Structural Characterization and Bioactivity of Exopolysaccharide Synthesized by Geobacillus sp. TS3-9 Isolated from Radioactive Radon Hot Spring
Lulu Wang1, Hong Zhang1, Long Yang1, Xinle Liang1*, Fuming Zhang2* and Robert J Linhardt2
1Department of Biochemical Engineering, Zhejiang Gongshang University, China
2Department of Chemical and Biological Engineering, Chemistry and Chemical Biology, Biology and Biomedical Engineering, Rensselaer Polytechnic Institute, USA
Submission: June 08, 2017; Published: July 21, 2017
*Corresponding author: Xinle Liang, Department of Biochemical Engineering, Zhejiang Gongshang University, Hangzhou 310025, China, Tel: 86571-8820-8582; Email: dbiot@mail.zjgsu.edu.cn
Fuming Zhang, Departments of Chemical and Biological Engineering, Rensselaer Polytechnic Institute, Troy, NY 12180, USA, Tel: 518-276-6839; Fax: 518-276-3405; Email: zhangf2@rpi.edu
How to cite this article: Lulu W, Hong Z, Long Y, Xinle L, Fuming Z, Robert JL. Structural Characterization and Bioactivity of Exopolysaccharide Synthesized by Geobacillus sp. TS3-9 Isolated from Radioactive Radon Hot Spring. Adv Biotech & Micro. 2017; 4(2): 555634. DOI: 10.19080/AIBM.2017.04.555635
Abstract
A thermophilic, endospore-forming, and radiation resistant bacterium, strain TS3-9, was isolated from the radon hot spring. Based on phenotype, fatty acid, G + C mol %, and phylogenetic analysis, TS3-9 was assigned to the genus GeobaciHus. In addition to surviving exposure to gamol/La irradiation (D10 = 10 kGy), TS3-9 produced an exopolysaccharide (EPS), the average molecular weight was approximately 3.2 x 106Da. The results of partial acid hydrolysis, GC-MS, FT-IR, and NMR analysis showed EPS was a heteropolysaccharide, composed of D-mannose, D-glucose and rhamnose as its principal monosaccharide composition in the relative proportions, 1:0.14:0.06. The uronic acids and acetyl contents (wt. %) were at 18.6 % and 6.07 %, respectively. Evaluation of antioxidant properties indicated that the EPS had good activities in scavenging hydroxyl radical, superoxide radical, and DPPH. Preliminary in vitro tests revealed that the EPS could significantly restrain the proliferation of hepatoma carcinoma cell.
Keywords: Polysaccharide; Geobacillus; Extremophile; Antioxidant activity
Introduction
Microbial exopolysaccharide (EPS) has a wide range of indus-trial (such as textiles, oil recovery, food additives, cosmetic and pharmaceutical) and medical applications. The collection of di-verse products from different microorganisms include: xanthan from Xanthomonas [1], gellan from Sphingomonas [2], heparason from E. coli [3], glucans from Leuconostoc, Streptococcus and Lactobacillus [4], cellulose from Acetobacter [5], hyaluronan from Pseudomonas and Streptococci group A and C. In recent years there has been an increased interest in EPS for pharmaceutical applications such as antioxidants, antitumor agents and im- munostimulants [6].
EPS producing extremophiles have been isolated from a va-riety of extreme environmental habitats, and include halophilic, psychropilic, acidophilic, alkaliphilic, thermophilic bacteria, and archaea. In contrast to the frequently pathogenic, mesophilic mi-crobial producers of EPS, extrermophiles are usually non-patho genic and offer biotechnological advantages, such as short fer-mentation times.
Application of biologically active polysaccharides, synthesized by extremophiles, offers promising opportunities in food, medicine, cosmetology, and in the environmental industry. The most inter-esting halophilic EPS is mauran, produced by Halomonas maura, which has a viscosifying activity similar to that of xanthan, and polymers from Salipiger mucosus, which is characterized by the presence of sulfate, phosphate and fucose and promises potential applications in medicine and as a supply of fucose [7]. Other halo- pilic EPS producers include Idiomarina, Alteromonas, and Palle- ronia [7].
Two psychropilic bacteria [8], Colwellia psychrerythraea and lleya marilimosa [9], reportedly produce EPS at temperature from - 8 to -14 °C. Acidophilic and alkaliphilic bacteria such as Acidithiobacillus and Leptospirillum produce EPS with novel chemical compositions and properties [10]. In the various ther- mophiles, such as Hyperthermophilic archaeon, Thermococcus litoralis, Thermotoga maritima and Geobacillus [11], the ability of EPS production is established in situ as a strategy for growth and survival in adverse conditions. A microbial glucan, sythesized by G. tepidanmans V264 was characterized [12] with a 111.4mg L-1 production and good stability at temperatures below 280 °C. This EPS is neutral towards normal metabolism of living organisms, suggesting its pharmaceutical utility in the development of novel class of anti-cytotoxic drugs.
Geobacillus sp. 4004 isolated from a marine hot spring can produce three fractions of EPS with a variety of compositions by mannose, glucose, galactose and arabinose [13]. Immunomodulatory and antiviral tests show that EPS from G. thermo denitrificans B3-72 can hinder HSV-2 replication in human peripheral blood mononuclear cells and partially restore the immunological disorders [1].
In the current study, we isolated a thermophilic and radiation resistant bacterium strain Geobacillus sp. TS3-9 from a radon hot spring. Radon hot springs offer extremely hostile environments for microorganisms. Only a few microorganisms, which have adapted to these special conditions, can be survive. This paper describes the classification of the isolated TS3-9, and the production, structural characteristics and biological activities of its EPS.
Methods
Isolation, identification and culture conditions of microorganism
The bacterial strains were isolated from Taishun radon hot spring (pH = 8.0, 55 °C, 55.5 Bq L-1 radon, and 20mg L-1 fluorine), located in Wuyan ling National Natural Preservation Zone in Tai- shun County, Zhejiang Province, China. Water samples were collected and inoculated in the medium TY containing peptone 1g L-1, yeast extract 0.5g L-1, and NaCl 0.2g L-1, pH 8.0-8.5, and cultivated overnight at 55 °C. After subsequent dilution in medium TY, 100μL of the culture was spread onto medium TY agar plates and incubated at 55 °C to obtain a pure colony. The purity of the isolates was confirmed microscopically and by the uniformity of colonies formed on solid medium TY plus 2% agar. The isolated pure cultures were stored in 30% glycerol at -70 oC.
The isolates were investigated for EPS production in synthetic medium (SMG), containing glucose 20g L-1, (NH4)2SO4 0.1g L-1, FeSO4.7H2O 0.03g L-1, Na2B407.10H20 0.04g L-1, yeast extract 0.1g L-1 [13]. Different sugars (lactose, galactose, xylose, maltose, fructose, mannose, sucrose, and sorbose, 20g L-1) were tested as carbon source by replacement of glucose. The effect of pH on the growth of the selected strains was determined in the range of 7.0-9.0. The optimum temperature for growth was determined by measuring the growth in the range of 45-70 °C.
Morphological, physiological and genetic tests
Colony morphology of the isolate TS3-9 was determined by Olympus DP51 camera. Cellular morphology was determined by scanning electron microscope (SEM) and Transmission Electron Microscope (TEM). Gram staining was performed according to Wensinck [14]. Growth was considered positive if the OD600 was greater than 0.1 after cultivating 24h. Gamol/La radiation-resis- tance of the bacterium was tested following protocols described by Rainey et al. [15] with minor modification. Deinococcus radi- odurans R1 and E. coli DH5α were used as positive and negative controls. Fatty acid analyses were performed as reported by Clayton & Claudio [16].
FAMEs were identified by comparison of sample FAME peak relative retention times with those of standards from Sigma (USA). Total genomic DNA of strain TS3-9 was extracted by using an EZ Spin Column Bacterial Genomic DNA Isolation Kit (Sango, China). The 16S rRNA gene of the isolate was amplified by polymerase chain reaction (PCR). Sequence analysis was performed using the BLAST program. MEGA, version 4.0 was used for phylogenetic analysis, distances were obtained using options according to the Kimura two parameter model [17] and clustering was performed by using the neighbour-joining method. Bootstrap values from 1000 replications were used to determine the confidence level of the branches. The G + C mol % content evaluated by modifying the procedure originally described by Dehainault [18].
Exopolysaccharides production
The EPS of TS3-9 was produced in synthetic medium by batch fermentation in shake flasks (150 rpm) at 60 °C for 48h. The fermented broth was collected and centrifuged at 4 °C, 11,000 x g for 20min and filtered through a 0.22μm membrane filter (Millipore, USA). Protein was precipitated by adding two volumes of 10 % trichloroacetic acid (TCA) at 4 °C overnight. The mixture was subjected to centrifugation (11,000 x g) at 4 °C for 30min. The super-natant was treated with three-volumes of cold absolute ethanol to precipitate EPS at -20 °C for 24h. The precipitated EPS was centrifuged (11,000 x g) at 4 °C for 30 min and the pellets were washed twice with ethanol and dissolved in distilled water, dialyzed using 5kD cut-off dialysis membrane and lyophilized. The samples were tested for carbohydrate, protein, and nucleic acid contents [19]. Triplicate cultures were grown and the experiments were repeated twice.
Purification of EPS
The crude lyophilized powders were dissolved in distilled water and applied to a DEAE Sepharose Fast Flow column (1.6 x 50cm, Amersham, USA), eluted firstly with distilled water and then a gradient of NaCl from 0 to 1M at a flow rate of 0.5mL min-1. Total carbohydrate content of the fractions (5mL/tube) was col-lected and determined by the phenol-sulfuric acid method [20]. The fractions with the highest carbohydrate content were combined, dialyzed, concentrated and applied to a Sephadex G-200 column (1.6 x 5cm, Amersham, US), eluted with 0.2M NaCl at a flow rate of 0.5mL min-1. The resulting fractions were combined according to the carbohydrate content, dialyzed and lyophilized to obtain the purified EPS.
Molecular weight determination of EPS
The molecular weight (Mw) of the sample was estimated on a High-Performance Size-Exclusion Chromatography (HPSEC) apparatus equipped with Ultrahydrogel linear (7.8x300mm) col-umn, coupled to a Waters 2424 ELS Detector (Waters Co., USA). The sample was dissolved at a concentration of 1mg mL-1 in distilled water. The carrier (dH2O) and sample solutions were rendered dust-free by passing through a 0.45μm Millipore filter, and degassed before use. The injection volume was 60μl and the flow rate was 0.5mL min-1. A mixture of dextrans (Mw: 5.6 x 103, 4 x 104, 3.6 x 105, 2.5 x 106 Da) was used for calibration curves.
Determination of monosaccharide compositions
Monosaccharide compositional analysis was performed by hydrolysis of EPS with 2M trifluoroacetic acid (TFA) at 120 °C for 6h and then trimethylsilylated. Unambiguous identification of sugar components was obtained by gas chromatography-mass spectrometry (GC-MS), using as standards: Glucose, galactose, fucose, mannose, xylose, arabinose, rhamnose (Sigma Aldrich, USA). GC-MS was performed on an Agilent 7890A-5975C instrument, equipped with a HP-5-MS column (60m x 0.25mm x 0.25m) and with a N2 flux of 50mL min-1; 126 °C 1min, from 126 to 250 °C at 3 °C min-1 was used as temperature program. The uronic acids and acetyl contents were determined according to a colorimetric method.
Nuclear magnetic resonance (NMR) spectroscopy
The lyophilized polysaccharide was kept over P2O5 in vacuum for several days and exchanged twice in D2O with intermediate lyophilization and then dissolved in 500μL of D2O to a final concentration of 40 mg mL-1. 1H and 13C NMR spectra were recorded with a Bruker Avance 500MHz spectrometer, operating frequencies 500.13MHz for 1H NMR and 125.75MHz for 13C NMR. Chemical shifts were reported relative to internal label tetramethylsi- lane (TMS) at 5 0.00ppm for 1H and 13C spectrum.
Fourier transform infrared (FT-IR) analysis
The infrared spectra of polysaccharides were recorded at room temperature with a Fourier transform infrared Bio-Rad spectrometer (NICOLET 380, Thermo) in the absorbance mode. Compressed pellets were prepared by mixing 2mg of EPS with 100mg KBr. Spectra were corrected for KBr background and re-corded in the range 4000-400 cm-1 using 32 scans [21].
Antioxidative activity in vitro
Hydroxyl radical scavenging assay: Reaction mixtures in a final volume of 4mL contained: 1mL H2O2 (8.8 mM L-1), 1mL FeSO4 (9 mM L-1), 1mL salicylic acid (9mM L-1), and 1mL EPS solution at concentrations of 0, 2.0, 4.0, 6.0, 8.0, and 10.0mg mL-1. The H2O2 was added to the mixture to initiate the reaction. The reaction mixture was incubated at 37 °C for 60 min and then centrifuged at 9,000 x g for 8min [16]. The absorbance of the reaction solutions at 510nm was measured and Vitamin C (Vc) was used as positive control. Scavenging of hydroxyl radicals was calculated according to: Scavenging ability (%) = (1-A/A0) x100, where A was absorbance of EPS and A0 is the absorbance of the blank.
Superoxide radical (O2-) scavenging assay: Superoxide anion radical scavenging activity was determined according to the method of Xiang [22]. Reaction mixtures containing 3ml of 0.05mol L-1 Tris-HCl buffers (pH 8.2) and 1mL of EPS solution at concentrations of 0, 2.0, 4.0, 6.0, 8.0, and 10.0mg mL-1 were incubated at 25 °C for 10min. Pyrogallol (200μL) was added to the mixture, and the reaction was kept at 25 °C for 4min. The reaction was terminated by addition of 0.5ml of HCl. The absorbance of the mixture was measured at 320nm against the blank. Vc was used as positive control. Scavenging of superoxide radicals was calculated according to
Scavenging ability (%) = (1-A/A0) x 100, where A was the absorbance of EPS and A0 was absorbance of the blank.
DPPH scavenging assay: Scavenging of DPPH radicals was measured according to the method of Dubuisson & Wergifosse [23]. The reaction mixture contained 3mL of a 0.004% ethanol solution of DPPH and 1mL of the EPS at concentrations of 0, 2.0, 4.0, 6.0, 8.0, and 10.0 mg mL-1, Absorbance at 517 nm was measured after 30min. Vc was used as positive control. Scavenging of DPPH radicals was calculated according to:
Scavenging ability (%) = (1-A/A0) x 100, where A was absorbance of EPS and A0 was the absorbance of DPPH solution.
In vitro anti-tumor activity against hepatoma carcinoma cell
The anti tumor activity assays of hepatoma carcinoma cell (HepG2) was evaluated in vitro using MTT assay [24]. Briefly, HepG2 in RPMI-1640 medium supplemented with 10% fetal calf serum were seeded in a 96-well flat-bottomed microplate with 1x105cells/ml. They were then stimulated by EPS at final concentrations of 4, 20, 100μg/mL. The cells were cultured for 44h at 37 °C under a humidified 5% CO2 atmosphere, each well was added 20μL of 5mg/mL of MTT and incubated for another 4h. Then the culture media were removed, 150μL of DMSO was added to each well. Absorbance at 490nm was detected by micro plate ELISA reader. The inhibition ratio of HepG2 proliferation was calculated as follows:
Where A is the absorbance value of negative control group, and A0 is that of sample group.
Statistical analysis
Data in all the bioassays were statistically evaluated by analy-sis of variance and P<0.05 was considered significant.
Results and Discussion
Isolation and identification of the thermophilic strain TS3-9
Strain TS3-9 was one of the isolates from the hot spring (pH = 8.0, 55 °C temperature, and 55.5Bq L-1 radon radiation). Its cells were Gram-negative, sporulating rods (5.5-6.0μm long and 0.3- 0.5μm wide) with terminal, ellipsoidal to cylindrical endospores (Figure 1a). Colonies are milky white, smooth and circular. TS3- 9 is thermophilic and facultative aerobe, exhibiting optimum growth temperature of 60 °C at pH 8.0. It can utilize glucose, lactose, galactose, xylose, maltose, fructose, mannose, sucrose, and sorbose when the medium was supplemented with yeast extract. It is positive for catalase, starch hydrolysis, sensitive to lysozyme, nitrate reduction. TS3-9 produces EPS in sugar media.
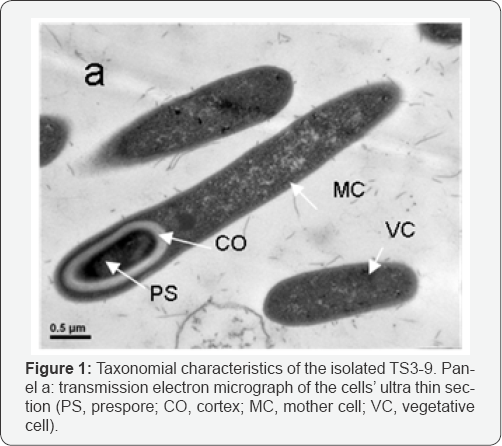
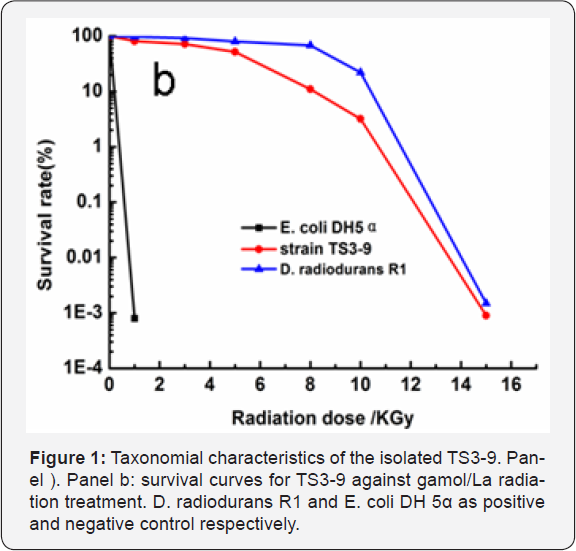
TS3-9 has a special biological characteristic in environmental resistance, which makes it to survive in the extremely hostile en-vironment. Its gamol/La radiation-resistance was compared with that of Deinococcus. Radiodurans R1 (as positive control) and E. coli DH 5α (as negative control) (Figure 1b). The lethal dose of ga- mol/La radiation for E. coli DH 5α was 2kGy, whereas strain TS3- 9 and Deinococcus radiodurans R1 could grow weakly under the radiation dose as high as 14kGy. These data suggest that TS3-9 is highly radiation-resistant. The fatty acid compositions were characterized by the abundance of branched acryl chains. The most abundant was iC16:0 (46.6%) and iC17:0 (37.2%), other components were nC15:0 (5.7%), nC17:0 (3.8%), nC18:0 (6.6%), which was different from those of Geobacilluse sp.
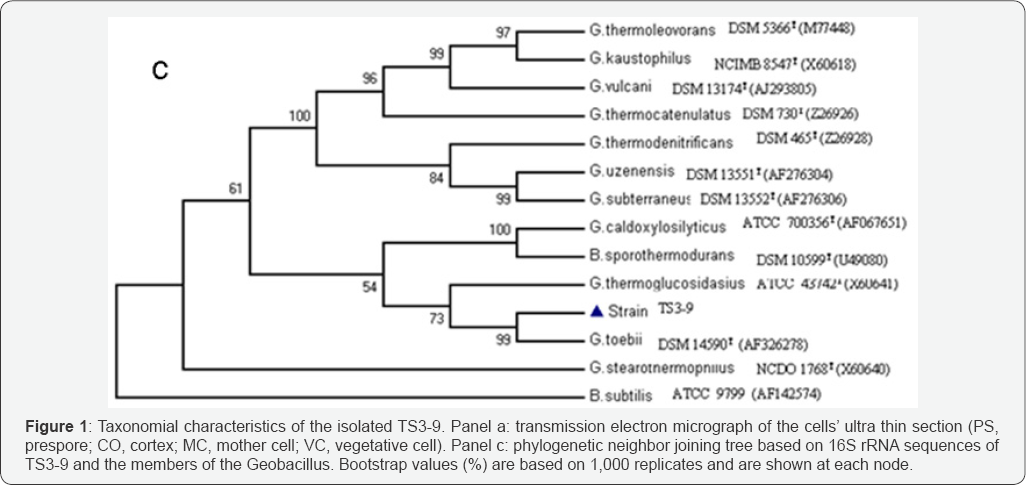
The DNA G + C content was determined to be 54±0.5mol %, distinguished from that of G. thermodenitrificans (58.5%), G. stea- rotermophilus (59.7%), and G. toebii SK-1T (43.9%). Compara-tive 16S rDNA gene sequence analysis showed that strain TS3-9 was phylogenetically most closely affiliated to the genus Geoba-cillus, and had a similarity level ranging 96-99% with respect to sequences of the type strains of recognized Geobacillus species (Figure 1c). In particular, the closer phylogenetic neighbor to isolate TS3-9 was G. toebii (98%, Genbank accession no. AF326278), and the more distant was G. stearothermophilus (96%, Genbank accession no. X60640). The phylogenetic tree analysis of closely related species showed that the strain TS3-9 fell within the cluster comprising Geobacillus species. Comparison of strain TS3-9 with other common Geobacillus strains revealed several unique fea-tures, making this new isolate a distinct member within the genus Geobacillus. Therefore the isolate TS3-9 is identified as Geobacillus sp., and named Geobacillus sp. TS3-9, which is deposited at the CCTCC (with accession number M 2010101) [24].
Synthesis and preparation of EPS
Strain TS3-9 showed a good yield of EPS in fermentation me-dium with glucose (30.6 mg L-1, sucrose (36.2mg L-1), galactose (36.8 mg L-1), and lactose (52.2mg L-1). The maximum yield of EPS by the isolate TS3-9 was reached in stationary phase after 25h of fermentation with the production of 87 mg L-1 in the optimal process (55 °C, pH 8.0 and 150rpm). In comparison, another EPS producer thermophilic strain 4009, isolated from muddy soil (Italy) with high temperature of 65 °C (Nicolaus et al., 2002), was induced by trehalose, reaching 60 mg L-1 EPS at the end of stationary phase. Another strain Geobacillus sp. V264 isolated from spring water sample with temperature 79 °C and pH 7.8, produced EPS of 60mg L-1 after 12h of cultivation [18]. High EPS producers reported include Bacillus thermantarcticus with 400mg L-1 max production [22]. Liu et al. [25] reports that Bacillus licheniformis strain 8-37-0-1 produces up to 47.45g L-1 EPS. These examples suggest that improvement of EPS production is essential for developing a successful biotechnological process.
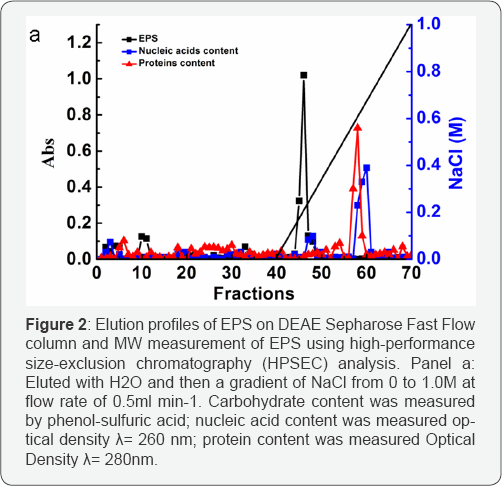
Crude EPS (80mg) was obtained from 1000ml of the fer-mented broth with cool ethanol precipitation, deproteination by pepsin, isopropanol precipitation, dialysis and lyophilization. Pu-rified EPS (15mg) was obtained from crude EPS by a sequential purification process (Figure 2a). Two fractions containing carbo-hydrates were separated and identified by DEAE Sepharose Fast Flow column. The first was eluted with distilled water, implying that it was of neutral character, and its yield was low. The second, an acidic fraction, was eluted in high yield with a NaCl gradient. All subsequent analyses were performed on the second acidic frac-tion. The purified EPS as a white powder was used for subsequent analysis. It had negative response to the Bradford test and no absorption at 280nm and 260nm in the UV spectrum, indicating the absence of protein and nucleic acid. As determined by colori-metric method, the polysaccharide fraction contains 18.6% uronic acid and 6.07% acetyl (wt. %). The HPSEC elution profile showed a single, symmetrical and sharp peak, revealing the EPS to be a homogeneous polysaccharide. The molecular size of EPS, estimat-ed by HPSEC using a calibration curve of standard dextrans, was approximately 3.2 x 106 Da.
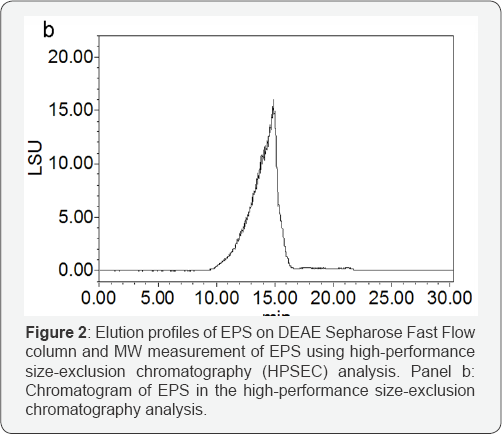
Structural characterization of EPS
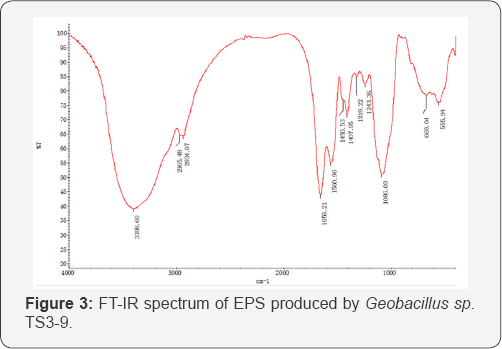
The infrared spectrum of EPS (Figure 3) is similar to those of other bacterial polysaccharides reported in the literature [21]. A strong band at 3398.60cm-1 was attributed to the hydroxyl stretching vibration of the polysaccharide. During 30002800cm-1 wave number region, two weak absorption peaks were detected due to C-H stretching vibration. A characteristic absorption, at 1086.09 cm-1, was also observed, indicating the presence of the pyranose ring of the sugar units. Another characteristic absorption, appearing at 1656.21 cm-1, was dominated by the ethanoyl linkage of (C=O) stretching vibration contribution.
Hydrolysis of EPS from isolate TS3-9 was conducted using 2M trifluoroacetic acid and yielded D-mannose, D-glucose, and rham- nose as principal monosaccharide compositions in a proportion of 1:0.14:0.06. The monosaccharide compositions of G. tepidamans V264 isolated from Bulgarian hot springs were glucose, galactose, fucose, and fructose in the relative proportions of 1:0.07:0.04:0.02 [12]; G. thermodenitrificans B3-72 isolated from Shallow, Marine Hydrothermal Vents of Flegrean Ares showed the presence of mannose : glucose : galactose: mannosamine in a relative proportions of 1:0.1:tr:tr (trace amount) [26]. The data showed that sugar constituents and the relative proportions of TS3-9 were distinct from those of Geobacillus.
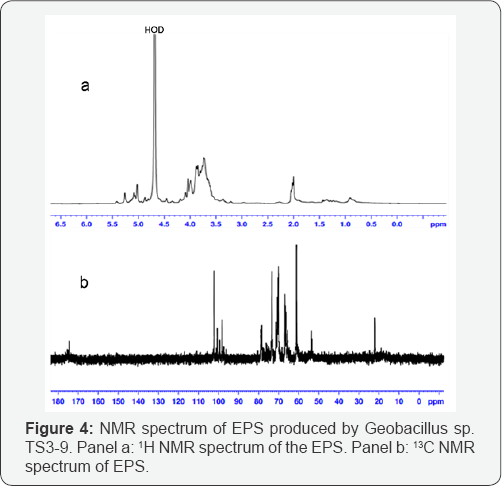
1H NMR spectrum of the biopolymer showed peaks in region from δ 5.0 to δ 5.4ppm and 13C NMR spectrum exhibited well-re-solved anomeric carbons from δ 98.0 to δ 102.0ppm (Figure 4). Based on these signals, the residues were determined to belong to pyranose ring with β-anomeric configuration. One interesting finding from the 1H and 13C NMR spectra was a peak at δ 2.0195 in 1H NMR and peaks at δ 22.16, δ 5174.40 in the 13C NMR spec-trum, demonstrating that the biopolymer was substituted by the acetyl groups [27], consistent with the analyses resulting from the colorimetric method.
Antioxidative activity in vitro
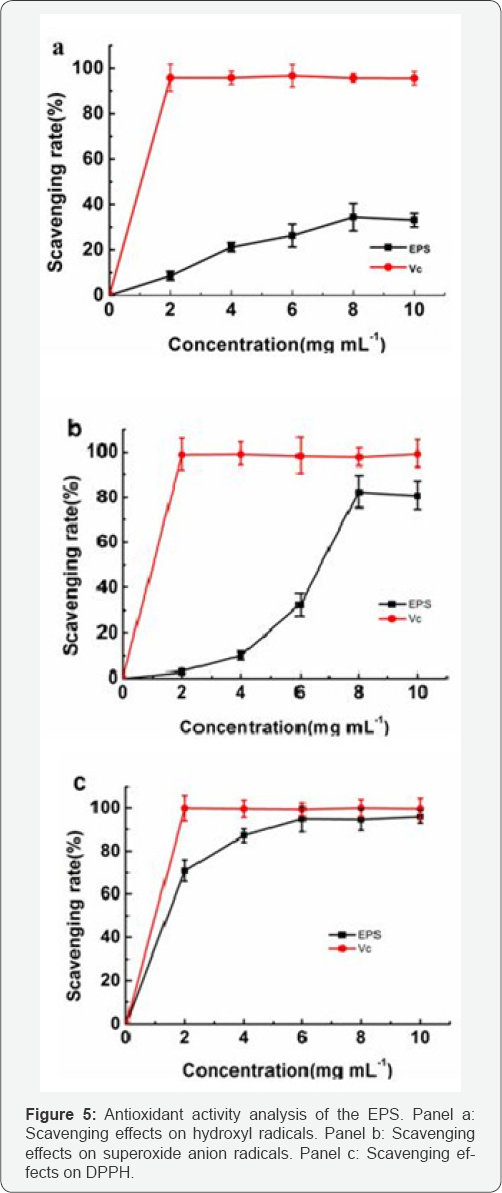
In the present experiment, the antioxidant activities of EPS in vitro were evaluated using different biochemical methods of superoxide anion, hydroxyl and DPPH. As shown in Figure 5a, the hydroxyl radical scavenging activity of EPS was concentration-dependent and slightly lower than that of Vc in the tested dosage range. The results of superoxide radical scavenging assay are showed in Figure 5b. The profile of EPS to scavenge superoxide radical was different from the hydroxyl radical scavenging activity: it had moderate activity at low concentration (<6mg mL-1), but it showed high activity at higher concentration (>8mg mL-1). The scavenging of EPS on the DPPH radical were measured and shown in Figure 5c. DPPH scavenging activity of the EPS was moderate, and the profile was different from the hydroxyl radical scavenging and superoxide radical scavenging assay.
Anti-tumor activity of EPS
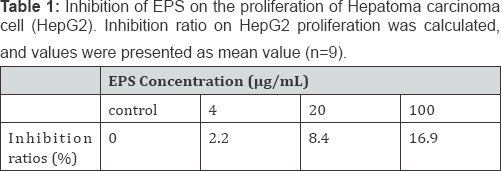
As shown in Table 1, the polysaccharide EPS was found to significantly inhibit the proliferation of HepG2 compared with the control (P<0.05). The inhibition effect of EPS on the proliferation of HepG2 was in a dose-dependent manner at concentrations from 4μg/mL to 100μg/mL, the inhibition ratio on HepG2 ranged from 2.2% to 16.9%. Further studies regarding the in vivo and in vitro antitumor activity effects of the EPS, as well as its mechanism, are needed in order to explore exopolysaccharide as a potential natural tumor inhibitor.
Conclusion
A thermophilic, radiation resistant bacterium, TS3-9, was iso-lated from radon hot springs and assigned to a distinct member of genus Geobacillus. TS3-9 produces a homogenous EPS with average molecular weight approximately 3.2 x 106Da. Chemical composition analysis on this EPS showed D-mannose, D-glucose, and rhamnose were major monosaccharide components. 1H and 13C NMR results demonstrated the structure complexity of the EPS. The EPS demonstrated good activities in antioxidant and restraint of proliferation of hepatoma carcinoma cell. Future investigation on TS3-9 will be focused on biochemical/molecular analysis to reveal the mechanism related to the thermal and radiation resistance features [28,29].
Acknowledgment
Acknowledgements the work was financially supported by the National Nature and Science Foundation of China (3117175) and Nature and Science Foundation of Zhejiang Province (Y3100609).
References
- Rau DC, Parsegian VA (1990) Direct measurement of forces between linear polysaccharides xanthan and schizophyllan. Science 249: 12781281.
- Wu X, Chen Y, Li Y, Li O, Zhu L, et al. (2011) Constitutive expression of Vitreoscilla haemoglobin in Sphingomonas elodea to improve gellan gum production. Appl Microbiol Biot 110: 422-430.
- Wang Z, Liu M, Zhang F, Zhong W, Sun A, et al. (2010) E. coli K5 fermentation and the preparation of heparosan, a bioengineered heparin precursor. Biotechnol Bioeng 107(6): 968-977.
- Coleman RJ (2008) Identification and organization of genes for diutan polysaccharide synthesis from Sphingomonas sp. ATCC 53159. J Ind Microbiol Biotechnol 35(4): 263-274.
- Kornmann H, Duboc P, Marison I, Stockar UV (2003) Influence of nutritional factors on the nature, yield, and composition of exopolysaccharides produced by Gluconacetobacter xylinus I-2281. Appl Environ Microb 69(10): 6091-6098.
- Arena A, Gugliandolo, Giovanna G, Pavone S, Iannello B, et al. (2009) An exopolysaccharide produced by Geobacillus thermodenitrificans strain B3-72: antiviral activity on immunocompetent cells. Immunol Lett 123(2): 132-137.
- Fernando MC, Emilia QM, Jose M, Inmaculada L, Victoria B (2005) Palleronia marisminoris gen. a moderately halophilic, exopolysaccharide producing bacterium belonging to the Alpha proteo bacteria, isolated from a saline soil. Int J Sys Evol Micr 55: 2525-2530.
- Ewert M, Deming JW (2011) Selective retention in saline ice of extracellular polysaccharides produced by the cold-adapted marine bacterium Colwellia psychrerythraea strain 34H J Glaciol 52: 111-117.
- Mancuso CN, Bowman JP, Guezennec J (2005) An exopylysaccharide- producing marine bacterium from the family Flavobacteriaceae, isolated from the Southern Ocean. Appl Environ Microb 5: 1557-1561.
- Soledad A, Ana O, Maria RF, Richard T, Emilia Q et al. (2003) An exopolysaccharide produced by the halophilic bacterium Halomonas maura, with a novel composition and interesting properties for biotechnology. Extremophiles 7(4): 319-326.
- Gugliandolo C, Lentini AV, Spano Maugeri TL (2012) New bacilli from shallow hydrothermal vents of panareaisland (Italy) and their biotechnological potential. J Gen. Appl Microbiol 112(6): 1102-1112.
- Kambourova M, Mandeva R, Dimova D, Poli A, Nicolaus B, et al. (2009) Production and characterization of a microbial glucan, synthesized by Geobacillus tepidamans V264 isolated from bulgarian hot spring. Carbohyd Polym 77: 338-343.
- Moriello SM, Lama L, Poli A, Gugliandolo C, Maugeri TL, (2003) Production of exopolysaccharides from a thermophilic microorganism isolated from a marine hot spring in flegrean areas. J Microbiol. Biotechn 30(2): 95-101.
- Wensinck F, Johanna JB (1957) Quantitative analysis of the gram reaction. J Gen Appl Microbiol 17: 401-413.
- Rainey FA, Ray K, Ferreira M (2005) Extensive diversity of ionizing radiation resistant bacteria recovered from sonoran desert soil and description of nine new species of the genus deinococcus obtained from a single soil sample. Appl Environ. Microbe 71(9): 5225- 5235.
- Clayton M, Claudio CO (2008) Optimization of the selectivity of a cyanopropyl stationary phase for the gas chromatographic analysis of Trans fatty acids. J Chromatography A 1194(1): 111-117.
- Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16: 111-120.?
- Dehainault C, Lauge' CA, Moncoutier VC, Berhouet SP (2004) Multiplex pcr/liquid chromatography assay for detection of gene rearrangements. Application to RB1 gene. Nucleic. Acids Res 32(18): 139-148.
- Manca MC, Lama L, Improta R, Esposito E, Gambacorta A, et al. (1996) Chemical composition of two exopolysaccharides from Bacillus hermoantarcticus Appl. Environ Microb 62(9): 3265-3269.
- Dubios M, Gilles K, Hamilton JK, Rebers PA, Smith F (1951) A colorimetric method for the determination of sugars. Nature 4265: 167.
- Gao CJ, Wang ZY, Su TT, Zhang J, Yang XH (2012) Optimisation of exopolysaccharide production by Gomphidius rutilus and its antioxidant activities in vitro. Carbohyd Polym 87: 2299-2305.
- Xiang Y (2012) Chemical properties and antioxidant activity of exopolysaccharides fractions from mycelial culture of inonotus obliquus in a ground corn stover medium. Food Chem 134: 1899-1905.
- Dubuisson M, Wergifosse B (2000) Antioxidative properties of natural coelenterazine and synthetic methyl coelenterazine in rat hepatocytes subjected to tert-butyl hydroperoxide-induced oxidative stress. Biochem Pharmacol 60(4): 471-478.
- Zhu Z, Liu N, Si C, Liu Y, Ding L, Jing C, et al. (2012) Structure and anti-tumor activity of a high-molecular-weight polysaccharide from cultured mycelium of Cordyceps gunnii. Carbohyd Polym 88(3): 10721076.
- Liu C, Lu J, Lu L, Liu Y, Wang F, Xiao M et al. (2010) Isolation, structural characterization and immunological activity of an exopolysaccharide produced by Bacillus licheniformis 8-37-0-1. Bioresource. Technol 101(14): 5528-5533.
- Nicolaus B, Lama L, Panico A, Moriello VS., Romano I, et al. (2002) Production and characterization of exopolysaccharides excreted by thermophilic bacteria from shallow, marine hydrothermal vents of flegrean ares (Italy). Syst Appl Microbiol 25(3): 319-325.
- Pan D, Wang LQ, Chen CH, Teng BS, Wang CD, et al. (2012) Structure characterization of a novel neutral polysaccharide isolated from ganoderma iucidum fruiting bodies. Food Chem 135: 1097-1103.
- Serrato RV, Sassaki GL, Gorin PAJ, Cruz LM, Pedrosa F, et al. (2008) Structural characterization of an acidic exoheteropolysaccharide produced by the nitrogen-fixing bacterium Burkholderia tropica. Carbohyd Polym 73(4): 564-572.
- Wang Z, Liu M, Zhang F, Zhong W, Sun A, et al. (2010) E. coli K5 fermentation and the preparation of heparosan, a bioengineered heparin precursor. Biotechnol Bioeng 107(6): 968-977.






























