Gastric Secretion and Upper Gastrointestinal Endoscopic Findings in End Stage Renal Disease Patients
Krishan L Gupta1*, Anil Gothwal1, Tarun Mittal1 and Usha Dutta2
1Department of Nephrology, Postgraduate Institute of medical Education and Research, India
2Department of Gastroenterology, Postgraduate Institute of medical Education and Research, India
Submission: August 31, 2017; Published: September 19, 2017
*Corresponding author: Usha Dutta, Department of Gastroenterology, Postgraduate Institute of medical Education and Research, India, Email: ushadutta@gmail.com
How to cite this article: Krishan L G, Anil G, Tarun M, Usha D. Gastric Secretion and Upper Gastrointestinal Endoscopic Findings in End Stage Renal Disease Patients. JOJ uro & nephron. 2017; 4(1): 555630. DOI: 10.19080/JOJUN.2017.04.555630
Abstract
Background: Impaired bicarbonate secretion is one of the proposed mechanisms for uremic gastrointestinal symptoms. This prospective case-control study was aimed to determine the effects of uremia on gastric acid and bicarbonate secretion in end stage renal disease (ESRD) patients on maintenance hemodialysis (MHD) and to correlate the bicarbonate secretion with morphological changes in the upper gastrointestinal tract and with the presence of H. pylori.
Methods and Materials: Gastric secretion analysis and upper gastrointestinal endoscopy (UGIE) were performed in 28 patients with ESRD on MHD with symptoms of acid peptic disease and 20 controls with similar symptoms but without evidence of kidney disease. Statistical analysis was done by student t-test and paired t-test.
Results: Rate of gastric juice secretion, acid output and volume of parietal cell secretion were significantly less in CKD patients as compared to patients without kidney disease. The non-parietal cell secretion, bicarbonate output and total acid secretion (acid output + bicarbonate output) were also significantly less in CKD patients as compared to controls. The acid bicarbonate output was not significantly different in CKD patients with endoscopic changes on UGIE as compared to CKD patients without significant endoscopic abnormalities.
Conclusion: The study indicates that there is a generalized secretory defect in ESRD patients leading to decrease in both acid and bicarbonate secretion. Symptoms of dyspepsia possibly occur because of an imbalance between acid and bicarbonate secretion rather than an absolute change in the amount of either acid or bicarbonate secretion.
Introduction
Gastrointestinal manifestations of uremia are non-specific and include nausea, vomiting, anorexia, epigastric pain, heart burn, hiccups and gastrointestinal hemorrhage. The pathogenesis of these abnormalities is poorly understood. Proposed mechanisms include increased back diffusion of hydrogen ions across the gastric mucosal barrier, hypergastrinemia, hyperacidity, hyper-parathyroidism, H. pylori infection and impaired defensive mechanisms like gastric bicarbonate secretion. The present study was undertaken to study the effects uremia on gastric acid and bicarbonate secretion in end stage renal disease (ESRD) patients on maintenance hemodialysis (MHD) and to correlate the bicarbonate secretion with morphological changes in the upper gastrointestinal tract and with the presence of H. pylori.
Patients and Methods
Twenty eight adults with ESRD on MHD with symptoms of acid-peptic disease were included as cases. Control group comprised of 20 patients with similar symptoms without evidence of kidney disease. After obtaining informed consent, all patients subsequently underwent estimation of gastric secretion and upper gastrointestinal endoscopy (UGIE).
For the purpose of estimating gastric bicarbonate secretion, patients were asked to report empty stomach. Use of antacids or anti-cholinergic drugs was prohibited. Ryle’s tube was introduced and positioned fluoroscopically in the gastric antrum. The overnight gastric juice was aspirated and discarded. The secretion in the next one hour was aspirated continuously by using a slow suction pump which works at a suction pressure of 3-5mmHg. Subjects were asked to spit out the saliva during the procedure so as to minimize the interference by salivary bicarbonate. If aspirate was bile stained then the entire sample was discarded and fresh one hour collection was done using after repositioning the Ryle’s tube. Venous blood was drawn for plasma osmolality at 30 minutes of the start of aspiration. Measurements included
i. Total volume of gastric juice to the nearest 0.5ml
ii. Osmolality of plasma and gastric juice and
iii. H+ ion concentration of gastric juice.
Osmolality was measured by freezing point depression method with the help of advanced digametic osmometer. Readings were taken in triplicate and the mean (expressed as milliosmoles per kilogram) taken for calculation H+ ion concentration was measured in-vitro by titration of gastric juice to pH 7.0 using 0.1N NaOH with the help of El top digital pH meter. All measurements were taken at room temperature.
Calculations were done by the method described by Feldman et al., two basic equations were used:
I. Measured acid output = Acid secreted - Acid neutralized
II. Measured osmolar output = Osmoles secreted - Osmoles neutralized
III. Bicarbonate secretion = Acid neutralized
UGIE was then performed on all CKD patients for visualization of lesions and taking biopsy (2cm) for rapid urease test (RUT) and histopathological examination for H. pylori.
Statistical analysis
The results were expressed as mean±SD. Statistical analysis was done by Student t-test and paired t-test.
Results
Amongst the case 21 (75%, n=28) were males where as all (100%, n=20) the patients in the control group were males. The mean age of cases was 36.3 years whereas it was 35.2 years for controls.
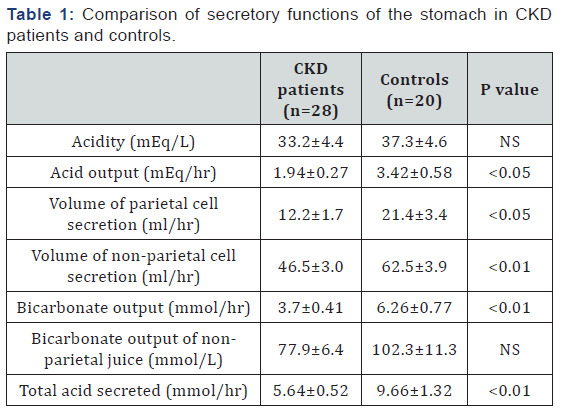
The rate of gastric juice secretion in CKD patients was 58.7±3.2ml/hr as compared to 83.8±6.1ml/hr in controls (p<0.05). Plasma osmolality in CKD patients was significantly higher (330.4±9.0mOsm/kg) in comparison to controls (294.4±3.4mOsm/k; p<0.01). Gastric juice osmolality followed the same pattern as that of plasma osmolality (CKD 218.2±8.1mOsm/kg, controls 156.7±10.6mOsm/kg; p<0.05). There was no difference in the gastric juice acidity, but the acid output and volume of parietal cell secretion were significantly less in gCKD patients as compared to controls (p<0.05) (Table 1). Five patients of CKD had achlorhydria.
The non-parietal cell secretion, bicarbonate output and total acid secretion (acid output + bicarbonate output) were significantly less in CKD patients as compared to controls. The bicarbonate concentration of the non-parietal juice was also less in CKD patients as compared to controls although the difference was not statistically significant (Table 1).
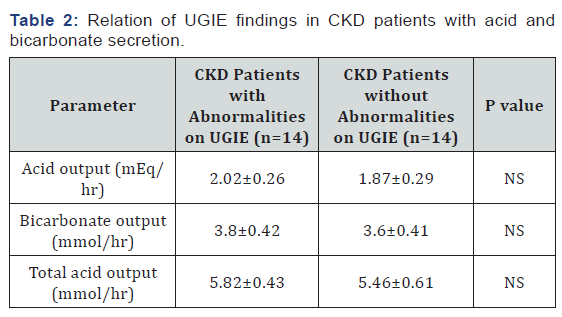
UGIE in CKD patients revealed no abnormality (n=14, 50%), esophagitis (n=10, 35.7%), gastritis (n=9, 32.1%), duodenitis (n=3, 10.7%) and duodenal ulcers (n=2, 7.1%). The acid output, bicarbonate output and total acid output were not significantly different in CKD patients with endoscopic changes on UGIE (n=14, 50%) as compared to those who had normal endoscopic findings (Table 2). Five patients (18.5%) of CKD had a positive rapid urease test and antral biopsies also revealed H. pylori. In patients with dyspepsia but no renal failure, H. pylori positivity was 45%.
Discussion
Although involvement of the gastrointestinal tract in CKD occurs frequently [1], its pathogenesis is poorly understood. Although gastric acid secretion and endoscopic studies have been done in uremic patients, data is scant on the correlation of gastric secretory function with endoscopic findings. Therefore, the present study was carried out to quantify both gastric acid and bicarbonate secretion and study their relation to gastric morphology.
In the present study, the basal acid output in ESRD patients was found to be significantly low as compared to subjects with dyspepsia and normal renal function. Earlier studies on acid output in kidney disease patients have shown conflicting results with some studies reporting hyperchlorhydria [2,3] while other showing normal or low acid output [4-6]. We also found that the bicarbonate output and bicarbonate concentration in the nonparietal cell secretion were low in CKD patients as compared to controls. These findings point towards a generalized secretory defect in ESRD patients leading to decrease in both acid and bicarbonate secretion. Symptoms of dyspepsia possibly occur because of an imbalance between acid and bicarbonate secretion rather than an absolute change in the amount of either acid secretion [7].
Abnormal findings on UGIE were observed in 50% of ESRD patients in this study. Previously, incidence of endoscopic abnormalities in CKD patients has been reported to be between 41.5-90.7% [8,9]. The reason for this wide variation in the incidence is not apparent but has been proposed to be due to differences in gastric acid status of different racial groups and partly due to subjective variation in interpretation of various inflammatory changes on endoscopy. Notwithstanding, the incidence of peptic ulcers was low (7.1%) in CKD patients, consistent with findings of other series (0-3.3%) [9,10].
The prevalence of H. pylori infection in CKD population reported in literature is quite varied (24%-88%) [10,11], with studies reporting both significantly higher [12] and lower [13] prevalence of H. pylori infection in uremic patients as compared to those with normal renal function. This discrepancy may be partly explained by the use of less sensitive and specific (techniques serologic or breath analysis) for the detection of H. pylori in a number of these studies. Using rapid urease test and antral biopsies for the detection of H. pylori, we found that the prevalence of H. pylori was less in CKD patients as compared to subjects with normal renal function [14-20].
Conclusion
The findings of the present study point towards a generalized secretory defect in ESRD patients leading to decrease in both acid and bicarbonate secretion. Symptoms of dyspepsia possibly occur possibly because of an imbalance between acid and bicarbonate secretion rather than an absolute change in the amount of either acid secretion.
References
- Khedmat H, Ahmadzad-Asl M, Amini M, Lessan-Pezeshki M, Einollahi B, et al. (2007) Gastro-duodenal lesions and Helicobacter pylori infection in uremic patients and renal transplant recipients. Transplant Proc 39(4): 1003-1007.
- Sullivan SN, Tustanoff E, Sluagfter DN , Linton AL, Lindsay RM, et al. (1976) Hypergastrinemia and gastric acid hypersecretion in uremia. ClinNephrol (5): 25-28.
- Dinoso VP, Murthy SN, Saris AL, Clearfield HR, Lyons P, et al. (1982) Gastric and Pancreatic function in patients with end-stage renal disease. J Clin Gastroenterol 4(4): 321-324.
- Prakash J, Agrawal BK (1991) Upper gastrointestinal mucosal lesions in chronic renal failure. Indian J Gastroenterol 10(4): 131-132.
- Reisman TN, Perez GO, Rogers AI (1976) Gastric secretory function in patients with chronic renal failure undergoing maintenance hemodialysis. Am J Dig Dis 21(12): 1044-1048.
- Mitchell CJ, Jewell DP, Lewin MR, McLaughlin JE, Moorhead JF (1979) Hypergastrinaemia in chronic renal failure. Br Med J 32(3): 208–213.
- Bacci MR, Chehter EZ (2013) Dyspepsia among patients with chronic kidney disease: a cross sectional study. Int Arch Med 6(1): 43.
- Margolis DM, Saylor JL, Geisse G, DeSchryver-Kecskemeti K, Harter HR, et al. (1978) Upper gastrointestinal disease in chronic renal failure. A prospective evaluation. Arch Intern Med 138(8): 1214-1217.
- Al-Mueilo SH (2004) Gastroduodenal lesions and Helicobacter pylori infection in hemodialysis patients. Saudi Med J 25(8): 1010-1014.
- Goenka MK, Kochhar R, Mehta SK, Nagi B, Malik AK, et al. (1989) Upper gastro-intestinal mucosal changes in patients with chronic renal failure. J Assoc Physicians India 37(9): 564-566.
- Davenport A, Shallcross TM, Crabtree JE, Davison AM, Will EJ, et al. (1991) Prevalence of Helicobacter pylori in patients with end-stage renal failure and renal transplant recipients. Nephron 59(4): 597-601.
- Karari EM, Lule GN, McLigeyo SO, Amayo EO (2000) Endoscopic finding and prevalence of Helicobacter pylori in chronic renal failure patients with dyspepsia. East Afr Med J 77(8): 406-409.
- Jaspersen D, Fassbinder W, Heinkele P, Kronsbein H, Schorr W, et al. [1995] Significantly lower prevalence of Helicobacter pylori in uremic patients than in patients with normal renal function. J Gastroenterol 30(5): 585-588.
- Loffeld RJ, Peltenburg HG, vd Oever H, Stobberingh E (1991) Prevalence of Helicobacter pylori antibodies in patients on chronic intermittent hemodialysis. Nephron 59(2): 250-253.
- Karari EM, Lule GN, McLigeyo SO, Amayo EO (2000) Prevalence of Helicobacter pylori in patients with chronic renal failure. Nephrol Dialysis Transplant 77(8): 406-409.
- Moustafa FE, Khalil A, Abdel-Wahab M (1997) Helicobacter pylori and uremic gastritis: a histopathologic study and a correlation with endoscopic and bacteriologic findings. Am J Nephrol 17(2): 165-171.
- Kang JY, Ho KY, Yeoh KG, Guan R, Wee A, et al. [199]Peptic ulcer and gastritis in uraemia, with particular reference to the effect of Helicobacter pylori infection. J GastroenterolHepatol ;14(8):771-778.
- Schoonjans R, Van VB, Vandamme W, Van HN, Verdievel H, et al. (2002) Dyspepsia and gastroparesis in chronic renal failure: the role of Helicobacter pylori. ClinNephrol 57(3): 201-207.
- Nakajima F, Sakaguchi M, Amemoto K, Oka H, Kubo M, et al. (2002) Helicobacter pylori in patients receiving long-term dialysis. Am J Nephrol 22(5-6): 468-472.
- Hammer J, Oesterreicher C, Hammer K, Koch U, Traindl O, et al. (1998) Chronic gastrointestinal symptoms in hemodialysis patients. Wien KlinWochenschr 110(8): 287-291.






























