Pitt-Hopkins Syndrome, Challenging Behaviour and Family Functioning: A Matched Control Pilot Study
Jonathan Mason1*, Bonnie Kirkman2, Annice Hill3 and Jannine D Cody4
1Associate Professor of Clinical Psychology, University of the Sunshine Coast, Maroochydore, Queensland, Australia
2Psychologist, University of the Sunshine Coast, Queensland, Australia
3Program Manager, Chromosome 18 Clinical Research Center, University Texas Health Science Center, San Antonio, USA
4Director, Chromosome 18 Clinical Research Center, University Texas Health Science Center, San Antonio, USA
Submission: May 14, 2021; Published: May 24, 2021
*Corresponding author:Jonathan Mason, Associate Professor of Clinical Psychology, University of the Sunshine Coast, Maroochydore, Queensland, Australia Glob
How to cite this article: Jonathan M, Bonnie K, Annice H, Jannine D C. Pitt-Hopkins Syndrome, Challenging Behaviour and Family Functioning: Ac Matched Control Pilot Study. Glob J Intellect Dev Disabil, 2021; 8(2): 555734. DOI:10.19080/GJIDD.2021.08.555734
Abstract
Background: Pitt-Hopkins syndrome (PTHS) is a rare genetic condition caused by the deletion of the TCF4 gene on the 18th Chromosome, and is associated with developmental delay, features of Autism Spectrum Disorder and communication difficulties. These characteristics are also known to be associated with an increased incidence of challenging behaviour, which in combination can lead to significant family stress.
Objective: The objective of this study was to investigate whether PTHS has an additional impact on challenging behaviour and family functioning when compared to people with similar damage to the 18th chromosome but where the TCF4 gene is intact.
Methods: Ten participants with abnormalities on the18th chromosome that included damage the TCF4 gene were compared to 10 participants with an intact TCF4 gene on measures of challenging behaviour and family functioning.
Results: Contrary to expectations, deletion of the TCF4 gene (PTHS) did not appear to increase challenging behaviour or significantly affect family functioning. Some differences in the hypothesised functions of behaviours between the two groups were observed, such that participants with PTHS were less likely to have challenging behaviour relating to escape, physical discomfort and social interaction.
Conclusion: The deletion of the TCF4 gene, which causes Pitt-Hopkins syndrome, does not appear to have an additional impact on the nature and level of challenging behaviour or family stress. Whilst the small variation found in the function of challenging behaviour might influence family functioning, other (non-genetic) mediating factors such as behaviour support skills and family resilience may be more important in determining outcomes.
Keywords: Challenging behaviour; Pitt hopkins syndrome; Autism spectrum disorder; Anxiety
Abbreviations: PTHS: Pitt Hopkins Syndrome; ASD: Autism Spectrum Disorder; ABC: Aberrant Behaviour Checklist
Introduction
The deletion or mutation of one copy of the TCF4 gene, located on the long arm of the 18th chromosome, causes Pitt Hopkins syndrome (PTHS) - a rare genetic condition thought to affect approximately one in 35,000 births [1. The syndrome is characterised by significant developmental delay, breathing difficulties, seizures, gastrointestinal issues, a lack of speech and distinctive facial features [1-3]. Ataxia and hypotonia are common, and most developmental milestones are significantly delayed, with the average age of walking four to six years of age [1]. Those with PTHS have been reported to display many of the symptoms commonly associated with Autism Spectrum Disorder (ASD) such as sensory issues, stereotypic movements, anxiety and difficulty adapting to changes in daily routine. Communication profiles vary widely, with some parents and caregivers reporting mainly non-verbal communication through gesturing, eye contact, noises and facial expressions.
Cohort studies have shown that common PTHS traits, such as poor communication skills, severe intellectual disability and ASD traits, increase the risk of challenging behaviour [4,5]. However, whereas hypotheses regarding the biological aetiology of challenging behaviour in people with chromosome 18 disorders have been tentatively explored in the literature, the communicative function served by the observed behaviours have not previously been investigated [2], and as a result, the relationship between PTHS and the function of challenging behaviours remains unclear. Understanding function is an important step in ensuring the efficacy of subsequent interventions designed to reduce challenging behaviour [6], and whilst this is usually individually assessed using techniques such as applied behaviour analysis, developing a preliminary understanding of whether there is a behavioural phenotype associated with PTHS that includes a common or more prominent function of behaviour is a useful step towards the possibility of interventions more specifically tailored to this subset of chromosomal disorders.
The relationship between chromosome 18 disorders and their impact on family functioning has also been neglected in the literature, although the relationship between some of the common features of chromosomal disorders in general (e.g., physical, motor, psychiatric and developmental impairments) have been shown to significantly contribute to family strain [7,8], impacting domains as diverse as finances, relationships and individual wellbeing. For example, the parents of children with pervasive developmental disorders are more likely to develop symptoms commonly associated with depression, lowering their confidence in their parenting role [8,9]. Parents’ personal strain is also influenced by their level of worry about the future [10,11]; parents have reported being worried about being able to care for their child when they became an adult, and the quality of life they would have, but also about their child’s development, their employment opportunities and their ability to adapt to adulthood [10,11]. The financial strain associated with managing the physical and mental health consequences of chromosomal disorders can also be significant, as can the impact on the ability of parents to balance the care requirements of their child with employment [11,12].
The aim of this study was to explore whether the deletion of the TCF4 gene significantly impacted challenging behaviour and family functioning over and above the levels experienced by a reference group of people with similar damage to the long arm of the 18th chromosome but where the TCF4 gene remains intact. It was hypothesised that TCF4 gene deletion (PTHS) would lead to greater levels of challenging behaviour and have a greater impact on family functioning than those in the reference group. Specific hypotheses about the function of behaviour were not made, with this aspect of the research considered exploratory.
Method
Design
Participants were recruited from the Chromosome 18 Clinical Research Centre’s ongoing longitudinal study of individuals with chromosome 18 abnormalities. Participants enter the main study by providing diagnostic information relating to their chromosome 18 abnormality and by undergoing detailed genetic testing [9]. They are also offered an opportunity to participate in ongoing research projects, of which the current study is one example.
Materials
Type and severity of challenging behaviour was assessed using the Aberrant Behaviour Checklist (ABC; [13] - a 58-item questionnaire with 5 sub-scales: Hyperactivity, Inappropriate Speech, Social Withdrawal, Irritability and Stereotypy. An extra subscale assessing Self-injury was created by totally items 2, 50, 52, following Ji, Capone, & Kaufmann [14]. Items are scored on a 4-point Likert scale, with higher scores indicative of higher levels of challenging behaviours.
The hypothesised function of each participant’s challenging behaviour was assessed using the Questions about Behavioural Function (QABF; [15]) - a 25-item questionnaire that consists of 5 sub-scales: Attention, Escape, Non-social, Physical and Tangible. Items are scored on a 4-point Likert scale, with higher scores indicating which function is considered most probable for each participant.
The family impact of caring for someone with chromosome 18 abnormalities was assessed using the Impact on Family Scale [16] - a 24-item questionnaire with 4 subscales measuring various components of family life (Finances, Social Relationships, Personal Strain and Mastery). Items are scored on a 4-point Likert scale, with higher scores indicating greater levels of (negative) impact.
Participants
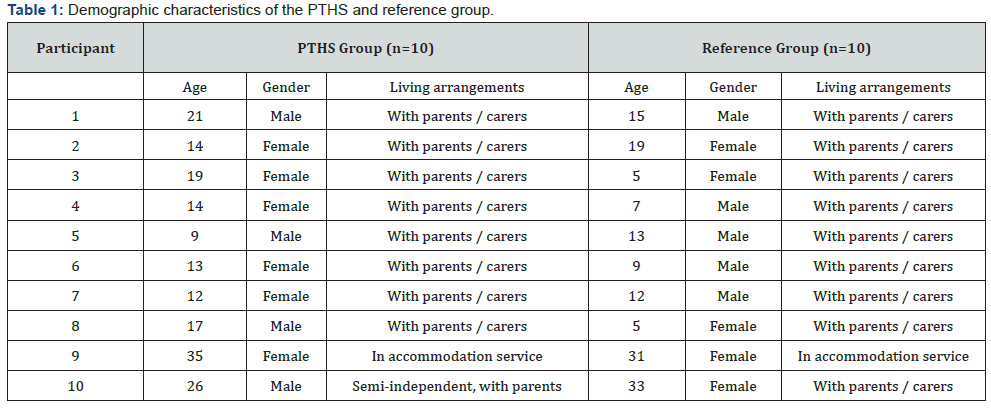
Ten participants with PTHS (Mage=14.9, SD=10) were identified and compared to 10 TCF4-intact reference group individuals (Mage=18, SD=7.7) with breakpoint deletions as close distally to the TCF4 gene as possible (location C18 21.2, following Hasi et al, [2]; see Figure 1). Individuals with both interstitial and terminal breakpoint deletions were included in the study due to recent findings that suggest no significant difference in behavioural functioning between these two groups [2]. Individuals with multiple chromosomal abnormalities were excluded.
Procedure
The research was approved by the research ethics committees of the two universities involved in the study. Families of young people with chromosome 18 abnormalities who had consented to be part of the Chromosome 18 Clinical Research Center’s longitudinal study were contacted via email. Informed consent was re-obtained prior to their participation.
Scoring and analysis
All participants included in the analysis provided complete data. ABC scores were reduced to 100 to facilitate comparisons. Kolmogorov-Smirnov and Shapiro Wilks statistics indicated that the data did not follow a normal distribution and a Mann-Whitney U test was used to assess between-group differences. The difference between mean ranks was interpreted due to the unequal distribution of score values between the two groups and asymptotic p values were analysed to allow correction for ties [17]. Effect sizes were calculated using partial eta squared [18]. Reliability analysis showed the ABC, QABF and IOFS had sound internal consistency, with Cronbach alphas ranging from 0.75 to 0.92. All analyses were completed using the IBM Statistical Package for Social Science (SPSS) Version 26.0.

Results
The demographic characteristics of the sample are summarised in table 1. Contrary to expectations, few significant differences in the type and severity of challenging behaviour between those with PTHS and the reference group were observed, and those that were noted did not occur in the predicted direction. The reference group were reported to have greater levels of inappropriate speech (Mdn=12.5) than the PTHS group (Mdn = 0; U = 21.5, z = -2.364, p = .018, ŋ2p -.5) and irritability (reference group Mdn =14.4, PTHS Group Mdn = 3.3; U = 21, z = -2.209, p = .027, ŋ2p -.5). Social attention was observed to be a more likely function for challenging behaviour in the reference group (Mdn = 2.5) than the PTHS group (Mdn = 0; U =25.5, z = -2.029, p = 0.042, ŋ2p -.5), as were escape (Reference group Mdn = 9.0, PTHS Mdn = 0 ; U =9.50, z = -3.212, p = 0.001, ŋ2p -.7), and physical discomfort (Reference Mdn =3.0 , PTHS Mdn =0; U =22.5, z = -2.278, p = 0.023, ŋ2p -.5). No significant differences were observed between the two groups on any of the Impact on Families subscales.
As eight of the 10 PTHS participants were noted to be young people aged 21yrs and under, a separate analysis was undertaken comparing these participants (Mage=10.6, SD=5.0) to eight participants aged 21yrs and under from the refence group (Mage = 14.9, SD=3.9) most closely matched for chromosomal breakpoint. Contrary to expectations, inappropriate speech was significantly higher in the reference group (Mdn=12.5) than the PTHS group (Mdn=0; U = 10.0, z = -2.555, p = 0.011, ŋ2p -.6); all other differences on the ABC were non-significant. Furthermore, the presence (reference group) or absence (PTHS group) of the TCF4 gene only appeared to influence one aspect of the function of challenging behaviour; Escape was observed to be more likely in participants in the reference group (Mdn = 7.0; U = 6.00, z = -2.919, p = 0.004, ŋ2p -.7) than the PTHS group (Mdn = 0). All other differences were non-significant. Finally, no significant differences were observed between the two groups on any of the Impact on Families subscales.
Discussion
Contrary to expectations, those with PTHS were found to have lower levels of challenging behaviour than a TCF4-intact reference group matched for the proximity of chromosomal breakpoint. The hypothesised function of behaviour varied between the groups, but no differences in the impact on family functioning were observed.
Whilst further research is needed to explore and validate these findings, they may reflect one of the general conceptions of people with PTHS as being friendly and non-aggressive [19,20], despite the syndrome’s close association with factors that typically correlate with higher levels of challenging behaviour (such as lower intellectual functioning and poor communication skills). In addition, young people with PTHS are generally thought to be non-verbal [2]; many of the items on the ABC that relate to the Inappropriate Speech subscale refer to speaking and the use of inappropriate words rather than more general vocalisations, potentially artificially deflating the score.
The differences observed in the attributed function of challenging behaviours between the two groups suggests that, at least on the face of it, there is some influence of the TCF4 gene on the underlying reasons for challenging behaviour. Whilst the higher scores in the reference group may simply reflect the generally higher levels of challenging behaviour also found in the that group, it remains possible that the lower scores found in the PTHS group across the assessed functions of behaviour reflect the existence of a set of very different behavioural functions that are not typically measured by standardised instruments such as the QABF. Other examples of rare genetic conditions with specific behavioural phenotypes exist, such as the severe lip, tongue and finger biting observed in Lesch-Nyhan syndrome [21]. Overall, however, a more plausible conclusion is that people living with PTHS are less prone to challenging behaviour than might be predicted from other features of the phenotype, and thus are less likely to have marked episodes of behaviour that can be attributed to clear behavioural functions. The finding from the secondary analysis that escape was a more common function of behaviour in young people in the reference group may at least in part reflect the less independent and more constricted lifestyle associated with being a young person living with PTHS, such that the reduction in opportunities for a normal life that are commonly associated with more severe levels of disability has the paradoxical effect of removing the need to use behaviour to reduce exposure to unwanted stimuli.
Deletion of the TCF4 gene did not appear to have a significant impact on family functioning, as those in the reference group with a child who had similar chromosomal damage but with the TCF4 gene intact reported similar levels of family functioning. Aside from the behavioural aspects of Pitt Hopkins syndrome, the physical complications associated with the phenotype can require significant family and medical support over the life of the affected person. Conversely, case studies of those with damage to the long arm of the 18th chromosome but who have the TCF4 gene intact have shown that some are able to complete high school and go on to gain employment [22]. It is perhaps surprising, therefore, that the carers of people with PTHS reported similar levels of family strain to those in the reference group. As in the case of challenging behaviour, concern over the physical wellbeing of a child, as well as the time and skill required to manage the various health complications and their consequences, may be significantly affected by the individual characteristics of the carers, resulting in a more complex interaction between client need and carer skill than was able to be measured in this study. There is also evidence to suggest that having a child with a chromosomal abnormality can have positive effects on the family [23-26] as can having a child with challenging behavior [27]. Parents report a sense of personal growth as they become more understanding, compassionate and less judgmental, as well as having a better sense of purpose and priority in their lives [23,25,26]. This sense of growth and development may also perceive inside the family unit, as they pool resources to work together, and they find themselves becoming stronger and closer, with better communication between every member of the.
Conclusion
Whilst the size and location of chromosomal breakpoint is an important factor in determining social, cognitive and functional outcomes, non-genetic mediating factors such as carer skill, personal and carer resilience and family and social support may be equally important when considering the development of behavioural problems and the impact of chronic disease on family functioning.
Limitations
Whilst the inclusion of 20 people with a rare chromosomal condition is positive, the cross-sectional design may have failed to obtain a representative sample, and whilst previous studies have not found phenotypic differences between PTHS resulting from TCF4 deletion versus TCF4 mutation, the rare nature of the syndrome makes firm conclusions in this regard difficult. The study did not take into consideration the variably of cognitive functioning between participants, which is a known correlate of both challenging behaviour [5,6] and aspects of family stress [7]. More detailed demographic information (e.g., family social economic status, engagement in previous behavioural or family interventions) may have strengthened the findings [28,29].
Acknowledgements
The authors wish to thank Ms. Hannah Anderson for her assistance with preparation of the manuscript.
References
- Sweetser DA, Elsharkawi I, Yonker L, Steeves M, Parkin K, et al. (2018) Pitt-Hopkins Syndrome. 2012 Aug 30. In: Adam MP, Ardinger HH, Pagon RA, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2020. Available from: https://www.ncbi.nlm.nih.gov/books/NBK100240/.
- Hasi M, Soileau B, Sebold C, Hill A, Hale DE, et al. (2011) The role of the TCF4 gene in the phenotype of individuals with 18q segmental deletions. Human Genetics 130(6): 777-787.
- Soileau B, Hasi M, Sebold C, Hill A, O’Donnell L, et al (2015) Adults with chromosome 18 abnormalities. Journal of Genetic Counseling 24(4): 663-674.
- Bowring DL, Totsika V, Hastings RP, Toogood S, Griffith GM (2017) Challenging behaviours in adults with an intellectual disability: A total population study and exploration of risk indices. The British journal of clinical psychology 56(1): 16-32.
- Lundqvist LO (2013) Prevalence and risk markers of behavior problems among adults with intellectual disabilities: A total population study in Örebro County, Sweden. Research in Developmental Disabilities 34(4): 1346-1356.
- Carr EG, Dunlap G, Horner RH, Koegel RL, Turnbull AP, et al. (2002) Positive behavior support: Evolution of an applied science. Journal of Positive Behavioral Intervention 4(1): 4-16.
- Blacher J, McIntyre L L (2006) Syndrome specificity and behavioural disorders in young adults with intellectual disability: Cultural differences in family impact. Journal of Intellectual Disability Research 50(3): 184-198.
- Giallo R, Wood CE, Jellett R, Porter R (2013) Fatigue, wellbeing and parental self-efficacy in mothers of children with an Autism Spectrum Disorder. Autism 17(4): 465-480.
- Heard PL, Carter EM, Crandall AC, Sebold C, Hale DE, et al. (2009) High resolution genomic analysis of 18q− using oligo‐microarray comparative genomic hybridization (aCGH). American Journal of Medical Genetics Part A 149(7): 1431-1437.
- Goble LA (2004) The impact of a child's chronic illness on fathers. Issues in Comprehensive Pediatric Nursing 27(3): 153-162.
- Nealy CE, O'Hare L, Powers J D, Swick DC (2012) The Impact of Autism Spectrum Disorders on the Family: A Qualitative Study of Mothers’ Perspectives. Journal of Family Social Work 15(3): 187-201.
- Vohra R, Madhavan S, Sambamoorthi U, St Peter C (2014) Access to services, quality of care, and family impact for children with autism, other developmental disabilities, and other mental health conditions. Autism 18(7): 815-826.
- Aman MG, Singh NN, Stewart AW, Field CJ (1985) The aberrant behaviour checklist: A behaviour rating scale for the assessment of treatment effects. American Journal of Mental Deficiency 89(5): 485-491.
- Ji NY, Capone GT, Kaufmann WE (2011) Autism spectrum disorder in Down syndrome: cluster analysis of Aberrant Behaviour Checklist data supports diagnosis. Journal of Intellectual Dsiability Research 55(11): 1064-1077.
- Paclawskyj TR, Matson JL, Rush KS, Smalls Y, Vollmer TR (2000) Questions about behavioral function (QABF): a behavioral checklist for functional assessment of aberrant behavior. Research in Developmental Disabilities 21(3): 223-229.
- Stein REK, Riessman CK (1980) The development of the Impact on Family Scale: Preliminary findings. Medical Care 18(4): 465-472
- McCrum-Gardner E (2008) Which is the correct statistical test to use? British Journal of Oral and Maxillofacial Surgery 46(1): 38-41.
- Tomczak M, Tomczak E (2014) The need to report effect size estimates revisited. An overview of some recommended measures of effect size. Trends in Sports Sciences 21: 19-25.
- Marangi G, Zollinio M (2015) Pitt-Hopkins syndrome and differential diagnosis: A molecular and clinical challenge. Journal of Paediatric Genetics 4(3): 168-176.
- Zweier C, Sticht H, Bijlsma EK, Clayton-Smith J, Boonen SE, et al. (2008) Further delineation of Pitt–Hopkins syndrome: phenotypic and genotypic description of 16 novel patients. Journal of Medical Genetics 45: 738-744.
- Nyhan WL (1976) Behavior in the Lesch-Nyhan syndrome. J Autism Dev Disord 6: 235-252.
- Cody JD, Sebold C, Heard P, Carter E, Soileau B, et al. (2015) Consequences of chromsome18q deletions. American Journal of Medical Genetics Part C: Seminars in Medical Genetics 169(3): 265-280.
- Bayat M (2007) Evidence of resilience in families of children with autism. Intellectual Disability Research 51(9): 702-714.
- Phelps KW, McCammon SL, Wuensch KL, Golden JA (2009) Enrichment, stress, and growth from parenting an individual with an autism spectrum disorder. Journal of Intellectual and Developmental Disability 34(2): 133-141.
- Stainton T, Besser H (1998) The positive impact of children with an intellectual disability on the family. Journal of Intellectual & Developmental Disability 23(1): 57-70.
- Taunt HM, Hastings RP (2002) Positive Impact of Children with Developmental Disabilities on Their Families: A Preliminary Study. Education and Training in Mental Retardation and Developmental Disabilities 37(4): 410-420.
- Griffith GM, Hastings RP (2014) ‘He's hard work, but he's worth it’. The Experience of Caregivers of Individuals with Intellectual Disabilities and Challenging Behaviour: A Meta-Synthesis of Qualitative Research. Journal of Applied Research in Intellectual Disabilities 27(5): 401-419.
- Giurgea I, Missirian C, Cacciagli P, Whalen S, Fredriksen T, et al. (2008) TCF4 Deletions in Pitt-Hopkins Syndrome. Human Mutation 29(11): 242-251.
- Carter E, Heard P, Hasi M, Soileau B, Sebold C, et al. (2015) Ring 18 molecular assessment and clinical consequences. American Journal of Medical Genetics Part A 167(1): 54-63.






























