Advance towards Host Mediated RNA Interference Insect Pest Management
Jagdish Jaba* and Preeti Sharma
Research Program-Asia, International Crops Research Institute for the Semi-Arid Tropics (ICRISAT), India
Submission: July 28, 2017; Published: August 21, 2017
*Corresponding author: Jagdish Jaba, Research Program-Asia, International Crops Research Institute for the Semi-Arid Tropics (ICRISAT), Patancheru 502324, Telangana, India, Email: jaba.jagdish@gmail.com
How to cite this article: Jagdish J, Preeti S. Advance towards Host Mediated RNA Interference Insect Pest Management. Adv Biotech & Micro. 2017; 5(4): 555666. DOI: 10.19080/AIBM.2017.05.555666
Abstract
Gene suppression via RNA interference (RNAi) provides an alternative strategy for insect pest management. Insect pests cost billions of dollars in the form of crop losses and insecticides, and farmers face an ever-present threat of insecticide resistance, fueling a continual search for alternative pest-control strategies. Of late, novel insect pest management strategies like transgenic plants expressing insecticidal crystal protein genes from the soil bacterium, Bacillus thuringiensis (Bt) are effective in managing the insect pests that belong to Lepidoptera and Coleoptera. But success of the above technology is threatened by accelerated development of resistance, which is also true with chemical insecticides. Therefore, there is a need for identifying some additional effective pest management strategies, which could also augment integrated pest management (IPM). In this scenario, RNA interference (RNAi) offers a great deal of hope in successful mitigation of various insect pests. This review will feature late endeavors to comprehend the boundaries to RNA conveyance in headstrong insect species, depict late advances in the commercial development of insect -protected crops and biological insecticides using RNAi, and talk about this methodology with regards to an integrated pest management approach.
Keywords: RNAi; Pest control; IPM; Biotechnology
RNA Interference-Introduction and Mechanism
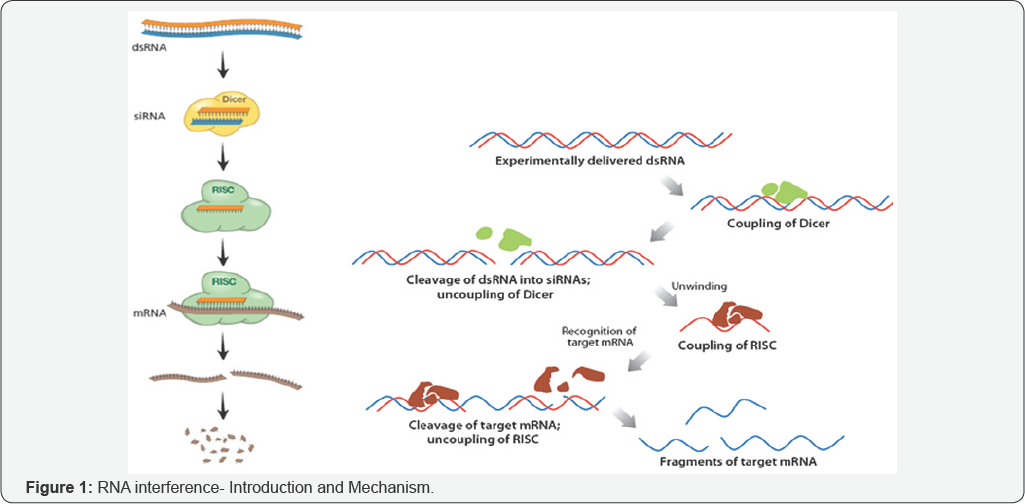
RNA interference (RNAi) is a potent method using only a few double stranded RNA (dsRNA) molecules per cell to silence the expression. RNA mediated silencing of specific gene(s) in pest insects through plant delivered RNA, offers the possibility to target genes necessary for their development, reproduction, or feeding success. Principally, this technology enables engineering of a new generation of pest-resistant GM crops. RNA interference (RNAi) is the specific downregulation of gene expression by double-stranded RNA (dsRNA) involving degradation of a target mRNA through the production of small interfering RNAs (siRNAs) from the dsRNA, which is cleaved by dsRNA-specific endonucleases referred to as dicers (Figure 1). One strand of the siRNA is assembled into an RNA-induced silencing complex (RISC) in conjunction with the Argonauts multi-domain protein, which contains an RNase H-like domain responsible for target degradation. The basis of this effect is thought to lie in the presence of an RNA-dependent RNA polymerase (RdRP) that is able to interact with the RISC complex and generate new dsRNA based on the partially degraded target template by using the hybridized siRNA strands as primers. The synthesized dsRNA is then acted on by the dicer enzymes to generate new siRNAs (secondary siRNAs), thus acting as an amplification step. In this way, once a dsRNA is introduced into a cell, its effect can persist over development; in addition, the ds RNAs can be exported to neighboring cells and thus spread the gene knockout effect through the organism [1].
RNAi and the genes that control in plants and animals
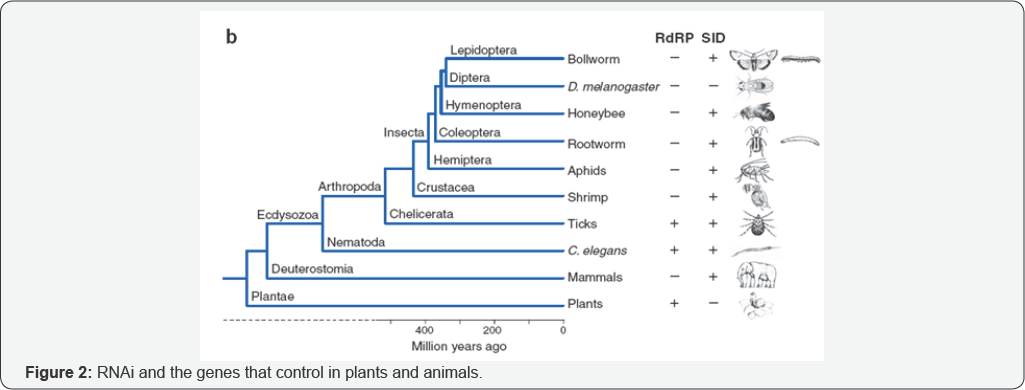
Evolutionary relationships of insects, higher animals and induces the endocytosis pathway of the dsRNA, in which case plants, indicating the presence or absence of genes encoding RdRP and the RNA channel transporter SID12. In arthropods,RdRP is restricted to the basal tick lineage, consistent with observations of systemic RNAi in these animals (as found in nematodes) [2]. The possibility of plant mediated RNAi to protect plants against insects has also been recognized for many years, but it was considered unfeasible initially, because in the complete genome sequence for the model insect Drosophila melanogaster [3]. In addition, the homologues of the C. elegans Systemic RNA Interference deficient mutant sid-1 gene have not been identified in the Drosophila genome [4]. Recent results have shown that many of these preconceptions were unduly pessimistic and that viable levels of insect resistance can be achieved by producing dsRNAs in plants (Figure 2).
Mechanism of dsRNA Uptake
Transmembrane channel-mediated uptake mechanism
The gene identified, systemic RNA interference deficient-1 (sid-1), is essential and sufficient to mediate systemic RNAi effect in C. elegans [5]. Two proteins involved. As (i) SID-2 modifies the SID- 1 molecule to activate the transport, or (ii) It binds the dsRNA from the environment and delivers it to SID-1, or (iii) It induces the endocytosis pathway of the dsRNA, in which case SID-1 delivers the dsRNA to the cytoplasm [6].
Endocytosis-mediated uptake mechanism
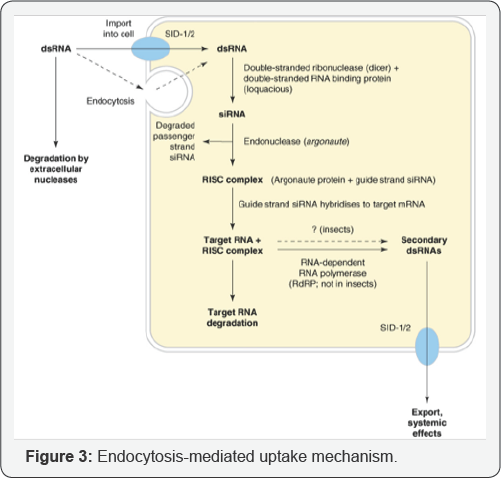
Apart from genes involved in cell-autonomous RNAi, a gene with a role upstream of the intracellular silencing events was identified: the clathrin heavy chain gene. It is known as a component of the endocytosis machinery. The role of vacuolar H+ ATPase was confirmed by Saleh et al. [6], and their pharmacological tests also showed that a combination of scavenger receptors participated in dsRNA uptake. An active control role for vacuolar H+ ATPase is proposed in breaking the normal endocytosis pathway to induce RNAi silencing in cells (Figure 3).
Link with immunity response
Link between antiviral immunity and RNAi was recently demonstrated in D. melanogaster. In this model infected insect cells release viral dsRNA which is taken up by uninfected cells through the dsRNA uptake pathway. Subsequently, an antiviral RNAi response, limiting virus replication, is elicited in those cells. Therefore it was concluded that immunity in multicellular organisms requires both cell-autonomous and systemic mechanisms to create pre-existing immunity to protect uninfected cells [7].
Factors influencing the silencing effect
- Concentration of dsRNA
- Nucleotide sequence
- Length of the dsRNA fragment
- Persistence of the silencing effect
- Life stage of the target organism
Production of dsRNA from an RNAi vectors
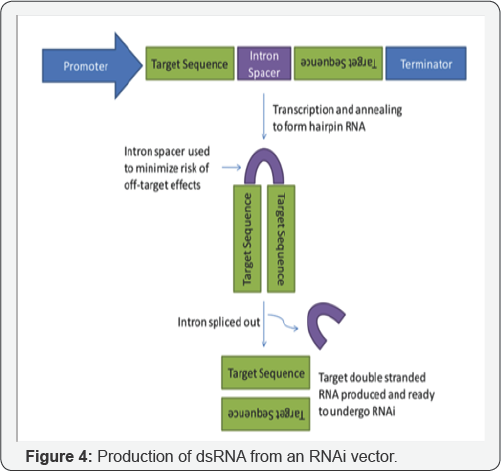
Vectors containing inverted repeats separated by an intron produce double-stranded RNA (dsRNA) or hairpin RNA (hpRNA) which effectively silences genes of interest. Producing dsRNA in RNase III deficient E. coli strains and extracting that product to treat plants as an alternative to RNAi transgenic plants has recently been accomplished. The key to an RNAi commercial product will be targeting genes that potently kill pests or inhibit toxin resistance [8]. Specific promoter, inverted repeats, and an intron spacer are used to produce the best RNAi effects. Insect systemic RNAi was first documented in another model insect Tribolium castaneum (flour beetle) and multiple genes such as Tc- ASH, Distalless, maxillopedia and proboscipedia were targeted by injection of specific dsRNA [9]. Turner offered experimental validation of this strategy on the larval stage of the light brown apple moth (Epiphyas postvittana). Transcript level of a larval gut-expressed gene (EposCXE1) and adult antennae expressed gene (EposPBP1) were reduced by feeding specific dsRNA [10]. Microinjection of dsRNA into adults and larvae has been used to silence genes in Tribolium castaneum. Similarly, dsRNA induced an RNAi response in Lepidopteran, such as Spodoptera litura (Fabri cius) (Lepidoptera: Noctuidae) and Epiphyas postvittana.No effective Bt toxins are known against sap-sucking homopteran pests such as aphids, leafhoppers, etc (Figure 4).
Delivery of ds RNA in plants
Biolistic and agroinfection delivery: When plant tissues are bombarded with gold or tungsten particles that have been coated with DNA or RNA, the nucleic acid is released and expressed in the cells where the particles come to rest. Bombarding cells with double-stranded (ds) RNA, siRNA, or DNA constructs that encode hairpin RNA can produce transient silencing of target reporter genes.
- Virus induced delivery
- Stable transformation with transgenes
Application of RNAi in Pest Control
A serious problem for insecticides is that they can kill nontargeted animals. To address this issue, the possibility of using RNAi to kill only the target animals by down-regulating essential gene functions in insects has been recognized for many years [1].
Cell line experiments
Cell line experiments can give additional information specific for the target gene in a simplified model. They can also inform on the possibility and effect of environmental RNAi when the dsRNA is applied through soaking of the cells in a dsRNA- enriched medium. This way the link between Cry1Ac insecticidal protein and an aminopeptidase N in the gut of larvae of the cotton bollworm (Helicoverpa armigera) (HaAPN1) was shown with RNAi cell line experiments. Sf21 cells were modified to express HaAPN1 and 48h after Haapn1 dsRNA treatment the expression of Haapn1 mRNA and its protein were reduced 70% compared to the controls, resulting in decreased sensitivity of the HaAPN1-expressing cells to Cry1Ac protein [11].
ds RNA feeding and in planta experiments
Silencing a cotton bollworm, P450 monooxygenase gene: Mao et al. [12] identified a P450 monooxygenase from the cotton bollworm (Helicoverpa armigera), it was named CYPAE14 and was involved in detoxification of the otherwise toxic allelochemical an indiscriminately toxic compound called gossypol-produced by cotton plant. This gene is induced by gossypol and its suppression reduced the larval tolerance to gossypol. The researchers transferred hairpin RNA constructs directed against CYP6AE14 into plant and fed cotton bollworm with the plant material. The model plants Nicotiana tabacum and Arabidopsis thaliana were modified with the cytochrome P450 gene of H. armigera. When the cotton bollworm larvae were fed transgenic leaves, levels of cytochrome P450 mRNA were reduced and larval growth retarded. As a glutathione-S- transferase gene (GST1) is silenced in GST1 dsRNA-expressing plants, feeding insects plant material expressing dsRNA may be a general strategy to trigger RNA interference and could find applications in entomological research and field control of insect pests.
Control of coleopteran pests through RNAi
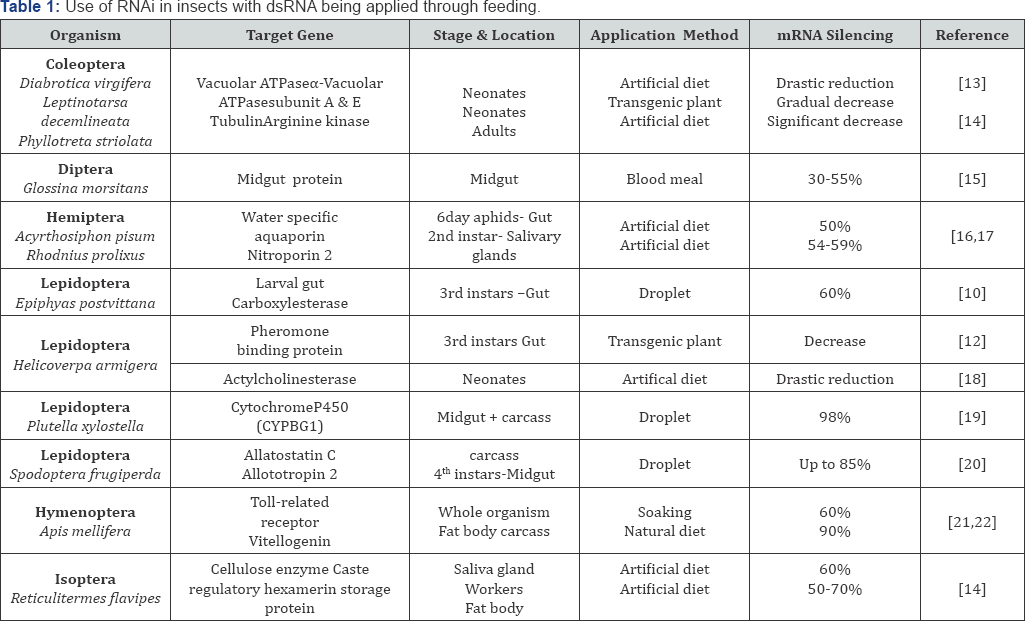
Baum et al. [13] utilized a screening approach where genes from Western corn root worm were identified in cDNA libraries, and genes encoding polypeptides predicted to provide an essential biological function were classified as 'targets'. dsRNAs directed against three target genes (b-tubulin, V-ATPase A and V-ATPase E) demonstrated an effective RNAi response in WCR that resulted in high larval mortality. Transgenic corn expressing one of these dsRNAs, targeted to a subunit of the midgut enzyme vacuolar ATPase (V-ATPase), showed protection against WCR infestation comparable to that provided by a Bt transgene. When dsRNAs designed to target WCR genes were tested in larvae from two other insect pests, mortality declined with decreasing sequence identity between the WCR genes and their orthologs in the other species, indicating that gene silencing was potentially very selective (Table 1) [13-22].
RNA i for the control of whiteflies by oral route
RNA interference mediated gene silencing was explored for the control of sap-sucking pest Bemisia tabaci. dsRNAs and siRNAs were synthesized from five different genes, actin ortholog, ADP/ ATP translocase, a-tubulin, ribosomal protein L9 (RPL9) and V-ATPase A subunit. A simplified insect bioassay method was developed for the delivery of ds/siRNA through the oral route, and efficacy was evaluated. ds/siRNA caused 29-97% mortality after 6 days of feeding. Knocking down the expression of RPL9 and V-ATPase A caused higher mortality with LC50 11.21 and 3.08|ig/ml, respectively, as compared to other genes [23].
Other applications
RNAi-based strategy being explored to fight IAPV infection is to feed bees siRNAs targeting specific IAPV sequences such that, following viral entry into bee cells, translation of viral proteins is blocked [24]. Kumar et al. [18] studied the effect of siRNA on larval development by selective targeting of the acetylcholinesterase (AChE) gene of Helicoverpa armigera. Chemically synthesized siRNA molecules were directly fed to H. armigera larvae along with the artificial diet. The siRNA treatment resulted in specific gene silencing of AChE and consequently brought about mortality, growth inhibition of larvae, reduction in the pupal weight, malformation and drastically reduced fecundity as compared to control larvae. Using chitin synthase gene A (SeCHSA) as the target gene, which is expressed in the cuticle and tracheae of the lepidopteran pest Spodoptera exigua, showed that the growth and development of S. exigua larvae fed Escherichia coli expressing dsRNA of SeCHSA was disturbed, resulting in lethality [25]. Whyard et al. [26] harnessed the sequence specificity of RNAi to design orally-delivered dsRNAs that selectively killed target insects. They found that D. melanogaster, T castaneum, pea aphids (Acyrthosiphon pisum), and tobacco hornworms (Manduca sexta) were selectively killed when fed species-specific dsRNA targeting vacuolar-type ATPase transcripts. For the aphid nymphs and beetle and moth larvae, dsRNA could simply be dissolved into their diets. However, to induce RNAi in the drosophilid species, the dsRNAs needed to be encapsulated in liposomes to help facilitate the uptake of the dsRNA [27]. Overcoming the specific delivery of dsRNA or siRNA into the cytoplasm of target cells is still an important issue for the use of RNAi for insecticides.
Bio-safety
A key advantage of RNAi-mediated resistance is that dsRNA has no inherent translational ability to produce a functional protein. This means that non-target effects should be minimal and lower than those of even highly specific transgenic proteins. Protection of non-target species that also feed on the plant can be provided by using dsRNA that has no effect when ingested by other invertebrates such as insects. One of the key advantages of this technology could be its high species specificity for the target pests. If the target sequence is carefully chosen, a specific gene or genes can be silenced. RNAi can also be used to achieve varying levels of gene silencing, using the same ihpRNA construct in different lines. This allows for selection of lines with varying degrees of gene silencing. In addition to this, the timing and extent of the gene silencing can be controlled, so that genes that are essential will only be silenced at chosen stages of growth or in chosen plant tissues. So, RNAi provides us with a great degree of flexibility in the field of functional genomics.
Limitations
There are also limitations however to RNAi. Unlike in insertional mutagenesis, for the use of RNAi the exact sequence of the target gene is required. Once this sequence information is available, the rest of the process is however relatively fast. Secondly, delivery methods for the dsRNA is a limiting step for the number of species which RNAi based approaches can be used easily. Due to this, improvement and further research into the kinds of vectors that can be used safely and reliably is needed.There have also been some reports that it has been difficult to detect mutants in which there has been subtle changes in gene expression. In plants, marker genes are being developed that will indicate if there has been a change in gene expression.
Conclusion and Future Perspectives
The RNA silencing pathways seem to have diversified during the evolution of eukaryotes, but the phenomenon of RNAi that regulates gene expression appears to be common among eukaryotic organisms. Therefore, once exogenous elicitor (dsRNA or siRNA) production from transgenic plants is delivered into pest cells, the endogenous cognate transcripts would be degraded. RNAi mediated plants resistance offer several advantages over conventional bio-engineering crops resistance. Firstly, in engineering broad-spectrum plant resistance even distinct lineages of plants pathogens have undergone convergent evolution and sharing homologues. Therefore multiple pathogens resistance can be achieved by silencing conserved and essential genes. Secondly, the resistance has the potential to be more durable. The RNAi-mediated resistance is based on RNA hybridization rather than protein-protein interaction; the molecular hybridization cannot be inhibited by minority nucleic acid mutation. Therefore, the possibility of the pests overcoming the resistance are likely difficult. In addition, this biotechnology represents a flexible means of developing pest resistant crops. In theory, all the pests' genes showing detrimental knockdown phenotype can be considered as potential targets. So this strategy would not be limited by the scarcity of resistance genes. Nevertheless, RNAi has opened up a new line of thinking in designing a futuristic approach which could result in paradigm shift in insect pest management strategies.
References
- Da Silva MR, Moraes ÉA, Lenquiste SA, Godoy AT, Eberlin MN, et al. (2014) Chemical characterization and antioxidant potential of Chilean chia seeds and oil (Salvia hispanica L.). LWT-Food Science and Technology 59(2): 1304-1310.
- Muñoz LA, Cobos A, Diaz O, Aguilera JM (2013) Chia seed (Salvia hispanica): an ancient grain and a new functional food. Food Reviews International 29(4): 394-408.
- Ullah R, Nadeem M, Khalique A, Imran M, Mehmood S, et al. (2016) Nutritional and therapeutic perspectives of Chia (Salvia hispanica L.): a review. J Food Sci Technol 53(4): 1750-1758.
- Coorey R, Tjoe A, Jayasena V (2014) Gelling Properties of Chia Seed and Flour. J Food Sci 79(5): E859-E866.
- Ali NM, Yeap SK, Ho WY, Beh BK, Tan SW, et al. (2012) The promising future of Chia, Salvia hispanica L. J Biomed Biotechnol 2012: 171956.
- Segura-Campos MR, Ciau-Solis N, Rosado-Rubio G, Chel-Guerrero L, Betancur-Ancona D (2014) Chemical and functional properties of chia seed (Salvia hispánica L.) gum. Int J Food Sci Nut 2014(2014): 1-5.
- Sargi SC, Silva BC, Santos HMC, Montanher PF, Boeing JS, et al. (2013) Antioxidant capacity and chemical composition in seeds rich in omega-3: chia, flax, and perilla. Food Sci Technol (Campinas.) 33(3): 541-548.
- https://ndb.nal.usda.gov/ndb/
- Reyes-Caudillo E, Tecante A, Valdivia-Lopez MA (2008) Dietary fibre content and antioxidant activity of phenolic compounds present in Mexican chia (Salvia hispanica L.) seeds. Food Chem 107(2): 656- 663.
- Rahman J, Camargo de AC, Shahidi F (2017) Phenolic and polyphenolic profiles of chia seeds and their in vitro biological activities. J Funct Foods 35: 622-634.
- Nadeem M, Abdullah M, Mahumd A, Hussain I, Inayat S (2013) Stabilization of butter oil with modified fatty acid profile by using Moringa oleifera extract as antioxidant. J Agric Sci Technol 15: 919–928.
- Nadeem M, Situ C, Mahmud A, Khalique A, Imran M, et al. (2014) Antioxidant activity of sesame (Sesamum indicum) cake extract for the stabilization of olein based butter. J Am Oil Chem Soc 91(6): 967–977.
- Haque MS, Ghoshal KK (1981) Floral biology and breeding system in the genus Salvia L. Proc Indian Natl Sci Acad 47(5): 716-724.
- Sreedhar RV, Kumari P, Rupwate SD, Rajasekharan R, Srinivasan M (2015) Exploring triacylglycerol biosynthetic pathway in developing seeds of Chia (Salvia hispanica L.): a transcriptomic approach. PLoS One 10 (4): 123580.
- Xue Y, Yin N, Chen B, Liao F, Win AN, et al. (2017) Molecular cloning and expression analysis of two FAD2 genes from chia (Salvia hispanica). Acta Physiologiae Plantarum 39(4): 95.






























