Exploring the Antifungal and Antibacterial Properties of Diethyl-4-Hydroxy-4-Methyl-2-(3-Nitrophenyl)-6-Oxocyclohexane-1,3-Dicarboxylate
Muhammad Shoaib1,2*
1Department of Microbiology, Faculty of Biology, Baku State University, Azerbaijan
2Department of Parasitology and Microbiology, PMAS Arid Agriculture University, Pakistan
Submission: October 09, 2019; Published: December 20, 2019
*Corresponding author: Muhammad Shoaib, Department of Microbiology, Faculty of Biology, Baku State University, Azerbaijan
How to cite this article: OMuhammad Shoaib. Exploring the Antifungal and Antibacterial Properties of Diethyl-4-Hydroxy-4-Methyl-2-(3-Nitrophenyl)-6- Oxocyclohexane-1,3-Dicarboxylate. 2019; 15(2): 555906. DOI: 10.19080/AIBM.2019.14.555906
Abstract
Due to increasing antimicrobial resistance, functionally substituted cyclohexane derivatives are being explored as antimicrobial agents. Agar well diffusion method was used to evaluate antibacterial and antifungal properties of diethyl-4-hydroxy-4-methyl-2-(3-nitrophenyl)-6-oxocyclohexane-1,3-dicarboxylate against Gram-positive bacteria, Gram-negative bacteria and fungi. Test compound exhibited moderate antibacterial properties against Gram-negative bacteria, while Gram-positive bacteria were found to be resistant. Candida tropicalis was found to be most sensitive yeast. Our findings reveal that tested compound can act as potential antimicrobial agent in the future.
Keywords: Antimicrobial resistance; Diethyl-4-hydroxy-4-methyl; Dicarboxylate; Cyclohexane derivatives; Bacteria; Dimethyl sulphoxide
Abbreviations: SDA: Sabouraud Dextrose Agar; DMSO: Dimethyl Sulphoxide; MIC: Minimum Inhibitory Concentration
Introduction
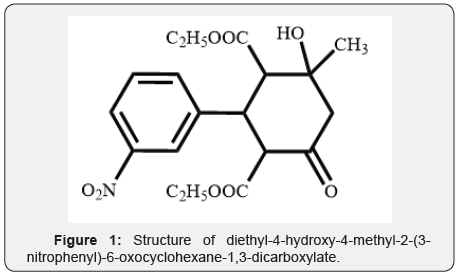
Ever increasing antimicrobial resistance is one of major challenges faced by medical community. Multi drug resistant bacteria and extensively drug resistant bacteria are major obstacle in eradication of infectious diseases. Pan drug resistant bacteria, which are resistant to all available drugs, have also emerged [1]. Unwise use of available antibiotics and shortage of new antimicrobial drugs are main reasons for development of antimicrobial resistance [2]. Drug development studies are focusing functionally substituted and hybrid organic molecules as potential antimicrobial agents of future. The higher anticipated antimicrobial activity of these molecules is conferred to their unique mode of action [3]. Functionally substituted derivatives of cyclohexane have potential antimicrobial properties and can act as probable antimicrobial agents in future [4]. In synthesis of organic compounds, diacetyl (diethoxy carbonyl) hydroxy cyclohexanones are used as construction blocks. Presence of numerous types of oxo groups in these blocks imparts higher chemical activity to them [5-9]. Functionally substituted cyclohexanones are widely explored for their potential antimicrobial properties. Keeping in mind the antimicrobial potential of functionally substituted cyclohexanones, here we report antifungal and antibacterialactivityofdiethyl-4-hydroxy-4-methyl-2-(3-nitrophenyl)-6-oxocyclohexane-1,3-icar boxylate. The test compound was synthesized according to known procedures [10]. The structure of compound is shown in (Figure 1).
Material and Methods
Test compound was screened for antibacterial and antifungal activities by using standard agar well diffusion assay [11]. Mueller-Hinton agar was used to determine antibacterial activity while Sabouraud Dextrose Agar (SDA) was used to determine antifungal activity. Microbial suspension (0.5 McFarland) was spread on agar plate with help of sterile glass spreader and wells were made using sterile glass pinchers. Test compound (100 μl) was poured in each well and plate was incubated for 24 hours and 72 hours for bacteria and fungi, respectively. After incubation, zone of inhibition was carefully measured. Test compound was screened against four Gram-positive bacteria (Staphylococcus aureus BDU- 23, Bacillus Subtilis BDU-50, Bacillus mesentericus BDU-51 and Bacillus megaterium BDU-N20) and four Gram-negative bacteria (Escherichia coli BDU-12, Klebsiella pneumoniae BDU-44, Acinetobacter baumannii BDU-32 and Pseudomonas aeruginosa BDU-49). Three species of Candida (Candida tropicalis BDU LK30, Candida pelliculosa BDU KT55 and Candida pseudotropicalis BDU MA88) were used to check anti-yeast properties of compound. All the test cultures were taken from our own collection at Department of Microbiology, Baku State University. Dimethyl Sulphoxide (DMSO) was used as solvent to dissolve the test compound. Three different concentrations of test compound (0.3%, 0.1% and 0.05%) were screened for antibacterial and antifungal activity. Due to inert nature of DMSO, it was used as control and all the experiments were repeated three times.
Results and Discussions
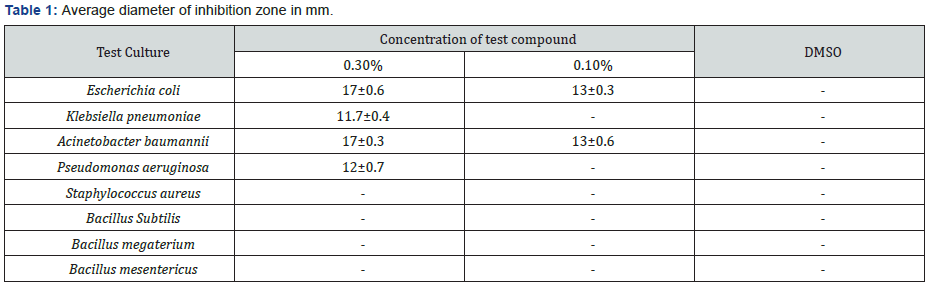
Test compound exhibited variable antibacterial and antifungal properties against different test cultures at concentration of 0.3% and 0.1%. All the tested bacteria and fungi were found to be resistant to test compound at concentration of 0.05%. Table 1 shows that test compound was found to be active against Gram-negative bacteria while Gram-positive bacteria were found to be resistant. These results are supported by [4], who also reported that cyclohexane tosyloxyimine derivative exhibited better antimicrobial properties against Gram-negative bacteria as compared to Gram-positive bacteria. This might be due to difference in structure of cell wall of Gram-positive bacteria and Gram-negative bacteria. Findings of [4,12,13] contradict our results which is due to difference in structure of our test compound and dialkyl carboxylate cyclohexane derivatives. Escherichia coli and Acinetobacter baumannii were most sensitive bacteria with zone of inhibition 17mm each. Test compound showed moderate antibacterial activity against Pseudomonas aeruginosa and Klebsiella pneumoniae with zone of inhibition 12 mm and 11.7 mm, respectively.
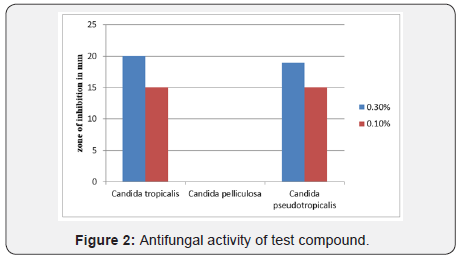
As shown in Figure 2, test compound exhibited better antifungal potential against Candida tropicalis and Candida pseudotropicalis with zone of inhibition 20 mm and 19mm, respectively. Candida pelliculosa was found to be resistant against tested compound. Our findings are similar to findings of [4], who showed that Candida pseudotropicalis was most sensitive yeast against functionally substituted cyclohexane derivatives. Our findings suggest that diethyl-4-hydroxy-4-methyl-2-(3-nitrophenyl)-6-oxocyclohexane- 1,3-dicarboxylate can act as probable antimicrobial agent in the future.
Conclusion
Shortage of new antimicrobial drugs and ever-increasing antimicrobial resistance accentuate the need of developing new antimicrobial drugs. Functionally substituted organic compounds are being considered as potential antimicrobial agents in future. Functionally substituted cyclohexane derivatives are being widely explored as probable antimicrobial agents. Antimicrobial profile of diethyl-4-hydroxy-4-methyl-2-(3-nitrophenyl)-6-oxocyclohexane- 1,3-dicarboxylate exhibits that it has potential to become effective antimicrobial substance. Therefore, Minimum Inhibitory Concentration (MIC) values must be determined to further strengthen the findings of current study.
References
- Bielawski K, Katarzyna L, Zbigniew K, Anna B, Olga M, et al. (2017) Synthesis and antimicrobial activity of chiral quaternary N-spiro ammonium bromides with 3′,4′-dihydro-1′h-spiro[isoindoline-2,2′isoquinoline] skeleton. Drug Des Devel Ther 11: 2015-2028.
- Hao H, Cheng G, Iqbal Z, Husssain HI, Ai X, et al. (2014) Benefits and risks of antimicrobial use in food-producing animals. Front Microbiol 5: 288.
- Tsemeugne J, Emmanuel SF, Jean-de-Dieu T, Taoufik R, Arnaud DN, et al. (2018) Synthesis, Characterization, and Antimicrobial Activity of a Novel Trisazo Dye from 3-Amino-4H-thieno[3,4c][1] benzopyran-4-one. Int J Med Chem 2018: 9197821.
- Shoaib M, Ganbarov G Kh (2019) Functionally Substituted Chemical Organic Compounds: Potential Antimicrobial Substances. J Microbiol Biotechnol 4(1): 000136.
- Ismiyev AI, Maharramov AM, Aliyeva RA, Askerov RK, Mahmudov KT, et al. (2013) Syntheses and some features of five new cyclohexane-1,3-dicarboxylates with multiple stereogenic centers. Journal of molecular structure 1032: 83-87.
- Poplevina NV, Kuznetsova AA, Krivenko AP (2010) Synthesis and structure of substituted triazoloquinazolines. Chem Heterocycl Compd 46(9): 1148-1150.
- Dyachenko VD, Sukach SM (2012) Synthesis of 1Hpyrazolo[3,4-c] isoquinolin-1-ones by the condensation of cyclohexanone derivatives with 3amino-1-phenyl-1H-pyrazol-5(4H)-one. Russian Journal of General Chemistry 82(2): 305-309.
- Magerramov AM, Ismiev AI, Kadyrova NA, Gadzhieva KE, Askerov RK, et al. (2013) Crystal structure of diethyl- 1- isobutyl- 9- hydroxy- 9- methyl- 7- phenyl- 1, 4- diazaspiro [4,5] decane - 6, 8 - dicarboxylate C25H38N2O5. Journal of Structural Chemistry 54(6): 1137-1139.
- Finar I (1961) The structure of 1,5-diketones. J Chem. Soc 2: pp. 674-679.
- Balouiri M, Sadiki M, Ibnsouda SK (2016) Methods for in vitro evaluating antimicrobial activity: A Review. J Pharm Anal 6(2): 71-79.
- Shoaib M (2019) Synthesis, Antibacterial and Antifungal Properties of Cyclohexane Tosyloxyimine Derivative. J Microbiol Biotechnol 4(3): 000150.
- Shoaib M, Ganbarov G, Israilova A, Babayeva I, Ismiyev A (2019) Synthesis and antimicrobial activity of new functionally substituted dialkyl carboxylate cyclohexane derivatives. German Science Herald 13-17.
- Ismiyev A, Shoaib M, Ganbarov G, Agayeva N (2019) Synthesis and antimicrobial activity of novel toluene sulfonyl derivatives of pyrazoles annelated with a polyfunctional cyclohexane ring. Advances in Biology & Earth Sciences 4 (2): 88-92.






























