Cannula, Conduit, Subway: Syringoma
Anubha Bajaji*
Histopathologist in A.B. Diagnostics, India
Submission: February 02, 2019; Published: July 19, 2019
*Corresponding author:Anubha Bajaji, Histopathologist in A.B. Diagnostics, New Delhi, India
How to cite this article: Anubha B. Cannula, Conduit, Subway: Syringoma. Adv Res Gastroentero Hepatol. 2019; 13(3): 555865. DOI: 10.19080/ARGH.2019.13.555865.
Keywords: Syringoma; Benign adnexal tumour; Supra-clavicular region; Neck; Anterior or posterior trunk; Marfan’s syndrome; Hyperthyroidism; Miniscule; Multiple; Skin coloured; Light brown; Axillae; Abdomen; Lower eyelids; Cheeks
Preface
The term “Syringoma” is a derivative of the Greek word “Syrinx” which connotes a pipe or a tube. Syringoma is a benign adnexal tumour commonly affecting adolescents’ females, delineated in an estimated 1% population and configuring well differentiated ducts and cellular strands of basaloid epithelium. The tumour recapitulates the glandular epithelial structures. The neoplasm usually articulates as an intra-dermal or subcutaneous nodule. As syringoma frequently appears at puberty it can manifest a significant cosmetic impact [1,2].
Clinical Elucidation
Individuals remains asymptomatic or rarely demonstrate pruritus while perspiring. Syringoma usually exhibits miniscule, multiple, skin coloured, light brown or mildly pigmented papules. The peri-orbital or supra-clavicular region, neck, anterior or posterior trunk are incriminated sites, particularly in the eruptive form. Adjunctive sites such as axillae, abdomen, lower eyelids, cheeks and extremities can be implicated.
Four major clinical variants are categorized by Friedman and Butler as i) localized, ii) familial, iii) a generalized subtype which incorporates eruptive and multiple lesions and iv) associated with Down’s syndrome [2,3]. Syringoma is also prominently associated with Down’s syndrome (22.2%), diabetes mellitus (2.1%), Ehlers Danlos syndrome, Marfan’s syndrome and hyperthyroidism. Syringoma appearing in conjunction with milia, cysts and atrophoderma vermiculata are cogitated as Nicolau- Balus syndrome [2,3].
Histological Elucidation
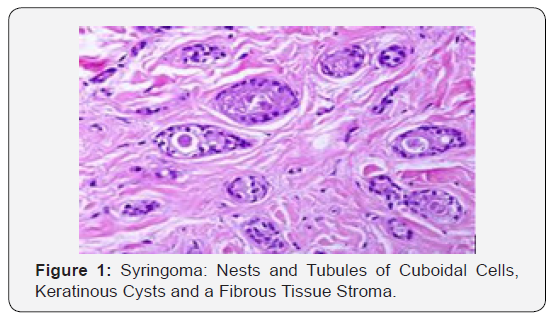
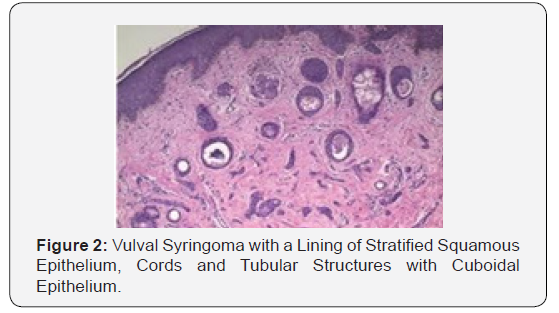
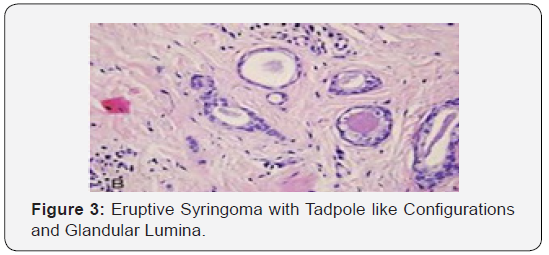
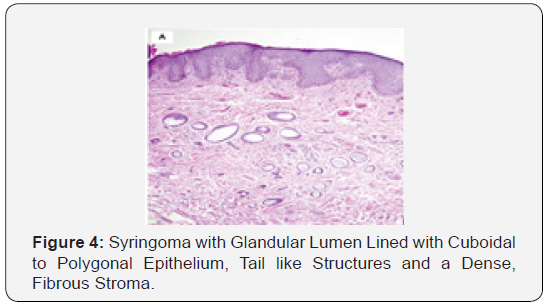
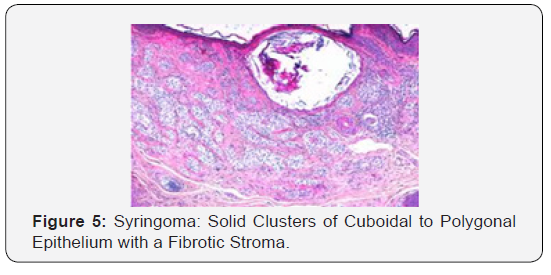
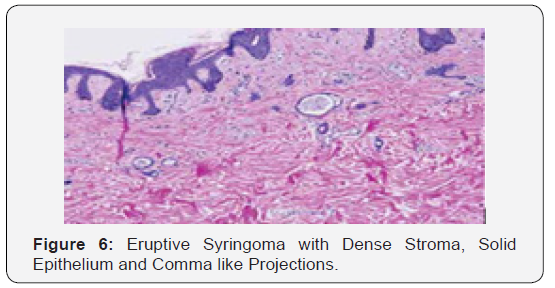
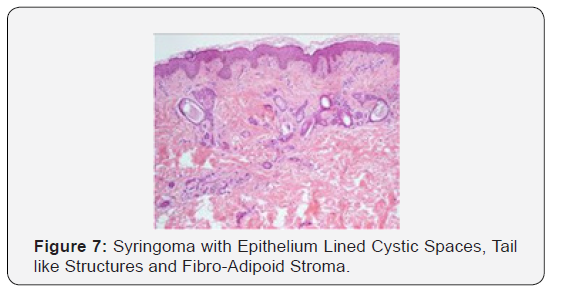
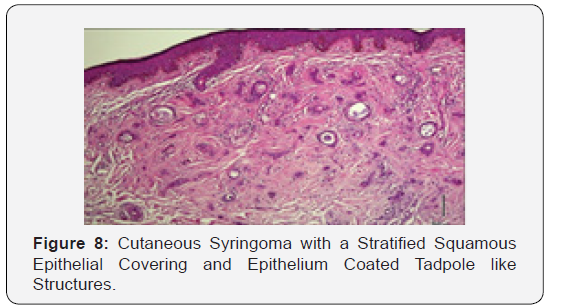
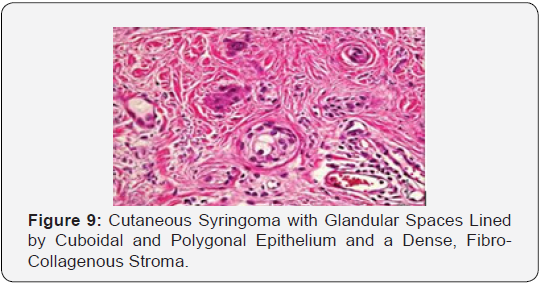
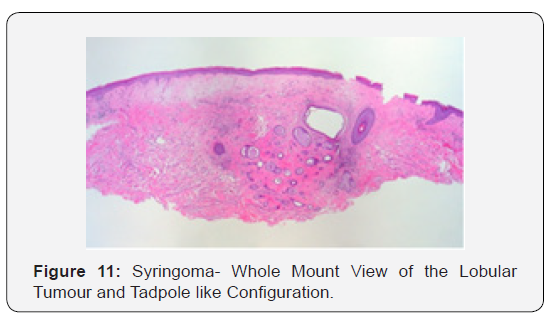
Syringoma as a benign adnexal tumour comprises of miniature, dilated and cystic spaces lined by a dual layer of cuboidal epithelium interspersed with solid epithelial strands and columns of identical cuboidal cells and ductal articulations embedded in a dense, fibrotic stroma. A predominant fibro-connective tissue stroma envelops numerous miniature ducts lined with a dual layer of epithelial cells. The cellular epithelium appears flattened in a majority of the cases. The internal row of epithelium occasionally displays vacuoles. The duct Lumina frequently contain amorphous cellular debris. A few ducts and cystic spaces exhibit miniscule tail like, comma shaped elongations of epithelial cell configurations resembling tadpoles. A cluster of such articulations can produce a distinctive paisley like pattern. In addition to the ducts, solid strands of basophilic epithelial cells can be elucidated [1,3].
Cystically dilated ductal Lumina are impacted with keratin and often depict keratohyaline granules. The duct Lumina display a lining epithelium and generally about the superficial epidermis. The keratin cysts may rupture and incur a prominent foreign body giant cell reaction with subsequent though infrequent calcification. Exceptionally, several tumour cells appear as clear cells on account of glycogen accumulation. A few ductular structures and epithelial cords with preponderant cellular islands of irregular outline and magnitude can co-exist. Precluding the peripheral cell layer, the lining epithelium is preponderantly comprised of clear cells. The lesions are conventionally situated in upper to mid-dermis [3,4].
Enzyme Histochemistry
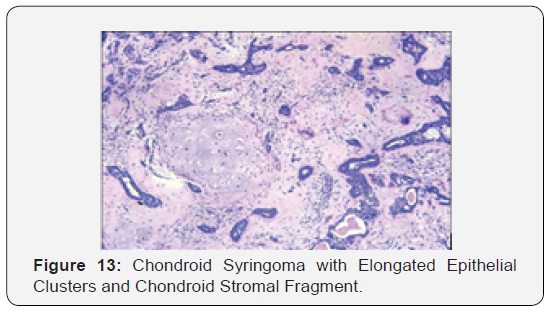
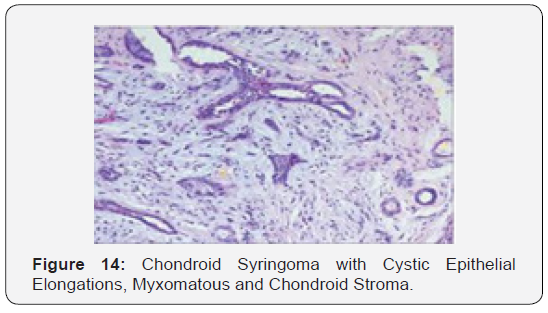
Discerns the syringoma as a tumor which differentiates towards intra epidermal eccrine sweat ducts and glands. Eccrine enzymes such as succinic dehydrogenase, phosphorylase and leucine aminopeptidase abundantly constitute the syringoma. The tumour cells display a weak reaction to lysosmal enzymes. Apocrine structures react to enzymes such as acid phosphatise and beta glucouronidase. Electron microscopic studies similarly cogitate the genesis of the tumour as being of eccrine sweat glands and ducts [4,5] (Figures 1-14).
Distinguishing Diagnosis
A clinical distinction of syringoma or a chondroid variant is mandated from lesions such as epidermal inclusion cyst, amelanotic nevus, sebaceous cyst, dermoid cyst, schwannoma, neurofibroma, pilomatrixoma and basal cell carcinoma [5,6]. An eruptive syringoma necessitates a demarcation from the clinically identical papular childhood dermatosis such as plane warts, acne vulgaris, lichen planus, granuloma annulare, papular sarcoidosis, milia, sebaceous hyperplasia, eruptive xanthoma, urticaria pigmentosa. Darier’s disease, pseudoxanthoma elasticuma and hidrocystoma.
Solid strands and columns of basophilic epithelial cells appear entrenched in a fibrous tissue stroma, a morphologically identical manifestation of a syringoma and a fibrosing basal cell epithelioma. However, fibrosing basal cell epithelioma is devoid of ductular configurations encompassing amorphous, eosinophilic material. Keratinous horn cysts abutting the epidermis delineated in syringoma often recapitulate the horn cysts of trichoepithelioma and can be misdiagnosed as the aforementioned tumours in a singular lesion. Although trichoepithelioma depicts solid columns and strands of basophilic epithelial cells and keratinous horn cysts, it frequently lacks ductal articulations [4,6]. Eruptive syringoma requires a histological distinction from conditions such as sclerosing (morphea- like) basal cell carcinoma and desmoplastic trichoepithelioma. Syringoma also necessitates a distinction from tumours of identical histology such as a microcystic adnexal carcinoma. The tumour tends to infiltrate the deep dermis and subcutaneous tissue [7,8].
Acknowledgements
Surgical excision, dermabrasion, cryotherapy, chemical peels particularly trichloro-acetic acid, topical atropine, carbon dioxide laser or pulsed dye laser can be beneficially adopted to treat syringomas. Post therapeutic adverse reactions such as scarring and dys-pigmentation can appear with employment of aforementioned options. Oral administration of isotretinoin in cumulative doses is advantageous and ensures a decline in the quantity and magnitude of syringomas.
Occasionally, administration of isotretinoin can be ineffective [8,9]. Several therapeutic strategies are available for managing eruptive syringoma or variants. However, the efficacy is restricted on account of the tumours being situated in the dermis with possible reoccurrences. Lesions can be eradicated by mild electrodessication or shaving. Carbon dioxide laser therapy with the pinhole technique and fractional thermolysis appears to be efficacious. Enlarged lesions necessitate a surgical extermination. Adjunctive modalities include cryosurgery, chemical peeling, dermabrasion and oral or topical retinoids. Appropriate for a syringoma or the chondroid variant is a comprehensive surgical excision with a prevention of future reoccurrences. Surgical extrication of the tumefaction appears effortless on account of the circumscribing capsule. A recalcitrant, enucleated or an incompletely excised tumour lobule enhances the risk of tumour reappearance and malignant transformation. Thus, an annual post-operative monitoring is a pre-requisite. Infrequently, tumours reappear following a comprehensive eradication within one to two years [9-11].
Therapeutic Options
Chondroid syringoma (CS) is an infrequent dermal tumour arising from the sweat glands, initially enunciated by Billroth in 1859. The neoplasm is a dual epithelial –mesenchymal lesion and is cogitated as the “mixed tumour”. Chondroid differentiation is cogent to the mesenchymal component of the tumour. The tumour morphologically recapitulates mixed tumours of the salivary and lacrimal glands. Hirsch and Helwig rephrased “myxoid” skin tumour with the terminology of “chondroid syringoma” in 1961 on account of the sweat gland elements articulating in a cartilaginous stroma [1,2]. The nomenclature demarcates betwixt the mixed tumours of salivary or lacrimal glands and the exceptional sweat gland tumours. Majority of the mixed sweat gland tumours are benign. As a mixed tumour, the neoplasm can be cogitated as a pleomorphic adenoma of skin. Reoccurrences, benign and malignant conformities are described
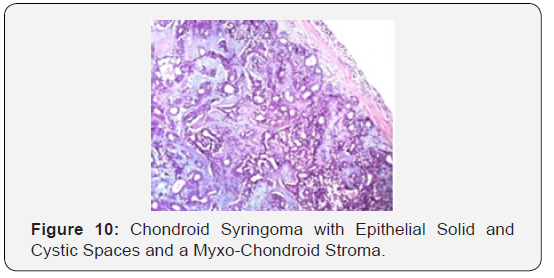
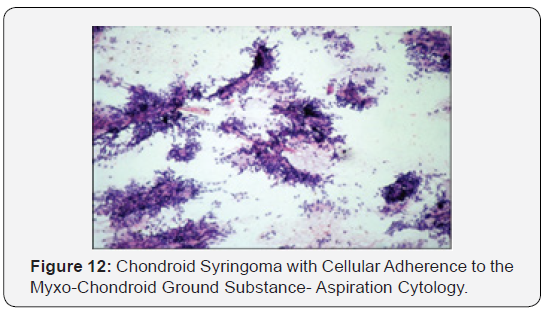
Head and neck region are frequently implicated, especially the cheek, eyelid, forehead, nose, upper lip and infrequently, the orbit. Chondroid syringoma also arises in the scalp, hand, foot, axilla, abdomen, penis, vulva and scrotum [3,4]. Chondroid syringoma has an estimated prevalence of 0.01% to 0.1% of the primary cutaneous tumours and is frequently elucidated at an average age of 50 years with a minimal male preponderance. The majority (80%) of the neoplasm are distributed in the head and neck preferentially on the nose, upper lip and eyebrows. Chondroid syringoma appears clinically as a gradually evolving, solitary, painless, well defined, firm, papule or tumour aggregate varies from 0.5 centimetre to 3.0 centimetre magnitude. The neoplasm is located in the dermis or sub-dermis. Superficial surface can be non-ulcerated, erythematous, purple or skin coloured and the nodule is unattached to the subjacent tissues [4,5]. On histology, a characteristic admixture of epithelial and mesenchymal constituents is confirmatory of the “mixed tumour” chondroid syringoma. Chondroid syringoma demonstrates dual histological variants -apocrine and eccrine. The apocrine subtype is frequent with randomly dispersed aggregates of squamous epithelial cells, tubular or branching glandular lumina and a double layered epithelial lining. The eccrine subtype typically depicts homogenous epithelial elements with miniscule, tubular and glandular lumina and a singular epithelial l lining.
Focal immune reactivity to keratin, vimentin, desmin and S-100 protein is elucidated. Distinct, diagnostic histological criterion for chondroid syringoma incorporate 1) clusters of cuboidal or polygonal epithelial cells 2) inter-digitating tubule-alveolar configurations with a dual or multi-layered cuboidal epithelial lining 3) ductal articulations lined by one or two columns of cuboidal cells 4) occasional keratinous cysts 5) a matrix composed of mesenchymal constituents in variable quantities. Tubules, alveoli and glandular articulations lined by two or more layers of cuboidal epithelium are exemplified. Islands of cuboidal or polygonal epithelium embedded in a fibro-adipoid, chondroid, hyaline or mucinous hypo-cellular stroma can be delineated [4,5].
A typical lesion of chondroid syringoma may display the entire spectrum of histological modifications or manifest only a few features. Electron microscopy, immune histochemistry and enzymatic analysis determine an apocrine or eccrine genesis of the tumour. Although benign, chondroid syringoma cogitates an exceptional malignant subtype. Atypical or malignant tumours elucidate a de novo emergence or may follow an inadequate resection. Malignant transformation in a chondroid syringoma is exceptional and is characterized by nuclear atypia, enhanced mitosis, infiltrative tumour margins, satellite tumour nodules, necrosis and rapid tumour progression. A neoplasm exceeding 3 centimeters in magnitude may possibly be malignant. Radiological assay of the tumour can analyse attributes such as anatomical expanse of the tumefaction, tumour invasion or adherence to adjacent anatomical structures. A chondroid syringoma necessitates a demarcation from adjunctive benign tumours of epidermal or mesenchymal appendages such as pleomorphic adenoma, dermoid cyst, neurofibroma, lymphangioma, cavernous haemangioma, lipoma, sebaceous cyst, lymphoma and histiocytic tumours [6,7].
Therapeutic strategies for managing chondroid syringoma include electrodessication and tumour vapourisation with carbon dioxide or Argon laser. However, a total surgical extermination remains the preferential therapy. Consistent monitoring for excluding tumour reoccurrence and malignant transformation is a pre-requisite. Chondroid syringoma may exemplify a salivary gland tumour and comprises of varying quantities of mucoid and cartilaginous stroma. Eruptive syringoma was first scripted by Jacquet and Darier in 1887. Eruptive syringoma as an exceptional tumefaction emerges as multitudinous, sequential lesions arising at puberty or childhood. Eruptive or disseminated syringoma typically develops hundreds of miniscule, poor to well demarcated, smooth surfaced, skin colored, pink, yellow or brownish papules ranging from one centimeter to five centimeters in magnitude. The lesions occur on the anterior chest, neck, eyelids, upper cheeks. upper abdomen, axilla, trunk, genitalia, extremities and peri-umbilical area. Eruptive syringoma arises in concordance with Down’s syndrome and Ehlers Danlos syndrome. Individuals with eruptive syringomas infrequently demonstrate a family history of identical lesions. Familial eruptive syringoma is an exceptional, autosomal dominant condition [5,6]. Eruptive syringoma is a disorder of obscure pathogenesis although a hyperplastic reaction of the eccrine duct to an inflammatory stimulus such as contact dermatitis, shaving, laser hair removal, alopecia areata or radiation dermatitis can be implicated. Alternatively, eruptive syringomas can emerge due to autoimmune damage to the acrosyringium and are cogitated as an acrosyringitis with ductal cysts. Eruptive syringoma can be cosmetically debilitating for individuals. A characteristic histology is diagnostic of syringoma. The lesions depict a follicular and non-follicular morphology [5,6].
Variants of Syringoma
References
- Wong JY, Becker SN (1982) Enterobius Vermicularis ova in routine cervicovaginal smears: Light and scanning electron microscopic observations. Acta Cytol 26(4): 484-487.
- Craggs B, De Waele E, De Vogelaere K, Wybo I, Laubach M, et al. (2009) Enterobius Vermicularis with tuboovarian abscess and peritonitis occurring during pregnancy. Surg Infect 10: 545-547.
- Parija SC (2006) Hookworms and other Bursate Nematodes, Oxyruid and Ascarid Nematodes. In: Parija SC, (Edt.), Textbook of Medical Parasitology- Protozology and Helminthology, (3rd edn.), New Delhi: All India, Publishers and Distributors, pp. 313-316.
- Mattia AR (1992) Perianal mass and recurrent cellulites due to Enterobius Vermicularis. Am J Trop Med Hyg 47: 811-815.
- Babady NE, Awender E, Geller R, Miller T, Scheetz G, et al. (2011) Enterobius Vermicularis in a 14-year-old girl’s eye. J Clin Microbiol 49(12): 4369-4370
- Sheety JB, Kulkarni DV, Prabhu VL (2012) Eggs containing larvae of Enterobius Vermicularis in vaginal smear. J Cytol 29(1): 94-96.






























