CO2 Mitigation Potentials of Microalgae: Its Expansion to a New Dimension for Closing the Loop of Carbon Between Source and Acquisition Through Food Chain of Phytophagous Fish in Open Ponds
B B Jana*
International Centre for Ecological Engineering, Centre for Human Resource Development (KSI), University of Kalyani, India
Submission: September 17, 2018; Published: January 03, 2019
*Corresponding author: B B Jana, International Centre for Ecological Engineering, University of Kalyani, Kalyani- 741235, West Bengal, India
How to cite this article: B B Jana. CO2 Mitigation Potentials of Microalgae: Its Expansion to a New Dimension for Closing the Loop of Carbon Between Source and Acquisition Through Food Chain of Phytophagous Fish in Open Ponds. Adv Biotech & Micro. 2019; 12(1): 555827. DOI: 10.19080/AIBM.2019.12.555827
Abstract
The burning issue of global warming and climate change has become a major worldwide concern of the day. Though numerous efforts are being made for mitigation of carbon, use of microalgae for capturing carbon in water bodies appear to be more attractive and promising because of their much higher bicarbonate driven carbon capture efficiency in water than atmospheric CO2 capture performance by terrestrial plants. Microalgae have been effectively used for biofuel and biochar production through biotechnological approach, the less explored area is its use in closing the loop of carbon cycle by trapping atmospheric carbon mediated through CO2- CO3- HCO3 system mechanism and production of nutrient regulated selective algae used for food for herbivorous fishes in open ponds. This is a win- win strategy towards carbon mitigation and low carbon foot print fish production in water bodies.
Keywords: Chlorella growth, aeration, C-sequestration, compressed CO2, open ponds
Introduction
Unbalanced emissions of atmospheric CO2 causing global warming and climate change pose major threat to sustainability.
Three major strategies that can reduce the carbon pool in the atmosphere below threshold level include:
a. reductions in CO2 emissions in the global atmosphere.
b. carbon capture for long-term storage using oceanic, geological, and chemical processes.
c. exploring alternative energy source for carbon-based fuel. Though oceanic, geological and chemical transformation methods are the major sources of carbon capture, biological sequestration especially using smart agriculture and aquaculture appear to be more attractive and promising because of ease, cost effective, and negative environmental impacts.
Despite microalgae have been extensively researched for biofuel or biochar production through biotechnological interventions [1, 2, 3]; the less explored area is its use in closing the loop of carbon between capture and smart carbon resilient aquaculture of herbivorous fishes in open ponds. This is a win-win strategy towards carbon mitigation and production of low carbon foot print fish especially in tropical countries. The purpose of the study is to overview the role of microalgae in global warming mitigation and to focus on closing the loop in carbon cycle by exploiting the trapped carbon through food chain of low carbon foot print fish in open ponds.
Microalgal photosynthesis
Microalgae have chlorophyll a though their several modifications have occurred through evolution in higher plants. Chlorophylls are made up of lipid-soluble hydrocarbon tail (C20H39 -), a hydrophilic head and a magnesium ion at the center. Photosynthesis is a process where conversion of photon energy into chemical energy occurs to form glucose and water and oxygen is released (Figure 1) as byproduct. Though visible spectrum is the source of energy, the maximum contributions are from the violet-blue, reddish orange- red wavelengths of the spectrum. The photoactivation is the first stage of photosynthesis when absorption of energy from a photon takes place in raising a molecule from its ground state or chromophore. As a result, water molecules are split off and transfer of energy to ATP and reduced NADP (nicotinamide adenine dinucleotide phosphate) are produced.
The chloroplast contained in the chlorophyll of microalgae, similar to other plants, is the site of photosynthetic reactions carried out in two separate steps of reactions - the biophysical and biochemical (Figure 1). The biophysical reactions take place in the thylakoid discs of the chloroplasts and water is oxidized, and oxygen is produced (Figure 2). The energy produced in the form of ATP and NADPH is used to fix or assimilate CO2 in the dark reactions. The biochemical reaction occurs in the chloroplast stroma resulting in the formation of primarily sugar molecules with some other organic molecules required for cell function and metabolism of microalgae.
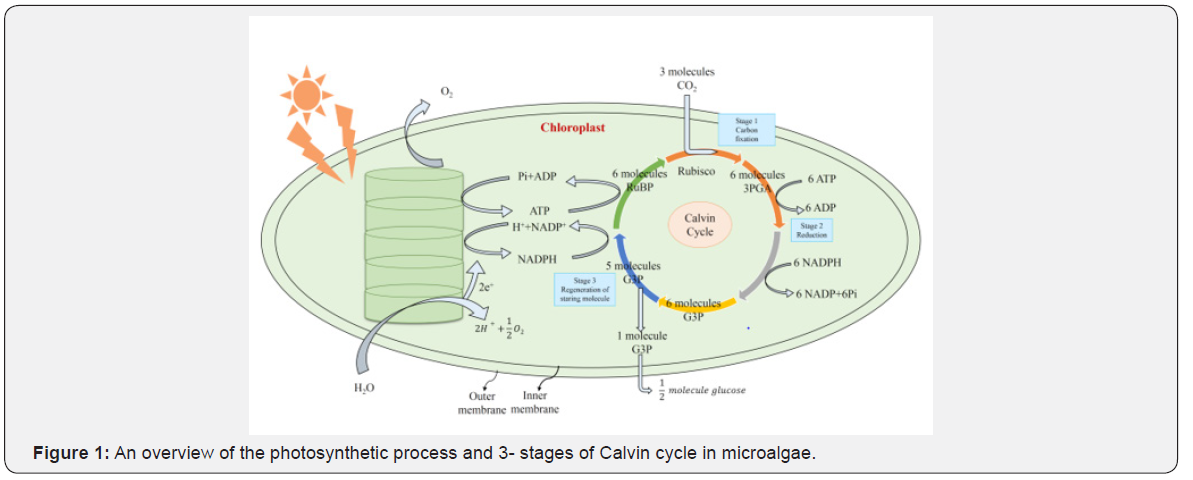

Advantage of carbon sequestration by microalgae in wetlands
The role of wetlands with a hot spot of green biodiversity has generally been underestimated despite their immense carbon capture and storage capacity potentials [4, 5, 6, 7]. Wetlands contain about 35 per cent of global terrestrial carbon though they comprise only 6 - 9% of the Earth’s surface. The carbon flux in water bodies depends on surface area, water concentrations and gas transfer velocity. However, the functional roles of pond metabolism finally determine the nature of pond soil to act as a sink or source of carbon. The net sinks for carbon in different wetlands have been estimated to be at rates up to 3 g C m-2 d-1 [8, 9, 10]. Sediments of aquaculture ponds are again very important in the carbon sequestration. Approx. 16.6 million tons of carbon is annually buried in aquaculture ponds of the world [11] and maximum sequestration occurs in Asia and particularly in China [12]. In other words, carbon sequestration by aquaculture ponds is about 0.21% of the annual global emissions. However, under unmanaged anaerobic conditions, wetlands can also emit greenhouse gases such as methane, nitrous oxide and hydrogen sulphide though this is limited in saline conditions or swampy areas. Further, the clearing or drainage of wetlands or aquaculture ponds can lead to large losses of stored organic carbon to atmospheric carbon dioxide. Notwithstanding the manifold positive impacts of wetlands overweighed towards beneficial environmental impacts and led to argue that wetland should not be destroyed or restored because of GHG emissions under limited conditions [13].
A major advantage of microalgae in aquatic environment is the 100% harvest of the production, and the carbon fixing rates of microalgae are much higher than in terrestrial plants. It is known that 1.6 and 2 grams of captured CO2 is used for every gram algal biomass [14]. As CO2 is present in atmosphere in concentration lower than nitrogen (N2) and oxygen (O2), this has created a thermodynamic barrier in CO2 capture. Nevertheless, high coefficient solubility of CO2 makes it far more soluble than atmospheric oxygen and nitrogen [15]. Carbon dioxide reacts with water; the carbon atom of CO2 is electron poor with an oxidation state of IV. The electron rich oxygen of water donates an electron pair to the carbon. After proton transfer from water to oxygen of the CO2 unit, carbonic acid is formed and then into bicarbonate and hydrogen, resulting in lowering of the pH - a favorable condition when CO2 gets dissolved in water.
One of the excellent abilities of aquatic cyanobacteria and eukaryotic algae is to use all forms of dissolved inorganic carbon (free CO2 - CO3- HCO3) at different pH conditions in aquatic environment. The CO2- CO3- HCO3 system acts as a buffer as well as a source of carbon for the photosynthetic microalgae at different pH conditions [16]. Among the three-chemical species of carbon in water, bicarbonate (HCO3) is most dominant (> 50%) at pH between 6.4 and 10.3, whereas carbonic acid (H2CO3) and carbonate (CO3) are dominant at pH < 6.4 and > 8.3, respectively [17, 18].
Carbon capture potentials of microalgae
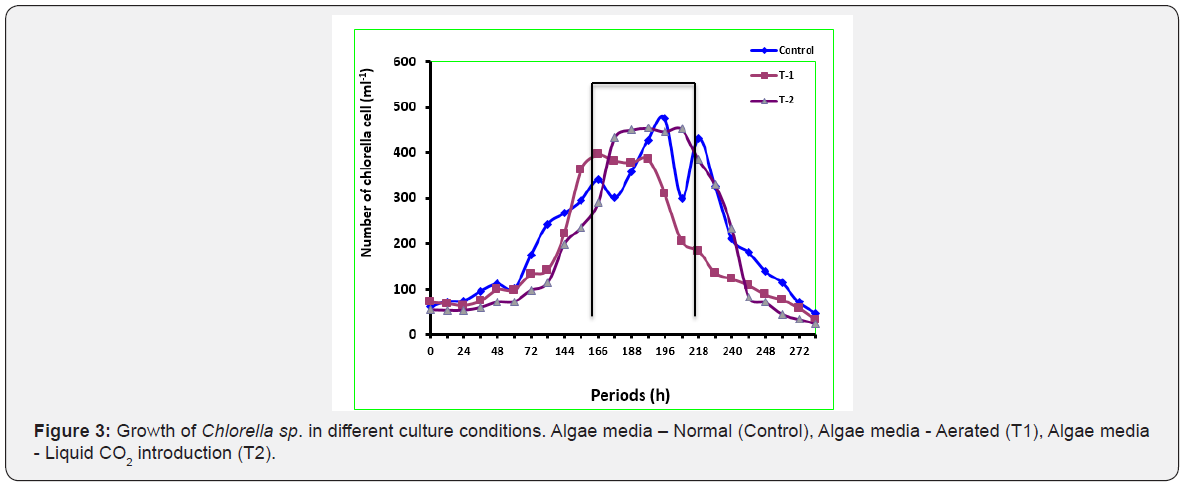
The microalgae are able to capture as high as 90% of carbon dioxide or bicarbonate in open ponds though the ability varies among species. Scenedesmus was better able to tolerate very high CO2 concentrations than Chlorella though both algae had about the same growth rate when the CO2 concentration remained in the range of 10 - 30%. Strains of microalgae that are able to tolerate about 20% CO2 (Scenedesmus, Chlorococum and Ankistrodesmus) formed several separate branches suggesting that certain groups or genotypes of algae tend to perform better under high CO2 level compared with other algal groups [19, 20]. demonstrated that Scenedesmus and Chlorella had much higher growth rates than the other three strains tested. The two algae again had very similar light response curves. Scenedesmus was completely inhibited by 100% CO2. A microalgal consortium consisting of Chlorella sp., Scenedesmus sp., Sphaerocystis sp. and Spirulina sp. procured from wastewater ponds revealed quite high CO2 sequestration (53- 100%; 150- 291 mg/g) [21]. While examining the growth of Chlorella under controlled laboratory culture medium (Hi Media) using aerated exogenously introduced compressed CO2 and control revealed no marked growth difference among the conditions, showing the peak between 166 - 218 hours of culture (Figure 3).
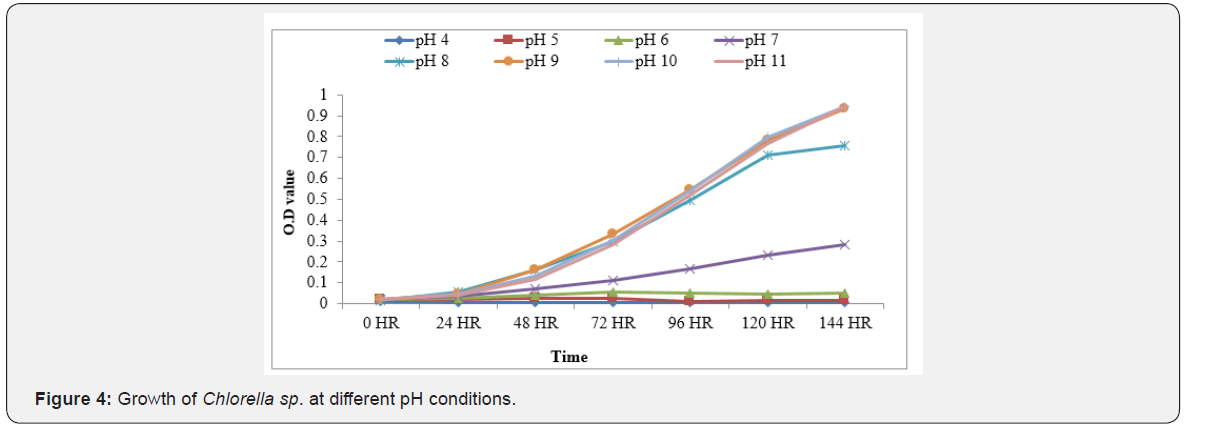
Comparing the growth responses of Chlorella sp. to different pH (4 - 11) of the algal culture medium revealed Chlorella growth higher between pH 9 and 11, whereas growth was retarded at pH 7 and hardly growth occurred at pH 4 - 6 (Figure 4).Though the growth of several algal species have been reported to be in the range of 7.5 - 8.5 [22, 23], Chlorella sp. in the present study and some other algae (Rhodobacter sphaeroides, Chlorella sorokiniana and Spirulinaplatensis) as reported by [24] were able to grow well at higher pH, where bicarbonate was the major source of carbon to microalgae (ICAR- NICRA Report, 2014). Among three culture media, growth was maximum in the aerated medium where the concentration of nitrate-N was highest and that of phosphate-P was the lowest or the nitrate- N was almost eight times higher than phosphate-P (Table 1).

The next ex situ experiment performed to examine the growth of microalgae at different N/P ratios (5, 20, 40, 50, 60, 70, 80, 90, 100) using urea and single super phosphate as sources of N and P revealed maximal growth at N/P ratio of 60 or 70 on day 7 of culture (NICRA - ICAR Report, 2014).This implied that this optimal nutrient condition can be maintained in open ponds by manipulating pond fertilization schedule for selection of right algae that would capture atmospheric carbon and would serve as natural food for herbivorous fishes regarded as low carbon foot print commercially important aquatic animal. It is also known that carbon-dioxide capture efficiencies as high as 80 to 99% are achievable under optimal conditions and with gas residence times as short as two seconds [25].
Biotechnological interventions
Biotechnology of algae has received much impetus since new species of microalgae that are being invented are capable of tolerate extremely high CO2 levels (40 - 100 vol%) and raised temperatures. Some of the mechanisms that regulate the cellular pH homeostasis of microalgae are: the photosynthetic apparatus state transitions that increase ATP generation, upregulation of H (+) -ATPases pumping protons out of the cell, rapid shutdown of CO2-concentrating mechanisms, and adjustment of membranes’ fatty acid composition [2].
The mechanism of carbon capture
There are different pathways of CO2 fixation in the photosynthesis of C3, C4 and CAM phototrophs. The first reaction is light dependent where protein rich chlorophyll pigments are held in chloroplast. The second reaction or the dark reaction or Blackman’s reaction is slower than light reaction and takes place in the stroma of chloroplast that embedded in the plasma membrane of photoautotrophs. The black reaction occurs also in the presence of light.
There are two cyclic reactions in dark reaction - Calvin cycle or C3 cycle and Hatch and Slack pathway or C4 cycle.
The Calvin cycle or C3 cycle, first observed by Calvin in unicellular green alga Chlorella, is a cyclic reaction occurring in the dark phase of photosynthesis when energy poor CO2 is converted into energy rich sugars during the process of carbon fixation or assimilation. The reactions of Calvin’s cycle occur in three phases: Carbonylative phase, Reductive phase and Regenerative phase.
The role of rubisco
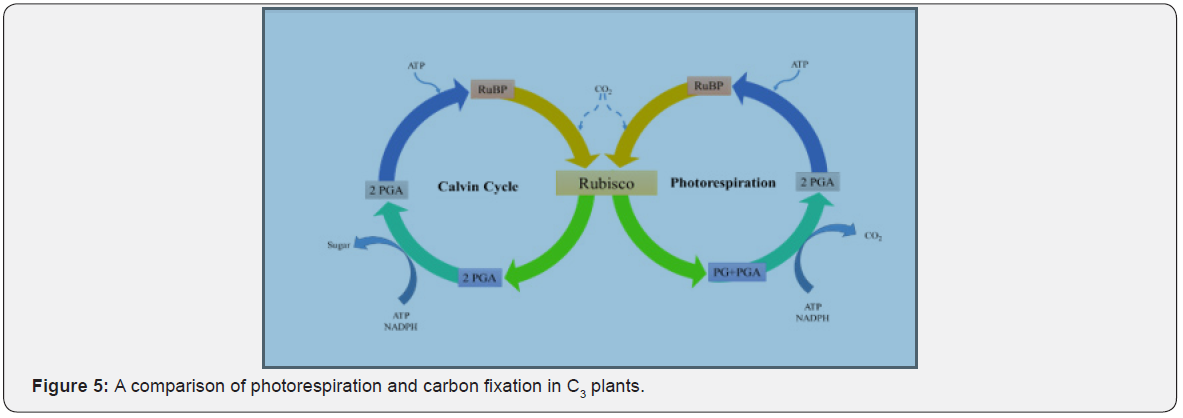
RuBisCO (Ribulose-1,5-bisphosphate carboxylase/oxygenase) is an enzyme and is involved in the first step of carbon fixation when carbon dioxide is converted to glucose. In this pathway, carboxylation of ribulose bis-phosphate (RuBP) occurs in presence of RuBisCO producing phosphoglycerate (PGA). The RuBisCO enzyme is capable of utilizing both CO2 and O2 leading to carboxylation in presence of CO2 and oxygenation of RuBP in presence of oxygen, respectively (Figure 5). In presence of high concentration of O2 is high and low level of CO2, RuBisCO acts as an oxygenase and further catalyzes the photorespiration reaction resulting in reduced carbon fixation due to production of 2-carbon molecule phosphogycolate [1].
In aquatic environment, algae capture carbon is pumped into the cell by bicarbonate transporters present in both the plasma membrane and in the chloroplast envelope of eukaryotic algae [26]. Inside the chloroplast, bicarbonate is concentrated, dehydrated spontaneously or by carbonic anhydrase through Calvin-cycle activity, finally yielding algal biomass. Pyrenoids of chloroplast which are analogous to cyanobacterial carboxysomes [27] played a great role the inhibition of photorespiration process thus aiding in increasing carboxylase activity of Rubisco helping in carbon fixation [1]. Although the structural configurations of pyrenoids may vary with species of algae, but in all cases, pyrenoids are present within the chloroplast embedded in plasma membrane of photoautotrophs, where RuBisCO molecules are found and help in concentrating CO2 around RuBisCO [1].
This loss of carbon reduces the Calvin cycle ability to regenerate the five-carbon sugar substrate ribulose bisphosphate which is essential for CO2 fixation by RuBisco. The process of photorespiration in the presence of light reduces photosynthetic carbon fixation efficiency by 20% to 30% [28]. For reducing the competitive inhibition of oxygen on carbon fixation by rubisco, microalgae actively pump sufficient bicarbonate from water phase into cells and to increase the internal CO2 concentrations to levels above those possible by equilibrium with air, and thus competitively inhibit photorespiration [29]. Other beneficial uses of algae are biochar and biofuel production.
Biochar
Other important strategies for carbon sink involve the formation of biochar which is geologically a stable form of carbon and can persist in soils for millions of years [30]. Biochar contains more than 90% carbon and is a by-product of pyrolysis at high temperatures in the presence of catalysts under anaerobic conditions [31].
Biofuel production
One of the important uses of microalgae is the production of fuel where high lipid content of microalgal biomass can be extracted and trasesertified into fuel. Many researchers worldwide are involved in this emerging field of innovative and cutting-edge research to explore the best strains of algae and developing the most efficient farming practices under different environmental conditions for the production of fuel [22,32].
Conclusion
Because of the excellent ability of microalgae to pump nongaseous bicarbonate into cell biomass in aquatic environment, microalgal culture is one of the lucrative tools for mitigation of global warming through trapping of atmospheric CO2 into the biomass of microalgae that served as natural food for zooplankton and herbivorous fishes. Selective microalgae can be grown by manipulation the pond fertilization schedule. This would help to grow selective microalgae which would capture atmospheric carbon and serve as food source for low carbon foot print fishes. Therefore, these optimum conditions can be applied for high growth of Chlorella. in open ponds.
Acknowledgement
I thank my student Mr. Debojyoti Kundu, Indian Institute of Technology, Kharagpur, West Bengal for his assistance during the preparation of MS.
References
- Peterhansel C, Horst I, Niessen M, Blume C, Kebeish R, et al. (2010) Photorespiration. The Arabidopsis Book 8: e0130.
- Solovchenko A, Khozin-Goldberg I (2013) High-CO2 tolerance in microalgae: possible mechanisms and implications for biotechnology and bioremediation. Bioethanol Lett 35(11): 1745-52.
- Nogia P, Kaur S, Mehrotra R, Mehrotra S (2016) Capturing atmospheric carbon: biological and nonbiological methods. International Journal of Low-Carbon Technologies11(2): 266-274.
- IPCC 2014 Climate change 2014: Impacts, Adaptations, and Vulnerability. Fifth Assessment Report of the Intergovernmental Panel on Climate Change.
- Jana BB, Lahiri S, Nandy S, Ghosh P, Bag SK, Biswas JK, Bhakta JN, Jana S (2015) Polyhouse driven raised temperature induces cichlid tilapia to breed during winter. Indian Journal of Animal Science 85(2):108-115.
- Jana BB, Nandy SK, Lahiri S, Bhakta JN, Biswas JK, et al. (2018) Heterogeneity of Water Quality Signature and Feedbacks to Carbon Sequestration in Wetlands Across Some Districts of West Bengal, India. J Water and Climate Change.
- Nandy S K, Jana BB, Lahiri S, Ghosh P, Bhakta, et al. (2018) CNP Ratio and Dose Regulated Production of Water Chestnut Trapa: Social and Environmental Implications. Russian Agricultural Sciences 44 (4): 318-325.
- Wetzel, R (2006) Transudation of elevated atmospheric CO2 and natural light trough altered carbon fluxes and higher trophic level production in aquatic ecosystems. Internationale Vereinigung für Theoretische und Angewandte Limnologie: Verhandlungen 29(3): 1112-1128.
- Bonneville MC, Strachan IB, Humphreys ER, Roulet NT (2008) Net ecosystem CO2 exchange in a temperate cattail marsh in relation to biophysical properties. Agricultural and Forest Meteorology 148(1): 69-81.
- Zhou L, Zhou G, Jia Q (2009) Annual cycle of CO2 exchange over a reed (Phragmites australis) wetland in Northeast China. Aquat Bot 91(2): 91- 98.
- Boyd CE, Wood C W (2010) Role of aquaculture pond sediments in sequestration of annual global carbon emissions. Environmental Pollution 158(8): 2537-2540.
- Verdegem MC, Bosma RH (2009) Water withdrawal for brackish and inland aquaculture, and options to produce more fish in ponds with present water use. Water Policy 111(S1): 52-68.
- Mitsch WJ, Bernal B, Nahlik AM, Mander U, Zhang L, Anderson CJ, et al. (2012) Wetlands, carbon, and climate. Landscape Ecology.
- Herzog H, Golomb D (2004) Carbon capture and storage from fossil fuel use. Encyclopedia of energy 1(6562): 277-287.
- Cole G A (1983) Text Book of Limnology Waveland Press, Inc, Illinois, USA 401 pp.
- Raven JA (2010) Inorganic carbon acquisition by eukaryotic algae: four current questions. Photosynth Res 106(1-2): 123-134.
- Jana BB (1998) The State-of-the-art of lakes in India: An overview. Archives fur Hydrobiology (Suppl) (Monograph Studies) 121: 1 - 89.
- Wetzel RG (2005) Limnology: Lake and river ecosystems. San Diego, CA: Academic Press.
- Zheng Liu, Fan Zhang, Feng Chen (2013) High throughput screening of CO2-tolerating microalgae using GasPak bags. Aquat Biosyst 9(1): 23.
- Hangata N, Takeuchi T, Fukuju Y (1992) Tolerance of microalgae to high CO2 and high temperature. Phytochemistry 31(10): 3345- 3348.
- Bhakta JN, Lahiri S, Pittman JK, Jana BB (2015) Carbon dioxide sequestration in wastewater by a consortium of elevated carbon dioxide tolerant microalgae. Journal of CO2 utilization 10: 105-112.
- Marcel J, Johannes T, Luuc RM, René HW (2003) Enclosed outdoor photobioreactors: light regime, photosynthetic efficiency, scale-up, and future prospects. Biotechnol Bioeng 81(2):193-210.
- Molina E, Fernández J, Acién FG, Chisti Y (2001) Tubular photobioreactor design for algal cultures. J Biotechnol 92(2):113-131.
- Ogbonna JC, Yoshizawa H, Tanaka H (2000) Treatment of high strength organic wastewater by a mixed culture of photosynthetic microorganisms. Journal of Applied Phycology 12(3-5): 277-284.
- Keffer JE, Kleinheinz GT (2002) Use of Chlorella vulgaris for CO2 mitigation in a photobioreactor. J Ind Microbiol Bioethanol 29(5): 275- 280.
- Spalding KL, Arner E, Westermark PO, Bernard S, Buchholz BA, et al. (2008) Dynamics of fat cell turnover in humans. Nature 453(7196): 783-787.
- Moroney JV, Ynalvez RA (2010) Proposed Carbon Dioxide Concentrating Mechanism in Chlamydomonas reinhardtii. Eukaryotic cell 6 (8): 1251- 1259.
- Zhu XG, Long SP, Ort DR (2008) What is the maximum efficiency with which photosynthesis can convert solar energy into biomass?. Curr opin biotechnol 19(2): 153-159.
- Badger MR, Price GD (1994) The role of carbonic anhydrase in photosynthesis. Annual review of plant biology 45(1): 369-92.
- Santin C, Doerr SH, Merino A, Bucheli TD, Bryant R, et al. (2017) Carbon sequestration potential and physicochemical properties differ between wildfire charcoals and slow-pyrolysis biochars. Scientific Reports. 7: 11233.
- Heilmann SM, Davis HT, Jader LR, Lefebvre PA, Sadowsky MJ, et al. (2010) Hydrothermal carbonization of microalgae. Biomass and Bioenergy 34(6): 875-882.
- Douskova I, Doucha J, Livansky K, Machat J, Novak P, et al. (2009) Simultaneous flue gas bioremediation and reduction of microalgal biomass production costs. Applied Microbiology and Biotechnology 82(1): 179-185.
- Mondal M, Goswami S, Ghosh A, Oinam G, Tiwari ON, et al. (2017) Production of biodiesel from microalgae through biological carbon capture: a review. Biotech 7(2): 99.






























