A Study of Elective or Therapeutic Neck Dissection in Oral Squamous Cell Carcinoma with N0 neck
Tadahide Noguchi1*, Akihiro Dohi1, Yasushi Sugiura1, Hirosaka Hayashi1, Manabu Yamada1,2, Natsuko Hichijo1 and Yoshiyuki Mori1
1Department of Dentistry, Oral and Maxillofacial Surgery, Jichi Medical University, Yakushiji, Shimotsuke-shi, Tochigi, Japan
2Department of Dentistry, Oral and Maxillofacial Surgery, National Hospital Tochigi Hospital, Nakatomatsuri, Utsunomia, Tochigi, Japan
Submission: March 08, 2023; Published: March 20, 2023
*Corresponding author: Tadahide Noguchi, Department of Dentistry, Oral and Maxillofacial Surgery, Jichi Medical University, Yakushiji, Shimotsuke-shi, Tochigi, Japan
How to cite this article: Tadahide Noguchi, Akihiro Dohi, Yasushi Sugiura, Hirosaka Hayashi, Manabu Yamada, et al. A Study of Elective or Therapeutic Neck Dissection in Oral Squamous Cell Carcinoma with N0 neck. Adv Dent & Oral Health. 2023; 16(1): 555929. DOI: 10.19080/ADOH.2023.16.555929
Abstract
Although there are many reports on the propriety of elective neck dissection in the initial treatment of N0 cases, the results are inconsistent. In this study, we report the significance of elective neck dissection in N0 cases of oral squamous cell carcinoma (OSCC) treated at our department. We included 225 patients who underwent initial surgical treatment for N0 cases of OSCC in our department for 10 years. Of the 225 patients, 88 underwent elective neck dissection including primary tumor resection (elective surgery group), and 137 underwent primary tumor resection alone (therapeutic surgery group). The elective surgery group included 2, 35, 11, and 40 cases with T1, T2, T3, and T4 stages, respectively. The therapeutic surgery group included 64, 68, 2, and 3 cases with T1, T2, T3, and T4 stages, respectively. In the elective surgery group, 32 patients had cervical lymph node histopathological metastasis. In the therapeutic surgery group, 40 patients had subsequent cervical lymph node metastasis. Postoperative adjuvant treatment was performed in 14 and 22 patients in the elective and therapeutic surgery groups, respectively. The 5-year survival rates were 80.7% and 83.9% in the elective and therapeutic surgery groups, respectively, with no significant difference between the groups. This result suggests that there is no difference in prognosis if early detection and treatment of subsequent cervical lymph node metastasis is performed with appropriate follow-up. However, there is still a need to consider the indication for elective neck dissection.
Keywords: Oral squamous cell carcinoma; Elective neck dissection; Therapeutic neck dissection; Occult lymph node metastasis
Keywords: OSCC: Oral squamous cell carcinoma; CT: Computed tomography; FDG-PET: fluorodeoxyglucose positron emission tomography; DOI: Depth of invasion
Introduction
In the treatment of oral squamous cell carcinoma (OSCC), control of the primary tumor and cervical lymph node metastasis are major factors that influence prognosis [1-3]. Although there are many reports regarding the presence or absence of elective neck dissection in the initial treatment of N0 cases exist, the results are not uniform [4-7]. In this study, we report on the significance of elective neck dissection in N0 cases of OSCC treated at our department.
Material and Methods
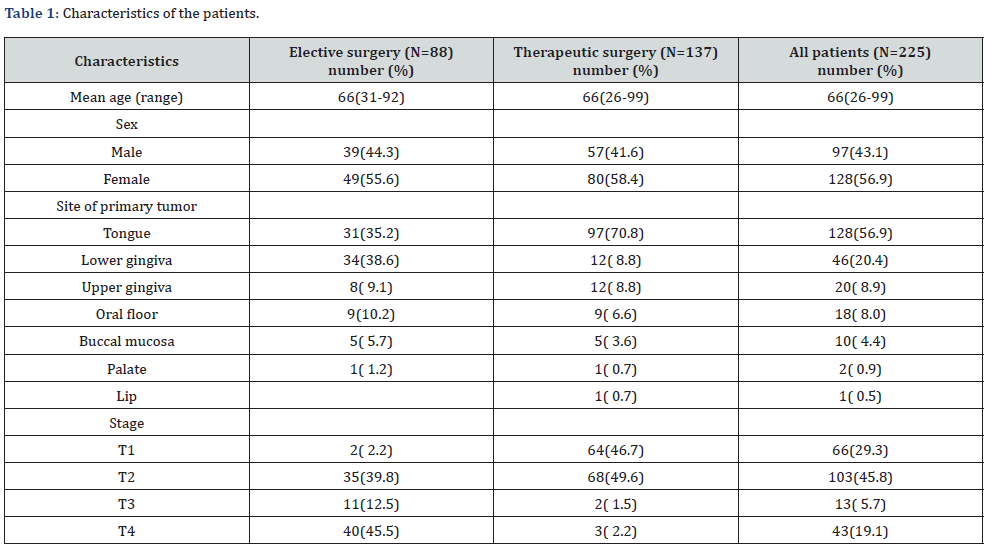
We included 225 cases of OSCC without cervical lymph node metastasis (clinical N0 cases) who underwent surgical treatment at our department between April 2009 and March 2019. Cervical lymph node metastasis was diagnosed by palpation, Computed tomography (CT), MR (Magnetic resonance), ultrasound, and fluorodeoxyglucose positron emission tomography (FDG-PET) examination. We excluded patients who underwent preoperative treatment with chemotherapy or radiotherapy. The selection of postoperative adjuvant therapy was based on the NCCN guidelines. Combined chemoradiotherapy was performed for high-risk factors for recurrence and metastasis (positive margins or extranodal extension: ENE), and intermediate-risk factors (pT4, pN2/3, perineural invasion, venous invasion, and lymphatic invasion) and radiotherapy alone were selected. There were 97 men (43.1%) and 128 women (56.9%), aged 26 to 99 years, with an average age of 66.0 years. There were 128 cases of tongue, 46 cases of mandibular gingiva, 20 cases of maxillary gingiva, 18 cases of oral floor, 10 cases of buccal mucosa, 2 cases of palate, and 1 case of lip involvement. In the T classification, T1 was 66, T2 were 103, T3 were 13, and T4 were 43 (Table 1).
Results
Of the 225 cases, 88 underwent elective neck dissection including primary tumor resection (elective surgery group), and 137 underwent primary tumor resection only (therapeutic surgery group) (Table 2). There were 88 patients (39.1%) in the elective surgery group, including 31 tongue, 34 mandibular gingiva, 8 maxillary gingiva, 9 oral floor, 5 buccal mucosa, and 1 palate locations. In the T classification, T1 was found in 2 cases (2.2%), T2 in 35 cases (39.8%), T3 in 11 cases (12.5%), and T4 in 40 cases (45.5%). There were 137 cases (60.9%) in the therapeutic surgery group, including 97 cases of tongue, 12 cases of mandibular gingiva, 12 cases of maxillary gingiva, 9 cases of oral floor, 5 cases of buccal mucosa, and 1 case each of palate and lip involvement. In the T classification, T1 was found in 64 cases (46.7%), T2 in 68 cases (49.6%), T3 in 2 cases (1.5%), and T4 in 3 cases (2.2%) (Table 1).

*pN: pathological neck (cervical lymph node)
There were 32 cases (36.4%) of cervical lymph node metastasis in the elective surgery group and 40 cases (29.2%) in the therapeutic group (137 cases), the results were higher in the elective surgery group (Table 2).
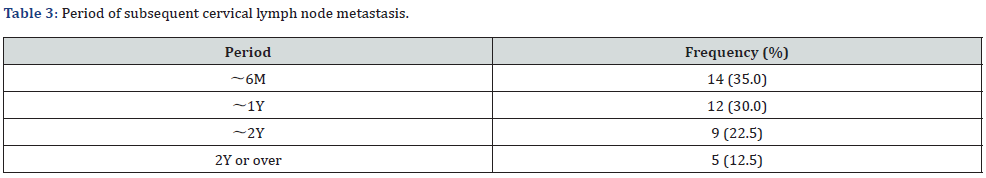
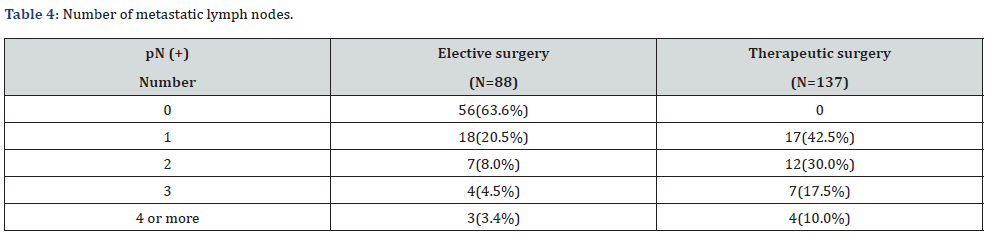
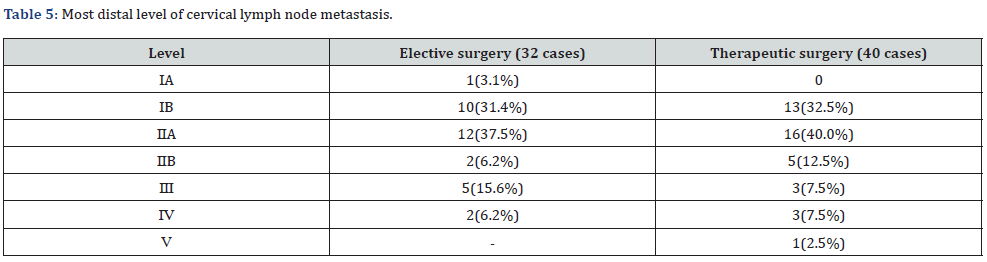
The period from primary tumor resection to subsequent cervical lymph node metastasis was < 6 months in 14 cases (35.0%), 6 months to < 1 year in 12 cases (30.0%), 1 year to < 2 years in 9 cases (22.5%), 2 years or more in 5 cases (12.5%), and 87.5% for 2 years or less (Table 3). In the elective surgery group, the number of metastatic lymph nodes were: 18 cases (20.5%) had 1, 7 cases (8.0%) had 2, 4 cases (4.5%) had 3, and 3 cases (3.4%) had 4 or more. In contrast, in the therapeutic surgery group (40 cases), 17 cases (42.5%) had 1, 12 (30.0%) had 2, 7 (17.5%) had 3, and 4 (10.0%) had 4 or more (Table 4). The most distal level of lymph node metastasis in each group was IIA region in 12 cases (37.5%), followed by IB in 10 cases (31.4%) in the elective surgery group, and IIA region in 16 cases (40.0%) and IB region in 13 cases (32.5%) in primary therapeutic surgery group. In both groups, IB and IIA accounted for about 70% (Table 5).
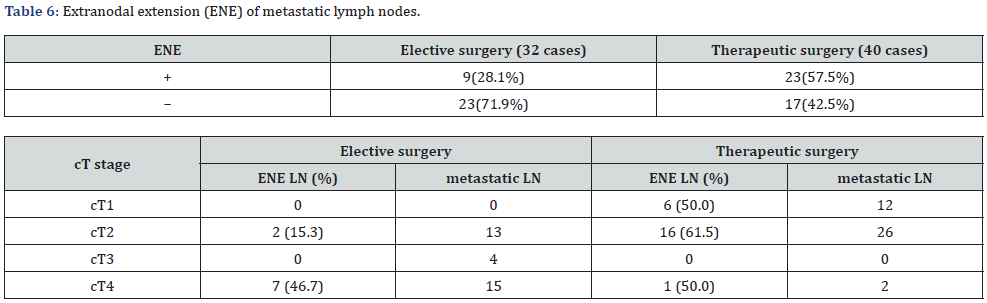
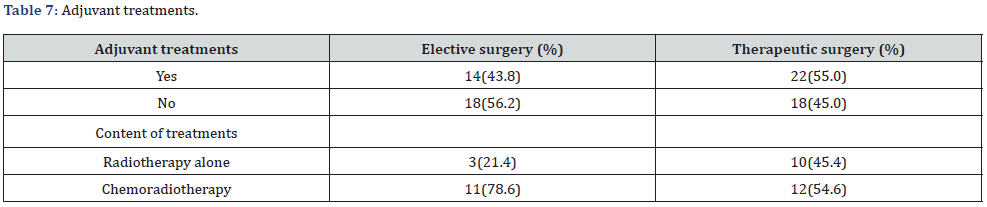
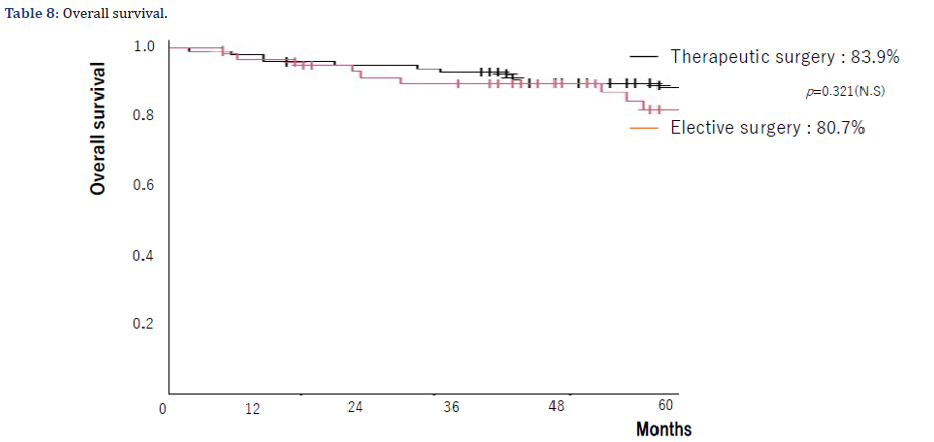
The rate of ENE was higher in the therapeutic surgery group (57.5%) than in the elective surgery group (28.1%). When examined by T classification, the ENE was observed in 2 of the 13 T2 (15.3%) and 7 of the 15 T4(46.7%) patients. The elective surgery group had a higher rate of ENE in advanced cases, whereas the therapeutic surgery group had a higher rate of ENE in all T classifications of 50% or more (Table 6). Regarding the presence or absence of postoperative adjuvant therapy, in the elective surgery group, 14 of the 32 patients with lymph node metastasis (43.8%) received postoperative adjuvant therapy, of which 11 (78.6%) received chemoradiotherapy. Three patients (21.4%) underwent RT alone. In the therapeutic surgery group, 22 of 40 (55%) patients with subsequent lymph node metastasis underwent adjuvant treatment. Twelve patients (54.6%) received chemoradiotherapy and 10 patients (45.4%) received radiotherapy alone. A majority of patients in both groups received adjuvant therapy; however, the elective surgery group had a higher rate of combined chemoradiotherapy (Table 7). Regarding prognosis, 7 of the 32 cases with cervical lymph node metastasis in the elective surgery group died from the primary disease and 1 died from other diseases, resulting in an OS rate of 80.7%. There was 1 case of neck recurrence. Of the 40 patients with cervical lymph node metastasis in the therapeutic surgery group, 6 died from primary disease and 2 died from other diseases, resulting in an OS of 83.9%. There were 2 cases of neck recurrence. The survival rate was slightly higher in the therapeutic surgery group; however, the difference was not statistically significant (p= 0.321) (Table 8).
Discussion
OSSC tends to metastasize to regional lymph nodes, and the presence or absence of lymph node metastasis is an independent poor prognostic factor [1-3]. If the tumor is locally confined, the survival rate is 80% or more, and the prognosis is relatively good. However, if the tumor spreads to the regional lymph nodes, the survival rate can drop to < 50% [8]. Reports of occult cervical lymph node metastasis vary among institutions; however, in many institutions the incidence of occult lymph node metastasis is relatively high, despite improvements in diagnostic imaging accuracy. The occult cervical lymph node metastasis rate is approximately 20-40% among institutions [9-17]. Some reports recommend elective neck dissection in early stages to control occult lymph node metastasis; however, neck dissection is unnecessary for approximately 70% of patients without occult lymph node metastasis, and because of the disadvantages of the aftereffects of the surgery (cosmetic problems due to incisions, facial nerve palsy, sensory disturbance and stiffness in the neck). In addition, it has been reported that if the primary tumor is resected and strict follow-up is performed, salvage is possible by performing neck dissection when cervical lymph node metastasis becomes clear [18]. During follow-up, occult lymph node metastasis may be found in an advanced state with extranodal extension; the prognosis might be poor. However, at present, there are no clear criteria or unified opinions regarding the indications for neck dissection in N0 cases, and the choice is left to each facility [4-7].
Randomized controlled trials have been conducted regarding elective neck dissection. However, there are problems such as difference in treatment methods for each facility and low diagnostic accuracy as postoperative CT imaging are not performed [7,15,19,20]. In 2015, D’Cruz et al. [21] carried out a large-scale randomized controlled trial in 469 T1-2 oral cancer cases and reported that of the two groups (elective versus therapeutic neck dissection) examined, the elective dissection group showed a significantly higher overall survival rate. However, subsequent lymph node metastasis occurred in 45.1% of the primary lesion resection group (therapeutic surgery group), of which 17.5% were judged as inoperable. In addition, ENE was found in 93% of the cases of subsequent lymph node metastasis that underwent neck dissection, and the rescue rate of the subsequent metastasis was reported to be as low as 48%. Therefore, because of these results, D’ Cruz et al. recommend elective neck dissection, although it is questionable whether this can be applied clinically in Japan. In Japan, a large-scale randomized controlled trial is ongoing in the fields of head and neck surgery and oral surgery, and results are expected.
Our results showed that occult neck lymph node metastasis was 36.4% in the elective surgery group and 29.2% in the therapeutic surgery group. Various modalities such as CT, MR, neck echo, and FDG-PET were used before surgery; however, the incidence of occult neck lymph node metastasis was relatively high. Occult neck cervical lymph node metastasis was higher in the elective surgery group, reflecting the fact that 19 (59%) of the 32 cases of neck metastasis in the elective surgery group were T3 and T4 cases. Regarding ENE of cervical lymph node metastasis, there were 9 of 32 patients (28.1%) in the elective surgery group and 23 of 40 patients (57.5%) in the therapeutic surgery group. It was suggested that this result might be related to the time lag from primary lesion resection to detection of subsequent cervical lymph node metastasis. If the detection time of occult lymph node metastasis is long, it might progress to ENE and the prognosis will be poor.
In this clinical study, there was no significant difference in the survival rate when the prognosis was compared between the elective surgery group and the therapeutic surgery group. This result suggests that salvage is possible (there is no difference in prognosis) if therapeutic neck dissection is performed when subsequent cervical lymph node metastasis becomes apparent after resection of the primary tumor. Therefore, elective neck dissection may not be necessary if strict follow-up using various modalities is possible. However, the incidence of ENE is higher in subsequent cervical lymph node metastasis, and postoperative adjuvant treatment is more indicated, leading to a burden on patients and a decrease in QOL. Therefore, it is necessary to examine the determinants of Depth of invasion (DOI), mode of invasion, and degree of differentiation the presence or absence of desmoplastic pattern, and genetic analysis in biopsy tissue in primary tumor. This will help in actively performing elective neck dissection. However, there is still a need to consider the indication for elective neck dissection.
Conclusion
In this clinical study, there was no significant difference in the survival rate when the prognosis was compared between the elective and therapeutic surgery groups. However, in actual clinical practice, the decision to perform elective neck dissection is made considering various conditions of the primary tumor, presence or absence of reconstruction, patient’s wishes, and clinician’s experience. A large-scale randomized controlled trial is ongoing in Japan, and we expect that the results will provide further evidence.
References
- Okura M, Aikawa T, Sawai NY, Iida S, Kogo M (2009) Decision analysis and treatment threshold in a management for the N0 neck of the oral cavity carcinoma. Oral Oncology 45(10): 908-911.
- Capote A, Escorial V, Muñoz-Guerra MF, Rodríguez-Campo FJ, Gamallo C, et al. (2007) Elective neck dissection early-stage oral squamous cell carcinoma– Does it influence recurrence and survival? Head Neck 29(1): 3-11.
- Kumar T, Patel MD (2013) Pattern of lymphatic metastasis in relation to the depth of tumor in oral tongue cancers: A clinicopathological correlation. Indian J Otolaryngol Head Neck Surg 65(Suppl 1): 59-63.
- Hakeem AH, Pradhan SA, Kannan R, Tubachi J (2016) Clinical outcome of surgical treatment of T1-2 N0 squamous ccell carcinoma of oral tongue with observation for the neck: Analysis of 176 cases. Ann Maxillofac Surg 6(2): 235-240.
- Haddadin KJ, Soutar DS, Oliver RJ, Webster MH, Robertson AG, et al. (1999) Improved survival for patients clinically T1/T2, N0 tongue tumors undergoing a prophylactic neck dissection. Head Neck 21(6): 517-525.
- Keski-Säntti H, Atula T, Törnwall J, Koivunen P, Mäkitie A (2006) Elective neck treatment versus observation in patients with T1/T2 N0 squamous cell carcinoma of oral tongue. Oral Oncol 42(1): 96-101.
- Yuen APW, Ho CM, Chow TL, Ng RW, Wei WI, et al. (2009) Prospective randomized study of selective neck dissection versus observation for N0 neck of early tongue carcinoma. Head Neck 31(6): 765-772.
- Grandi C, Alloisio M, Moglia D, Podecca S, Sala L, et al. (1985) Prognostic significance of lymphatic spread in head and neck carcinomas: therapeutic implications. Head neck Surg 8(6): 67-73.
- Dias FL, Kligerman J, Matos de Sá G, Arcuri RA, Freitas EQ, et al. (2001) Elective neck dissection versus observation in stage I squamous cell carcinomas of the tongue and floor of the mouth. Otolaryngol Head Neck Surg 125(1): 23-29.
- Ferlito A, Rinaldo A, Silver CE, Gourin CG, Shah JP, et al. (2006) n EM, Pellitteri PK, Werner JA, Myers EN: Elective and therapeutic selective neck dissection. Oral Oncol 42(1): 14-25.
- Franceschi D, Gupta R, Spiro RH, Shah JP (1993) Improved survival in the treatment of squamous carcinoma of the oral tongue. Am J Surg 166(4): 360-365.
- Gourin CG, Conger BT, Porubsky ES, Sheils WC, Bilodeau PA, et al. (2008) The effect of occult nodal metastases on survival and regional control in patients with head and neck squamous cell carcinoma. Laryngo scope 118(7): 1191-1194.
- Huang SF, Kang CJ, Lin CY, Fan KH, Yen TC, et al. (2008) Neck treatment of patients with early stage oral tongue cancer: comparison between observation, supraomohyoid dissection, and extended dissection. Cancer 112(5): 1066-1075.
- Kaya S, Yilmaz T, Gürsel B, Saraç S, Sennaroglu L (2001) The value of elective neck dissection in treatment of cancer of the tongue. Am J Otolaryngol 22(1): 59-64.
- Kligerman J, Lima RA, Soares JR, Prado L, Dias FL, et al. (1994) Supraomohyoid neck dissection in the treatment of T1/T2 squamous cell carcinoma of oral cavity. Am J Surg 168(5): 391-394.
- Mira E, Benazzo M, Rossi V, Zanoletti E (2002) Efficacy of selective lymph node dissection in clinically negative neck. Otolaryngol Head Neck Surg 127(4): 279-283.
- Stuckensen T, Kovács AF, Adams S, Baum RP (2000) Staging of the neck in patients with oral cavity squamous cell carcinomas: a prospective comparison of PET, ultrasound, CT and MRI. J Craniomaxillofac Surg 28(6): 319- 324.
- Hirashima Soichi, Yoshioka Izumi, Habu Manabu, Furuta Norihiko, Kokuryo Shinya, et al. (2009) Evaluation of prophylactic neck dissection in Stage I and II oral squamous cell carcinoma. J Jpn Soc Oral Oncology 55: 622-628.
- Hanai N, Asakage T, Kiyota N, Homma A, Hayashi R (2019) Comtroversies in relation to neck management in N0 early oral tongue cancer. Jpn J Clin Oncol 49(4): 297-305.
- Fakih AR, Rao RS, Borges AM, Patel AR (1989) Elective versus therapeutic neck dissection in early carcinoma of the oral tongue. Am J Surg 158(4): 309-313.
- D'Cruz AK, Vaish R, Kapre N, Dandekar M, Gupta S, et al. (2015) Elective versus Therapeutic Neck Dissection in Node-Negative Oral Cancer. N Engl Med 373(6): 521-529.






























