Extracted Tooth: Can it be A Bone Graft Substitute?
In Woong Um*
R&D Institute, Korea Tooth Bank, Seoul, Korea
Submission: February 01, 2017; Published: February 28, 2017
*Corresponding author: In-Woong Um, DDS, Ph.D, R&D Institute, Korea Tooth Bank, Seoul-In Dental Clinic, 622 Eonju-ro, Gangnam-gu, Seoul 135-832, Republic of Korea, Tel:+82-2-548-2228 ; Fax: + 82-2-548-2228; E-mail:h-bmp@hanmail.net
How to cite this article: In Woong U. Tooth and Bone Graft Substitute Extracted Tooth: Can it be A Bone Graft Substitute?. Adv Dent & Oral Health. 2017; 002 4(1): 555627. DOI: 10.19080/ADOH.2017.04.555627
Abstract
This article introduces the use of Demineralized Dentin Matrix (DDM) scaffold, which are fabricated from the patient’s own extracted tooth to repair the alveolar bone defect under the view point of clinical implant dentistry. DDM is natural collagen based scaffold with similar organic and inorganic component of autogenous bone. The possibilities of DDM as bone graft substitute are compared to well known demineralized bone matrix (DBM) which has a little limitations. Based on in vitro and in vivo studies on DDM, clinical applications of DDM as three dimensional scaffolds for alveolar bone regeneration have shown predictability as well as possibilities of future advancement to the carrier of growth factors and stem cells.
Keywords: Demineralized dentin matrix, Demineralized bone matrix
Abbreviation: DDM: Demineralized Dentin Matrix; DBM: Demineralized Bone Matrix
Introduction
Dentin
Autogenous bone graft has been generally carried out for the repair of alveolar bone defect as a gold standard. To avoid and overcome the disadvantages of not only donor site morbidity and complications but also the limited supply, the development for bone substitutes as an alternative of autogenous bone have begun. And various artificial materials including ceramic material, synthetic biological material nano-material have been developed.
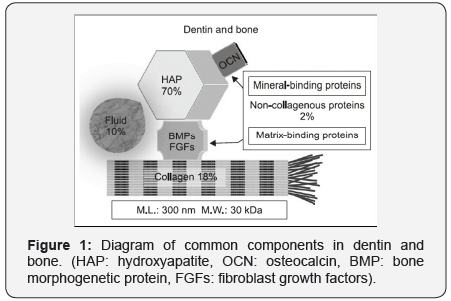
One of the alternatives recently developed is dentin, which has been discarded after extraction. Because the structure and composition of dentin are similar to that of bone, consisting of collagen (20%), hydroxyapatite (70%), and body fluid (10%) in weight (Figure 1) [1-5]. Bone inducing molecules, such as bone morphogenetic proteins (BMPs), was also identified from rat, rabbit and bovine derived bone and dentin [6,7]. Moreover, BMPs derived from human dentin induced new bone formation [7,8].
Apart from these molecules, dentin collagen is the most highly insoluble matrix due to its cross-linking [9], with osteoinductive growth factors that provide an appropriate environment for osteoinduction [8,10]. Dentin matrix-derived BMP is not exactly same as bone matrix-derived BMP, but they are very similar. In other words, two types of BMP exhibit the same action in the body [11].
The collagenous dentin matrix is a repository of bone morphogenetic proteins (BMPs), transforming growth factor-β (TGF-β), insulin-like growth factor (IGF), and basic fibroblast growth factor (bFGF). In addition, the concentration of TGF-β, insulin growth factor-I (IGF-I), insulin growth factor-II (IGF-II) fibroblast growth factor (aFGF, bFGF), and platelet derived growth factor (PDGF) were detected in human DDM. However, the concentrations of these growth factors in dentin were lower, compared to bone [10,12].
Several non-collagenous proteins such as osteocalcin (OCN) and osteopontin (OPN) are common in dentin and bone, while dentin phosphoprotein (DPP) (so-called phosphophoryn) is a specific non-collagenous protein in dentin (Figure 1). DSP is a dentin-specific non-collagenous protein involved in the calcification of dentin and suggested that dentin has high affinity for and marked osteoconductive effect on the jaw bone [5,13-16].
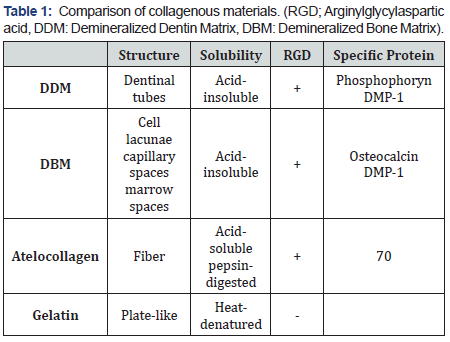
Clinically, it also can be defined as reprocessed natural collagenous scaffold with endogenous growth factors among acid-soluble, acid-insoluble, heat-denatured gelatin and pepsin digested atelocollagen (Table 1) [5].
Demineralized bone matrix (DBM) vs demineralized dentin matrix (DDM)
The idea of DDM is basically comes from DBM. DBM is prepared by demineralization of allograft bone [17]. It results in the elimination of the major part of the mineral phase and the immunogenic components of bone [18,19].
It has been speculated that the osteoinductive growth factors contained in the dentin matrix are more easily accessed and unshielded once the mineral phase of the dentin has been removed by demineralization [20]. Demineralization with 0.6N HCL results in the elimination of the major part of the mineral phase retaining a very low fraction of mineral in dentin (10-30% approx.) and the immunogenic components, but retains Type I collagen that provides an osteoconductive scaffold and a soluble protein fraction comprising several GFs, BMPs among them [21-23]. As a result, DDM increased their osteoinductivity and decreased their antigenicity after demineralization [17-20].
DBM by itself is presented in the form of a powder, but is usually mixed with other suitable materials (e.g. hyaluronan, poloxamer) to form a paste that can be easily molded at the bone defect [19,21]. DDM powder is prepared by crushing the dentin into 300-800mm sized particles. Each DDM powder possesses dentinal tubule-derived micropore spaces (diameter: 0.8-1.0μm, approximately 50,000 tubes/mm2). This pore size is too small for the infiltration and ingrowth of cells. Once dentin is demineralized, the dentinal tubule would become wider and serve as a channel for releasing essential proteins, which may promote growth and differentiation of osteoblasts. Enlarged dentinal tubules and loosened collagen by demineralization enhanced surface microroughness or micro-texture to help easy releasing of signaling factors and remodeling [1,18,22]. For an average follow-up of at least 5 years, DDM powder showed that cortico-cancellous bone had been maintained successfully after functional loading, which illustrates the remodeling capacity of DDM powder [23].
DDM block, fabricated from root dentin, is a biomimetic of cortical bone that exhibits slow creeping substitution properties with 3 to 5lm innate micropores (dentinal tubules) and 0.2 to 0.3mm macropores. DDM block is well incorporated and remodeled into cortico-cancellous bone with dental implant after average 44 months of follow-up [24,25].
DBM can be supplemented with additional growth factors in an attempt to boost their regenerative capacity. DBM has been mainly loaded with BMPs and particularly, with BMP-2 for bone tissue engineering [26,27]. DDM can also be supplemented with rhBMP-2 to enhance bone forming capacity by its endogenous and exogenous growth factors [28-30].
Conclusion
Human DDM, prepared by demineralization process of extracted teeth either orthodontic or pathologic origin, can be defined as acid-insoluble, cross-linked collagen with natural growth factors, absorbable, and elastic material with original dentin structures. Reprocessed dentin scaffolds represent probably the most advanced scaffolds available because it has not only the complex composition, but also the structure of the native tissues. This cannot be achieved with the scaffolds prepared from synthesis and tissue engineering such as those presented before.
Acknowledgment
Human DDM, prepared by demineralization process of extracted teeth either orthodontic or pathologic origin, can be defined as acid-insoluble, cross-linked collagen with natural growth factors, absorbable, and elastic material with original dentin structures. Reprocessed dentin scaffolds represent probably the most advanced scaffolds available because it has not only the complex composition, but also the structure of the native tissues. This cannot be achieved with the scaffolds prepared from synthesis and tissue engineering such as those presented before.
References
- Kim YK, Kim SG, Byeon JH, Lee HJ, Um IW, et al. (2010) Development of a novel bone grafting material using autogenous teeth. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 109(4): 496-503.
- Murata M, Akazawa T, Mitsugi M, Um IW, Kim KW, et al. (2011) Human dentin as novel biomaterial for bone regeneration. Intech publisher, Croatia, pp. 127-140.
- Murata M, Akazawa T, Mitsugi M, Kabir MA, Um IW, et al. (2013) Autograft of dentin materials for bone regeneration. Intech publisher, Croatia, pp. 391-403.
- Kim YK, Lee JK, Kim KW, Um IW, Murata M (2013) Healing Mechanism and Clinical Application of Autogenous Tooth Bone Graft Material. Intech publisher, Croatia, pp. 405-435.
- Murata M (2012) Collagen biology for bone regenerative surgery. J Korean Assoc Oral Maxillofac Surg 38(6): 321-325.
- Urist MR (1965) Bone: formation by autoinduction. Science 150(3698): 893-899.
- Kawai T, Urist MR (1989) Bovine tooth derived bone morphogenetic protein. Journal of Dental Research 68(6): 1069-1074.
- Murata M, Kawai T, Kawakami T, Akazawa T, Tazaki J, et al. (2010) Human acid-insoluble dentin with BMP-2 accelerates bone induction in subcutaneous and intramuscular tissues. Journal of the Ceramic Society of Japan 118(2010): 438-441.
- Xu C, Wang Y (2010) Cross-linked demineralized dentin maintains its mechanical stability when challenged by bacterial collagenase. Journal of Biomedical Materials Research 96(2): 242-248.
- Finkelman RD, Mohan S, Jennings JC, Taylor AK, Jepsen S, et al. (1990) Quantitation of growth factors IGF-I, SGF/IGF-II, and TGF-beta in human dentin. Journal of Bone and Mineral Research 5(7): 717-723.
- Bessho K, Tanaka N, Matsumoto J, Tagawa T, Murata M (1991) Human dentin-matrix-derived bone morphogenetic protein. J Dent Res 70(3): 171-175.
- Ito K, Arakawa T, Murata M, Tazaki J, Takuma Te, et al. (2008) Analysis of bone morphogenetic protein in human dental pulp tissues. Archives of Bioceramics Research (8): 166-169.
- Qin C, D’ Souza R, Feng JQ (2007) Dentin matrix protein 1 (DMP1): new and important roles for biomineralization and phosphate homeostasis. J Dent Res 86(12): 1134-1141.
- Sun Y, Lu Y, Chen S, Prasad M, Wang X, et al. (2010) Key proteolytic cleavage site and full-length form of DSPP. J Dent Res 89(5): 498-503.
- Qin C, Brunn JC, Jones J, George A, Ramachandran A, et al. (2001) A comparative study of sialic acid-rich proteins in rat bone and dentin. Eur J Oral Sci 109(2):133-141.
- Nampo T, Watahiki J, Enomoto A, Taguchi T, Ono M, et al. (2010) A new method for alveolar bone repair using extracted teeth for the graft material. J Periodontol 81(9):1264-1272.
- Urist MR (1965) Bone: formation by autoinduction. Science 150(3698): 893-899.
- Reddi AH (1998) Role of Morphogenetic Proteins in Skeletal Tissue Engineering and Regeneration. Nature Biotechnology 16(3): 247-252.
- Dinopoulos HT, Giannoudis PV (2006) Safety and Efficacy of Use of Demineralised Bone Matrix in Orthopaedic and Trauma Surgery. Expert Opinion in Drug Safety 5(6): 847-866.
- Reddi AH (1974) Bone matrix in the solid state: geometric influence on differentiation of fibroblasts. Adv Biol Med Phys 15(0): 1-18.
- Juang Ming Yee A, Bae HW, Friess D, Robbin M, Johnstone B, et al. (2003) Augmentation of Rabbit Posterolateral Spondylodesis Using a Novel Demineralized Bone Matrix-Hyaluronan Putty. Spine 28(21): 2435-2440.
- Koga T, Minamizato T, Kawai Y, Miura K, Nakatani Y, et al. (2016) Bone Regeneration Using Dentin Matrix Depends on the Degree of Demineralization and Particle Size. PLoS ONE 11(1): e0147235.
- Kim YK, Lee JH, Um IW, Cho WJ (2016) Guided Bone Regeneration Using Demineralized Dentin Matrix: Long-Term Follow-Up. J Oral Maxillofac Surg 74: 515.e1-515.e9.
- Kim YK, Um IW, Murata M (2014) Tooth bank system for bone regeneration-safety report. J Hard Tissue Biol 23: 371-376.
- Kim YK, Pang KM, Yun PY, Leem DH, Um IW (2016). Long-term followup of autogenous tooth bone graft blocks with dental implants. Clinical Oral Implants Research 5(2): 108-118.
- Lin H, Zhao Y, Sun W, Chen B, Zhang J, et al. (2008) The Effect of Crosslinking Heparin to Demineralized Bone Matrix on Mechanical Strength and Specific Binding to Human Bone Morphogenetic Protein-2. Biomaterials 29(9): 1189-1197.
- Lammens J, Nijs J, Schepers E, Ectors N, Lismont D, et al. (2009) The Effect of Bone Morphogenetic Protein-7 (OP-1) and Demineralized Bone Matrix (DBM) in the Rabbit Tibial Distraction Model. Acta Orthop Belg 75(1): 103-109.
- Kim YK, Um IW, An HJ, Kim KW, Hong KS, et al. (2015) Effects of Demineralized Dentin Matrix Used as an rhBMP-2 Carrier for Bone Regeneration. J Hard Tissue Biology 23(3): 415-422.
- Kim YK, Kwon KH, Lee ES, Kim CH, Kim MY, et al. (2015) Experimental Study on Human Demineralized Dentin Matrix as rhBMP-2 Carrier In Vivo. J Dent App 2(7): 269-273.
- Kim YK, Jang HJ, Um IW. (2016) A Case Report of Allogenic Demineralized Dentin Matrix loaded with Recombinant Human Bone Morphogenetic Proteins for Alveolar Bone Repair. Journal of Dental and Oral Health 2(6): 45-50.






























