An In-Vitro Trial on Antifungal Effect of Xanthium Strumarium Leaf Extract on the Growth of the Mycelial form of Histoplasma Capsulatum Var Farciminosum Isolated from Horse
Fentahun Wondmnew1*, Kindu Wondmnew1, Feqadu Regasa2 and Kiflu Belete2
1Department of Veterinary Medicine, Adis Ababa University, Ethiopia
2Adis Ababa University, Ethiopia
Submission: June 18, 2016 ; Published: July 5, 2016
*Corresponding author: Fentahun Wondmnew, Department of Veterinary Medicine, Adis Ababa University, Adis Ababa, Ethiopia
How to cite this article: Fentahun W, Kindu W, Feqadu R, Kiflu B. An In-Vitro Trial on Antifungal Effect of Xanthium Strumarium Leaf Extract on the Growth of the Mycelial form of Histoplasma Capsulatum Var Farciminosum Isolated from Horse. Adv Biotech & Micro. 2016; 1(2): 555557. DOI: 10.19080/AIBM.2016.01.555557
Abstract
Epizootic lymphangitis (EL) has deleterious effect on both welfare and health of the horses and mules so that it has a serious negative impact on the livelihoods of cart-horse owners/drivers in the affected parts of Ethiopia. Unfortunately, antifungal drugs for the treatment of EL are costly and mostly unavailable in such areas. The aim of this study was to investigate into the effect of Xanthium Strumarium leaf extract (XSLE) on the Growth of the mycelial form of Histoplasma capsulatum var Farciminosum which has been reported to have a strong antifungal activity in previous studies, in vitro. Histoplasma Capsulatum var Farciminosum was isolated from a horse with a cutaneous form of EL. Agar dilution assay was performed to evaluate the anti HCF activity of XSLE and measure the minimum inhibitory concentration (MIC). Ketoconazole was used as a positive control. Xanthium Strumarium extract inhibited the growth of HCF at concentrations of 0.068 mg/ml. The result indicates that XSLE can potentially be used for the treatment of EL provided that convenient methods of preparation, dose and route of administration are established through rigorous in vitro and in vivo trials.
Keywords: Epizootic lymphangitis; Xanthium Strumarium leaf extract; Horses; Mule; Ethiopia
Abbreviations: MIC: Minimum Inhibitory Concentration; SPANA: Society for the Protection of Animals Abroad; HCF: Histoplasma capsulatum var Farciminosum; SDA: Sabouraud’s Dextrose Agar; XSLE: Xanthium Strumarium Leaf Extract
Introduction
Histoplasma capsulatum var Farciminosum is the causal agent of epizootic lymphangitis, or preferably histoplasmosis, a chronic disease of horses and other Equidae [1] generally affecting the subcutaneous lymph nodes and the lymphatics of the neck, chest and legs but it can also be present as an ulcerating conjunctivitis of the palpebral conjunctiva, or rarely as a multifocal pneumonia [2]. In all cases, the lesions are nodular and granulomatous in character, and the organism, once established, spreads locally by invasion and then via the lymphatics [3]. Epizootic lymphangitis is a contagious disease which can infect humans [4]. The yeast form of the fungus is found in the infected host and the mycelial saprophytic stage in the environment [3,5]. Epizootic lymphangitis has a serious negative impact on the livelihoods of cart horse owners/drivers in the affected areas and also compromises the welfare of working horses [6]. The disease is endemic in countries bordering the Mediterranean, particularly in Italy and North Africa and is also found in Central and Southern Africa, and in regions of Asia and Russia [4].
The mode of transmission of the disease is not well established [7]. Direct contact with infective materials through injured skin or through cutaneous abrasions is the most common mode of infection [8]. In Ethiopia the disease was reported to have an overall prevalence of 18.8% in 28 towns being more common in mid altitude areas (1500 to 2300 meter above sea level) [9].
Recommended treatments for EL include intravenous sodium iodide or intravenous amphotercin B [4]. Another treatment regimen involves local treatment of skin lesions by incising nodules, draining the pus and infusing with 4% iodine tincture followed by oral potassium iodide (30 g for a horse of 200–250 kg) daily for 5 days and then every other day for a further 3–4 weeks or longer if owners comply [10]. This regimen, devised by the Society for the Protection of Animals Abroad (SPANA) clinic at the College of Veterinary Medicine and Agriculture of Addis Ababa University, was reported to result in a recovery rate of 85% [11].
Ethno pharmacologists, botanists, microbiologists, and natural products chemists are combing the Earth for phytochemicals and “leads” which could be developed for treatment of infectious diseases [12]. Medicinal plants would be the best source to obtain a variety of drugs in developed countries about 80 of plants are used in traditional medicine [13]. Over 50% of modern clinical drugs are natural product of medicinal plants and these natural products play an important role in drug development programs in the pharmaceutical industry [14] humans are using natural product of animals, plants and microbial sources for thousands of years either in the pure form or crude extracts [15].
In Ethiopia the medicinal plants are widely used in the society. This has been examined to assure the plants efficacy in treating disease by some researchers. For instance an in-vitro study conducted by [6] indicated that fresh garlic extract has some degree of growth inhibitory effect on the mycelial form of Histoplasma capsulatum var Farciminosum [16] has conducted evaluation of berries of phytol accado decandra for growth inhibition of Histoplasma capsulatum var Farciminosum and treatment of cases of epizootic lymphangitis in Ethiopia and that was effective.
The works mentioned above are good indicators that Xanthium Strumarium leaf extract may have a potent inhibitory effect on Histoplasma capsulatum var Farciminosum (HCF). There was no research that has been done so far in this plant extract to assure whether it has antifungal effect for the treatment of EL. Therefore, the objectives of this study were to investigate into the effect of xanthium strumarium leaf extract on the growth of HCF and to determine the minimum inhibitory concentration (MIC) in vitro.
Materials and Methods
Study area
The study was conducted at the College of Veterinary Medicine and Agriculture of Addis Ababa University between November 2013 and May 2014.
HCF Isolate
The Histoplasma capsulatum var Farciminosum isolate was found from Aklilu Lemma Institute of Pathobiology, Addis Ababa University in which it was isolated according to the methods described by [3]. The isolate was then sub cultured into Sabouraud’s dextrose agar (SDA) slants (2.5% glycerol and 0.005% chloramphenicol) and incubated for 21 days at 27 °C.
Agar dilution assay
Medicinal plant used for study
Xanthium strumarium / rough cocklebur: is broad leaved, tap rooted herbaceous annual plant which is in family Asteraceae, sub family asteroideae tri beheliantheae, genus xanthium and species xanthium strumarium. It grows as weed throughout on waste lands. Cockleburs are short day plants and they can also flower in the tropics where the day length is constant. The herb is reputed as medicine in Europe, China, Indo-china, Malaysia and America also [17]. Stem is erect, ridged, rough and hairy and frequently branched resulting somewhat bushy plants from 30-120cm tall. It has small greed unisexual flower occurring in separate cluster at the end of the brunches and main stems. The fruit is brown, hard, woody, bur from 0.4-0.8 inch long and coved with stout, hooked bristle. Its seed are produced in hard, spiny, globes or oval double chambered single seeded bur [18]. Beside its medicinal value, if small quantity of parts of the mature plant is consumed, the seeds and seedlings will cause intoxication because extremely toxic chemical carboxyatratyloside is contained in them [19].
Collection of medicinal plant
The Collection of medicinal plant was done in Kemissie Amhararigion of Oromia zone. Fresh plants were collected and shade then powdered. Powder was preserved in zip lock bags at room temperature.
Preparation of Xanthium strumarium leaf extract (XSLE)
25 grams of xanthium strumarium were mixed with 250 ml of 95% ethanol in separate bottle and shacked gently. After this was done the mixtures were left in the laboratory class at room temperature for 7 days. After that the solutions were filtered by filter pepper into another pure jar. Then it was put in vacuum drying machine and ethanol and plant extract were separated in different container. The plant extracts were put on Petri dish and set in the incubator for 24 hours. The extract can be changed to the stock solution any time after 24 hours incubation. Therefore the stock solution was prepared after two weeks by diluting with distilled water.
Preparation of the test media
Serial dilutions of the XSLE stock solution were made by using sterile distilled water as a diluents to obtain solutions with the following concentrations: 4.91 mg/ml, 2.18 mg, 1.11 mg/ml, 0.55 mg/ml, 0.27 mg/ml, 0.14 mg/ml, 0.068 mg/ml and 0.034 mg/ml. 2 ml of distilled water was added into each 7 universal bottles and into the first universal bottle 2ml of stock solution was added. From 8% XS 2ml of the extract was taken and dropped into the first three universal bottles. Then three serial dilutions were made for the remaining 7 universal bottles. Then into each 8 universal bottles 14ml of SDA was added and mixed well and slant was made by allowing solidifying in an inclined position and kept at 4 °C for 5 hours until inoculation with the test fungus. Then the agar dilution assay and inoculation of the test fungus were repeated three times to see the reputability of the experiment.
Preparation of the control media
Ketoconazole in the range of 0.03125 μg/ ml to 16 μg/ ml (standard) and saline water were inoculated with the test fungus. Antifungal susceptibility testing broth macro dilution testing was performed in accordance with the guidelines in NCCLS document [20].
Inoculums preparation
A pure sub culture of the mycelial colony of HCF on SDA (with 2.5% glycerol and 0.005% chloramphenicol) (Figure 1) was used for the tests. The inoculums suspension was prepared by transferring a portion of the fungal colony to a sterile saline solution in a sterile test tube using a sterile wire loop. The turbidity of the suspension was adjusted by adding fungal colony or saline and vigorously mixing using a Vortex until turbidity approximates that of a 0.5 McFarland standard [21].
Inoculation of the media
The inoculums suspension was inoculated into triplicates of the test media and the controls using sterile swabs. After dipping into the suspension, the swab was uniformly streaked all over the agar surface.
Determination of minimum inhibitory concentration (MIC)
Minimum inhibitory concentration was defined as the lowest concentration of drug that completely inhibited visible growth [22]. The MIC was determined after incubating the inoculated media at 27 °C for 21 days for sub cultured HCF colony.
Results
HCF isolate
The Histoplasma capsulatum var Farciminosum isolate which was found from Aklilu Lemma Institute of Pathobiology, Addis Ababa University was reexamined with gram staining (Figure 2) and sub cultured in which the mycelial form of the organism grows slowly under aerobic conditions at 27oC on a media, enriched Sabouraud’s dextrose agar, 2.5% glycerol and 0.005% chloramephenicol (SDA) (Figure 1) for its activeness and showing characteristic morphology of the target fungal colony. Organisms are usually surrounded by a ‘halo’ when stained with Gram stain [3].

Agar dilution assay
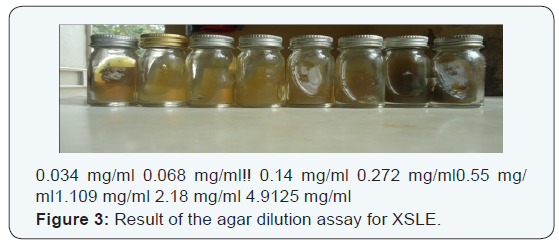
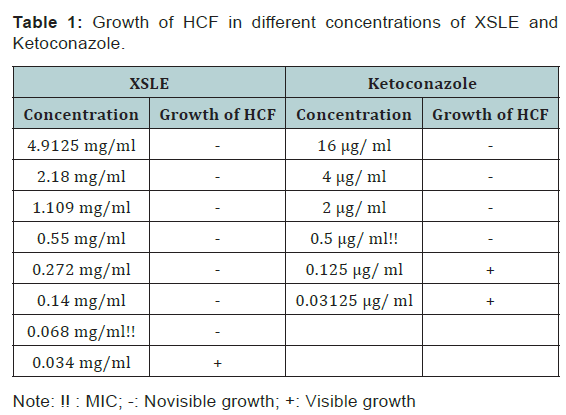
Fungal growths were not observed in media containing the XSLE at concentrations of 4.91 mg/ml, 2.18 mg, 1.11 mg/ml, 0.55 mg/ml, 0.27 mg/ml, 0.14 mg/ml, and 0.068 mg/ml (Figure 3). The MIC of the XSLE against HCF was 0.068 mg/ml (Figure 3) and that of the positive control was 0.5 μg/ ml (Figure 4). The highest concentration of XSLE without growth inhibitory effects was 0.034 mg/ml (Figure 5). The results of the agar dilution assays are summarized in (Table 1).


Discussion
The isolate was checked by gram staining and sub culturing so that it was characteristic HCF both in gram staining and colony morphology as indicated in [5,8,3]. The interesting foundation that was encountered while sub culturing the HCF colony was that well visible growth has been seen in 21 days even if growth is relatively slow and most isolates require from four to eight weeks for development of characteristic colonies [4]. Histoplasma capsulatum var Farciminosum produces moderately growing, white to buff brown, velvety colonies on most mycological media at 25 to 30 °C so that it was grown with this characteristics on SDA (with 2.5% glycerol and 0.005% chloramphenicol) [21].
Whole plant of Xanthium strumarium as well as all parts separately is used in medicine [17]. The genus xanthium also possess antibacterial, antiviral, antimalarial, fungicidal, insecticidal, and cytotoxic activities against cancer cell lines [23]. The inhibitory effect of Ketoconazole is 136 times potent than XSLE on the growth of the mycelial form of HCF in which it is more potent medicinal plant.
Antifungal activity was determined by the agar diffusion method. Test samples were diluted in Sabouraud dextrose agar followed by solidification in slanting positions. Test fungal cultures were inoculated on the slant and were incubated at 29°C for 3-7 days [24,25]. Among dermatophytes, the most susceptible strain was microsporumcanis which showed 50% and 60% inhibition against crude extract and ethylacetate fraction, respectively. Aspergillus flavus was the second most susceptible fungi, exhibited 50% inhibition against n-hexane fraction [26].
The MIC of XSLE obtained in this study was 0.068 mg/ml. However, the difference between the MIC and the maximum non inhibitory concentration (0.034 mg/ml) reported in this study suggest that the MIC is higher. The principal compounds isolated from Xanthium strumarium leaves were found to contain, isoxanthanol, hydroquinone, caffeyolquinic acids, xanthanol, anthraquinone, cardenolide, leucoanthocyanin, simple phenolics, triterpenoids and thiazinedione [17]. Xanthium strumarium produces secondary metabolites such as alkaloids, tannins, terpenoids, flavonoids, chloroform and n-hexane fractions whose activity has been demonstrated to be antifungal [27]. Antifungal activity of these molecules from xanthium strumarium exhibited 60% and 50% inhibition activity against the major dermatophyte fungi, microsporumcanis [14]. Even if the effect of XS has not been studied on HCF secondary metabolites especially Chloroform (CHCl3) and n-hexane (C6H14) group from Xanthium strumanrium are cytotoxic with an average 9-16 mm zone of inhibition. Further investigations are, however, necessary to explore mechanism(s) of action involved in these pharmacological activities [28].
Conclusion
My study revealed that XSLE strong inhibitory effect on the growth of the mycelial form of HCF. XSLE inhibited the growth of HCF at concentrations ≥0.068 mg/ml. The result indicates that XSLE can be included in the treatment of EL provided that convenient methods of preparation, dose and route of administration should be established through rigorous in vitro and in vivo trials
Recommendation
Based on the above conclusions the following recommendations have made
- Further studies on phyto-chemical analyses should be conducted on this plant and the active chemical should be identified.
- In-vivo studies must be conducted so that the safety margin, toxicity and cure rates will be known in order to use them commercially.
References
- The Center for Food Security and Public Health (2009) Epizootic Lymphangitis. Iowa State University College of Veterinary Medicine, pp. 1-4.
- Chandler FW, Kaplan W, Ajello L (1980) A colour atlas and textbook of histopathology cases of histoplasmosis farciminosi in horses and its morphological characteristics. Wolfe Medical Publications Ltd London 13: 507-508.
- OIE (2008) Terrestrial Manual for Epizootic Lymphangitis. Office International des Epizooties 852-857.
- Al-Ani FK (1999) Epizootic lymphangitis in horses: a review of the literature. Rev Sci Tech 18(3): 691-699.
- Weeks RJ, Padhye AA, Ajello L (1985) Histoplasma capsulatum variety farciminosum. A new combination for Histoplasmafarciminosum. Mycologia 77(6): 964-970.
- Mesfin M (2012) The effect of fresh garlic extract on the growth of the mycelial form of histoplasma capsulatum var farciminosum- an In Vitro Trial (unpublished DVM thesis at College of Veterinary Medicine and Agriculture, Addis Ababa University).
- Singh T (1995) Studies on epizootic lymphangitis I Modes of infection and transmission of equine histoplasmosis (epizootic lymphangitis). Indian J Vet Sci 35: 102-110.
- Selim SA, Soliman R, Osman K, Padhye AA, Ajello L (1985) Studies on histoplasmosis farciminosii (epizootic lymphangitis) in Egypt. Isolation of Histoplasma farciminosum from cases of histoplasmosis farciminosii in horses and its morphological characteristics. Eur J Epidemiol 1(2): 84-89.
- Ameni G (2006) Epidemiology of equine histoplasmosis (epizootic lymphangitis) in carthorses in Ethiopia. Vet J 172(1): 160-165.
- Jones J (2006) Epizootic lymphangitis: the impact on subsistence economies and animal welfare. Vet J 172(3): 402-404.
- Aklilu N, Zerfu A (2010) Socio economic impact of epizootic lymphangitis (EL) on horse drawn taxi business in central Ethiopia. Proceedings of the 6th International Colloquium on Working Equids, pp. 83-86.
- Cowan MM (1999) Plant products as antimicrobial agents. Clinical Microbiology Reviews 12(4): 564-582.
- World Health Organization (1997) Quality assurance of pharmaceuticals: a compendium of guidelines and related materials. Geneva 1: 31-37.
- Bharathi B, Padmini R, Swamidoss D, Gorge (2010) Phytochemichal analysis and antibacterial activity of solanumtrilobatum against HIV associated bacterial pathogens. Int J P H SCI 2(2): 598-605.
- Parekh j, Chanda S (2007) In-vitro antimicrobial activity of Trapanatans Linn. Fruit rind extracted in different solvents. Afr J Biotechnol 6(6): 766-770.
- Negesse M, Eyasu M, Nigatu A, Gobena A (2012) evaluation of berries of phytolaccadodecandra for growth inhibition of histoplasmacapsulatum var farciminosum and treatment of cases of epizootic lymphangitis in Ethiopia. Asian Pac J Trop Biomed 2(7): 505-510.
- Bhogaonkar PY, Ahmad SA (2012) Pharmacognostic studies on xanthium strumarium L- A Folk Unani Medicinal Herb. Bioscience Discovery 3(1): 101-106.
- Agharkar SP (1991) Medicinal plant of Bombay presidency pbl. Scientific publishers, Jodhpur (India). 1: 852-857.
- Madalln RL, Sing JP (2001) Diseases, competitors moulds and physiological disorders of button mushroom and their management. Journal of Chafan Singh Haryana 30(1): 193-194.
- Ren-kai L, Meral AC, Nicole N, Lester P, David WW, et al. (2000) In Vitro Activities of Voriconazole, Itraconazole, and Amphotericin B against Blastomyces dermatitidis, Coccidioides immitis, and Histoplasma capsulatum. Antimicrobial agents and chemotherapy 44(6): 1734- 1736.
- Deanna AS, Padhye AA, Standard PG, Michael GR (1997) An aberrant variant of histoplasma capsulatum var capsulatum. J Clin Microbiol 35(3): 734-735.
- Li RK, Ciblak MA, Nordoff N, Pasarell L, Warnock DW, et al. (2000) A clinical and laboratory update. Journal of Epidemiology 1: 84-89.
- Sravani PS, Mohana L, Kumar AS (2010) Evaluation of Diuretic Activity of Xanthium strumarium. International Journal of Preclinical and Pharmaceutical Research 1(1): 31-34.
- Paxton JD (1991) Assay for antifungal activity. Academic Press, London. Wolfe Medical Publications Ltd. London, England. New combination for Histoplasma farciminosum. Mycologia 77: 964-970.
- Nisar M, Kaleem WA, Qayum M, Abrar H, Zia-ul-haq M, et al. (2010) Biological screening of zizyphus oxyphylla Edgew leaves. Pak J Bot 42: 4063-4069.
- Fazli K, Zafar I, Ayub K, Zakiullah F, Muhammad SK (2012) validation of some of the ethnopharmacological uses of xanthium strumarium and duchesnea indica. Pak J Bot 44(4): 1199-1201.
- Gujar J, Talwankar D (2012) Antifungal potential of crude plant extract on some pathogenic fungi. World Journal of Science and Technology 2(6): 58-62.
- Nasrin F (2014) Antimicrobial and cytotoxic activity of ethyl acetate, chloroform and n-hexane extracts of cucumis sativus leaves. The Experiment 21(3): 1480-1486.






























