Using A Metered Dose Inhaler (Mdi) With Spacer or Nebulizer for Managing Children Under 5 Years of Age with Exacerbation of Wheezing or Asthma
Kimberly Dimino DNP, RN, CCRN*
William Paterson University, Wayne NJ, Hackensack Meridian Health, Hackensack NJ, USA
Submission: December 09, 2019; Published: December 16, 2019
*Corresponding author: Kimberly Dimino, William Paterson University, Hackensack NJ, USA
How to cite this article: Dimino, K. Using A Metered Dose Inhaler (Mdi) With Spacer or Nebulizer for Managing Children Under 5 Years of Agewith Exacerbation of Wheezing or Asthma. 2019; 11(2): 555815.10.19080/JOJNHC.2019.11.555815
Abstract
Childhood asthma presents burdens on the child with asthma, their families, society, and healthcare. Inhaled short acting bronchodilators are the most common first line medications used to treat exacerbation of wheezing or asthma. Nebulizers and metered dose inhalers (MDIs) with spacers are among the most widely used devices to deliver these medications. Although evidence supporting MDIs as an efficient, more convenient and cost-effective way to administer inhaled medications to children, nebulizers continue to be seen as the gold standard for managing children under 5 years of age with exacerbation of wheezing or asthma.
Keywords: Asthma exacerbation Wheezing Management Preschool Metered dose inhaler with spacer Nebulizer
Abbrevations: MDI: Metered Dose Inhaler; MDI-spacer: Metered Dose Inhaler with Spacer
Introduction
Asthma is the third leading cause of hospitalization in children [1]. It is estimated that approximately 6 million children in the United States have asthma. Children under the age of 5 years have the highest rate of potentially preventable hospital stays for asthma [2]. The yearly direct healthcare cost of asthma care in the United States is estimated to be $50.1 billion [3]. Hospital stays demonstrate the largest portion of that cost. Indirect costs (e.g. loss of productivity-missed school days, parents missed workdays) make up another $5.9 billion for a total of $56 billion dollars per year. While the CDC (2018) reports that asthma exacerbations have gone down in children of all races and ethnicities from 2001 through 2016, emergency department and urgent care visits related to asthma exacerbations continue to be high among Non-Hispanic black children ages 0-4 years. Childhood asthma presents burdens on the child with asthma, their families, society, and healthcare [4]. Inhaled short acting bronchodilators are the most common first line medications used to treat wheezing or asthma exacerbations [5]. Nebulizers and metered dose inhalers (MDIs) are among the most widely used devices to deliver these medications. Although administration of bronchodilators via MDI is the most preferred and widely used for older children and adults, nebulizers continue to be seen as the “gold standard” for delivering bronchodilators in office and hospitals for treatment of wheezing or asthma exacerbations in children under 5 years of age [6-8]. In the past it was presumed by the medical community that infants and young children were unable to use MDIs effectively because they were unable to coordinate inhalation with medication administration [9]. The introduction of spacers, plastic tubes that hold inhaled medications in a chamber and allows a patient to inhale medications while taking normal breaths through a facemask or mouthpiece, helped solve this problem [10-12]. Adding a spacer onto an MDI increases the amount of inhaled drug that reaches the lung by 20% and reduces local and systemic side effects of inhaled medications [13]. In addition, MDIs used with spacers (MDI-spacers) are easier to use, less expensive, are portable, require less time and effort, do not require medication preparation or electricity, and require lower medication doses compared to nebulizers. In contrast, nebulizers are more expensive, inconvenient to use, require a power source and longer time for administration, require maintenance, and have been shown to have imprecise dosing. Additionally, nebulizers potentially pose a risk of infection [14]. In 2003 nebulizer use was associated with an outbreak of severe acute respiratory syndrome (SARS). Transitions from nebulizer to MDI-spacers in older children and adults have been found to decrease supply and labor costs and total number of treatments necessary to manage wheezing or asthma exacerbations [15,16]. Most recently [17] conducted a meta-analysis to compare the efficacy of pediatricasthma exacerbation and wheezing treatment in the emergency department by nebulizer and MDI-spacers and identified no differences in cardiorespiratory frequency, O2 saturation, and asthma scores upon between inhalation the two treatment techniques. Additionally, most current pediatric wheezing and asthma management guidelines recommend the use of MDIspacers and age-appropriate masks or holding chambers for all children [18]. Despite evidence identifying MDI-spacers as at least as effective as nebulizers in the delivery of short acting bronchodilators to manage children under 5 years of age with exacerbation of wheezing or asthma, nebulizers are still widely used as the modality of choice to administer bronchodilators to children under the age of five.
The purpose of this article is to identify and discuss literature identifying the use of a MDI-spacer as at least as effective as nebulizers in the delivery of short acting bronchodilators to manage children under 5 years of age with exacerbation of wheezing or asthma.
Literature search
English language publications (2000 to 2019) randomized clinical trials were retrieved from CINAHL, Scopus, and Cochrane databases. Seven journal articles met the specific criteria for this systematic review (2 meta-analysis and 5 randomized control trials). The primary outcome measure was efficacy of asthma exacerbation management in children under 5 years of age.
Results
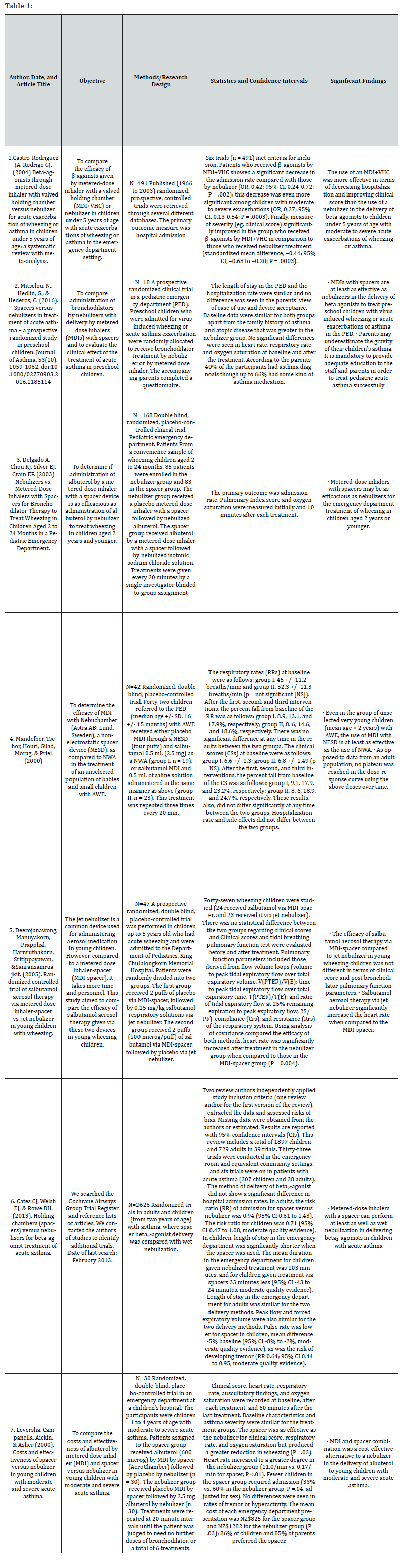
Castro-Rodriguez JA et al. [19] conducted a meta-analysis to compare the efficacy of a bronchodilator given by MDIs with spacers or nebulizers in children under 5 years of age with exacerbations of wheezing or asthma in an emergency department setting. Castro-Rodriguez JA et al. [19] analyzed databases published (1966 to 2003) randomized, prospective, controlled trials with a primary outcome measure of hospital admissions and identified six trials (n = 491) that met criteria for inclusion. Patients who received a bronchodilator by MDI with spacer showed significant decreases in hospital admission rates compared with those treated with nebulizers (OR, 0.42; 95% CI, 0.24-0.72; P = .002); decreases were noted to be more significant among children with moderate to severe exacerbations (OR, 0.27; 95% CI, 0.13-0.54; P = .0003). Finally, measured severity (eg, clinical score) and identified significant improvement in the group who received a bronchodilator by MDI with spacer in comparison to those who received nebulizer treatments (standardized mean difference, −0.44; 95% CI, −0.68 to −0.20; P = .0003).
Niki Mitselou et al. [20] compared administration of bronchodilators by nebulizers to MDIs with spacers and evaluated the clinical effect of thesss treatment of acute asthma in preschool children. A prospective randomized clinical trial in a pediatric emergency department (PED) was employed. Preschool children who were admitted for virus induced wheezing or acute asthma exacerbation were randomly allocated to receive bronchodilator treatment by nebulizer or by MDI with spacer. Their parents completed questionnaires. Identified no differences in parents’ views of ease of use and device acceptance, length of stay in the pediatric emergency department, and rates of hospitalization rates. Additionally, significant differences were not seen in respiratory rates, oxygen saturations, or heart rate at baseline or after treatment. Parents reported that 40% of the young children had a diagnosis of asthma while up to 66% were taking some type of asthma medication. Sought, to examine if efficacy of administration of albuterol, a bronchodilator, by MDI with a spacer vs. nebulizer to treat wheezing in children aged 2 years and younger. Double blind, randomized, placebo-controlled clinical trial was employed. A convenience sample of wheezing children aged 2 to 24 from a pediatric emergency were studied. Eighty-five patients were enrolled in the nebulizer group and 83 in the spacer group (N=168). The nebulizer group received a placebo MDI with a spacer followed by nebulized albuterol. The spacer group received albuterol by a MDI with a spacer followed by nebulized isotonic sodium chloride solution. Treatments were given every 20 minutes by a single investigator blinded to group assignment. Admission rate was the primary outcome measured. Pulmonary Index score and oxygen saturation were measured initially and 10 minutes after each treatment. Delgado, Chou, Silver, and Crain (2003) found that the nebulizer group had a significantly higher mean (SD) initial Pulmonary Index score compared with the spacer group (7.6 [2.5] vs. 6.6 [2.0]; P = .002). With the initial Pulmonary Index score controlled, children in the spacer group were admitted less (5% vs. 20%; P = .05). Analyses revealed an interaction between group and initial Pulmonary Index score and lower admission rates in the spacer group were found primarily in the children experiencing severe asthma exacerbations. Mandelber et al. [21] sought to determine the efficacy of MDIs with spacers compared to nebulizers in the treatment of a population of babies and small children with exacerbation of asthma. The design of the study was a randomized, double blind, placebocontrolled trial. Forty-two children referred to the PED (median age ± SD, 16 ± 15 months) with exacerbation of asthma received either placebo MDI with spacer (four puffs) and salbutamol 0.5 mL (2.5 mg) as a nebulizer (group I, n = 19), or salbutamol MDI and 0.5 mL of saline solution administered in the same manner as above (group II, n = 23). This treatment was repeated three times every 20 min. Found that respiratory rates (RRs) at baseline were as follows: group I, 45 ± 11.2 breaths/min; and group II, 52.3 ± 11.3 breaths/min (p = not significant [NS]). After the first, second, and third interventions, the percent fall from baseline of the RR were as follows: group I, 8.9, 13.1, and 17.9%, respectively; group II, 8.6, 14.6, and 18.6%, respectively. There was no significant difference at any time in the results between the two groups. The clinical scores (CSs) at baseline were as follows: group I, 6.6 ± 1.3; group II, 6.8 ± 1.49 (p = NS). After the first, second, and third interventions, the percent fall from baseline of the CS were as follows: group I, 9.1, 17.9, and 23.2%, respectively;group II, 8.6, 18.9, and 24.7%, respectively. These results, also, did not differ significantly at any time between the two groups. Hospitalization rate and side effects did not differ between the two groups. Deeronjanawong J [22] aimed to compare the efficacy of bronchodilator therapy administered via MDI with spacer and nebulizer in wheezing children up to 5 years old. A prospective randomized, double blind, placebo-controlled trial was employed. Patients were randomly divided into two groups. The first group received 2 puffs of placebo via MDI-spacer, followed by 0.15 mg/ kg salbutamol respiratory solutions via jet nebulizer. The second group received 2 puffs (100μg/puff) of salbutamol via MDIspacer, followed by placebo via jet nebulizer. Clinical scores and tidal breathing pulmonary function test were evaluated before and after treatment. Pulmonary function parameters included those derived from flow volume loops (volume to peak tidal expiratory flow over total expiratory volume, VPTEF/VE; time to peak tidal expiratory flow over total expiratory time, TPTEF/TE; and ratio of tidal expiratory flow at 25% remaining expiration to peak expiratory flow, 25/PF), compliance (Crs), and resistance (Rrs) of the respiratory system. The efficacy of both methods was compared using analysis of covariance. Forty-seven wheezing children were studied (24 received salbutamol via MDI-spacer, and 23 received it via nebulizer). No statistical differences were noted in clinical scores and pulmonary function between the two groups. However, heart rates were noted to significantly increase after treatment in the nebulizer group when compared to those in the MDI-spacer group (P = 0.004).
Cates CJ [23] conducted a meta-analysis to assess the effects of MDI with spacers compared to nebulizers for the delivery of bronchodilators for acute asthma. Cochrane Airways Group Trial Register and reference lists of articles were searched. Randomized trials in adults and children (from two years of age) with asthma, where spacer bronchodilator delivery was compared with wet nebulization. Two review authors independently applied study inclusion criteria (one review author for the first version of the review), extracted the data and assessed risks of bias. Results are reported with 95% confidence intervals (CIs). A total of 1897 children and 729 adults in 39 trials were included in this review. Thirty-three trials were conducted in the emergency room and equivalent community settings, and six trials were on in patients with acute asthma (207 children and 28 adults). Methods of delivery of bronchodilators did not show significant differences in hospital admission rates. In adults, the risk ratio (RR) of admission for spacer versus nebulizer was 0.94 (95% CI 0.61 to 1.43). The risk ratio for children was 0.71 (95% CI 0.47 to 1.08, moderate quality evidence). Length of stay in the emergency department was significantly shorter for children when the spacer was used (mean duration in the emergency department for children given nebulized treatment was 103 minutes, and for children given treatment via spacers 33 minutes less) (95% CI -43 to -24 minutes, moderate quality evidence). No significant differences were noted in length of stay in the emergency department, orpeak flow and forced expiratory volume between the two devices. Pulse rates were noted to be lower in children who used spacers (mean difference -5% baseline (95% CI -8% to -2%, moderate quality evidence), as was the risk of developing tremor (RR 0.64; 95% CI 0.44 to 0.95, moderate quality evidence).
Leversha [24] sought to compare the costs and effectiveness of albuterol by MDI and spacer versus nebulizer in young children with moderate and severe acute asthma. Randomized, double blind, placebo-controlled trial was conducted in an emergency department at a children’s hospital. Children 1 to 4 years of age with moderate to severe acute asthma participated in this study. Children assigned to the spacer group received albuterol by MDI with spacer (Aero Chamber) followed by placebo by nebulizer (n = 30). The nebulizer group received placebo MDI with spacer followed by 2.5 mg albuterol by nebulizer (n = 30). Treatments were administered at 20-minute intervals until their status improved and they did not need further doses, or a total of 6 treatments were administered. Baseline clinical score, heart rate, respiratory rate, auscultatory findings, and oxygen saturation were recorded at the beginning, after each treatment, and 60 minutes after the last treatment. Both groups had similar characteristics and asthma severity. The spacer was found to be as effective as the nebulizer for clinical score, respiratory rate, and oxygen saturation but produced a greater reduction in wheezing (P =.03). Heart rate increased to a greater degree in the nebulizer group (11.0/min vs. 0.17/min for spacer, P <.01). Fewer children in the spacer group required admission (33% vs. 60% in the nebulizer group, P =.04, adjusted for sex). No differences were seen in rates of tremor or hyperactivity. The mean cost of each emergency department presentation was $825 for the spacer group and $1282 for the nebulizer group (P =.03); 86% of children and 85% of parents preferred the spacer.
Discussion
The primary objective of this article was to identify and discuss literature identifying the use of MDI-spacer as at least as effective as nebulizers in the delivery of short acting bronchodilators for managing children under 5 years of age with exacerbation of wheezing or asthma. All seven studies in this review compared the use of MDI-spacers and nebulizers for managing children under 5 years of age with wheezing or asthma exacerbations. The two meta-analysis reported results with 95% confidence intervals. Overall, all of the studies identified that MDIs used with spacers were at least as effective if not better than nebulizers in the delivery of bronchodilators to treat children under the age of 5 years with exacerbations of wheezing or asthma. Among these studies, one revealed that the use of an MDI-spacer decreased hospitalization and improved clinical scores, decreases were noted to be more significant among children with moderate to severe exacerbations. Lower admission rates were also noted in one of the studies in an MDI with spacer group. Length of stay in the emergency department was found to be significantly shorterin one of the studies when an MDI-spacer was used. Three studies showed that bronchodilator therapy via nebulizer increased the heart rate when compared to the MDI-spacer and amongst them one noted decrease in risk of developing tremors. Another study revealed that although the MDI-spacer was as effective as the nebulizer for clinical score, respiratory rate, and oxygen saturation, the MDI-spacer significantly reduced wheezing. This review also identified that an MDI-spacer was a more cost-effective alternative to a nebulizer in the delivery of inhaled medication. Interestingly, one study found that 40% of the preschool children who participated in the study had a diagnosis of asthma however, 66% of them were on some type of asthma medication. This finding speaks to the continued mismanagement of pediatric asthma. One of the studies also noted that 86% of the preschool children and 85% of parents preferred the MDI-spacer to the nebulizer. One of the studies noted the importance of understanding that parents may underestimate the gravity of their children’s asthma and recommended that adequate education be provided to staff and parents in order to treat pediatric exacerbations of asthma successfully (Table 1).
Summary Points
a) MDI-spacers are at least as effective if not better than nebulizers in the delivery of bronchodilators used to treat children under the age of 5 years with exacerbations of wheezing or asthma
b) MDI-spacers decreased hospitalization and improved clinical scores
c) Lower admission rates were identified in MDI-spacers group compared to a nebulizer group
d) Length of stay in the emergency department was found to be significantly shorter when an MDI-spacer was used compared to a nebulizer
e) Bronchodilator therapy via nebulizer increased the heart rate and risk of developing tremor when compared to the MDI-spacer
f) MDI-spacer significantly reduced wheezing when compared to the nebulizers
g) MDI-spacer were found to be a more cost-effective alternative to a nebulizer in the delivery of inhaled medication
h) 40% of the preschool children who participated in one of the studies had an actual diagnosis of asthma while 66% of the preschool were taking some type of asthma medication
i) 86% of the preschool children and 85% of parents who participated in one of the studies preferred the MDI-spacer over the nebulizer
j) Parents may not recognize the seriousness of their child’s diagnosis of asthma
Barriers
Frequent emergency department visits for pediatric asthma care is associated with lack of education regarding diagnosis and medication use [25,26] found children frequently used MDIsspacers incorrectly and recommended that repeated education would result in better medication therapy. A study [27] found that caregivers of urban, minority children with persistent asthma lacked proper techniques for effective use of MDIs with spacers which suggests the potential value of educational interventions. Recommended interventions for successful adherence promotion include providing reinforcement for caregivers, providing feedback on progress, tailoring education to individual needs and circumstances, standardizing therapy, and continuity of care and education [28].
Conclusion
The MDI-spacer is an efficient, more convenient and costeffective way to delivering first line bronchodilators to children under the age of 5 years with exacerbations of wheezing or asthma. Despite evidence and guidelines, nebulizers continue to be the device of choice. It is well documented that children under the age of 5 years have the highest rate of potentially preventable hospital stays for asthma. While asthma can be a manageable disease, children under the age of 5 years, particularly those who are minorities and/or whose family’s incomes fall below poverty lines, are not, as a population, receiving evidence based, patient and family centered asthma care [29,30]. Therefore, it is imperative to identify, implement, and disseminate evidenced based approaches to ensure improvement and high standards of asthma care among all children, especially those under the age of 5 years with exacerbations of wheezing or asthma. This review ofthe most current evidence supports recommended guidelines and provides further evidence of the efficacy of using an MDI-spacer compared to nebulizers for children under the age of 5 years with exacerbations of wheezing or asthma. The identification and implementation of evidence-based practice, a systematic approach to clinical decision making that aims to provide the most current, consistent and best possible care to patients, can improve outcomes for young children with asthma [31]. Although the use of MDI-spacers is an efficient, more convenient and cost-effective way to administer inhaled medications to children under the age of 5 years with wheeze or asthma exacerbations, it is not widely used. There is a need for well-designed pediatric asthma studies that assess outcomes of implementing evidence based approaches and costs for improving asthma care. Patient satisfaction also needs to be considered as it is an important aspect of patient and family centered care and has been found to significantly correlate with better outcomes [32-38]. Efforts need to be targeted to improve pediatric management in urban communities, decrease risks of exacerbations, standardize and improve therapy, and reduce costs by reducing overuse of healthcare resources.
References
- Centers for Disease Control and Prevention (CDC) (2010) Behavioral Risk Factor Surveillance System Survey Data.
- Fingar K, Washington R (2015) Potentially Preventable Pediatric Hospital Inpatient Stays for Asthma and Diabetes 2003-2012 HCUP Statistical Brief.
- Barnett, Nurmagambetov (2011) Costs of Asthma in the Unites States: 2002-2007. Journal of Allergy and Clinical Immunology 127(1): 145-152.
- Tjard R, Bart P, Reiner P, Richard P, Hans T (2002) Randomized controlled economic evaluation of asthma self-management in primary health care. American Journal of Respiratory and Critical Care Medicine 166(8): 1062- 1072.
- Global strategy for asthma management and prevention (2006) Global initiative for asthma (GINA).
- National Institute of Health (2002) Guidelines for the diagnosis and management of asthma. National Institutes of Health, National Heart, Lung, and Blood Institute National Asthma Education and Prevention Program.
- British Thoracic Society and others (2003) Guidelines on the management of asthma: management of acute asthma. Thorax 58(Suppl): i32-50.
- Global Initiative for Asthma Global Strategy for Asthma Management and Prevention (2016).
- Scichilone N (2015) Asthma Control: The Right Inhaler for the Right Patient. Advances in Therapy 32(4): 285-292.
- Ortiz-Alvarez O, Mikrogianakis A (2012) Managing the paediatric patient with an acute asthma exacerbation. Canadian.
- Chou KJ, Cunningham SJ, Crain EF (1995) Metered dose inhalers with spacers vs nebulisers for paediatric asthma. Arch Paediatr Adolesc Med 149(2): 201-205.
- Delgado A, Chou KJ, Silver EJ, Crain EF (2003) Nebulizers vs Metered-Dose Inhalers with Spacers for Bronchodilator Therapy to Treat Wheezing in Children Aged 2 to 24 Months in a Pediatric Emergency Department. Arch Pediatr Adolesc Med 157(1): 76-80.
- Moore R (2015) The use of inhaler devices in children.
- Lee D, Hui A, Wu P, Chan P, Cameron, et al. (2003) A major outbreak of severe acute respiratory syndrome in Hong Kong. N Engl J Med 348(20): 1986-1994.
- Tenholder MF, Bryson MJ, Whitlock WL (1992) A model for conversion from small volume nebulizer to metered dose inhaler aerosol therapy. Chest 101(3): 634-637.
- Turner, Gafni, Swan, Fitzgerald (1996) A review and economic evaluation of bronchodilator delivery methods in hospitalized patients. Arch Intern Med 156(18): 2113-2118.
- Roncada C, Andrade J, Bischoff LC, Pitrez PM (2018) Comparison of two inhalational techniques for bronchodilator administration in children and adolescents with acute asthma crisis: a meta-analysis. Revista paulista de pediatria: orgao oficial da Sociedade de Pediatria de Sao Paulo 36(3): 364-371.
- American Heart Association (AHA) (2016) Pediatric advanced life support.
- Castro-Rodriguez JA, Rodrigo GJ (2004) Beta-agonists through metered-dose inhaler with valved holding chamber versus nebulizer for acute exacerbation of wheezing or asthma in children under 5 years of age: a systematic review with meta-analysis. J Pediatr 145(2): 172-177.
- Niki Mitselou, Gunilla Hedlin, Carl-Axel Hederos (2016) Spacers versus nebulizers in treatment of acute asthma a prospective randomized study in preschool children. Journal of Asthma 53(10): 1059-1062.
- Mandelber A, Tsehori S, Houri S, Gilad E, Morag B, et al. (2000) Nebulized Aerosol Treatment Necessary in the Pediatric Emergency Department? Chest 117(5): 1309-1313.
- Deeronjanawong J, Manuyakorn W, Prapphal N, Harnruthakorn C, Sritippayawan S, et al. (2005) Randomized controlled trial of salbutamol aerossol therapy via metered dose inhaler-spacer vs. jet nebulizer in young children with wheezing. Pediatr Pulmonol 39(5): 466-472.
- Cates CJ, Welsh EJ, Rowe BH (2013) Holding chambers (spacers) versus nebulisers for beta-aganist treatment of acute asthma. Cochrane Database of Systematic Reviews 13(9): CD000052.
- Leversha, Campanella, Aickin, Asher (2000) MI Costs and effectiveness of spacer versus nebulizer in young children with moderate and severe acute asthma. J Pediatr 136(4):497-502.
- Al-Muhsen S, Horanieh N, Dulgom S, Aseri ZA, Vazquez-Tello A, et al. (2015) Poor asthma education and medication compliance are associated with increased emergency department visits by asthmatic children. Annals of Thoracic Medicine 10(2): 123-131.
- Harnett CM, Hunt EB, Bowen BR, O'Connell OJ, Edgeworth DM, et al. (2014) A study to assess inhaler technique and its potential impact on asthma control in patients attending an asthma clinic. J Asthma 51(4): 440-445.
- Reznik, Silver, Cao (2014) Evaluation of MDI-spacer utilization and technique in caregivers of urban minority children with persistent asthma. Journal of Asthma 51(2): 149-154.
- Eakin MN, Rand CS (2012) Improving Patient Adherence with Asthma Self-Management Practices: What Works? Annals of Allergy, Asthma & Immunology: Official Publication of the American College of Allergy, Asthma, & Immunology 109(2): 90-92.
- Centers for Disease Control and Prevention (CDC) (2016) Measures to Identify and Track Racial Disparities in Childhood Asthma: Asthma Disparities Workgroup Subcommittee Recommendations. Standards Subcommittee of the Asthma Disparities Workgroup.
- Environmental Protection Agency (EPA) (2012) Coordinated Federal Action Plan to Reduce Racial and Ethnic Asthma Disparities.
- Cunningham S, Logan C, Lockerbie L, Dunn MJ, McMurray A, et al. (2008) Effect of an integrated care pathway on acute asthma/wheeze in children attending hospital: Cluster randomized trial. J Pediatr 152(3): 315-320.
- Bjermer (2014) The importance of continuity in inhaler device choice for asthma and chronic obstructive pulmonary disease. Respiration 88(4): 346-352.
- Chapman, Boulet, Rea, Franssen (2008) Sub- optimal asthma control: prevalence, detection and consequences in general practice. Eur Respir J 31(2): 320-325.
- Leo H, Song B (2017) Spacers Versus Nebulizers in Treatment of Acute Asthma - A Prospective Randomized Study in Preschool Children. Pediatrics 140(3): S218.
- National Heart, Lungs, Blood Institute (2007) Blood Guidelines for the Diagnosis and Management of Asthma (EPR-3).
- Pravikoff, Tanner, Pierce (2005) Readiness of U.S. nurses for evidence-based practice. American Journal of Nursing 105(9): 40-51.
- Turkel, Yilmaz, Yuksel (2016) Metered dose inhaler-spacer use education effects on achieve asthma control in children. Tuberk Toraks 64(2): 105-111.
- Virchow JC, Crompton GK, Dal Negro R, Pedersen S, Magnan A, et al. (2008) Importance of inhaler devices in the management of airway diseases. Respir Med 102(1): 10-19.






























