Use of Cold Plasma Technology in Biomaterials and Their Potential Utilization in Controlled Administration of Active Substances
R ReynaMartínez1, RI Narro Céspedes1*, MC Ibarra Alonso2 and YK Reyes Acosta1
1Faculty of Chemical Sciences, Autonomous University of Coahuila, Blvd V Carranza e Ing, México
2Conacyt, Autonomous University of Coahuila, Blvd V Carranza e Ing, México
Submitted: May 26, 2018; Published: June 06, 2018
*Corresponding author: Abraham Tamir, Emeritus Professor of Ben Gurion, University of the Negev, Israel.
How to cite this article: R ReynaMartínez, R N Céspedes, M I Alonso, Y R Acosta. Use of Cold Plasma Technology in Biomaterials and Their Potential Utilization in Controlled Administration of Active Substances. JOJ Material Sci. 2018; 4(5): 555649. DOI: 10.19080/JOJMS.2018.04.555649
Abstract
Currently the controlled supply of drugs or active substances, in biomedical applications, has reached a high scientific and technological interest, mainly due; to the ability to maintain the optimal concentration of an active substance in the desired location, the greatest attention is directed towards biopolymers and bioceramics, due to the various qualities they exhibit such as: biocompatibility, bioactivity, biodegradability, etc. On the other hand, it is well known that the release profile depends crucially on the surface of these biomaterials. To enhance the advantages of the use of these biomaterials in the controlled dosage of drugs, they can be treated by non-thermal plasma, thus improving their surface characteristics (greater functionality and porosity), without altering the integral properties of the biomaterial. The plasma treatment for surface modification of materials is; green, economical, fast and efficient, by varying the parameters in plasma reactors can be adjusted physical and chemical characteristics in treated materials. This review highlights the most recent approaches to the treatment of plasma applied to different biomaterials, which aims to elucidate the outlook that exists regarding its potential application in the area of administration of drugs or active substances, so that this review can be used as a general guide for the selection of future biomaterials treated by plasma with application in controlled dosing.
Keywords: Cold plasma technology; Biomaterials; Drug delivery systems
Abbrevations: DBD: Dielectric Barrier, RF: Radio Frequency; PP: Polypropylene; CTAB: Hexadecyl Trimethyl Ammonium Bromide; PLA: Polylactic Acid; E-RL: Eudragit RL; DOX: Drug Doxorubicin
Introduction
Research in plasma science and technology, particularly non-thermal or cold plasma, has been increasing in recent years, demonstrating a very significant potential for various applications. Currently, surface modification by cold plasma has been widely diversified, being used in industries such as textiles, automotive, polymers, electronics and among others, which use the modification of various surfaces for the solution of different problems or improvements that It requires a specific product made to measure. One of the advantages of this technology is that surface properties of the materials can be manipulated according to the specific needs of the client. As a result of this, surface modification by plasma has become a phenomenon of scientific interest since it has many different benefits ranging from improving physical properties, modifying thermal properties, inhibiting microbial growth, as well as various applications in biomedicine. This field of study involves materials with very high intrinsic requirements that require certain specific physicochemical properties in addition to biocompatibility, bioactivity, sterilization and in some cases biodegradability, and that are generally not available in nature so, to face this problematic, composite materials are used that combine the specifications of their components. The cold plasma technology can offer alternatives to deal with this problem by implementing efficient surface modification routes that lead to obtaining materials with the desired requirements. All the advantages mentioned above can be exploited in the application of release of active substances, since through the treatment of plasma the kinetics of drug release can be altered, either by introducing reactive groups or by erosion on the surface: This is undoubtedly generating a new perspective in drug delivery systems that can offer greater efficacy or provide a more beneficial therapy
Background
The term plasma refers to an almost neutral ionized gas, composed mainly of photons, ions and free electrons, as well as atoms in the fundamental states or excited with a neutral net [1]., it was introduced by Langmuir in 1929 and is known as the fourth state of matter [1]. Other authors refer to it as the ionized state of a gas, where atoms, molecules, radicals and electrons are in a highly excited state, emitting UV and visible radiation [1]. Also, it is known that plasma is found abundantly in the universe, in the stars, nebula, however under earth conditions it is scarce. Plasma can be classified according to different parameters, one of the main ones being temperature, in this way it can be classified in two types; thermal plasma (of high temperature) that is characterized by a high percentage of ionization that it generates, the high temperatures and overall the thermodynamic equilibrium that is found [2]; and non-thermal plasma (of low temperature) presents a thermal diphase that makes it be in a thermodynamic disequilibrium. The pressures with which the latter work with are relatively low [2]. Coming up next the main advantages of using non-thermal or cold plasma are described and the sources involved in the surface modification of biomaterials.
Non-Thermal Plasma
Previously, the cold plasma was generated in low-pressure that limited its applications. However, the recent advances in plasma engineering has allowed the generation of this system at atmospheric pressure, which has taken it to a higher investigation in various applications of science and engineering [3]. In Figure 1 a summary of the main applications of cold plasma technology.

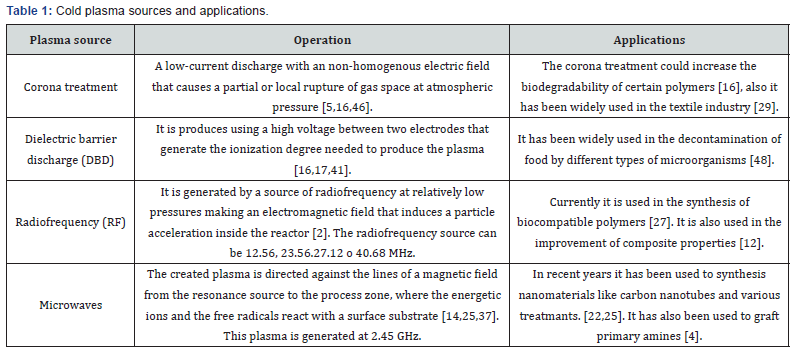
Main Sources of Cold Plasma
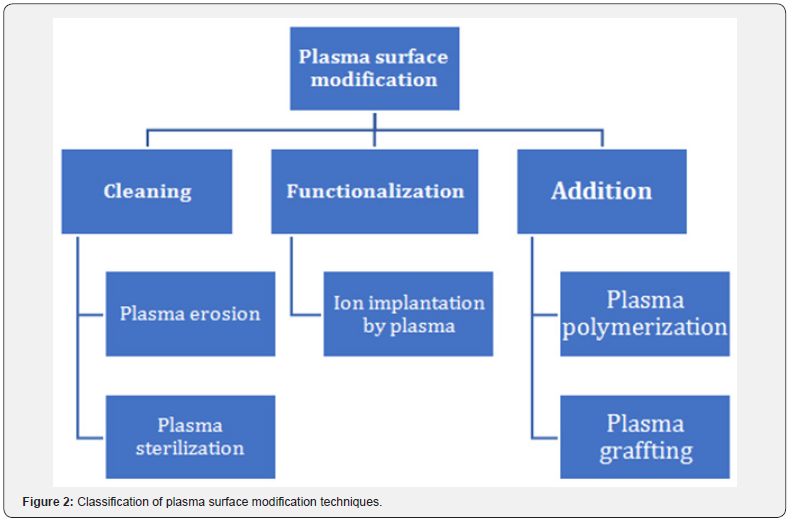
Various types of cold plasma generation systems exist and have been used for different applications. In Table 1 some plasma sources are shown. In general, the plasma modification techniques can be classified in three categories (Figure 2), depending on the transformation characteristics of the surface [4]. Each technique alters the properties of biomaterial surfaces in a different degree going from surface cleaning, surface functionalization and/or the addition of materials that confer wanted properties.
Biomaterials
A biomaterial is defined as any material natural or synthetic that constitutes a part of a biological system or a biomedical device that does, improves or replaces a natural function without provoking toxic, immunogenic, trombogenic, carcinogenic or irritating reaction [5,6]. Once these properties are satisfied it can be said that a biomaterial is and should be biocompatible, however, according to the needs and the understanding that has been accomplished with the interaction of these materials with live systems, the concept of biomaterial has evolved with time [7-9]. In the last years, biomaterial science has investigated different types of materials and its applications that replace o restore tissue functions, compromised or degenerated organs [10]. However, many materials that have essential mechanical properties for the fabrication of prosthesis show a deficient integration with human tissues or biological systems, thus the induce blood clots and strange reactions in the body that lead to implantation failure [11]. Examples of failure and negative induced include tissue necrosis, infection, calcification, extensive fibrosis and scar formation. However, the human body response to a biomaterial can be partially optimized by the chemical change of its surface.
Various methods of surface engineering can be classified in two main groups: physicochemical and biological. In the physicochemical methods are the wet chemical processing, the grafting of functional groups, the methods that use material irradiation like cold plasmas, ion or electron beams, lasers, photolithography [12, 13]. Between all the used techniques for surface engineering, those bases on plasma technology have demonstrated being extremely efficient in improving the surface properties of a material without affecting general mass properties of the materials [14]. The surface properties can be finely defined varying plasma parameters adapted to produce different types of surfaces depending on the specific application and requisites [15]. Also, the plasma process is controllable and reproducible, it provides sterile surfaces and can be transferred easily to the industrial production, in Figure 3 some properties obtained thanks to the plasma technology are shown.

Plasma and Biomaterials
The effectiveness of plasma treatments is influenced by diverse factors, like plasma source and reactor design, the functioning conditions of plasma, type of precursor used and the nature of the processed material [10]. The selection of these factors allow the adequate surface modification of various materials opening various application doors, this is due to the plasma treatments leading to the surface modification, deposition and incrustation of functional groups, also by plasma treatment the surface rugosity can be controlled, reticular formation, polymerization grafting and polymeric surface covering with a thin film [16]. In Figure 4 a scheme of surface modifications by plasma treatments is shown. After reviewing some technical aspects of plasma and biomaterial backgrounds, examples of material processing by this technology will be analyzed. These examples will show how the compatibility of the biological system and the material can be influenced greatly by the surface properties and how these depend on the treatment that is done to the material.

Application of Plasma in Biomaterials
The improvement of surface properties modified with plasma has fortified many promising biomedical applications, including surgical tools and medical devices for orthopedic, dental and cardiovascular implants. The versatility of the plasma techniques provides a broad range of surface modification capacities, that allows applications that go from the atomic level cleaning to the biomedical implant restructuring [4,17]. The non-reactive plasma gases like air and argon, have been used to clean and sterilize surgical tools and medical devices by the elimination of proteinic contaminants on the surface and in this wat avoid potential infection in the implantation site [17-19]. The surface of a material can be sterilized by the interaction of different gas species [20] or atmospheric air [21,22]. The functioinalization by plasma can be applied in order to disinfect and sterilize biomedical devices than can be damaged by chemical or extreme temperature conditions [22]. For example, some applications that have used argon plasma treatment at low pressure is that used for dental cleaning dental [23], and it has been used for in cold plasma polymerization against cutaneous tuberculosis [24]. The deposition of molecular plasma can be used to deposit biomolecules like aminoacids in the form of biased substrate solutions or suspensions, to improve the bioactivity of the metallic implants [25]. The bioactive implants have also united covalently deposited films by plasma like heptafluorobutyric metacrylate films for endovascular antitrombogenic stents [26]. Said implants modified with plasma to be bioresorbable can generate soft issued, membranous tissues and organs like cardiovascular tissues, skin, ligands, tendons, periodontal fibers or correction of hernias that can induce fibrosis [27].
In 2018 Canullo et al. [23] did a study to improve the murine osteoblasts adhesion over different grafted materials with nonatmospheric argon plasma. In the study it was demonstrated that the argon plasma treatment is capable of increasing the protein adsorption and the cellular adhesion in different classes of grafted materials like hydroxyapatite and calcium dibasic phosphate [23]. It is important to mention that the protein adsorption plays a fundamental role in the production of biocompatible implants. The biocompatibility of materials depends in great measure of the interactions that occur between the surface and the biological environment. It is known that the content of a live body with a material induces the adsorption of proteins on its surface, creating an interphase, in which other proteins and cells are adsorbed [28]. Many studies have demonstrated that surface functional groups can affect the adsorption of proteins and the following cell responses [29]. Therefore the biomaterials with an adequate and adhered surface layer can improve the biocompatibility of the medical devices changing the adsorption of proteins proteins [30]. Plasma polymerization can be used to create hydrophilic and antithrombogenic surfaces [4], and plasma treatment can also be used to induce the death of cancer cells [31-33]. For example, Partecke et al. demonstrated the possible use of atmospheric plasma (Jet plasma) of argon in cancer therapy in vitro, as well as in vivo [34]. The authors hypothesized that this approach can potentially be applied to increase the apoptosis of residual cancer cells after the resection of malignant tumors, they found that plasma treatment in this type of tumors caused cell death in pancreatic cancer cells in In vitro, it took up to 10 seconds of treatment to induce apoptosis, while the longer application caused non-apoptotic cell death, in addition to this all effects were leveled after approximately 72 hours after treatment (Figure 5).

Plasma treatment can also be used to decrease bacterial adhesion and improve the wettability of contact lenses, thus providing greater comfort for the user [23,35-38]. This can be achieved by increasing the hydrophilicity of the surface of the biomaterial with the oxidation of the plasma or by depositing a layer of thin film on its surface [37]. In this way, plasma technology has been used to improve the antimicrobial properties of some biomaterials, due to the high requirements of biomaterials in terms of biosecurity, another example of this is the study by Feng et al. [39] In 2016, they modified by plasma and a beam of metallic biomaterial ions such as titania incorporating silver ions to improve the antibacterial properties without compromising the compatibility with osteoblastic cells, in this material implanted with silver ions, the electron storage capacity of the nanoparticles can facilitate the transfer of electrons between the titania membranes and the bacteria that results in the death of these microorganisms. These reactions simultaneously promote cell growth, proliferation and differentiation (Figure 6).

It has also been reported the manufacture of improved surgical meshes incorporating antibiotics within its structure for the local treatment of possible postoperative infections [40]. With such an approach, the comfort of the patient can be improved by eliminating the systemic administration of the drug and the dosage of the required drug can be reduced, thus decreasing the possible side effects of the drug. For example, polypropylene (PP) surgical meshes can be applied for the surgical repair of pathologies and, in particular, for the repair of hernias. Labay et al. [41] They used two techniques of plasma irradiation (Air Plasma and Argon Plasma) in order to improve the loading capacity of the meshes, as well as to maintain their antibacterial activity. The first air plasma treatment allowed a three-fold increase in the amount of incorporated ampicillin and decreased adhesion of fibroblasts. In general, the loading capacity of the drug of the surgical meshes of the PP was achieved and a good biocompatibility was maintained. Biomaterials treated via plasma and its potential application in the administration of drugs or active substances.
Both, surface functionalization and porosity are variants that directly affect the release kinetics. A common problem in terms of drug release is that the release process cannot be adjusted beyond its intrinsic capacity, which is related to its porosity [42]. One approach that has been used to prolong the release of the drug is by creating thin films by plasma polymerization on the surface of the materials, these act as a barrier in the dosage affecting the release profile [43]. A few works have investigated the polymerization of plasma to produce surface coatings in biomaterials and thus control the speed of drug release, another approach lies in studies in which ceramic materials are treated with non-thermal plasma technology to make them more porous as studied by Palaniappan et al. [44] in which they carried out a synthesis of mesoporous silica (SiO2) particles by the sol-gel method, giving it a plasma treatment of argon, in which they obtained the elimination of the organic residues produced from CTAB (hexadecyltrimethylammonium bromide), thus replacing the calcination treatment. The efficient elimination of CTAB residues, provides a greater biocompatibility of the silica particles. Plasma treatment can be an extremely efficient tool during the manufacture of drug delivery devices [16,25,45], because it modifies the surface of materials, for example; surface barrier formation by plasma polymerization, erosion of the material leading to an increase in porosity which allows greater surface area, which can be very useful in the loading and dosing stage of drugs or active substances. On the other hand, the functionalization of the surface through of plasma treatment allows the incorporation of reactive groups that lead to greater wettability on the surface, which increases the degree of degradation, which in turn leads to a change in the release profile.
Depending on the biomedical application that is required, different release profiles of active substances can be established, for example; rapid release of a drug may be desirable for rapid pain relief, while sustained and significantly slower release will be necessary for hormonal and anticancer therapies [46,47]. Through the use of plasma, we can control the release profiles, such as the research work carried out in 2015 by Myung et al. [48] they used the polymerization by radiofrequency plasma of acrylic acid to immobilize a drug Doxorubicin (DOX) and control its release. The DOX was covalently immobilized on the surface of the deposited glass with a thin film of plasma-polymerized acrylic acid containing the carboxyl group, the thin film of acrylic acid was coated on a glass surface at a pressure of 1.33 Pa and a radio frequency (RF) discharge power of 20 W for 10 min. The DOX layer coated with the thin film of polymerized acrylic acid reduced the rate of DOX release. They concluded that plasma polymerization is a method with a wide range of possibilities in its application as an aid in the release of drugs.
Another study was carried out by Stloukal et al. [49], in which the effect of the plasma treatment on the release kinetics of the drug Temozolomide (TMZ) was investigated from biodegradable polyester thin films, comprising polylactic acid (PLA) and polyester urethane. The authors used two plasma treatment systems, the first being diffuse coplanar barrier discharge, applying air as a gaseous medium, while the other involved radiofrequency plasma capacitively coupled under an argon atmosphere with hexamethyldisiloxane. The results showed that both forms of plasma treatment positively reduced the undesirable burst effect and benefited the TMZ release rate. Another example of the action of plasma technologies in drug delivery systems is that studied by Nakagawa et al. [50] in 2006 where they prepared an intragastric floating drug administration system by irradiation with plasma applied to the 5-fluorouracil double-tablet tablet (5-FU) as a central material with an outer layer composed of povidone (PVP), Eudragit RL (E-RL) and NaHCO3. The flow of the plasma caused the thermal decomposition of NaHCO3 to generate carbon dioxide and the resulting gases were trapped in the outer layer phase, so that the tablets acquired a lower density than the gastric content and remained floating in the simulated gastric fluid for a prolonged period of time. In addition, the release of 5-FU from the tablet is maintained by the appearance of a plasma-induced crosslinking reaction in the outer layer of the tablet and the release rate of 5-FU could be well controlled by the plasma operating conditions with an operation of 70 to 100 W at 13.56MHz.
Simovic et al. [51] in 2009 coated vancomycin loaded in nanoporous anodic aluminum oxide with allylamine plasma layers. The authors showed that by gradually increasing the thickness of the plasma polymerization layer, the amount of drug released decreases in a certain period of time. Other studies go back to the year 1993 where Kametani et al [52] developed a drug release system using plasma technology by oxygen irradiation that caused the formation of a porous structure in the polycarbonate which was synthesized from bisphenol, so that had a cross-linked intramolecular bifunctionality. The dissolution profiles showed that the release of theophylline (drug) at different speeds was feasible, depending on the set of conditions chosen for the manufacture of tablets and the operating conditions of the plasma [53-60].
Conclusion
Plasma treatment is a promising method to improve the surface properties of various biomaterials, which is already well known in recent decades. In this review, we studied the ability of the techniques of plasma modification used to treat different materials and the perspectives that exist to this day to be exploited in the technology of controlled release of drugs or active substances. The choice of plasma treatment technique will be dictated by the application of the material. The analysis of the literature opened the panorama to note the efficiency of different techniques of modification by plasma as versatile tools for biomedical applications [61-67].
References
- De Geyter N, Morent R (2014) Cold plasma surface modification of biodegradable polymer biomaterials. Wood head Publishing Limited.
- Thiry D, Konstantinidis S, Cornil J, Snyders R (2016) Plasma diagnostics for the low-pressure plasma polymerization process: A critical review. Thin Solid Films 606: 19-44.
- Gordillo Vázquez FJ (2008) Air plasma kinetics under the influence of sprites. J Phys D Appl Phys 41(23).
- Wen C (2015) Surface Coating and Modification of Metallic Biomaterials.
- Basu B (2017) Biomaterials for Musculoskeletal Regeneration.
- Zhang YS, Yao J (2017) Imaging Biomaterial-Tissue Interactions. Trends Biotechnol, p. 1-12.
- Blackwood DJ (2003) Biomaterials: Past Successes and Future Problems. Corros Rev 21: 97-124.
- Ratner BD, Bryant SJ (2004) Biomaterials: Where We Have Been and Where We Are Going. Annu Rev Biomed Eng 6: 41-75.
- Rodil SE (2009) Modificación Superficial De Biomateriales Metálicos. Rev Latinoam Metal y Mater 29: 67-83
- Minati L, Migliaresi C, Lunelli L, Viero G, Dalla Serra M, et al. (2017) Plasma assisted surface treatments of biomaterials. Biophys Chem 229: 151-164
- Franz S, Rammelt S, Scharnweber D, Simon JC (2011) Immune responses to implants - A review of the implications for the design of immunomodulatory biomaterials. Biomaterials 32(28): 6692-6709.
- Govindarajan T, Shandas R (2014) A survey of surface modification techniques for next-generation shape memory polymer stent devices. Polymers (Basel) 6(9): 2309-2331.
- Zhou T, Zhu Y, Li X, Liu X, Yeung KWK, et al. (2016) Surface functionalization of biomaterials by radical polymerization. Prog Mater Sci 83: 191-235.
- Zhao Y, Yeung KWK, Chu PK (2014) Functionalization of biomedical materials using plasma and related technologies. Appl Surf Sci 310: 11-18.
- Chu P (2002) Plasma-surface modification of biomaterials. Mater Sci Eng R Reports 36(5-6): 143-206.
- Yoshida S, Hagiwara K, Hasebe T, Hotta A (2013) Surface modification of polymers by plasma treatments for the enhancement of biocompatibility and controlled drug release. Surf Coatings Technol 233: 99-107.
- Althuri A, Gujjala LKS, Banerjee R (2017) Partially consolidated bioprocessing of mixed lignocellulosic feed stocks for ethanol production. Bioresour Technol 245: 530-539.
- Mai Prochnow A, Murphy AB, McLean KM, Kong MG, Ostrikov K (2014) Atmospheric pressure plasmas: Infection control and bacterial responses. Int J Antimicrob Agents 43(6): 508-517.
- Liao X, Liu D, Xiang Q, Ahn J, Chen S, et al. (2017) Inactivation mechanisms of non-thermal plasma on microbes: A review. Food Control 75: 83-91.
- Miao H, Yun G (2011) The sterilization of Escherichia coli by dielectricbarrier discharge plasma at atmospheric pressure. Appl Surf Sci 257(16): 7065-7070.
- Sohbatzadeh F, Hosseinzadeh Colagar A, Mirzanejhad S, Mahmodi S (2010) E. coli, P. aeruginosa, and B. cereus Bacteria Sterilization Using Afterglow of Non-Thermal Plasma at Atmospheric Pressure. Appl Biochem Biotechnol 160(7): 1978-1984.
- Peng P, Chen P, Schiappacasse C, Zhou N, Anderson E, et al. (2018) A reviews on the non-thermal plasma-assisted ammonia synthesis technologies. J Clean Prod 177: 597-609.
- Canullo L, Peñarrocha D, Clementini M, Iannello G, Micarelli C (2015) Impact of plasma of argon cleaning treatment on implant abutments in patients with a history of periodontal disease and thin biotype: Radiographic results at 24-month follow-up of a RCT. Clin Oral Implants Res 26(1): 8-14.
- Daeschlein G, Napp M, Majumdar A, Richter E, Rüsch Gerdes S, et al. (2017) In vitro killing of mycobacteria by low temperature atmospheric pressure plasma and dielectric barrier discharge plasma for treatment of tuberculosis. Clin Plasma Med 5(6): 1-7.
- Bilek MM, McKenzie DR (2010) Plasma modified surfaces for covalent immobilization of functional biomolecules in the absence of chemical linkers: Towards better biosensors and a new generation of medical implants. Biophys Rev 2(2): 55-65.
- Santos M, Bilek MMM, Wise SG (2015) Plasma-synthesised carbonbased coatings for cardiovascular applications. Bio surface and Biotribology 1:146-160.
- Zarrintaj P, Manouchehri S, Ahmadi Z, Saeb MR, Urbanska AM, et al. (2018) Agarose-based biomaterials for tissue engineering. Carbohydr Polym 187: 66-84.
- Estupiñan Duran HA, Peña Ballesteros DY (2011) Adsorption study of proteins on surfaces of poly-lactic acid by using electrochemical and microgravimetric techniques. Electroanal Chem 78(169): 167-175.
- Tang L, Thevenot P, Hu W (2008) Surface Chemistry Influences Implant Biocompatibility. Curr Top Med Chem 8(4): 270-280.
- Silva Bermudez P, Rodil SE (2013) An overview of protein adsorption on metal oxide coatings for biomedical implants. Surf Coatings Technol 233: 147-158.
- Bae J, Lee M, Park S, Jeong MG, Hong DY, et al. (2017) Investigation of intermediates in non-oxidative coupling of methane by non-thermal RF plasma. Catal Today 293(294): 105-112.
- Vandamme M, Robert E, Lerondel S, Sarron V, Ries D, et al. (2012) ROS implication in a new antitumor strategy based on non-thermal plasma. Int J Cancer 130(9): 2185-2194.
- Vandamme M, Robert E, Pesnel S, Barbosa E, Dozias S, et al. (2010) Antitumor effect of plasma treatment on u87 glioma xenografts: Preliminary results. Plasma Process Polym 7(3-4): 264-273.
- Partecke LI, Evert K, Haugk J, Doering F, Normann L, et al. (2012) Tissue Tolerable Plasma (TTP) induces apoptosis in pancreatic cancer cells in vitro and in vivo. BMC Cancer 12: 1-10
- Chen M, Zhang Y, Sky Driver M, Caruso AN, Yu Q, et al. (2013) Surface modification of several dental substrates by non-thermal, atmospheric plasma brush. Dent Mater 29(8): 871-880.
- López Alemany A, Compañ V, Refojo MF (2002) Porous structure of Purevision TM versus Focus® Night & Day TM and conventional hydrogel contact lenses. J Biomed Mater Res 63(3): 319-325.
- Yin SH, Wang YJ, Ren L, Zhao LN, Chen H, et al. (2009) Surface Hydrophilicity Improvement of RGP Contact Lens Material by Oxygen Plasma Treatment. Mater Sci Forum 610-613: 1268-1272.
- Yong Seung K (2009) Surface Modification of Rigid Gas Permeable Contact Lens Treated by Using a Low-Temperature Plasma in Air. J Korean Phys Soc 55: 2436.
- Feng H, Wang G, Wu G, Jin W, Wu H, et al. (2016) Plasma and ion-beam modification of metallic biomaterials for improved anti-bacterial properties. Surf Coatings Technol 306: 140-146.
- Yerdel MA, Akin EB, Dolalan S, Turkcapar AG, Pehlivan M, et al. (2001) Effect of single-dose prophylactic ampicillin and sulbactam on wound infection after tension-free inguinal hernia repair with polypropylene mesh. The randomized, double-blind, prospective trial. Ann Surg 233(1): 26-33.
- Labay C, Canal JM, Modic M, Cvelbar U, Quiles M, et al. (2015) Antibiotic-loaded polypropylene surgical meshes with suitable biological behaviour by plasma functionalization and polymerization. Biomaterials 71: 132-144.
- Bergamasco DA, De Carvalho Campos JS (2017) Corona Treatment Applied in the Processing of Silk Waste. Study of Properties Such as Hydrophilicity and Tensile Strength. Procedia Eng 200: 18-25.
- Szili ERSDSJB (2018) Surface modification of biomaterials by plasma polymerization. Surf Modif Biomater Methods Anal Appl 3: 9
- Palaniappan A, Zhang J, Su X, Tay FEH (2004) Preparation of mesoporous silica films using sol-gel process and argon plasma treatment. Chem Phys Lett 395(1-3): 70-74.
- McInnes SJP, Michl TD, Delalat B, Al Bataineh SA, Coad BR, et al. (2016) Thunderstruck: Plasma-Polymer-Coated Porous Silicon Microparticles As a Controlled Drug Delivery System. ACS Appl Mater Interfaces 8(7): 4467-4476.
- Lee IC, He JS, Tsai MT, Lin KC (2015) Fabrication of a novel partially dissolving polymer microneedle patch for transdermal drug delivery. J Mater Chem B 3: 276-285.
- Rocca Smith JR, Karbowiak T, Marcuzzo E, Sensidoni A, Piasente F, et al. (2016) Impact of corona treatment on PLA film properties. Polym Degrad Stab 132: 109-116.
- Myung SW, Jung SC, Kim BH (2015) Immobilization and controlled release of drug using plasma polymerized thin film. Thin Solid Films 584: 13-17.
- Stloukal P, Novák I, Mičušík M, Procházka M, Kucharczyk P, et al. (2018) Effect of plasma treatment on the release kinetics of a chemotherapy drug from biodegradable polyester films and polyester urethane films. Int J Polym Mater Polym Biomater 67: 161-173.
- Nakagawa T, Kondo SI, Sasai Y, Kuzuya M (2006) Preparation of floating drug delivery system by plasma technique. Chem Pharm Bull 54(4): 514-518.
- Simovic S, Losic D, Vasilev K (2010) Controlled drug release from porous materials by plasma polymer deposition. Chem Commun 46(8): 1317.
- Kametani T, Umezawa O (1993) NII-Electronic Library Service. Chem Pharm Bull 14: 369-375
- Bedel S, Lepoittevin B, Costa L, Leroy O, Dragoe D, et al. (2015) Antibacterial poly(ethylene terephthalate) surfaces obtained from thymyl methacrylate polymerization. J Polym Sci Part A Polym Chem 53: 1975-1985.
- Canal C, Khurana K, Gallinetti S, Bhatt S, Pulpytel J, et al. (2016) Design of calcium phosphate scaffolds with controlled simvastatin release by plasma polymerisation. Polym (United Kingdom) 92(1): 170-178.
- Cao H, Qiao Y, Liu X, Lu T, Cui T, et al. (2013) Electron storage mediated dark antibacterial action of bound silver nanoparticles: Smaller is not always better. Acta Biomater 9(2): 5100-5110.
- Cho SC, Han C, Choi H, Kim HS, Jin S, et al. (2016) Synthesis and consolidation behavior of Al/AlN composite powders by reactive RF thermal plasma spraying. Powder Technol 287: 395-402.
- Czylkowski D, Hrycak B, Miotk R, Jasiński M, Dors M, et al. (2018) Hydrogen-enriched gas production from kerosene using an atmospheric pressure microwave plasma system. Fuel 215: 686-694.
- Desmet T, Morent R, De Geyter N, Leys C, Schacht E, et al. (2009) Nonthermal plasma technology as a versatile strategy for polymeric biomaterials surface modification: A review. Biomacromolecules 10(9): 2351-2378.
- Erfani R, ZareBehtash H, Hale C, Kontis K (2015) Development of DBD plasma actuators: The double encapsulated electrode. Acta Astronaut 109: 132-143.
- González E, González F (2008) Síntesis por radiación con microondas de nanotubos de carbono. Univ Sci 13(3): 258-266.
- Ixix JLG, Mex E, Stephen M, Alvarez M Parámetros del Plasma, p. 0-5.
- Kasálková NS, Slepička P, Bačáková L, Sajdl P, Švorčík V (2013) Biocompatibility of plasma nanostructured biopolymers. Nucl Instruments Methods Phys Res Sect B Beam Interact with Mater Atoms 307: 642-646.
- Labay C, Canal JM, Navarro A, Canal C (2014) Corona plasma modification of polyamide 66 for the design of textile delivery systems for Cosmetic therapy. Appl Surf Sci 316: 251-258.
- Moisan M, Sauve G, Zakrzewski Z, Hubert J (1994) An atmospheric pressure waveguide-fed microwave plasma torch: The TIA design. Plasma Sources Sci Technol 3(4): 584-592.
- Pankaj SK, Keener KM (2017) Cold plasma: background, applications and current trends. Curr Opin Food Sci 16: 49-52.
- Petlin DG, Tverdokhlebov SI, Anissimov YG (2017) Plasma treatment as an efficient tool for controlled drug release from polymeric materials: A review. J Control Release 266: 57-74.
- Sakr AA, El-Shaer MA, Ghaly MF, Abdel-Haliem MEF (2015) Efficacy of dielectric barrier discharge (DBD) plasma in decontaminating Streptomyces colonizing specific Coptic icons. J Cult Herit 16(6): 848- 855.






























