Effectiveness of Acanthopanax Senticosus Harms in a Mouse Model of Nickel Allergy
Dai Tsuchida1, Tadahide Noguchi2*, Maki Tanaka3, Yusuke Takahashi3, Masaki Fujishima4, Eri Okumura4, Hideo Takekoshi4, Hidekatsu Takeda5 and Masahiro Iijima1
1Division of Orthodontics and Dentofacial Orthopedics, Department of Oral Growth and Development, School of Dentistry, Health Sciences University of Hokkaido, Japan
2Department of Dentistry, Oral and Maxillofacial Surgery, Jichi Medical University, Japan
3Department of Clinical Laboratory Science, School of Medical Technology, Health Sciences University of Hokkaido, Japan
4Sun Chlorella Corp., Production & Development Department, Japan
5Department of Physical Therapy, Sapporo Medical University of Medicine, Japan
Submission: August 08, 2022; Published: August 16, 2022
*Corresponding author: Tadahide Noguchi, Department of Dentistry, Oral and Maxillofacial Surgery, Jichi Medical University, Tochigi, Japan
How to cite this article: Dai T, Tadahide N, Maki T, Yusuke T, Masaki F, et al. Effectiveness of Acanthopanax Senticosus Harms in a Mouse Model of Nickel Allergy. Adv Dent & Oral Health. 2022; 15(4): 555918. DOI: 10.19080/ADOH.2022.15.555918
Abstract
Although metal allergies are thought to develop in a T cell-dependent manner, no appropriate animal model currently exists. Further, since the molecular mechanisms of the allergic reactions are not completely understood, a fundamental treatment method has not been established. Acanthopanax senticosus Harms (ASH) is a plant known for its anti-inflammatory and antioxidant effects. This study aimed to investigate the therapeutic effects of ASH using a mouse model of nickel allergy. Nickel was selected since it is frequently used for dental metals and metal ornaments. The mice were divided into a group to which a regular solid diet (control group) was given and a group to which ASH was added to the regular solid diets (ASH group). A mixed solution of Lipopolysaccharide (LPS) and nickel chloride (NiCl2) was intraperitoneally administered to sensitize. Seven days after sensitization, a NiCl2 solution was injected intradermally into the pinna to induce nickel allergy. For the evaluation of allergic reactions, the thickness of the pinna, the histopathological evaluation of the pinna, and the measurement of cytokines in the serum of mice were performed. In the ASH group, pinna swelling was significantly reduced at 24 and 48 h time points and lymphocyte infiltration was attenuated, compared with those in the control group. In vitro examinations showed that ASH suppressed TNF-α and interleukin-6 (IL-6) production in human peripheral blood. These results of this study suggest that ASH has a preventive effect on nickel allergy.
Keywords:Acanthopanax senticosus Harms; Metal Allergy; Nickel Allergy Mouse Model
Abbreviations:ASH: Acanthopanax senticosus Harms; LPS: Lipopolysaccharide; ACD: Allergic contact dermatitis; ELISA: Enzyme-linked immunosorbent assay; DAB: Diaminobenzidine; CIA: Collagen-induced arthritis
Introduction
Allergic contact hypersensitivity to metals is classified as a delayed type of hypersensitivity.
Although various metals are known to cause allergic reactions, the precise molecular mechanisms responsible for this allergy remain unclear, and no treatment for metal allergy has been established. The metal most commonly involved in such allergies is nickel, which is frequently used in dental metals and metallic ornaments, causing contact dermatitis. It occurs when metals come in direct contact with the skin and induce palmoplantar pustulosis, lichen planus, and pompholyx eczema due to the absorption of metals into the body. Oral lichen planus-like lesions may appear as lace-like keratotic lesions in the vicinity of metals in the oral cavity, which may be difficult to diagnose and treat.
Metal allergy is a disorder in which certain metallic elements act as haptens to cause localized or systemic contact dermatitis. Metal-derived contact allergic reactions can progress to allergic contact dermatitis (ACD) when exposure exceeds an individual’s threshold. Studies on the prevalence of metal allergies have been conducted in various countries, and in all of them, nickel, cobalt, and chromium have shown high favorable rates in patch tests. It is estimated that up to 17% of women and 3% of men are allergic to nickel [1]. The prevalence of metal allergy is even higher in patients with dermatitis [1]. Nickel exposure primarily occurs through consumer metal products. The use of accessories such as necklaces and earrings, belt hardware, jeans buttons and zippers, and cell phones increases the risk of sensitization to nickel and other metals [1-4]. Metal restorations in dentistry are also essential factors in causing metal allergies, and reports of such allergies are rising. The removal of metal restorations can be quite time-consuming and financially burdensome. Since CD4 and CD8-positive T cells are involved in the development of metal allergy, it is thought that modulating signal transduction to immune cells, such as epithelial cells, may inhibit the progress of allergies [5]. Acanthopanax senticosus HARMS (ASH) has various medicinal effects such as anti-anxiety, reduction of sexual behavior, the proliferation of learning function, suppression of lipid peroxidation, lowering blood pressure, induction of β-endorphin and growth hormone, increase in natural killer (NK) cell activity, and suppression of tumor cell proliferation [6-10]. This study aims to investigate the preventive and therapeutic effects of ASH by using the nickel (Ni) allergy mouse model, which is frequently used for dental metals and metal ornaments.
Materials and Methods
Mice
4-6 weeks old female BALB/c mice were purchased from HOKUDO (Hokkaido, Japan). Drinking water was provided ad libitum. The mice were acclimatized to our facility for at least seven days before the experiment. All experiments were conducted in accordance with The Animal Experiment Board and approved by the Health Sciences University of Hokkaido animal experiments. (Approval number, 19-022).
Reagents
ASH was obtained from Sun Chlorella Corp. (Kyoto, Japan). Nickel (II) chloride was purchased by Wako (Osaka, Japan). Lipopolysaccharide (LPS, derived from Escherichia coli serotype 055: B5) was purchased from Sigma-Aldrich (St. Louis, MO, USA). All reagents used in this experiment were adjusted to the specified concentrations using saline solution, immediately before dosing.
Dosing of ASH
Solid diets (Oriental Yeast Co., Ltd.) containing 5% of ASH extract were fed freely to animals of treatment group, while the control group was fed regular solid diets. Body weight was measured every week. After 28 days of treatment, nickel was administered.
Nickel allergy model mice
Sensitization phase. 9-11 weeks old BALB/c female mice (after the treatment) were intraperitoneally injected with 250 uL of 5.0 μg/mL LPS and 5.0 mM nickel chloride (NiCl2) solution (LPS-NiCl2), using a 26G needle. Elicitation phase. Seven days after sensitization, 20 uL of 5 mM NiCl2 solution was intradermally injected into the right pinna, under 5% isoflurane inhalation anesthesia to induce Ni allergy, using a 33G needle.
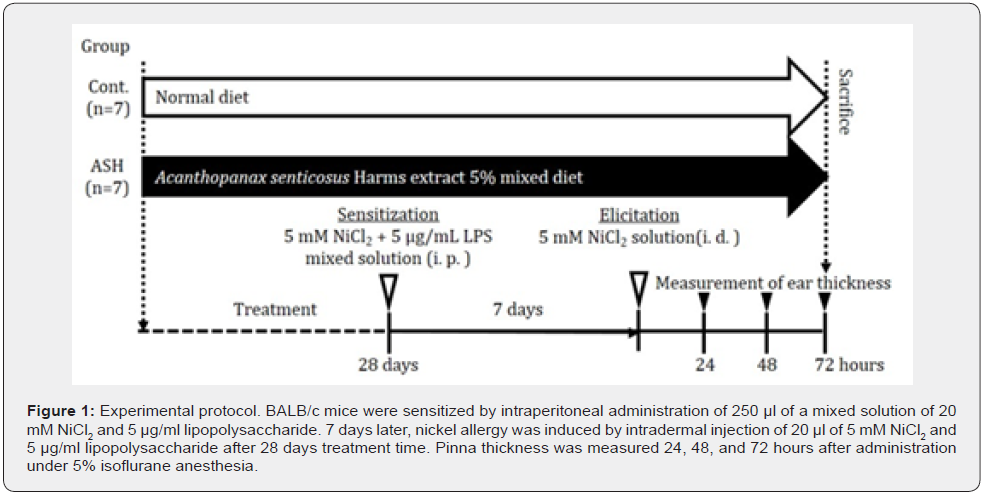
The thickness of the pinna was measured using a peacock dial thickness gauge. Three measurements were taken at 24, 48, and 72 hours after the challenge (Figure 1). The rate of swelling was calculated as the increase in ear thickness after attraction, relative to the thickness immediately before attraction (mean ± standard error, n=7). We decided to use ipsilateral pinna thickness before attraction, as the control data because the pathogenesis involves a systemic immune response, despite inducing a local inflammatory response. The mice were then anesthetized with 5% isoflurane, euthanized using CO2 gas, and blood was collected by cardiac puncture. The blood was stored at -80°C after serum separation. The right pinnae were excised and immersed in a fixative solution (10% formalin) for 12 hours, embedded in paraffin blocks.
Histological evaluation
Samples for all histopathological evaluations were prepared 72 hours after elicitation using the pinna. Paraffin-embedded blocks were sliced and stained with hematoxylin-eosin according to the standard method. Immunostaining was performed using the Goat anti-Rat IgG antibody Vectastain ABC-HRP kit (Vector Laboratories, Burlingame, CA) and diaminobenzidine (DAB) as a chromogen according to the manufacturer’s protocols. CD4 monoclonal antibody as anti CD4 antibody (4SM95, CD4:1/100,Invitrogen, Carlsbad, CA) was used. Histopathological studies were performed by the New Drug Research Center (Hokkaido, Japan).
Measurement of cytokines in serum by enzyme-linked immunosorbent assay (ELISA)
Blood samples from mice were centrifuged at 3000 rpm for 10 min at 4 °C, and serum was collected. Quantitative evaluation of proinflammatory cytokines (TNF-α and IL-6) in the serum of all mice was performed using a commercially available ELISA kit (R&D Systems). The absorbance (primary wavelength 450 nm, reference wavelength 620 nm) of all wells was measured using an absorptiometer (Infinite® F200, Tecan).
Statistical analyses
Statistical analysis was performed using statistical analysis software (SPSS Statics 25, IBM). Student’s t-test was used to test the difference between the means of each group. P-values <0.05 were considered statistically significant.
Results
Components of ASH
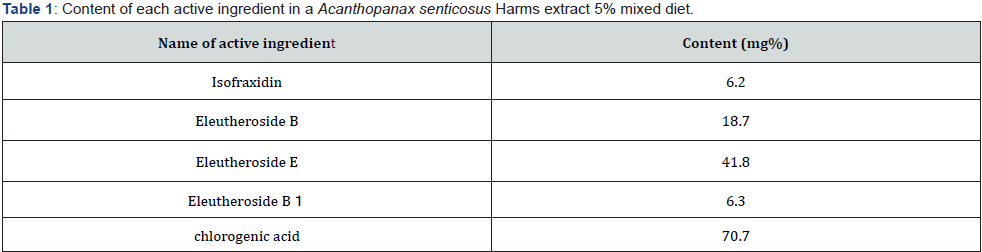
Table 1 shows the composition of the 5 % ASH extract blended chow. The content of each active ingredient in 100 g of feed was 6.2 mg of isofraxidin, 18.7 mg of eleutheroside B, 41.8 mg of eleutheroside E, 6.3 mg of eleutheroside B1, and 70.7 mg of chlorogenic acid.
Influence of ASH administration on nickel allergy model mice
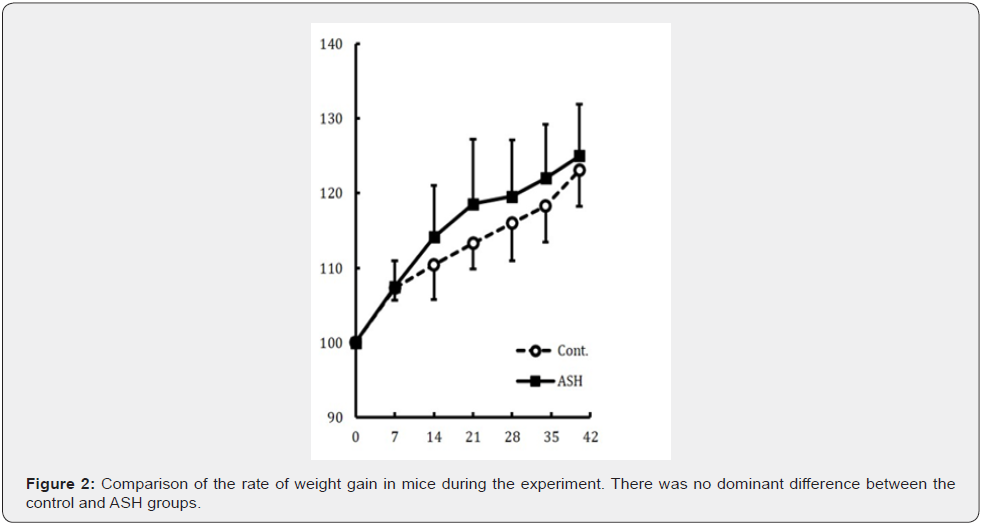
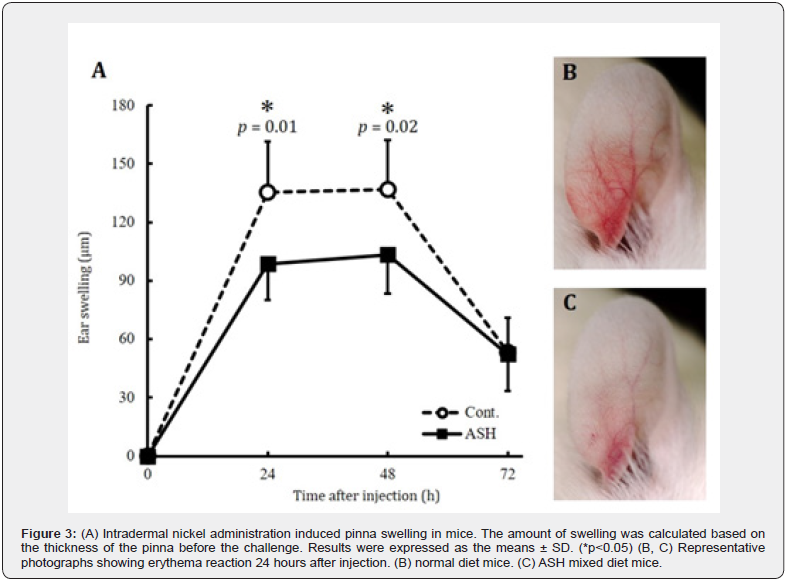
Influence on pinna swelling: No dead mice were identified during all experimental periods, and no significant changes in body weight were observed (Figure 2). Significant inhibition of swelling at 24 and 48 h time points was observed in the ASH group (Figure 3A). Photographs of representative pinna from each group at the 24-h time point after nickel allergy challenge are shown in Figure 3B & 3C. The extent of redness in the ASH group was limited and milder, compared with that in the control group.
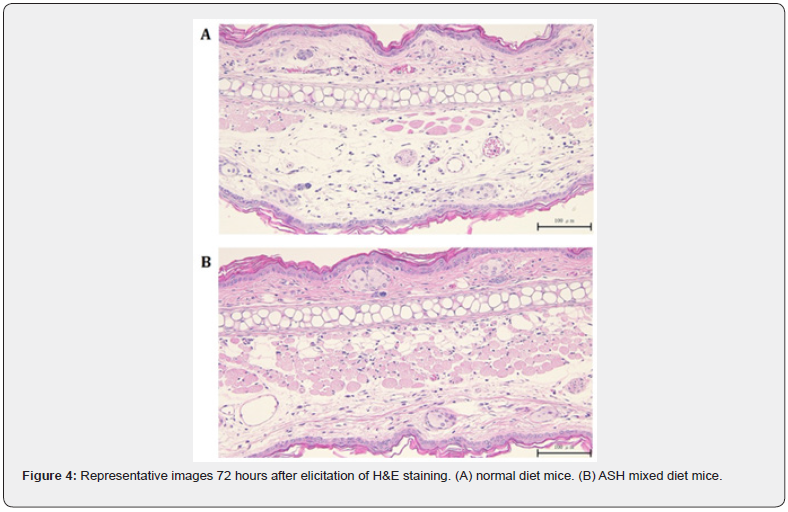
Histopathological evaluation of the pinna: H&E staining (×200 magnification) revealed chronic inflammatory cell infiltration with predominance of lymphocytes and subcutaneous edema, but both were milder in the ASH group, compared with those in the control group (Figure 4A, B).
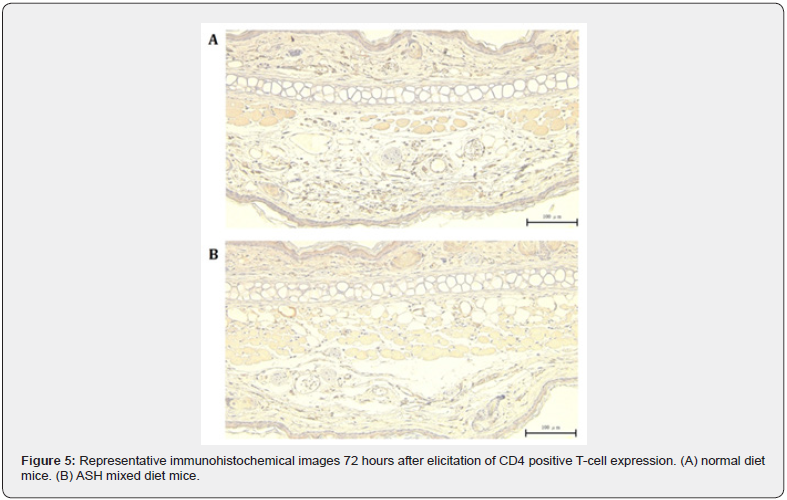
Immunohistochemical staining revealed lesser infiltration of CD4 positive T cells in the blood vessels of the ASH group, compared with that in the control group (Figure 5A & 5B).
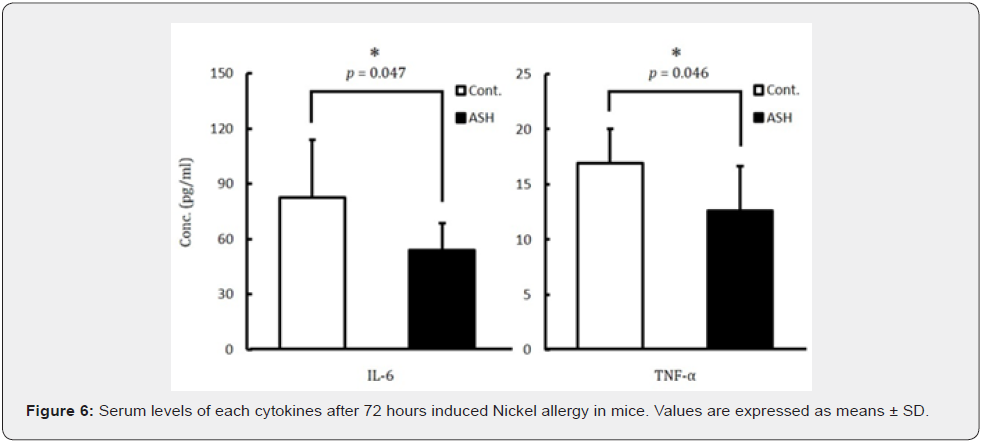
Cytokine levels in the blood serum: The inflammatory cytokine levels in murine blood serum are shown in Figure 6. The mean IL-6 and TNF-α production in the control group was 82.7 pg/mL and 16.9 pg/mL, respectively, and in the ASH group it was 53.7 pg/mL and 12.6 pg/mL, respectively. A significant (p<0.05) reduction of approximately 65.0 % for IL-6 and 15.2 % for TNF-α was observed in the ASH group, on comparison with the control group.
Discussion
Upon direct contact with metal accessories, the metal ions eluted from the product by sweat, penetrate directly into the skin. Dental metals, restored in the oral cavity, are eluted and ionized by food, saliva and metabolites of oral bacteria and are taken up by the oral mucosa. Ionized metals bind to self-proteins in the body and are antigenized. These ions are then taken up by antigen-presenting cells, transported to regional lymph nodes, and recognized and sensitized by hapten-specific T cells. Subsequent exposure to the same metal activates T cells and produces inflammatory cytokines, leading to allergic symptoms. Contact dermatitis (mucositis) is followed by systemic contact dermatitis, in which the antigen is transported to the wholebody via blood circulation [1,11-15]. However, no treatment has been established yet due to difficulties in identifying the allergen metals. The removal and re-restoration of the metal restoration are accompanied by considerable time and economic burden, with no guaranteed improvement in the disease state.
CD4 and CD8 positive T cells contribute to the cellular immunity to the disease state manifestation during a metallic allergy [16- 18]. This can be suppressed by altering the signal transduction of these immunocompetent cells. We investigated the therapeutic effects of ASH against metal allergy using a mouse model of Ni allergy. Takahashi et al. [9] reported the therapeutic effects of ASH in a mouse model of collagen-induced arthritis (CIA); they observed a delay in the onset of CIA and reduction in its severity. They concluded the enhanced effects of anti-TNF-α-antibody therapy in CIA mouse models. ASH suppressed the production of TNF-α, IL-6, superoxide, and hydroxyl radicals. The production of both cytokines was suppressed at the transcriptional level. Therefore, ASH enhanced the therapeutic effect of the anti-TNFα antibody on rheumatoid arthritis through the anti-inflammatory and antioxidative effect [9].
Our study showed that ASH significantly alleviated pinna swelling and inhibited the invasion of CD4 positive cells in a nickel allergy mouse model. Cytokine levels were significantly reduced in murine serum. Liriodendrin, isolated using ethyl acetate (EtOAc) from the bark of ASH by activity-induced fractionation, inhibits vascular permeability [19]. This may have resulted in alleviated edema and CD4 invasion by vascular permeability inhibition in the ASH group. Oral mucosal diseases in which cell-mediated immunity is thought to be involved in the pathogenesis, includes oral lichen planus and GVHD besides metallic allergy. Lichen planus is a recalcitrant chronic inflammatory keratosis that appears on the skin and mucous membranes. In the oral cavity, lichen planus occurs frequently on the buccal mucosa and gingiva and may become cancerous by several percent. Histopathologically, thickening with keratinization of the epithelium and zonal infiltration of inflammatory cells (mainly T cells) just beneath the basal cells are characteristic findings, and the interaction between infiltrated lymphocytes and epithelial cells seems to contribute to the pathogenesis [20-22]. Various modifiers are complicatedly concerned in the disease state formation of oral lichen planus besides the cell-mediated immunity mechanism, and the scavenger such as free radical and antioxidant enzyme is also considered to contribute to this [23].
ASH inhibited the generation of superoxide and hydrogen peroxide in murine peritoneal macrophages in vitro and in vivo [24], and it acts as a radical scavenger [9,25-27]. In addition, GVHD, expressed in the oral mucosa, has clinicopathological and histological findings like oral lichen planus, which occur when donor-derived T cells are activated and attack the oral mucosal epithelium of the transplant donor [28]. Furthermore, ASH extract is commercially available in many countries, and no obvious side effects have been reported when administered as a single agent [6]. Therefore, we would like to expect ASH to be clinically applied as a treatment for such refractory oral mucosal diseases.
Conclusion
In modern society, where we have the opportunity to come into contact with many metals, many people around the world are suffering from the symptoms of contact hypersensitivity. In the current situation where a fundamental treatment method has not been established for metal allergies, our research results suggest that administration of ASH is useful for the prevention and treatment of metal allergies.
References
- Thyssen JP, Menne T (2010) Metal Allergy-A Review on Exposures, Penetration, Genetics, Prevalence, and Clinical Implications. Chem Res Toxicol 23(2): 309-318.
- Thyssen JP, Johansen JD, Zachariae C, Menne ́T (2008) The outcome of dimethylglyoxime testing in a sample of cell phones in Denmark. Contact Dermatitis 59(1): 38-42.
- Thyssen JP, Hald M, Avnstorp C, Veien N, Lauerberg G, et al. (2009) Characteristics of nickel allergic dermatitis patients seen in private dermatology clinics in Denmark: A questionnaire study. Acta Derm-Venereol 89(4): 384-388.
- Heim KE, McKean BA (2009) Children’s clothing fasteners as a potential source of exposure to releasable nickel ions. Contact Dermatitis 60(2): 100-105.
- Kalish RS, Askenase PW (1999) Molecular mechanisms of CD8+T cell–mediated delayed hypersensitivity: Implications for allergies, asthma, and autoimmunity. J allergy Clin Immunol 103(2Pt 1): 192-199.
- Huang L, Zhao H, Huang B, Zheng C, Peng W, et al. (2011) Acanthopanax senticosus: Review of botany, chemistry and pharmacology. Pharmazie 66(2): 83-97.
- Jung HJ, Park HJ, Kim RG, Shin KM, Ha J, et al. (2003) In vivo anti-inflammatory and antinociceptive effects of liriodendrin isolated from the stem bark of Acanthopanax senticosus. Planta Med 69(7): 610-616.
- Fujikawa T, Yamaguchi A, Morita I, Takeda H, Nishibe S (1996) Protective effects of Acanthopanax senticosus harms from Hokkaido and its components on gastric ulcer in restrained cold water stressed rats. Biol Pharm Bull 19(9):1227-1230.
- Takahashi Y, Tanaka M, Murai R, Kuribayashi K, Kobayashi D, et al. (2014) Prophylactic and therapeutic effects of Acanthopanax senticosus Harms extract on murine collagen-induced arthritis. Phytother Res 28(10): 1513-1519.
- Yoon T, Yoo Y, Lee S, Shin K, Choi W, et al. (2014) Anti-metastatic activity of Acanthopanax senticosus extract and its possible immunological mechanism of action. J Ethnopharmacol 93(2-3): 247-253.
- Schmalz G, Garhammer P (2002) Biological interactions of dental cast alloys with oral tissues. Dental Materials 18(5): 396-406.
- Saito M, Arakaki R, Yamada A, Tsunematsu T, Kudo Y, et al. (2016) Molecular Mechanisms of Nickel Allergy Int J Mol Sci 17(2): 202.
- Pigatto PD, Guzzi G (2008) Systemic allergic dermatitis syndrome caused by mercury. Contact Dermat 59: 66.
- Yoshihisa Y, Shimizu T (2012) Metal allergy and systemic contact dermatitis: An overview. Dermatol Res Pract 749561.
- Yokozeki H, Katayama I, Nishioka K, Kinoshita M, Nishiyama S (1992) The role of metal allergy and local hyperhidrosis in the pathogenesis of pompholyx. J Dermatol 19(12): 964-967.
- Saint-Mezard P, Berard F, Dubois B, Kaiserlian D, Nicolas JF (2004) The role of CD4+ and CD8+ T cells in contact hypersensitivity and allergic contact dermatitis. Eur J Dermatol 14: 131-138.
- Sumiwi YA, Soesatyo MH, Sosroseno W (2010) The role of CD4+ T cells in the induction of contact hypersensitivity to mercury in a murine model. Cutan Ocul Toxicol 29(1): 30-33.
- Kawano M, Nakayama M, Aoshima Y, Nakamura K, Ono M, et al. (2014) NKG2D+ IFN-γ+ CD8+ T cells are responsible for palladium allergy. PLoS One 9: e86810.
- Yang L, Li D, Zhuo Y, Zhang S, Wang X, et al. (2016) Protective Role of Liriodendrin in Sepsis-Induced Acute Lung Injury. Inflammation 39: 1805-1813.
- Kramer IR, Lucas RB, Pindborg JJ, Sobin LH (1978) Definition of leukoplakia and related lesion: an aid to studies on oral precancer. Oral Sugery, Oral Medicine, Oral Pathology 46(4): 518-539.
- van der Meji EH, Schepman KP, van der Waal I (2003) The possible premalignant character of oral lichen planus and oral lichenoid lesions: a prospectine study. Oral Surgery, Oral Medicine, Oral Pathlogy, Oral Radiology, and Endodontology 96(2): 164-171.
- Shklar G (1972) Lichen planus as an oral ulcerative disease. Oral Surgery, Oral Medicine, Oral Pathlogy 33(3): 376-388.
- Noguchi T, Jinbu Y, Mitamura J, Tsukinoki K, Kusama M (2015) Immunohistochemical study on SOD in oral lichen planus. J Jpn Oral Muco Membr 11: 48-53.
- Lin QY, Jin LJ, Cao ZH, Lu YN, Xue HY, et al. (2008) Acanthopanax senticosus suppresses reactive oxygen species production by mouse peritoneal macrophages in vitro and in vivo. Phytother Res PTR 22(6): 740-745.
- Chen C, Ribaya-Mercado J, McKay D, Croom E, Blumberg J (2010) Differential antioxidant and quinone reductase inducing activity of American, Asian, and Siberian ginseng. Food Chem 119(2): 445-451.
- Lee S, Son D, Ryu J, Lee Y, Jung S, et al. (2004) Antioxidant activities of acanthopanax senticosus stems and their lignan components. Arch Pharm Res 27(1): 106-110.
- Liang Q, Yu X, Qu S, Xu H, Sui D (2010) Acanthopanax sentico-sides B ameliorates oxidative damage induced by hydrogen peroxide in cultured neonatal rat cardiomyocytes. Eur J Pharmacol 627(1): 209-215.
- Al-hashimi I, Schifter M Lockhart PB, Wray D, BrennanM, Migliorati CA, Axéll T, et al. (2007) Oral lichen planus and lichnoid lesion: diagnostic and therapeutic considerations. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology 103: S25.e1-e12.






























