Breast And Cervical Cancer-Related PTSD: Randomized Controlled Trial on the ASSYST Treatment Intervention
Ross J1*, Navarro F1, Mainthow N2, Givaudan M2 and Jarero I2
1Ágape, Desarrollo Integral, AC, Puebla, México
2Department of Research, EMDR Mexico, Mexico City, Mexico
Submission: November 09, 2023; Published: January 08, 2024
*Corresponding Address: Ross Jessica, Ágape, Desarrollo Integral, AC, Puebla México
How to cite this article: Ross Jarero*, Navarro F, Mainthow N, Givaudan M and Jarero I. Breast And Cervical Cancer-Related PTSD: Randomized Controlled Trial on the ASSYST Treatment Intervention. Canc Therapy & Oncol Int J. 2024; 25(5): 556175. DOI:10.19080/CTOIJ.2024.25.556175
Abstract
The aim of this randomized controlled trial with an intention-to-treat analysis was to evaluate the effectiveness, efficiency, and safety of the Acute Stress Syndrome Stabilization Individual (ASSYST-I) treatment intervention in reducing posttraumatic stress disorder (PTSD), anxiety, and depression symptoms in adult females with breast or cervical cancer. A total of 30 adult females met the inclusion criteria and participated in the study. Participants’ ages ranged from 37 to 62 years old (M =48.63 years old). A two-arm randomized controlled trial (RCT) design was applied. PTSD, anxiety, and depression symptoms were measured in three time points for all participants in the study. Repeated-measures ANOVA showed that the intervention had a significant effect for time with a large effect size (F (2,56) = 76.68 p <.000, η² = .738), and a significant interaction with a large effect size between time and group, (F (2, 56) = 101.21, p <.000, η² = .738), on PTSD. For anxiety, results differed significantly across time points (F (2,56) = 3.16, p= .05, η² =. 102). Mean scores on depression showed a significant interaction between time and group (F (2, 56) =5.94, p =.000, η² =. 317).
Results on the Reliable Change Index (RCI) and the Clinically Significant Change (CSC) Margin showed that the ASSYST-I treatment intervention exhibited reliable change in PTSD symptom reduction and clinically significant change, indicating that the treatment group participants are more likely to belong to the non-PTSD population after the intervention. Conversely, the control group participants are more likely to belong to the PTSD population. All this data supports the effectiveness, efficiency, and safety of the ASSYST-I treatment intervention in reducing PTSD, anxiety, and depression symptoms in adult females with breast or cervical cancer. No adverse effects or events were reported by the participants during the treatment procedure administration or at follow-up. None of the participants showed clinically significant worsening/exacerbation of symptoms after treatment. Participants in the control group were invited to receive the intervention treatment after the follow-up assessment, fulfilling our ethical criteria.
Keywords: Acute Stress Syndrome Stabilization; ASSYST; Posttraumatic stress disorder (PTSD); Anxiety; Depression; Breast Cancer; Cervical Cancer
Abbreviations: PTSD: Posttraumatic Stress Disorder; RCT: Randomized Controlled Trial; ASSYST-I: Acute Stress Syndrome Stabilization Individual; RCI: Reliable Change Index; CSC: Clinically Significant Change; FCR: Fear of Cancer Recurrence; DSM: Diagnostic and Statistical Manual; AIP: Adaptive Information Processing; EMDR-PRECI: EMDR Protocol for Recent Critical Incidents and Ongoing Traumatic Stress; ASD: Acute Stress Disorder; SPIRIT: Standard Protocol Items Recommendation for Interventional Trials; CONSORT: Consolidated Standards of Reporting Trials; ANOVA: Analyses of variance; HADS: Hospital Anxiety and Depression Scale; CAPS-5: Clinician-Administered PTSD Scale-5; PCL-5: Posttraumatic Stress Disorder Checklist for DSM-5
Introduction
As of 2020, female breast cancer is the most prevalent type of cancer, as well as one of the deadliest among females, with 7.8 million women diagnosed and 685,000 deaths, while cervical cancer is the fourth most common cancer in women, with about 570,000 women diagnosed and about 311, 000 deaths in 2018 [1-2]. The diagnosis of female breast cancer can have catastrophic implications and severe life impediments, no matter the developmental life cycle of the female diagnosed, particularly with younger females and those with children [3]. From diagnosis to treatment, the experience of breast cancer for women is distressing and disruptive, causing psychological and functional impairment that is found to not only remain, but increase over time, long after treatment has been terminated, ranging from fear of cancer recurrence (FCR) to posttraumatic stress disorder symptoms (PTSD) [4-6], while cervical cancer has also been linked to PTSD symptoms [7]. It is important to note that while the diagnosis of a life-threatening illness is considered a criterion A event for PTSD in the Diagnostic and Statistical Manual (DSM)-IV, and the diagnosis of cancer met criterion A, there is discrepancy as to whether or not breast or cervical cancer diagnosis meets criterion A for PTSD, as the diagnosis of a life-threatening illness must be considered “sudden and catastrophic,” and the diagnosis of cancer is not considered a PTSD criterion A event in the DSM-5 [8-9]. However, breast cancer diagnosis is sudden and catastrophic for most women, given the high mortality rate, treatment trajectory, and disruption of daily life [5]. This discrepancy does not negate the evidence for the rampancy of PTSD intrusion symptoms and their detrimental effects on the quality of life and functioning of female breast or cervical cancer patients. For the purposes of this paper, we will explore the recruitment and treatment of PTSD, anxiety, and depression symptoms provoked by breast cancer or cervical cancer diagnosis and treatment (e.g., surgery and chemotherapy).
Findings consistently show the correlation between cancer diagnosis, particularly female breast cancer, and PTSD symptoms [10-16]. Studies show that PTSD intrusion symptoms, most reported as, but not limited to nightmares and extreme psychological and physiological distress related to FCR, are the most pervasive symptoms of the experienced symptom clusters among cancer patients, with a prevalence of 11–45%. Also, 34.1% of the patients who developed PTSD symptoms shortly after their breast cancer diagnosis had no change or had exacerbated symptoms. Based on these findings, the literature has concluded that PTSD symptoms in this population do not subside with time and that treatment intervention procedures for cancer-related PTSD symptoms are required [4-6,17]. It is surprising to find that given the amount of evidence correlating female breast cancer, cervical cancer, and other types of cancer and PTSD symptoms, there are no treatment guidelines for cancer-related PTSD symptoms. In a systematic review of 508 studies, only eight met the inclusion criteria, albeit with study limitations, concluding that insufficient evidence supports the effectiveness of psychological interventions in treating cancer-related PTSD symptoms [18]. Treatment intervention procedures to reduce or eliminate cancer-related PTSD symptoms could have long-lasting benefits for female breast or cervical cancer patients.
Acute Stress Syndrome Stabilization Individual Treatment Intervention
The Acute Stress Syndrome Stabilization Individual (ASSYST-I) treatment intervention is an adaptive information processing (AIP)-informed, evidence-based, symptom-focused, trauma-sensitive approach, which contains the core components (the most therapeutically active) of the EMDR Protocol for Recent Critical Incidents and Ongoing Traumatic Stress (EMDR-PRECI) [19-31]. The ASSYST-I requires less time than the full EMDR standard protocol or the EMDR-PRECI, making it less expensive, less complex, effective, efficient, and safe, enhancing the feasibility of delivering brief treatment in primary care settings worldwide. This treatment intervention is specifically designed to provide in-person or online support to clients who present PTSD or Acute Stress Disorder (ASD), intense psychological distress, and/ or physiological reactivity caused by the disorders’ intrusion symptoms associated with the memories of the traumatic event(s) or adverse experience(s). Intrusion symptoms are core ASD and PTSD dimensions and may cause extreme distress, functional impairment, or dissociation from present surroundings. Therefore, focusing on this domain could identify targets specifically related to trauma [32], as “reducing intrusion symptoms may also have useful downstream effects since they are hypothesized to be a mediator of other PTSD symptom clusters” [33,p.2].
Previous ASSYST Treatment Intervention Studies
Eight previous studies on the ASSYST treatment interventions with 362 participants among different populations have proven their efficacy and safety:
i. General population in lockdown and with ongoing traumatic stress during the COVID-19 Pandemic.
ii. TeleMental Health counseling to the general population after adverse experiences.
iii. Mental Health Professionals working during the COVID-19 Pandemic with patients suffering from trauma-related disorders and stressors.
iv. General population with non-recent pathogenic memories.
v. Adult Syrian refugees living in Lebanon.
vi. Adult Females with Adverse Childhood Experiences.
vii. Public sector workers during the COVID-19 Pandemic.
viii. Female children polytraumatized by adverse childhood experiences, neglect, and maltreatment [34-41].
Reliable Change Index and Clinically Significant Change Margin
To know whether PTSD symptom change does indeed indicate a reliable and clinically significant change in this study, we used the Reliable Change Index (RCI) and the Clinically Significant Change (CSC) Margin. The RCI determines if the magnitude of observed change over time on a given measure is beyond what should be attributed to measurement error. The CSC is used to determine if an observed end score on a measure of symptomatology indicates that the respondent is more likely to belong to the nondisordered population than the disordered population. For the Posttraumatic Stress Disorder Checklist for DSM-5 (PCL-5), the more conservative value of the RCI is 18 points and an end score of ≤ 28 for the CSC margin [42].
Objective
The objective of this randomized controlled trial with an intention-to-treat analysis was to evaluate the effectiveness, efficiency, and safety of the Acute Stress Syndrome Stabilization Individual (ASSYST-I) treatment intervention in reducing PTSD, anxiety, and depression symptoms in adult females with breast or cervical cancer.
Method
Study Design
To measure the effectiveness of the ASSYST-I on the dependent variable PTSD, anxiety, and depression symptoms, this study, with an intention-to-treat analysis, used a two-arm randomized controlled trial (RCT) with a waitlist no-treatment control group design. PTSD, anxiety, and depression symptoms were measured at three-time points for all participants in the study: Time 1. Pretreatment assessment; Time 2. Post-treatment assessment; and Time 3. Follow-up assessment. For ethical reasons, all participants in the control group were invited to receive treatment intervention after the follow-up assessment was completed.
Ethics and Research Quality
The study protocol and detailed procedures were reviewed and approved by the EMDR Mexico International Research Ethics Review Board (also known in the United States of America as an Institutional Review Board) in compliance with the International Committee of Medical Journal Editors recommendations, the Guidelines for Good Clinical Practice of the European Medicines Agency (version 1 December 2016), and the Helsinki Declaration as revised in 2013. The research quality of this study was based on the Consolidated Standards of Reporting Trials (CONSORT) 2010 Statement and the Standard Protocol Items Recommendation for Interventional Trials (SPIRIT) 2013 checklist [43,44].
Participants
This study was conducted in Puebla City, Mexico, from June to September 2023, with the Mexican (Latina) adult female population with a breast or cervical cancer diagnosis and subsequent treatment (surgery and chemotherapy). Thirty-five potential participants were recruited. Inclusion criteria was:
a. Being an adult female
b. Having received a breast or cervical cancer diagnosis and subsequent treatment
c. Voluntarily participating in the study
d. Not receiving specialized trauma therapy
e. Not receiving pharmacotherapy for PTSD symptoms
f. Having a PCL-5 total score of 31 points or more. Exclusion criteria was:
a. Ongoing self-harm/suicidal or homicidal ideation
b. Diagnosis of schizophrenia, psychotic, or bipolar disorder
c. Diagnosis of a dissociative disorder
d. Organic mental disorder
e. A current, active chemical dependency problem
f. Significant cognitive impairment (e.g., severe intellectual disability, dementia)
g. Presence of uncontrolled symptoms due to a medical illness.
Five of the 35 potential participants were excluded for not wanting to participate in the study. A total of 30 adult females met the inclusion criteria and participated in the study. Participants’ ages ranged from 37 to 62 years old (M =40.63 years). Participation was voluntary, with the participants signing informed consent before treatment per the Mental Capacity Act 2005.
Instruments for Psychometric Evaluation
a. To measure PTSD symptom severity and treatment response, we used the Posttraumatic Stress Disorder Checklist for DSM-5 (PCL-5) provided by the National Center for PTSD (NCPTSD), with the time interval for symptoms to be the past week. The instrument was translated and back translated to Spanish. It contains 20 items, including three new PTSD symptoms (compared with the PTSD Checklist for DSM-IV) [45,46]: blame, negative emotions, and reckless or self-destructive behavior. Respondents indicated how much they have been bothered by each PTSD symptom over the past week (rather than the past month), using a 5-point Likert scale ranging from 0=not at all, 1=a little bit, 2=moderately, 3=quite a bit, and 4=extremely. A total symptom score of zero to 80 can be obtained by summing the items. The sum of the scores yields a continuous measure of PTSD symptom severity for symptom clusters and the whole disorder. Psychometrics for the PCL-5, validated against the Clinician- Administered PTSD Scale-5 (CAPS-5) diagnosis, suggest that a score of 31-33 is optimal to determine probable PTSD diagnosis [47].
b. To measure anxiety and depression symptom severity and treatment response, we used the Hospital Anxiety and Depression Scale (HADS), extensively used to evaluate these psychiatric comorbidities in various clinical settings at all healthcare services and with the general population. The instrument was translated and back translated to Spanish. It is a 14-item self-report scale to measure Anxiety (7 items) and Depression (7 items) of patients with both somatic and mental problems using a 4-point Likert scale ranging from 0 to 3. The response descriptors of all items are Yes, definitely (score 3); Yes, sometimes (score 2); No, not much (score 1); No, not at all (score 0). A higher score represents higher levels of Anxiety and Depression: a domain score of 11 or greater indicates Anxiety or Depression; 8–10 indicates a borderline case; 7 or lower indicates no signs of Anxiety or Depression [48,49].
Procedure
Randomization, Allocation Concealment Mechanism, and Blinding Procedure
A computer-generated simple randomization with a 1:1 allocation ratio was used. Two independent assessors blind to treatment conditions conducted the randomization process to avoid allocation influence. The treatment random allocation sequence was concealed using sequentially numbered, opaque, sealed, and stapled envelopes opened only after they were irreversibly assigned to the participants. The safekeeping of the envelopes and the assignment of participants to each arm of the trial (implementation of the random allocation sequence) was overseen by a person not involved in the research study and independent of the enrollment personnel. The participants’ treatment allocation was blinded to the research assistants who conducted the intake interview, initial assessment, and enrollment and for the independent assessors who conducted the follow-up assessments. Participants were instructed not to reveal their treatment allocation to those conducting the assessments. Fifteen participants were allocated to the treatment group (TG) and fifteen to the control group (CG). See Figure 1. Flow Diagram.

Enrollment, Assessments Times, Blind Data Collection, and Confidentiality of Data
Treatment group (TG) and control group (CG) participants completed the instruments in person and on an individual basis during distinct assessment moments. During Time 1, research assistants formally trained in all of the instruments’ administration, who were not blind to the study but blind to the participant’s treatment allocation, conducted the intake interview, collected demographic data (e.g., name, age, gender, and contact information), assessed potential participants for eligibility based on the inclusion/exclusion criteria, obtained signed informed consent from the participants, conducted the pre-treatment application of instruments, enrolled participants in the study, and randomly assigned each treatment group participant to one of the treatment providers formally trained in the ASSYST-I that participated in this study. The research assistants (all EMDR therapy trained clinicians) also assisted the participants in identifying their worst cancer-related adverse experience to be treated with the ASSYST-I. Each identified adverse experience was written down by the research assistants on the Memory Record Sheets that were utilized by the treatment providers during the ASSYST-I treatment intervention and utilized by participants during the three assessments times to ensure participants were focusing on the same cancer-related adverse experience when they received the treatment intervention, as well as at the specific assessment time when they completed the assessment tools.
To obtain maximally interpretable PCL-5 and HADS scores, research assistants and independent assessors:
a. Discussed with each participant the purpose of the instruments in detail.
b. Encouraged attentive and specific responses.
c. Invited participants to read each question carefully before responding and select the correct answer.
d. Clarified their questions about some of the symptoms.
e. Reworded conceptually complex symptoms when necessary.
f. Reminded participants of last week’s symptoms’ time frame, as well as.
g. To only report symptoms related to the pathogenic memory of their worst adverse experience and not based on their everyday general distress.
During Time 2 (post-treatment assessment seven days after treatment) and Time 3 (follow-up assessment 30 days after treatment), assessments were conducted for all participants by blind-to-treatment allocation independent assessors with formal training in the administration of the instruments. The data safekeeper independent assessor received the participants’ assessment instruments that were answered during Times 1, 2, and 3. All data was collected, stored, and handled in full compliance with the EMDR Mexico International Research Ethics Review Board requirements to ensure confidentiality. Each study participant consented to access their data, which was strictly required for study quality control. All procedures for handling, storing, destroying, and processing data followed the Data Protection Act 2018. All persons involved in this research project were subject to a signed professional confidentiality agreement.
Withdrawal from the Study and Missing Data
All research participants had the right to withdraw from the study without justification at any time and with assurances of no prejudicial result. If participants decided to withdraw from the study, they were no longer followed up in the research protocol. There were no withdrawals or missing data during this study.
Treatment
Treatment Providers and Treatment Fidelity
The ASSYST-I was provided in person to each participant by 12 licensed thanatologists formally trained in this treatment intervention. Treatment providers received ongoing supervision and clinical feedback from the research project Clinical Director (MN) through daily online group supervision and completing detailed session summary forms for each session with each participant that they were assigned that were designed specifically for the ASSYST-I treatment intervention to guide, elicit, monitor, and facilitate clinicians’ treatment fidelity.
Treatment Description and Treatment Safety
A symptom trajectory-based stepped-care approach to adverse experiences was used during this study. This means a stepped progression of mental health care provided in an intensified manner based on the ongoing monitoring of the PTSD symptom trajectory during the study. The main objectives of this approach are:
a. To strategize treatment
b. To provide treatment interventions according to the progression of pathophysiology
c. To improve symptom relief and clinical outcomes.
The participants’ treated pathogenic memories were an average of 2.09 years old and received six in-person sessions, averaging 49 minutes per session. The ASSYST-I treatment intervention started with the worst pathogenic memory produced by the cancer-related worst adverse experience (e.g., surgery for the extirpation of affected organs, chemotherapy side effects) selected during the T1. Pre-treatment assessment. Therefore, at the beginning of the first treatment session, the participants were asked to run a mental movie of the previously selected cancerrelated worst adverse experience and then choose the worst part (this pathogenic memory was like a still photo, not a movie). Once the worst pathogenic memory reached subjective levels of disturbance of zero or one (ecological/realistic), the patients were instructed to run the mental movie again and select any other disturbing part. This procedure was repeated until the entire adverse experience could be visualized without highly distressing parts. The treatment intervention was considered complete when the participant’s subjective levels of disturbance associated with all the worst adverse experience disturbing parts decreased to zero or one (ecological/realistic). The ASSYST treatment intervention was provided to all the treatment group participants in an intensive treatment modality with two 60-minute (max) sessions provided per day over three consecutive days. Treatment safety was defined as the absence of adverse effects, events, or symptoms worsening. Therefore, participants were instructed by their treatment providers to immediately report any adverse effects (e.g., dissociative symptoms [derealization/ depersonalization], fear, panic, freeze, shut down, collapse, fainting); events (e.g., suicidal ideation, suicide attempts, selfharm, homicidal ideation); or symptoms worsening during the entire study time frame. The TG participants reported no adverse effects or events during the treatment procedure administration or at the thirty-day follow-up. None of the participants in the TG showed clinically significant worsening/exacerbation of symptoms on the PCL-5 after completion of treatment.
Examples of the Pathogenic Memories Treated with the ASSYST-I
Examples of pathogenic memories treated during the ASSYST-I sessions were:
a. Looking at herself in front of a mirror with scars instead of breasts.
b. Watching her hair fall out in uncontrolled clumps while taking a shower.
c. Imagining her children homeless living on the street because she died and there is no one to take care of them.
d. Fainting on the street after chemotherapy.
e. Feeling like she is burning inside.
f. Feeling the pain from the catheter after it had already been removed.
Statistical Analyses
Analyses of variance (ANOVA) for repeated measurements comparing two groups: treatment group (TG) vs control group (CG) was applied to analyze the effects of the treatment across time at three-time measurements: Time 1. Pre-treatment assessment, Time 2. Post-treatment assessment and Time 3 Follow-up assessment to analyze the effect of the treatment intervention ASSYST-I on: PTSD, Anxiety, and Depression; eta squared (η²) is reported to show the effect size. Comparison of means analyses were carried out using t test for independent samples and t test within groups. Cohen´s d is included to report effect size for t test results.
PTSD
Results showed that the intervention had a significant effect for time on PTSD with a large effect size (F (2,56) = 76.68 p <.000, η² = .738), a significant effect for group with a middle effect size was also found (F (1, 28 = 31.16, p<.000, η² = .527) and a significant interaction with a large effect size between time and group, (F (2, 56) = 101.21, p <.000, η² = .738). Comparison of means between groups did not show significant differences for Time 1. Pre-treatment assessment (M = 39.66, SD = 8.45 vs M = 38.86, SD = 11.57). For Time 2. Post-treatment assessment, significant differences between the treatment group (TG) and control group (CG) were found, with a medium effect, t (28) = - 7.481, p=.000, d = .64, (M = 15.40, SD = 7.29 vs M = 40.93, SD=11.02). For Time 3. Follow-up assessment, significant differences between the treatment group (TG) and control group (CG) were also found, with larger effect, t (28) = - 8.876, p=.000, d = - 2.31, (M = 14.26, SD = 6.78 vs M = 41.86, SD=9.94). Intragroup comparisons of means for treatment group (TG) showed significant differences between Time 1. Pre-treatment assessment and Time 2. Posttreatment assessment with a large effect, t (14) = 9.73, p=.00, d = 2.17 and between Time 2. Post-treatment assessment and Time 3. Follow-up assessment, t (22) = 3.37, p =.005, d = .11. Intragroup comparison of means for control group (CG) showed significant differences between Time 1 and Time 2, t (14) = -2.69, p =.01, d = .07. See Table 1 and Figure 2.

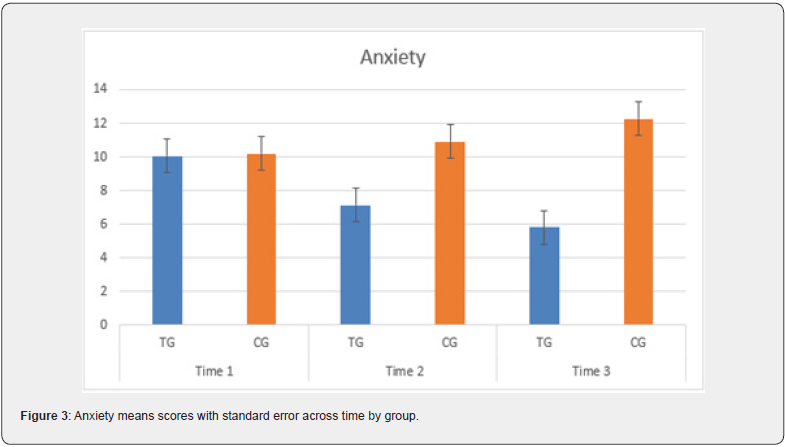
Anxiety
Repeated-measures ANOVA determined that mean scores on Anxiety differed significantly across time points with (F (2,56) = 3.16, p= .05, η² =. 102). Significant effect for group was also observed (F (1,28 = 7.16, p<.05, η² = .204) and a significant interaction between time and group, (F 2, 56) = 76.83, p <.00, η² = .415). Comparison of means between groups did not show significant differences for Time 1. Pre-treatment assessment (M = 10.06, SD = 3.39 vs M =10.20, SD = 4.05). For Time 2. Posttreatment assessment, significant differences between treatment group (TG) and control group (CG) group were found, with a medium effect, t (28) = - 2.40, p =.01, d = -.64, (M = 7.13, SD = 4.35 vs M = 10.93, SD = 4.02) as well as Time 3. Follow-up assessment, with a large effect, t (28) d = - 4.76, p =.000, d = - 1.22, (M = 5.80, SD = 3.68 vs M = 12.26, SD = 3.75). Intragroup comparison of means for the Treatment Group (TG) showed significant differences between Time 1. Pre-treatment assessment and Time 2. Post-treatment assessment, with a medium effect, t (14) = 2.51, p<.05, d = .45 and between Time 2. Post-treatment assessment and Time 3. Follow-up assessment, t (14) =3.69, p< .005, d = .18. Intragroup comparison of means for the control group (CG) showed significant differences between Time 2. Post-treatment assessment and Time 3. Follow-up assessment, with a small effect, t (14) = - 4.00, p = .001, d = .07. See Table 1 and Figure 3.


Depression
ANOVA for repeated measurements revealed that that mean scores on Depression did not differ significantly across time points. Significant interaction between time and group was founded, (F (2, 56) =5.94, p =.000, η² =. 317). Inter-subject comparisons show a significant effect for this variable by group (F (1,28) =5.94, p<.05, η² =. 175. Comparison of means between groups for Depression did not show significant differences for pretest Time 1. Pre-treatment assessment, (M= 7.60, SD= 4.11 vs M= 7.53, SD= 3.22). For Time 2. Post-treatment assessment, significant differences between the treatment group (TG) and the control group (CG) group were found, t (28) = - 2.60 p =.01, d = -0.64, (M = 5.00, SD = 4.03 vs M = 8.66, SD = 3.67). For Time 3. Followup assessment, significant differences between the treatment group (TG) and the control group (CG) group were found, t (28) = - 43.12 p =.000, d = - 1.23, (M = 4.06, SD = 3.19 vs M = 9.00, SD = 3.07). Intragroup comparison of means for the treatment group (TG) showed significant differences between Time 1. Pretreatment assessment and Time 2. Post-treatment assessment, t (14) = 2.23, p <.05, d = .45 and between Time 2. Post-treatment assessment and Time 3. Follow-up assessment, t (14) = 3.28, p=.005, d = .16. Intragroup comparison of means for the control group (CG) showed significant differences between Time 2P.osttreatment assessment and Time 3. Follow-up assessment, t (19) = 3.50, p=.002, d = .53. See Table 1 and Figure 4.
Discussion
The aim of this randomized controlled trial with an intentionto- treat analysis was to evaluate the effectiveness, efficiency, and safety of the ASSYST-I treatment intervention in reducing PTSD, anxiety, and depression symptoms in adult females with breast or cervical cancer. A total of 30 adult females met the inclusion criteria and participated in the study. Participants’ ages ranged from 37 to 62 years old (M =48.63 years old). A two-arm randomized controlled trial (RCT) design was applied. This research design was useful in demonstrating the effect of the ASSYST-I treatment intervention in reducing PTSD, anxiety, and depression symptoms. No significant differences were found between the groups at Time 1, confirming their equivalence before treatment and the large and maintained effect in the treatment group at followup. Results showed that the ASSYST-I treatment intervention had a significant effect in the different comparisons carried out through ANOVA for repeated measurements. The stronger effect was observed for Time 3. Follow-up assessment for the three dependent variables: PTSD, anxiety and depression. Regarding the Reliable Change Index (RCI), all TG participants exhibited reliable change in symptom reduction, with an average of 25.40 points for PTSD symptom reduction, 18 points being the more conservative value. This is indicative that the ASSYST-I treatment intervention reduced PTSD symptom severity beyond what is attributable to measurement error. In reference to the Clinically Significant Change (CSC) margin, all TG participants exhibit clinically significant change, indicating that these participants are more likely to belong to the non-PTSD population.
Conclusion
Given the ubiquity of PTSD symptoms among women who have experienced a cancer diagnosis, especially breast cancer, one would determine that there would be a multitude of studies demonstrating effective treatment intervention procedures treating cancer-related PTSD symptoms. While a significant amount of literature provides evidence for the correlation between PTSD symptoms and breast cancer, and to some extent, cervical cancer diagnosis and treatment, there appears to be a deficit in evidence-based research studies on treatment intervention procedures to target cancer-related intrusion symptoms. The promising results of this study fill at least part of the gap in the present literature lacking in terms of actual treatment of cancerrelated PTSD symptoms.
The ASSYST-I treatment intervention goes beyond palliative management of psychological distress in cancer-related PTSD symptoms, and what is of particular importance to cancerrelated PTSD intrusion symptoms is that the ASSYST treatment intervention procedures are specifically designed to treat these symptoms. Also, this treatment intervention includes in the assessment phase the instruction, “or even looking into the future,” allowing the patient to target and process the FCR, which is unique to and prominent among cancer patients. The FCR causes extreme distress among this population, particularly in the younger population, who may fear the cancer will return later in life, or those with children, who fear what will happen to their children if the cancer returns. This allows processing the maladaptive and often catastrophic fear of the future, specifically the cancer recurrence, even after treatment has been terminated. This emphasizes the appropriateness of the ASSYST-Individual as a cancer-related PTSD symptoms treatment intervention. This study has shown the efficiency, effectiveness, and safety of the ASSYST-I as an appropriate cancer-related PTSD, anxiety, and depression symptoms treatment intervention for female breast or cervical cancer patients. Due to the evidence of PTSD intrusion symptoms as the dominant symptom cluster negatively impacting female breast cancer patients, this is of vital importance for the holistic treatment of female breast cancer and cervical cancer patients.
Limitations and Future Directions
The small sample size and the follow-up assessment at 30 days due to ethical reasons (providing treatment to the CG participants as soon as possible) are limitations of this study. We recommend future multicenter randomized controlled trials with an intentionto- treat analysis, a larger sample, follow-up assessment at six months, and following the Consolidated Standards of Reporting Trials (CONSORT) 2010 Statement and the Standard Protocol Items Recommendation for Interventional Trials (SPIRIT) 2013 checklist.
Conflict of Interest and Funding
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgment
We want to express our gratitude to the research assistants and treatment providers who participated in this study: Amalia Osorio Vigil, Isabel Guadalupe Rodríguez Salazar, Pilar Osorio Vigil, Malena Abraham Gutiérrez, Julieta Fernández López, Alejandra Goytia Vázquez de Mercado, Liliana Ramos Martínez, Tere Gómez Cortés, Valeria Sandoval Centeno, María de Lourdes Guadalupe Lavalle, Leonor Elena Careaga Sartorius, María Amparo Alonso Soberón, María Fernanda Zambrano, Lisis Marlen Vera Tlachés, Claudia Montserrat Denetro García, Argelia Vázquez García, Gemma Fuentes Góngora, María Abiti Maldonado, Jenny Hidalgo Ramé, Jessica Vasconcelos Beltrán, Josefina Pérez Gómez, Alma Rosa Martínez Chávez.
References
- (2023) World Health Organization. Breast Cancer. World Health Organization.
- (2023) World Health Organization. Cervical Cancer. World Health Organization.
- Valazquez D, Rosenberg S, Gelber S, Kathryn J Ruddy, Morgan E, et al. (2020) Posttraumatic stress in breast cancer survivors was diagnosed at a young age. Psycho-oncology, J Psychol Behav Soc Dimen Cancer 29(8): 1312-1320.
- Carletto S, Camillo Porcaro, Carmen S, Vincenzo V, Maria RS, et. al (2019) Neurobiological features and response to eye movement desensitization and reprocessing treatment of posttraumatic stress disorder in patients with breast cancer. European J Psychotraumatology 10(1).
- Abdi N, Malekzadeh M, Fereidouni Z, Behnammoghadam M, Parisa Z, et al. (2021) Efficacy of EMDR Therapy on the Pain Intensity and Subjective Distress of Cancer Patients. J EMDR Practice Res 15(1).
- Brown Lawren C, Amy R Murphy, Chloe S Lalonde, Preeti D Subhedar, Andrew H Miller, et al. (2020) posttraumatic stress disorder and Breast Cancer: Risk Factors and the Role of Inflammation and Endocrine Function. Cancer 126(14): 3181-3191.
- Adellund Holt K, Jensen PT, Gilså Hansen D, Elklit A, Mogensen O (2016) Rehabilitation of women with gynecological cancer: the association between adult attachment, post-traumatic stress disorder, and depression. Psycho-oncology, J Psychol Behav Soc Dimen Cancer 25(6): 691-698.
- Kangas M (2013) DSM-5 Trauma and Stress-Related Disorders: Implications for Screening for Cancer-Related Stress. Front Psychiatry 4(122): 691-698.
- American Psychiatric Association (2022) Diagnostic and statistical manual of mental disorders (5th edn., text rev.).
- Xin Wu, Jieru Wang, Reuben Cofie, Atipatsa C Kaminga, Aizhong Liu (2016) Prevalence of Posttraumatic Stress Disorder among Breast Cancer Patients: A Meta-analysis. Iran J Public Health 45(12): 1533-1544.
- Oliveri S, Paola Arnaboldi, Silvia Francesca Maria Pizzoli, Flavia Faccio, Alice V Giudice et al. (2019) PTSD Symptom Clusters Associated with Short- and Long-term Adjustment in Early Diagnosed Breast Cancer Patients. Ecancermedicalscience 13(917).
- Hong-Ming Chen, Vincent Chin-Hung Chen, Han-Pin Hsiao, Yi-Ping Weng, Ya-Ting Hsu, et al. (2019) Correlations and Correlates of Post-Traumatic Growth and Post-Traumatic Stress Symptoms in Patients with Breast Cancer. Neuropsychiatr Dis Treat 15: 3051-3060.
- Arnaboldi P, Claudio Lucchiari, Luigi Santoro, Claudia Sangalli, Alberto Luini, et al. (2014) PTSD Symptoms Because of Breast Cancer Diagnosis: Clinical Implications. SpringerPlus 3(392).
- Bulotienė G, Matuizienė J (2014) Posttraumatic Stress in Breast Cancer Patients. Acta Medica Lituanica 21(2): 43-50.
- Varinka V, Franziska Neufeld, Judith Kaste, Markus Bühner, Philipp Sckopke, et al. (2017) Clinically Assessed Posttraumatic Stress in Patients with Breast Cancer During the First Year After Diagnosis in the Prospective, Longitudinal, Controlled Cognicares Study. Psycho-Oncology, J Psychol Behav Soc Dimen Cancer 26(1): 74-80.
- Mallet J, Olivier Huillard, Francois Goldwasser, Caroline Dubertret, Yann Le Strat, et al (2018) Mental Disorders Associated with Recent Cancer Diagnosis: Results from a Nationally Representative Survey. Eur J Cancer 105: 10-18.
- Šimunović M, Ljubotina D (2020) Prevalence, structure and predictors of posttraumatic stress disorder in Croatian patients following breast cancer. Psychiatr Danub 32(2): 187-196.
- Dimitrov L, Moschopoulou E, Korszun A (2019) Interventions for the Treatment of Cancer-related Traumatic Stress Symptoms: A Systematic Review of the Literature. Psycho-oncology 28(5): 970-979.
- Shapiro F (2018) Eye movements desensitization and reprocessing. Basic principles, protocols, and procedures (Third edition). Guilford Press.
- Hase M, Balmaceda UM, Ostacoli L, Liebermann P and Hofmann A (2017) The AIP Model of EMDR Therapy and Pathogenic Memories. Front Psychol 8: 1578.
- American Psychiatric Association (2013) Diagnostic and Statistical Manual of Mental Disorders (5th) Arlington, VA, United States.
- Jarero I, Artigas L, Luber M (2011) The EMDR Protocol for Recent Critical Incidents: Application in a Disaster Mental Health Continuum of Care Context. J EMDR Practice Res 5(3): 82-94.
- Jarero I, Uribe S (2011) The EMDR Protocol for Recent Critical Incidents: Brief Report of An Application in a Human Massacre Situation. J EMDR Practice Res 5(4): 156-165.
- Jarero I, Uribe S (2012) The EMDR Protocol for Recent Critical Incidents: Follow-up REPORT of An Application in a Human Massacre Situation. J EMDR Practice Res 6(2): 50-61.
- Jarero I, Amaya C, Givaudan M, Miranda A (2013) EMDR Individual Protocol for Paraprofessionals Use: A Randomized Controlled Trial Whit First Responders. J EMDR Practice Res 7(2): 55-64.
- Jarero I, Uribe S, Artigas L, Givaudan M (2015) EMDR Protocol for Recent Critical Incidents: A Randomized Controlled Trial in a Technological Disaster context. J EMDR Practice Res 9(4): 166-173.
- Jarero I, Schnaider S, Givaudan M (2019) EMDR Protocol for Recent Critical Incidents and Ongoing Traumatic Stress with First Responders: A Randomized Controlled Trial. J EMDR Practice Res 13(2): 100-110.
- Encinas M, Osorio A, Jarero I, Givaudan M (2019) Randomized Controlled Clinical Trial on the Provision of the EMDR-PRECI to Family Caregivers of Patients with Autism Spectrum Disorder. Psychol Behav Sci Int J 11(1): 1-8.
- Estrada BD, Angulo BJ, Navarro ME, Jarero I, Sánchez-Armass O (2019) PTSD, Immunoglobulins, and Cortisol Changes after the Provision of the EMDR- PRECI to Females Patients with Cancer-Related PTSD Diagnosis. Am J Applied Psychol 8(3): 64-71.
- Jiménez G, Becker Y, Varela C, García P, Nuño MA, et al. (2020) Multicenter Randomized Controlled Trial on the Provision of the EMDR-PRECI to Female Minors Victims of Sexual and/or Physical Violence and Related PTSD Diagnosis. Am J Applied Psychol 9(2): 42-51.
- Jarero I (2021) ASSYT Treatment Procedures Explanation. Technical Report. Research Gate.
- Kleim B, Graham B, Bryant RA, et al. (2013) Capturing Intrusive Re-Experiencing in Trauma Survivors’ Daily Lives Using Ecological Momentary Assessment. J Abnorm Psychol 122(4): 998-1009.
- Astill Wright L, Horstmann L, Holmes EA et al. (2021) Consolidation/Reconsolidation Therapies for the Prevention and Treatment of PTSD and Re-Experiencing: A Systematic Review and Meta-Analysis. Transl Psychiatry 11(1): 453.
- Becker Y, Estévez ME, Pérez MC, Osorio A, Jarero I, et al. (2021) Longitudinal Multisite Randomized Controlled Trial on the Provision of the Acute Stress Syndrome Stabilization Remote for Groups to General Population in Lockdown During the COVID-19 Pandemic. Psychol Behav Sci Int J 16(2): 1-11.
- Smyth-Dent K, Becker Y, Burns E, Givaudan M (2021) Acute Stress Syndrome Stabilization Remote Individual (ASSYST-RI) for Tele Mental Health Counseling After Adverse Experiences. Psychol Behav Sci Int J 16(2): 1-7.
- Magalhães SS, Silva CN, Cardoso MG, Jarero I, Pereira Toralles MB (2022) Acute Stress Syndrome Stabilization Remote for Groups Provided to Mental Health Professionals During the Covid-19 Pandemic. J Med Bio Sci 21(3): 637-643.
- Mainthow N, Pérez MC, Osorio A, Givaudan M, Jarero I (2022) Multisite Clinical Trial on the ASSYST Individual Treatment Intervention Provided to General Population with Non-Recent Pathogenic Memories. Psychol Behav Sci Int J 19(5): 1-9.
- Smith S, Todd M, Givaudan M (2023) Clinical Trial on the ASSYST for Groups Treatment Intervention Provided to Syrian Refugees living in Lebanon. Psychol Behav Sci Int J 20(2): 1-8.
- Mainthow N, Zapien R, Givaudan M, Jarero I (2023) Longitudinal Multicenter Randomized Controlled Trial on the ASSYST Individual Treatment Intervention Provided to Adult Females with Adverse Childhood Experiences. Psychol Behav Sci Int J 20(3): 556040.
- Magalhães SS, Guimarães ACF, Silva CN, Souza JSS, Carasek L, et al. (2023) Randomized Clinical Controlled Trial on the ASSYST Treatment Intervention Provided to Public Sector Workers During the COVID-19 Pandemic. Psychol Behav Sci Int J 20(5): 1-11.
- Carretero KP, Delgadillo A, Villarreal AM, Roque JP, Poiré A, et al. (2023) Randomized Controlled Trial on the ASSYST Treatment Intervention with Female Children Polytraumatized by Adverse Childhood Experiences, Neglect and Maltreatment. Academ J Pediatr Neonatology 12(5): 1-9.
- Marx BP, Lee DJ, Norman SB, Bovin MJ, Sloan DM, et al. (2021) Reliable and Clinically Significant Change in the Clinician-Administered PTSD Scale for DSM-5 and PTSD Checklist for DSM-5 Among Male Veterans. Psychological Assessment. Advance online publication.
- (2010) Consolidated Standards of Reporting Trials (CONSORT) 2010 Statement. BMJ 340: c869.
- (2013) Standard Protocol Items Recommendation for Interventional Trials (SPIRIT) 2013 Checklist.
- Weathers FW, Litz BT, Keane TM, Palmieri PA, Marx BP, et al. (2013) The PTSD Checklist for DSM-5 (PCL-5).
- Bovin MJ, Marx BP, Weathers FW, Gallagher MW, Rodriguez P, et al. (2016) Psychometric properties of the PTSD checklist for diagnostic and statistical manual of mental disorders- Firth edition (PCL-5) in veterans. Psychol Assess 28(11): 1379-1391.
- Franklin CL, Raines AM, Cucurullo L-A, Chambliss JL, Maieritsch KP, et al. (2018) 27 ways to meet PTSD: Using the PTSD-checklist for DSM-5 to examine PTSD core criteria. Psychiatry Res 261: 504-507.
- Zigmond AS, Snaith RP (1983) The Hospital Anxiety and Depression Scale. Acta Psychiatrica Scandinavica 67: 361-370.
- Ying Lin C, Pakpour AH (2017) Using Hospital Anxiety and Depression Scale (HADS) on patients with epilepsy: Confirmatory factor analysis and Rasch models. Seizure 45: 42-46.






























