Saliva Flow Rate and Caries-Related Micro Organisms of Obese Patients Before and 6 Weeks after Bariatric Surgery: A Pilot Study
Freja Freedman1, Richard Marsk2 and Lena Karlsson1*
1Department of Dental Medicine, Karolinska Institutet, Stockholm, Sweden
2Division of Surgery, Department of Clinical Sciences, Danderyds Hospital, Karolinska Institute, Stockholm, Sweden
Submission: August 13, 2023; Published: August 22, 2023
*Corresponding author: Lena Karlsson, Department of Dental Medicine, Karolinska Institute, Sweden
How to cite this article: Freja F, Richard M, Lena K. Saliva Flow Rate and Caries-Related Micro Organisms of Obese Patients Before and 6 Weeks after Bariatric Surgery: A Pilot Study. Adv Dent & Oral Health. 2023; 16(3): 555940. DOI: 10.19080/ADOH.2023.16.555940
Abstract
Objectives: Obesity is a worldwide health issue that is increasing in prevalence where bariatric surgery is the most effective treatment. The impact of bariatric surgery on oral health is largely unknown, and different postoperative side effects could have a negative impact on oral health with an increased risk of dental problems. The aim of this study was to investigate if saliva and caries-related microorganisms of obese patients are different before and after bariatric surgery.
Material and methods: Twenty patients (n=20) planned for bariatric surgery were included in the study; eight patients planned for a gastric sleeve procedure and twelve for a gastric bypass. Unstimulated saliva and stimulated saliva flow rate were sampled before surgery at the dietitian’s visit and after surgery at the six weeks follow up appointment. Caries-related microorganisms Streptococcus Mutans (S. Mutans) and Lactobacillus spp. were detected using a cultivation method.
Results: There was a significant decrease in unstimulated saliva flow rate, median value of 0.0133 ml/min, (P=0.010), of anaerobic Lactobacillus spp. (P= 0.002) and a borderline significant (P=0.05) of S. Mutans, after bariatric surgery.
Conclusion: Within the limitations of the present study, findings showed that obese patients having undergone weight loss surgery have a significant decrease of unstimulated saliva flow rate which could lead to impaired oral health.
Keywords: Bariatric Surgery; Oral health; Salivary Flow Rate, Streptococcus Mutans, Lactobacillus Spp.
Introduction
Obesity is a worldwide health issue increasing in prevalence. It has been estimated that 12% of the adults and 5% of the children in the world are obese and that these figures are increasing [1]. A Body Mass Index (BMI) equal to or larger than 30 kg/m² is considered to fall into the range of obesity. Obesity has been proven to have adverse effects on overall health but also to be a socioeconomic burden both for the individual and for society [2]. Type 2 diabetes (T2D), hyperlipidemia, hypertension, heart disease, obstructive sleep apnea, stroke and depression as well as many types of cancer are just some of the few conditions that are associated with obesity [3]. Bariatric surgery has been proven to have a significant effect on weight loss, resulting in, on average, 20-40 kg weight loss, depending on method of choice. The most common bariatric surgeries are gastric bypass, (Figure 1a) and sleeve gastrectomy, (Figure 1b). Both procedures work by altering levels of gut hormones and also by restricting the amount of food that can be consumed at a given time.
Bariatric surgery has also shown, in many cases, to result in total resolution of the comorbid diseases associated with obesity and an overall significantly enhanced life quality [4]. However, the impact of bariatric surgery on oral health is largely unknown, and different postoperative side effects could have a negative impact on oral health with an increased risk of dental problems. The chronic pathological condition dental caries, characterized by the demineralization of the teeth due to cariogenic microorganisms such as Streptococcus Mutans (S. Mutans) and Lactobacillus spp., carbohydrate diet and dental plaque is one of the biggest challenges in dental care. Post-surgery, the ability of eating larger meals is greatly reduced. Thus, the patients are instructed to eat smaller meals 6-8 times a day, avoid drinking in conjunction with food intake and rather drinking in between meals. A diet consumption pattern of frequent intake of food and drinks is a well-known potential risk factor and may contribute to a higher risk for dental caries [5-7]. However, it is hard to determine the relationship between caries and obesity, whether it’s due to oral hygiene, diet, nutrition, saliva or genetics [8]. Another potential side effect of bariatric surgery is hyposalivation [9,10,5]. This fact, along with postoperative dietary changes, makes these patients vulnerable to a multitude of potential complications. A study made by Greenway and Greenway [11] showed a relation between post-operative bariatric surgery patients and the increase of root surface caries. The authors stated that this might be caused by the reduced saliva flow and buffering capacity. Knaś et al. [12] measured the salivary flow rate, stimulated and unstimulated, in obese patients about to undergo bariatric surgery.
Before the surgery a reduced stimulated and unstimulated salivary flow was observed in obese patients compared to control group with a normal BMI. Six months after the surgery, only the unstimulated salivary flow was restored to normal values and the stimulated salivary flow rate was still significantly reduced. Hashizume et al. [13] evaluated S. Mutans and actobacillus spp. present in obese patient before and after bariatric surgery and found that S. Mutans was increased at 6 months after bariatric surgery, while there was no significant increase in Lactobacillus spp. Because the number of bariatric surgeries has increased extensively in recent years, a question has been raised about whether changes in oral health condition after bariatric surgery or unsatisfactory oral health conditions before bariatric surgery might worsen after the surgery. The aim of the present study was therefore to investigate if saliva and caries-related microorganisms of obese patients is different before and after bariatric surgery and test the hypothesis that the salivary and caries-related microorganisms of obese patients undergoing surgery will be similar to those before surgery.
Materials and Methods
Study design
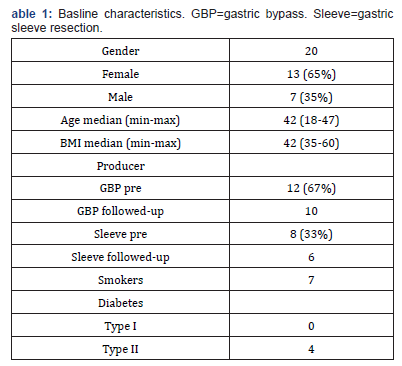
Twenty patients (n=20) planned for bariatric surgery were included to take part in this prospective non-randomized clinical study; eight patients planned for a gastric sleeve procedure and twelve patients for a gastric bypass, (Table 1). The Ethics Committee in Stockholm, Sweden (2019-01232) approved the study. The declaration of World Medical Association of Helsinki [14] as well as human rights, has been taken into consideration throughout the study. The authors have no conflict of interest to declare.
Study group
The study participants were consecutively recruited from Danderyd’s Hospital, Sweden, during the period 8th of May until 15th of November 2020, where they had been scheduled and qualified for surgery according to national guidelines for bariatric surgery (BMI>35 and comorbid disease, age >18 years, non-pregnancy, previous serious attempts of weight loss by conservative measures). Patients were given oral and written information before giving their consent at their preoperative first visit to the dietitians. Signatures for written consent were then collected at their visit at the dietitians before surgery.
Salivary and bacterial test
The collection of unstimulated saliva and stimulated saliva was performed before surgery at the dietitian’s visit and after surgery at the 6 week follow up appointment. Unstimulated and stimulated saliva was collected between 10am and 4pm, at least one hour after fasting using a sialometry test supervised during the procedure by author FF. Unstimulated saliva was collected in a private room where the patients were instructed to sit comfortably with the head slightly tilted in a forward position. The patient was instructed, after an initial swallowing action, to passively let the saliva drool from the lower lip without any masticatory or gustatory stimulus and into a plastic container for 15 minutes. The stimulated saliva secretion was thereafter collected including the same steps described and by letting the patient chew on a piece of paraffin tablet frequently until it became soft, producing as much saliva as possible, spitting it into a plastic container for 5 minutes. The volume of the unstimulated and stimulated saliva was measured by a 10 ml syringe and then transferred to a 20 ml test tube for each sample. Flowrate was calculated by dividing saliva volume by time of secretion and expressed as milliliters per minute.
One milliliter of the stimulated saliva was thereafter taken from the test tube and transferred to a small glass vial containing VMG-II solution, a transport medium to stabilize and preserve the oral microbiome composition. Directly after sampling, the saliva was stored in a -18°C freezer at Danderyd’s Hospital for future analysis. The frozen VMG-II tubes with stimulated saliva were sent to the department of microbiology at Karolinska Institutet where cultures of S. Mutans and Lactobacillus spp. were analyzed. Sample serial dilutions from 10−1 to 10−4 in PBS were distributed on to the plates and incubated for 48h at 37°C anaerobically. Selection media for S. Mutans and anaerobic Lactobacilli species were Mitis Salivarius Bacitracin (MSB)-agar, Rogosa agar, respectively. After cultivation on respective selective medium, the colonies fulfilling the colony morphology for each species were counted. Result given as CFU/mL saliva was calculated using the equation: No. of colonies x volume inoculated x the dilution factor x (sample volume divided by total sample volume (VMGII medium + saliva). All CFU/ml <150 was undetected and therefore counted as equal to null.
Statistical methods and analysis
All data was analysed using the Wilcoxon Signed Rank Test for categorical data and paired t-test for normally distributed numerical data. Statistical calculations were processed using Sigmaplot™. (Systat software inc., San Jose, Ca, USA). Means and t-tests were used when normal distribution was ascertained by the statistical software. P-value < 0.05 was considered statistically significant.
Results
A total of 16 patients fulfilled the study. Three patients were excluded from analysis because of the time restrictions of the study where their surgeries were planned after the end-ofinclusion, but had their saliva collected six weeks after surgery for future studies. One patient was excluded due to the decision to decline further participation in the present study post-surgery. All patients except one, that had his oral diabetic medication removed after surgery, had the same medication before and 6 weeks after bariatric surgery.
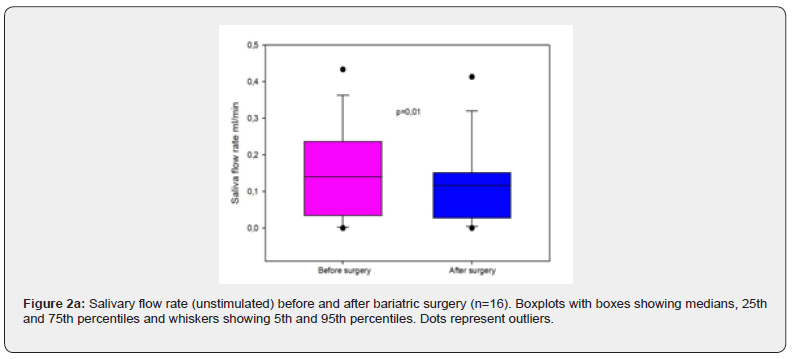
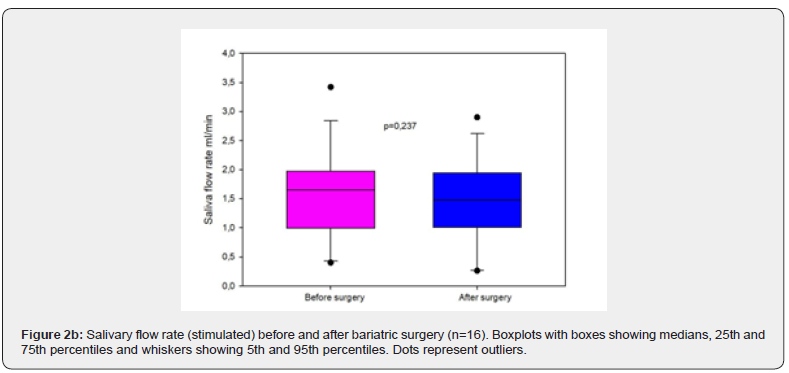
The results showed a significant decrease in unstimulated saliva flow rate after bariatric surgery (P=0.010), (Figure 2a). Changes in stimulated saliva after bariatric surgery could not exclude the possibility that the difference is due to chance (P=0.237), (Figure 2b). When comparing types of surgery, gastric sleeve and gastric bypass, the differences in stimulated and unstimulated saliva flow rate was not significant. There was a significant difference in numbers of anaerobic Lactobacillus spp. after bariatric surgery (P= 0.002), (Figure 3a). When comparing S. Mutans in stimulated saliva there was a borderline significant difference (P=0.05), shown in Figure 3b.
Discussion
The present study was undertaken to investigate the possible changes in salivary flow rate and caries-related microorganisms in obese patients before and after bariatric surgery. Our main findings were a significant decrease in unstimulated saliva flow rate and a significant decrease in numbers of Lactobacillus spp., post-surgery. Saliva has many protective functions for dental hard and soft tissues and contains many important substances, including electrolytes, mucus, antibacterial compounds, and various enzymes. A reduction in saliva flow rate is often accompanied by major changes in saliva composition, resulting in increased susceptibility to oral infections such as dental caries [5]. The impact of dry mouth and its complication on quality of life is also documented [15]. Some of the side effects after bariatric surgery are malabsorption, dehydration, and vomiting [16], which may be contributing factors to a dry mouth [17].


This corresponds very well to Netto et al. [18] findings which showed a high number of patients, 2-years after bariatric surgery, with hyposalivation and a disease development in the oral cavity. A potential explanation to the reduced saliva flow rate postsurgery is that the exocrine function of the saliva gland is partially regulated by hormones, but also the mechanical stimulation by chewing, salivation. When undergoing bariatric surgery, the hormonal activity is affected, partially by the lower account of hormonal receptors in the gut which could lead to reduced saliva flow rate. Bariatric surgery leads to weight loss by the reduced stomach area by restricting the patient from overeating, this will cause signals to the brain that the stomach is full and end of the salivation [19,20].
This is a wanted result in the aspect of weight loss but could lead to changes in the oral environment and impaired oral health. However, the material available about salivary flow rate and post bariatric surgery is limited, sometimes contradictory and to the best of our knowledge, material where mostly stimulated saliva flow rate has been investigated. Gambus et al. [21] found no difference in stimulated saliva flow rate when evaluating the sialometric changes in morbidly obese patients before and after bariatric surgery. Marsicano et al. [7] studied stimulated salivary flow in bariatric patients before, and up to after 3 months and 6 months post-surgery.
In this study, saliva flow was reduced in patients before surgery and increased after surgery. The authors reported that the use of medications for hypertension, diabetes, hyperlipidemia, and antidepressants decreased significantly after only 3 months of bariatric surgery, and the reduction in the use of medication most likely influenced the improvement in stimulated salivary flow. Marsicano et al. [22] reported in a later published study that obese patients and operated patients had a lower stimulated salivary flow rate, regardless of surgery. Other studies [23] also show that as the BMI increases, in individuals not having bariatric surgery, the saliva flow decreases and is explained as a consequence of medication and the nutrition deficit induced by a diet rich in simple carbohydrates and fat.
Questions have been raised whether changes in salivary flow rate is due to obesity and unsatisfactory general health conditions, to bariatric surgery and the significant weight loss with possible malabsorption and dehydration as a side effect, and/or to the health benefits of bariatric surgery with a reduction in the use of medication and comorbid diseases associated to obesity. Within the limitation of the present study with a small patient cohort (n=16) and saliva collection procedures taken at various time points on the day, even though efforts were taken to schedule the sampling at the same time of day whenever it was possible, we found a significant decrease in unstimulated saliva flow rate which warrant further investigation.
The present study also investigated the possible changes in caries-related microorganisms in obese patients before and after bariatric surgery. Numbers of studies [24-28] have shown that caries is associated with increases in the proportions of acidogenic (acid-producer) and aciduric (acid-tolerating) bacteria, especially S. Mutans and Lactobacillus spp. However, there is scarce research investigating the possible changes of caries-related microorganisms, before and after bariatric surgery. We found, in the present study, a borderline significant difference with decreased level of S. Mutans post-surgery. As a comparison, the only study [19] found in the literature that evaluated stimulated salivary and microbiological condition of morbidly obese patients (n=27) undergoing bariatric surgery, reported that S. Mutans increased post-surgery.
The limited number of participants could possibly explain the contradictory result found in our study, whereas reduced salivary flow is often accompanied by a concomitant increase on cariogenic microorganisms in the oral cavity. Another weakness of the present study was that only two bacteria types were analyzed; S. Mutans and Lactobacillus spp., which have been associated with dental caries for over a century. New insights and OMICS approaches [29] emphasizes that some long-held caries paradigms should be revised, since the identification of previously unknown bacterial genera/species appears to be associated with dental caries.
Conclusion
To the best of the authors’ knowledge our study is the first investigating both unstimulated- and stimulated salivary flow rate and caries-related microorganisms before and after bariatric surgery. We did find differences regarding these variables and our hypothesis was therefore rejected. These findings justify the need for the present and further studies. Our results also indicate a need for interdisciplinary collaboration between medical and dental staff to establish preventive dental protocols and promote oral health in patients undergoing bariatric surgery.
References
- GBD 2015 (2017) Obesity Collaborators, Afshin A, Forouzanfar MH, Reitsma MB, Sur P, Estep K, et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N Engl J Med 377(1): 13-27.
- Tremmel M, Gerdtham UG, Nilsson PM, Saha S (2017) Economic Burden of Obesity: A Systematic Literature Review. Int J Environ Res Public Health 14(4): 435.
- Cummings S, Pratt J (2015) Metabolic and bariatric surgery: Nutrition and dental considerations. J Am Dent Assoc146(10): 767-772.
- Köhler H, Dorozhkina R, Gruner-Labitzke K, de Zwaan M (2020) Specific Health Knowledge and Health Literacy of Patients before and after Bariatric Surgery: A Cross-Sectional Study. Obes Facts 13(2): 166-178.
- Hague AL, Baechle M (2008) Advanced caries in a patient with a history of bariatric surgery. J Dent Hyg 82(2): 22.
- Jastrzębska-Mierzyńska M, Ostrowska L, Wasiluk D, Konarzewska-Duchnowska E (2015) Dietetic recommendations after bariatric procedures in the light of the new guidelines regarding metabolic and bariatric surgery. Rocz Panstw Zakl Hig 66(1): 13-9.
- Marsicano JA, Grec PG de M, Belarmino LB, Ceneviva R, Peres SH de CS (2011) Interfaces between bariatric surgery and oral health: a longitudinal survey. Acta Cir Bras 26(Suppl2):79-83.
- Słotwińska SM, Słotwiński R (2015) Host response, obesity, and oral health. Cent Eur J Immunol 40(2): 201-205.
- Shikora SA, Kim JJ, Tarnoff ME (2007) Nutrition and gastrointestinal complications of bariatric surgery. Nutr Clin Pract 22(1): 29-40.
- Karlsson L, Carlsson J, Jenneborg K, Kjaeldgaard M (2018) Perceived oral health in patients after bariatric surgery using oral health-related quality of life measures. Clin Exp Dent Res 4(6): 230-240.
- Greenway SE, Greenway FL (2000) Root surface caries: a complication of the jejunoileal bypass. Obes Surg 10(1): 33-36.
- Knaś M, Maciejczyk M, Sawicka K, Hady HR, Niczyporuk M, et al. (2016) Impact of morbid obesity and bariatric surgery on antioxidant/oxidant balance of the unstimulated and stimulated human saliva. J Oral Pathol Med 45(6): 455-464.
- Hashizume LN, Bastos LF, Cardozo DD, Hilgert JB, Hugo FN, et al. (2015) Impact of Bariatric Surgery on the Saliva of Patients with Morbid Obesity. Obes Surg 25(8): 1550-1555.
- Shrestha B, Dunn (2020) The Declaration of Helsinki on Medical Research involving Human Subjects: A Review of Seventh Revision. L.J Nepal Health Res Counc 17(4): 548-552.
- Flink H, Tegelberg Å, Arnetz JE, Birkhed D (2020) Self-reported oral and general health related to xerostomia, hyposalivation, and quality of life among caries active younger adults. Acta Odontol Scand 78(3): 229-235.
- De Moura-Grec PG, Yamashita JM, Marsicano JA, Ceneviva R, de Souza Leite CV, et al. (2014) Impact of bariatric surgery on oral health conditions: 6-months cohort study. Int Dent J 64(3): 144-149.
- Heling I, Sgan-Cohen HD, Itzhaki M, Beglaibter N, Avrutis O, et al. (2006) Dental complications following gastric restrictive bariatric surgery. Obes Surg 16(9): 1131-1134.
- Netto BDM, Moreira EAM, Patiño JSR, Benincá JP, Jordão AA, et al. (2012) Influence of Roux-en-Y gastric bypass surgery on vitamin C, myeloperoxidase, and oral clinical manifestations: a 2-year follow-up study. Nutr Clin Pract 27(1): 114-121.
- Holst JJ, Madsbad S, Bojsen-Møller KN, Svane MS, Jørgensen NB, et al. (2018) Mechanisms in bariatric surgery: Gut hormones, diabetes resolution, and weight loss. Surg Obes Relat Dis 14(5): 708-714.
- Dimitriadis GK, Randeva MS, Miras AD (2017) Potential Hormone Mechanisms of Bariatric Surgery. Current Obesity Reports 6(3): 253-265.
- Gambus LC, Johann AC, Bettega PV, Rosa EA, Morimoto IM, et al. (2018) Sialometric and Sialochemical Changes in Morbidly Obese Patients before and after Bariatric Surgery. J Contemp Dent Pract 19(7): 756-761.
- Marsicano JA, Sales-Peres A, Ceneviva R, de C Sales-Peres SH (2012) Evaluation of oral health status and salivary flow rate in obese patients after bariatric surgery. Eur J Dent 6(2): 191-197.
- Flink H, Bergdahl M, Tegelberg A, Rosenblad A, Lagerlöf F (2008) Prevalence of hyposalivation in relation to general health, body mass index and remaining teeth in different age groups of adults. Community Dent Oral Epidemiol 36(6): 523-531.
- Selwitz RH, Ismail AI, Pitts NB (2009) Dental caries The Lancet 369(9555): 51-59.
- Pannu P, Gambhir R, Sujlana A (2013) Correlation between the salivary Streptococcus mutans levels and dental caries experience in adult population of Chandigarh, India [Internet]. European Journal of Dentistry 7(2): 191-195.
- Renke W (2016) Saliva and Dental Caries. Dental Caries p. 59-69.
- Salonen L, Allander L, Bratthall D, Hellden L (1990) Mutans Streptococci, Oral Hygiene, and Caries in an Adult Swedish Population. Journal of Dental Research 69(8): 1469-1475.
- Marsh PD (2003) Are dental diseases examples of ecological catastrophes? Microbiology 149(Pt 2): 279-294.
- Nascimento MM, Zaura E, Mira A, Takahashi N, Ten Cate JM (2017) Second Era of OMICS in Caries Research: Moving Past the Phase of Disillusionment, Journal of Dental Research 96(7): 733-740.






























