens strain BDIFST240014. Molecular Biology Reports 52(1).
- Ardila N, Daigle F, Heuzey MC, Ajji A (2017) Antibacterial activity of neat chitosan powder and flakes. Molecules 22(1).
- Dawood DH, Elmongy MS, Negm A, Taher MA (2020) Extraction and chemical characterization of novel water-soluble polysaccharides from two palm species and their antioxidant and antitumor activities. Egyptian Journal of Basic and Applied Sciences 7(1): 141–158.
- Sarkar S, Cabrera-Barjas G, Singh RN, Fabi JP, Breig SJM, et al. (2024) Unveiling a novel exopolysaccharide produced by Pseudomonas alcaligenes Med1 isolated from a Chilean hot spring as biotechnological additive. Scientific Reports 14(1): 25058.
- Barker SA, Bourne EJ, Stacey M, Whiffen DH (1954) Infra-red Spectra of Carbohydrates. Part I. Some Derivatives of D-Glucopyranose pp: 171-175.
- Ahmed Z, Wang Y, Anjum N, Ahmad H, Ahmad A, et al. (2013) Characterization of new exopolysaccharides produced by coculturing of L. kefiranofaciens with yoghurt strains. International Journal of Biological Macromolecules 59: 377–383.
- Ismail B, Nampoothiri KM (2010) Production, purification and structural characterization of an exopolysaccharide produced by a probiotic Lactobacillus plantarum MTCC 9510. Archives of Microbiology 192(12): 1049–1057.
- Sasikumar K, Kozhummal Vaikkath D, Devendra L, Nampoothiri KM (2017) An exopolysaccharide (EPS) from a Lactobacillus plantarum BR2 with potential benefits for making functional foods. Bioresource Technology 241: 1152–1156.
- Wang J, Zhao X, Tian Z, Yang Y, Yang Z (2015) Characterization of an exopolysaccharide produced by Lactobacillus plantarum YW11 isolated from Tibet Kefir. Carbohydrate Polymers 125: 16–25.
- Ayyash M, Abu-Jdayil B, Olaimat A, Esposito G, Itsaranuwat P, et al. (2020) Physicochemical, bioactive and rheological properties of an exopolysaccharide produced by a probiotic Pediococcus pentosaceus M41. Carbohydrate Polymers 229: 115462.
- Krishnamurthy M, Jayaraman Uthaya C, Thangavel M, Annadurai V, Rajendran R, et al. (2020) Optimization, compositional analysis, and characterization of exopolysaccharides produced by multi-metal resistant Bacillus cereus KMS3-1. Carbohydrate Polymers 227: 115369.
- Yu L, Ye G, Qi X, Yang Y, Zhou B, et al. (2023) Purification, characterization and probiotic proliferation effect of exopolysaccharides produced by Lactiplantibacillus plantarum HDC-01 isolated from sauerkraut. Frontiers in Microbiology 14: 1210302.
- Lu X, Zhao Y, Wang C, Zhang H, Liu P (2024) Necessity of Different Lignocellulose on Exopolysaccharide Synthesis and Its Hypoglycemic Activity In Vitro of Inonotus obliquus. Applied Biochemistry and Biotechnology 196(6): 3420-3440.
- Saha S, Santra HK, Chattopadhyay S, Banerjee D (2024) Production of Exopolysaccharide from an Endophytic Fusarium sp. Glos2 and Documentation of its In vitro Antioxidative Potentialities. Journal of Pure and Applied Microbiology 18(4): 2911-2924.
- Saravanan C, Shetty PKH (2016) Isolation and characterization of exopolysaccharide from Leuconostoc lactis KC117496 isolated from idli batter. International Journal of Biological Macromolecules 90: 100–106.
- Srinivash M, Krishnamoorthi R, Mahalingam PU, Malaikozhundan B, Keerthivasan M (2023) Probiotic potential of exopolysaccharide producing lactic acid bacteria isolated from homemade fermented food products. Journal of Agriculture and Food Research 11: 100517.
- Pulsawat W, Leksawasdi N, Rogers PL, Foster LJR (2003) Anions effects on biosorption of Mn (II) by extracellular polymeric substance (EPS) from Rhizobium etli. Biotechnology Letters 25: 1267-1270.
- Saleh AK, El-Gendi H, Soliman NA, El-Zawawy WK, Abdel-Fattah YR (2022) Bioprocess development for bacterial cellulose biosynthesis by novel Lactiplantibacillus plantarum isolate along with characterization and antimicrobial assessment of fabricated membrane. Scientific Reports 12(1).
- Patel VH, Shah K, Patel FR (2023) Physical Characterization and Structure Elucidation of Exopolysaccharide from Marine Halotolerant Bacterium Enterobacter cloacae VHP-34. Industrial Biotechnology 19(6): 326-336.
- Marimuthu S, Rajendran K (2023) Structural and Functional Characterization of Exopolysaccharide Produced by a Novel Isolate Bacillus sp. EPS003. Applied Biochemistry and Biotechnology 195(7): 4583–4601.
- Cao C, Liu Y, Li Y, Zhang Y, Zhao Y, et al. (2020) Structural characterization and antioxidant potential of a novel exopolysaccharide produced by Bacillus velezensis SN-1 from spontaneously fermented Da-Jiang. Glycoconjugate Journal 37(3): 307–317.
- Jiang G, He J, Gan L, Li X, Xu Z, et al. (2022) Exopolysaccharide Produced by Pediococcus pentosaceus E8: Structure, Bio-Activities, and Its Potential Application. Frontiers in Microbiology 13: 923522.
- Du R, Yu L, Sun M, Ye G, Yang Y, et al. (2023) Characterization of Dextran Biosynthesized by Glucansucrase from Leuconostoc pseudomesenteroides and Their Potential Biotechnological Applications. Antioxidants 12(2).






























