Comparative Evaluation of Antimicrobial Efficacy of Calcium Hydroxide, Chitosan, Chlorhexidine and Their Combinations Against Enterococcus Fecalis and Candida Albicans Using Quantitative Real Time Polymerase Chain Reaction – An In Vitro Study
Archana Srinivasan*, Srirekha A, Kusum Bashetty, Teja Deepthi C, Ashis Mohanty and Ashwija Shetty
Department of Conservative dentistry & Endodontics, The Oxford Dental College and Hospital, Karnataka, India
Submission: September 27, 2019; Published: October 31, 2019
*Corresponding author: Archana Srinivasan, Department of Conservative dentistry & Endodontics, The Oxford Dental College and Hospital, Karnataka, India
How to cite this article: Archana Srinivasan, Srirekha A, Kusum Bashetty, Teja Deepthi C, Ashis Mohanty, Ashwija Shetty. Comparative Evaluation of Antimicrobial Efficacy of Calcium Hydroxide, Chitosan, Chlorhexidine and Their Combinations Against Enterococcus Fecalis and Candida Albicans Using Quantitative Real Time Polymerase Chain Reaction – An In Vitro Study. Adv Dent & Oral Health. 2019; 11(4): 555817. DOI:10.19080/ADOH.2019.11.555817
Abstract
Objectives: To comparatively evaluate the antimicrobial efficacy and synergistic effect of calcium hydroxide, chlorhexidine gluconate, chitosan and their combinations against E. fecalis and using Quantitative real time PCR.
Materials and Method: 120 human extracted single rooted maxillary premolar teeth were selected. Access cavity preparation was done followed by cleaning and shaping. Middle third of the root was cut using a rotary diamond disc. The specimens were inoculated with E. faecalis and Candida Albicans for 14 days and divided into two groups with sixty teeth each
a. Group 1: E. Fecalis.
b. Group 2: Candida Albicans.
This was followed by placement of Intracanal medicament and the two groups were further subdivided into six subgroups with ten teeth each.
c. Group 1 & 2 A: 2%Chlorhexidine.
d. Group 1 & 2 B: Calcium Hydroxide and 2% Chitosan.
e. Group 1 & 2 C: 2%Chitosan and 2%Chlorhexidine.
f. Group 1 & 2 D: 2%Chitosan.
g. Group 1 & 2 E: Calcium Hydroxide.
h. Group 1 & 2 F: Control.
The dentinal chips were collected, following which DNA isolation was done. The specimens were analyzed using real-time PCR.
Result: In the E. Fecalis group, ANOVA test statistical analysis showed significant difference in the quantity between subgroup 1C and subgroup 1F(P>0.05), whereas, in the Candida Albicansgrou, Kruskal Alli’s test showed a statistically significant difference between subgroup 2C and subgroup 2F (P<0.02).
Conclusion: Combination of 2% Chlorhexidine and 2% Chitosan are the most effective intracanal medicament against Enterococcus Fecalis and Candida Albicans.
Clinical significance: 2% Chlorhexidine and 2% Chitosan, as intracanal medicament, due to their probable synergistic antimicrobial effect against E. Faecalis and C. Albicans, can show promising results in reducing the endodontic treatment failure cases.
Keywords: E. Faecalis; C. Albicans; Polymerase Chain Reaction; Chlorhexidine; Chitosan; Calcium Hydroxide
Introduction
Bacterial invasion of root canal system is crucial for the onset and maintenance of periapical diseases. Hence, one of the goals of the endodontic treatment is to kill micro-organisms in the root canal system. Although, biomechanical preparation and root canal shaping effictively reduces the microbiota, these procedures do not completely eliminate bacteria in the lateral and accessory root canals, isthmi, and apical deltas. Thus, intracanal medication between appointments is recommended to further reduce bacteria in the root canal system. Candida albicans is the most common species of fungi cultured from root canals of teeth with failed endodontic treatment [1]. Because of collagenolytic activity, it may be possible for the yeast to use dentin as a nutrient source and promote colonization in the root canal. Another organism commonly found in cases of failed endodontic infections and endodontic flare-ups is Enterococcus faecalis. It has the ability to survive in root canal system as a single organism without the support of other bacteria and is small enough to proficiently invade and live within the dentinal tubules [1]. Calcium hydroxide is one of the most commonly used substances in Endodontics and its antibacterial property stems from its ability to increase a solution’s pH. Chlorhexidine has emerged as an intracanal medicament because of its antimicrobial spectrum, its ability to maintain its antibacterial action for a prolonged duration when adhered to anionic substrates and its slow release as its concentration decreases. Chitosan is a natural polysaccharide comprising of copolymers of glucosamine and N-acetyl glucosamine.
In endodontics its role as antifungal and antibacterial agent has not been subjected to adequate scrutiny. There is increasing scientific evidence regarding the role of C. albicans and E. faecalis particularly as members of secondary or persistent infections associated with failed endodontic therapy and their resistance against commonly used intracanal medicaments [2]. PCR assays are very sensitive and enable rapid identification of microbial species and strains that are difficult or even impossible to culture. Also, in cases with small number of bacteria, negative culture may result, even though viable bacteria may be present because of low sensitivity of culturing technique [3]. Thus, a more sophisticated and sensitive molecular technique like PCR was used to assess the effect of antimicrobial agents against E. faecalis [4].
Materials and Methods
Preparation of Dentine Specimens
The model proposed by Haapasalo & Ørstavik was modified [5]. One hundred and twenty single-rooted human maxillary premolar teeth freshly extracted for orthodontic reasons were selected. A rotary diamond disc was used to decoronate the teeth below the cementoenamel junction and the apical part of the root to obtain 6 mm of the middle third of the root. Gates Glidden drills no. 3 (Mani Inc, Tachigi-ken, Japan) in a slow-speed handpiece was used to standardize the internal diameter of the root canals. The specimens were placed in an ultrasonic bath of 17% ethylenediaminetetraacetic acid for 5 min followed by 3% NaOCl for 5 min to remove organic and inorganic debris. The traces of chemicals used were removed by immersing the dentine specimens in an ultrasonic bath containing distilled water for 5 min. All the specimens were sterilized in an autoclave for two cycles. The first cycle was at 121 0C and the second was with the specimens immersed in 1 mL of tryptone soya (TS) broth in individual microcentrifuge tubes [6].
Contamination of Specimens
The test organism used for this study was E. faecalis, which is a gram-positive facultative anaerobic bacterium that is common in root filled teeth with posttreatment infection. E. faecalis (ATCC 29212) was grown in tryptone soya agar for 24 h. The culture was suspended in 5 mL of TS broth and incubated for 4 h at 37 _C and its turbidity adjusted to 0.5 McFarland standard. Each dentine block was placed in pre-sterilized microcentrifuge tubes containing 1 mL of the TS broth. Fifty micro liters of the inoculum containing the E. faecalis was transferred into each of the microcentrifuge tubes. At the end of 24 h, the dentine specimens were transferred into fresh broth containing E. faecalis. All procedures were carried out under laminar flow. Purity of the culture was checked [6].
Antimicrobial Assesment
At the end of 14th day, the blocks were irrigated with 5 mL of sterile water to remove the inoculated broth. 120 sample teeth were divided into 60 teeth each for E. Faecalis and Candida Albicans. This was followed by placement of Intracanal medicament and the two groups were further subdivided into six subgroups with ten teeth each.
a. Group 1 & 2 A: 2% Chlorhexidine.
b. Group 1 & 2 B: Calcium Hydroxide and 2% Chitosan.
c. Group 1 & 2 C: 2% Chitosan and 2%Chlorhexidine.
d. Group 1 & 2 D: 2% Chitosan.
e. Group 1 & 2 E: Calcium Hydroxide.
f. Group 1 & 2 F: Control.
Teeth after placement of intracanal medicament were sealed with a temporary restorative material and incubated in an aerobic environment at 37 degrees celcius. An antimicrobial assessment was performed at the end of 14 days after placing intracanal medicament using quantitative real time PCR.
Extraction of RNA
All the plastic wares and glass wares used for this experiment were made RNase free by treating with DEPC for overnight and autoclaving twice at 121 oC until the traces of DEPC removed. These were then dried in Hot Air Oven at 90 oC before use. The samples were vortexed, the dentine samples were taken out, the broth was centrifuged, and the pellet was collected for the study. The pellet was suspended in 0.5ml of Trizol Reagent (Takara Bio Inc, Japan), mixed, transferred to a 2ml microfuge tube and incubated at room temperature for 15 – 20 mins. It was then centrifuged at 10,000 rpm (Sigma) for 5 min and the supernatant was transferred to a fresh sterile microfuge tube. Equal volume of Chloroform: Isoamyl alcohol (24:1) was added, mixed gently and incubated in ice for 2 minutes. After centrifuging the above at 12,000 rpm for 5 minutes the supernatant was transferred to a fresh vial and then incubated in ice for 2 minutes after adding 500 μl of isopropanol. The RNA was pelleted down by centrifuging the above for 10 minutes at 10,000 rpm and the pellet was vortexed gently with 300 μl of 70% of ethanol. Centrifugation at 10,000 rpm was repeated for 10 minutes and the pellet was air dried. 20μl of RNase free water was added to the pellet and heated gently (60 oC) to dissolve the pellet in water.
DNase Treatment
The extracted RNA was treated with DNase enzyme to remove any traces of DNA contamination. One micro liter of DNase was added to above isolated RNA and incubated for 1hr at 37 oC and after the temperature was raised to 70 oC for 5 minutes to inactivate the enzyme. The RNA was stored at -20 oC for later use.
RNA Quantification
The concentration and purity of RNA was assessed using a spectrophotometer (Sartorius). A 1μL aliquot of RNA was pipetted onto the apparatus pedestal. RNA with an absorbance ratio at 260 and 280 nm (A260/A280) between 1.8 and 2.2 was deemed indicative of pure RNA.
cDNA Synthesis
After quantification, RNA was reverse transcribed using oligo dT (Sigma Aldrich). Hundred nano gram of RNA was aliquoted to a fresh sterile microfuge tube and 2μl of oligo dT was added and incubated at 70 oC for 5 minutes and immediately transferred to ice. To this 2μl of dNTPs, 1μl of Reverse Transcriptase enzyme (Biolabs, New England) and 2 μl of 10x Reverse transcriptase buffer was added and made up the volume to 25μl using RNase free water. This mixture was incubated at 42 oC for 90 minutes and reaction was terminated by incubating at 70 oC for 15 minutes.
Real-time PCR (qPCR)
Relative Quantification using Real Time PCR. The quantification was carried out in Applied Biosystems Step One Real Time PCR using the SYBR Green Chemistry.
Statistical Analysis
Since the E. Faecalis group sample was normally distributed, parametric test ANOVA was applied for the comparison between the groups, followed by Tukey multiple comparison means to check the difference in bacterial inhibition between groups (P < 0.05). For the Candida Albicans group since the sample was not normally distributed Kruskal Walli’s test was applied followed by post hoc comparison using Dunn test to check the difference in microbial inhibition between groups (P < 0.02).
Results
E. Faecalis Group
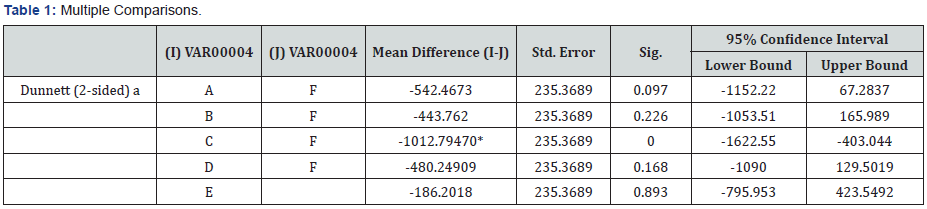
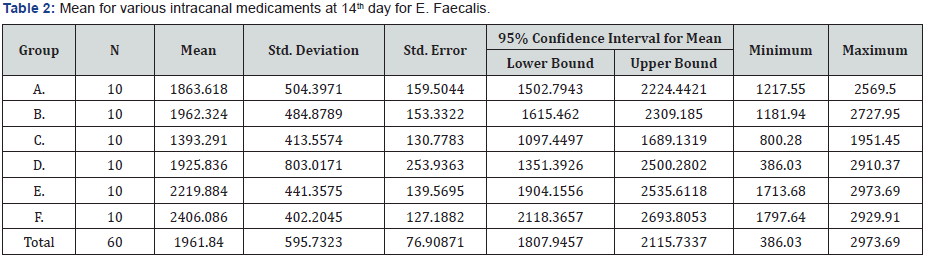
Mean and Median for the samples were calculated and since there was a statistically significant difference among the groups, Tukey Post-Hoc was applied for multiple comparisons. There was a statistically significant difference between Group C (Chlorhexidine +Chitosan) and Group F (Control). In the E. Faecalis group, statistical analysis showed significant difference in the quantity between subgroup 1C (chlorhexidine and chitosan combination) and subgroup 1F(control) (P>0.05) (Table 1). Maximum reduction of the bacteria was seen in the third group, i.e, chlorhexidine and chitosan combination (Table 2), whereas the least reduction was see in the control group, i.e, without any intracanal medicament (Table 2). Following the third group, the next group to show reduction in the bacteria was the chlorhexidine group, followed by chitosan group. Least reduction was seen with the Calcium hydroxide, calcium hydroxide and chitosan combination and the Control group (Table 2).
Candida Albicans Group
Table 3: Median, and interquartile range for various intracanal medicaments at 14th day for Candida Albicans
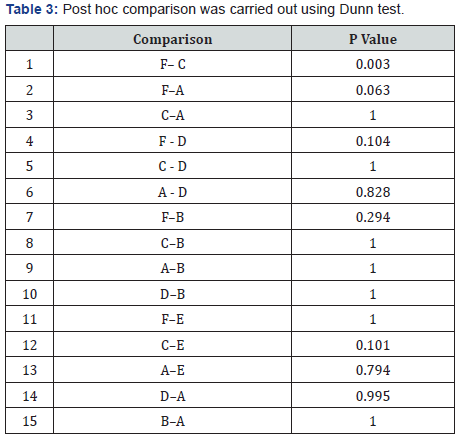
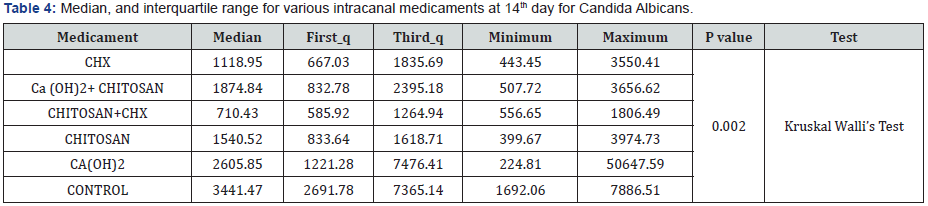
In Candida Albicans group, a statistically significant difference was seen between subgroup 2C (Chlorhexidine and Chitosan combination) and subgroup 2F(control) (P<0.02) (Table 3). Maximum reduction of the microorganism was seen in the third group, i.e, chlorhexidine and chitosan combination, whereas the least reduction was see in the control group, i.e, without any intracanal medicament (Table 4).
Discussion
Enterococcus faecalis has been chosen for the study as it is one of the most persistent microorganisms, with a prevalence of 24 to 77% in root filled teeth with periradicular lesions [7]. Studies have shown E. faecalis to be viable inside the root canal dentin up to a period of 12 months [8]. Therefore, it is important to consider treatment regimens aimed at eliminating or preventing the infection of E. faecalis during all the phases of root canal treatment. Similarly, Candida albicans was chosen as it has shown to play a major role in the endodontic treatment failure than other fungi [9]. Disinfection of pulp space is an important step during and after cleaning and shaping. Intracanal medicaments are used for root canal disinfection [9]. The use of intracanal medications possessing antimicrobial properties between appointments may reduce or eliminate bacteria in the root canal system and significantly increase the success of root canal treatment.
In the present study, a combination of 2% Chlorhexidine and 2% Chitosan showed statistically significant values (P < 0.05 level for E. Faecalis and P < 0.02 for C. Albicans). Maximum reduction of E. Faecalis (1393.2±413.5 bacterial count) and Candida Albicans (710.43) were seen when the above combination was used as an intracanal medicament (Tables 2 & 4) respectively. This is in accordance with the study conducted by Ballal et al. [2]. The possible reason could be the ant bactericidal dosage of 2% and increased diffusion of the medicament into the dentinal tubules [10]. The combination could possibly have a synergistic effect on viability of E. Faecalis and C. Albican [11]. Though the other groups had no statistically significant difference, considering the data provided for the reduction in the count of the two micro-organism, 2% Chlorhexidine gluconate showed to be more effective against C. Albicans and E.Faecalis than 2% Chitosan gel when used alone( 1863±504.3<1925.8±803) (Tables 2 & 4) respectively. Possible reason could be the bactericidal activity of 2% chlorhexidine [12] as compared to the bacteriostatic activity of Chitosan [13]. Hence, Chitosan gel alone may not be very useful as an antimicrobial agent against C. albicans and E. faecalis.
Chlorhexidine in gel formulation has been chosen for this study due to its low toxicity on the periapical tissues, solubility in water and viscosity that keeps the active agent in contact with the root canal walls and dentinal tubules [14-16]. In this work, the results corroborated previous study and indicate that treatment with Ca(OH)2 alone has minor effect on C. Albicans and E.Faecalis as compared to the treatment with chlorhexidine alone[17,18] (2605.85>1118.95 – C. Albicans) (2406±402> 1863±504.3 – E. Faecalis)and the combination of chlorhexidine and Chitosan(2605.85>710.43 – C .Albicans and (2406±402 > 1393.2±413.5– E. Faecalis) (Tables 2 & 4) respectively. Possible reason could be that although both calcium hydroxide and chlor hexidine exert their bactericidal activity by disintegrating membranes, additionally, chlorhexidine may induce reactive oxygen species production in alkaline environment, which would inhibit the E.Faecalis growth because of the destruction of cell wall and the plasma membrane mediated by nitric oxide [18]. Whereas, the combination of Calcium Hydroxide and Chitosan have shown better results for both the groups of micro-organisms as compared to calcium hydroxide alone. (C. Albicans –1874.8<2605.85), E. Faecalis – (2219.8±441 <2406±402) (Tables 2 & 4) respectively. Possible reason could be the antimicrobial action of chitosan that could have enhanced the antimicrobial action of calcium hydroxide. Hence, this study demonstrated that combining Chlorhexidine gluconate with Chitosan had a good anti-microbial effect against C. albicans and E. faecalis. However, further investigations are required to prove the results in vivo.
Conclusion
A combination of 2% Chlorhexidine and 2% Chitosan showed better antibacterial efficacy against E. faecalis and antifungal activity against Candida Albicans by using real- time PCR.
Acknowledgment
The study was supported by grants from the Rajiv GandhiUniversity Of Health Sciences, Bangalore (ORDER NO.RGU: Adv. Res.: Proposal-D-06:2015-16). The authors also wish to thank Dr. Savitha for the support.
References
- Jaheer S, Roopadevi G, Bolla N, Varri S, Thumu J, et al. (2014) Comparative evaluation of antimicrobial efficacy of triple antibiotic paste and calcium hydroxide using chitosan as carrier against Candida albicans and Enterococcus faecalis: An in vitro study. J Conserv Dent 17(4): 335-339.
- Ballal NV, Kundabala M, Bhat KS, Acharya S, Ballal M, et al. (2009) Susceptibility of Candida albicans and Enterococcus faecalis To Chitosan, Chlorhexidine gluconate and their combination in vitro. Aust Endod J 35(1): 29-33.
- Kandaswamy E, Nagendrababu V, Kalaiselvam R, Deivanayagam K (2013) Dentinal tubule disinfection with 2% chlorhexidine, garlic extract, and calcium hydroxide against Enterococcus faecalis by using real-time polymerase chain reaction: In vitro study. J Conserv Dent 16(3): 194-198.
- Williams JM, Trope M, Caplan DJ, Shugars DC (2006) Detection and quantitation of E. Faecalis by real-time PCR (qPCR), reverse transcription- PCR (RT-PCR), and cultivation during endodontic treatment. J Endod 32(8): 715-721.
- Haapasalo M, Ørstavik D (1987) In vitro infection and disinfection of dentinal tubules. Journal of Dental Research 66(8): 1375-1379.
- Kandaswamy, Venkateshbabu N, Gogulnath D, Kindo AJ (2010) Dentinal tubule disinfection with 2% chlorhexidine gel, propolis, morinda citrifolia juice, 2% povidine iodine, and calcium hydroxide. Int Endod J 43(5): 419-423.
- Rocas IN, Siqueira JF, Santos KRN (2004) Association of Enterococcus faecalis with different forms of periradicular diseases. J Endod 30(5): 315-320.
- Sedgley CM, Lennan SL, Appelbe OK (2005) Survival of Enterococcus faecalis in root canals ex vivo. Int Endod J 38(10): 735-742.
- Ambikathanaya (2014) Intracanal antiseptic medications; A review. Ujmds 2(3): 136-142.
- Krithikadatta J, Indria R, Dorothykalyani AL (2007) Disinfection of dentinal tubules with 2% Chlorhexidine, 2% Metronidazole, Bioactive Glass when compared with Calcium Hydroxide as intracanal medicaments. J Endod 33(12): 1473-1476.
- Natasha J, Dakshita S, Udai P, Kanwardeep S, Urja J, et al. (2017) Evaluation of antibacterial efficacy of Chitosan, Chlorhexidine, Propolis and Sodium hypochlorite on Enterococcus faecalis biofilm: An in vitro study. J Clin Exp Dent 9(9): 1066-1074.
- Mohammadi Z, Abbott PV (2009) The properties and applications of Chlorhexidine in endodontics. Int Endod J 42(4): 288-302.
- Kmiec M, Pighinelli L, Tedesco MF, Silva MM, Reis V (2017) Chitosan-properties and applications in dentistry. Adv Tissue Eng Regen Med Open Access 2(4): 205-211.
- Ferraz CCR, Gomes BPFA, Zaia AA, Teixeira FB, Souza FJ (2001) In vitro assessment of the antimicrobial action and the mechanical ability of chlorhexidine gel as an endodontic irrigant. J Endod 27(7): 452-455.
- Greenstein G, Berman C, Jaffin R (1986) Chlorhexidine. An adjunct to periodontal therapy. J Periodontol 57(6): 370-376.
- Gomes BPFA, Ferraz CCR, Vianna ME, Berber VB, Teixeira FR, et al. (2001) In vitro antimicrobial activity of several concentrations of sodium hypochlorite and chlorhexidine gluconate in the elimination of Enterococcus faecalis. Int Endod J 34(6): 424-428.
- Ronan J, Thais H, Carla R, Claudia R, Ivaldo G, et al. (2013) Antimicrobial activity of calcium hydroxide and chlorhexidine on intratubular Candida albicans International Journal of Oral Science 5(1): 32-36.
- Delgado RJ, Gasparoto TH, Sipert CR, Pinheiro CR, Moraes IG, et al. (2010) Antimicrobial effects of callcium Hydroxide and Chlorhexidine on E Faecalis. J Endod 36(8): 1389-13993.






























