Abstract
Background: Adhesive capsulitis is a common shoulder disorder characterized by pain and restricted range of motion (ROM). Corticosteroid injections (CIs) are widely used in management; however, the optimal injection site-intra-articular (IA) or subacromial (SA)-remains unclear.
Purpose: To compare the effectiveness of IA and SA corticosteroid injections in improving pain and shoulder function in patients with adhesive capsulitis.
Methods: A systematic review of Level I and II studies published up to January 23, 2026, was conducted using PubMed, MEDLINE, and EMBASE.
Eligible studies included human participants receiving IA or SA corticosteroid injections and reporting outcomes related to pain or ROM. Nine
studies met the inclusion criteria. Data on study characteristics, outcomes, and risk of bias were extracted and synthesized.
Results: Both IA and SA injections consistently improved pain and ROM in the short term across all included studies. Multiple analyses—
including those by Chen et al. [1], Cho et al. [2], Shang et al. [3], Shin and Lee [4], Buchbinder et al. [5], and Challoumas et al. [6]—reported no
significant difference between IA and SA injections in pain reduction or functional improvement. Some studies (Kitridis et al. [7], Wang et al. [8]
suggested that IA injections may provide slightly greater benefit, particularly when combined with physiotherapy. SA injections were associated
with fewer minor adverse events in some reports. Sensitivity and subgroup analyses generally supported the robustness of these findings.
Conclusion: IA and SA corticosteroid injections are both effective and safe treatment options for adhesive capsulitis, with no clear superiority of one injection site over the other. Treatment selection may depend on patient characteristics, clinician preference, and potential adverse effects. Combining corticosteroid injections with physiotherapy may further enhance outcomes. High-quality randomized controlled trials with
standardized protocols and long-term follow-up are needed to establish definitive recommendations.
Keywords:Nonsteroidal Anti-Inflammatory Drugs; Corticosteroid Injection; Subacromial; Idiopathic; While Subacromial
Abbreviations:IA, Intra-Articular; SA: Subacromial; CI: Corticosteroid Injection; NSAID: Nonsteroidal Anti-Inflammatory Drugs; ROM: Range of Motion
Case Description
Adhesive capsulitis of the shoulder is a pathological condition characterized by fibrosis and adhesion formation within the glenohumeral joint capsule, leading to progressive pain, stiffness, and functional limitation [9]. It affects approximately 2–5% of the population, predominantly women, with peak incidence between 40 and 60 years of age [10]. Patients typically present with gradual onset of shoulder discomfort and restricted range of motion. The condition has been described using various terms, including frozen shoulder, scapulohumeral periarthritis, shoulder contracture, and humeroglenoid acromioclavicular syndrome [11]. Adhesive capsulitis is classified as either primary (idiopathic), when no underlying cause is identified, or secondary, often associated with diabetes mellitus, rotator cuff pathology, or trauma [12]..
Multiple treatment modalities have been proposed, including physical therapy, pharmacologic management, suprascapular nerve block, and arthroscopic capsular release [13,14]. Among these, corticosteroid injections (CIs) are widely regarded as a first-line intervention for primary adhesive capsulitis due to their ability to reduce synovial inflammation, alleviate pain, and accelerate early functional recovery. Intra-articular (IA) injections are commonly used and can be performed without imaging guidance, while subacromial (SA) injections are also frequently utilized, particularly when symptoms overlap with subacromial pathology.
However, uncertainty remains regarding which injection site provides superior clinical outcomes. Given the variability in clinical practice and the lack of consensus on the optimal injection site, a systematic review is warranted to evaluate the comparative effectiveness of IA and SA corticosteroid injections. This review synthesizes the available evidence and highlights the implications for clinical decision-making. Purpose The purpose of this study is to systematically compare the efficacy of intra-articular (IA) and subacromial (SA) corticosteroid injections in patients with adhesive capsulitis, focusing on improvements in pain and range of motion (ROM).
Objectives
• To identify and review Level I and II evidence evaluating
IA and SA corticosteroid injections for adhesive capsulitis.
• To compare the effectiveness of IA and SA injections in
reducing pain and improving shoulder function.
• To assess the quality and risk of bias of the included
studies.
• To provide evidence-based recommendations for clinical
practice regarding the use of IA and SA corticosteroid injections.
Methods
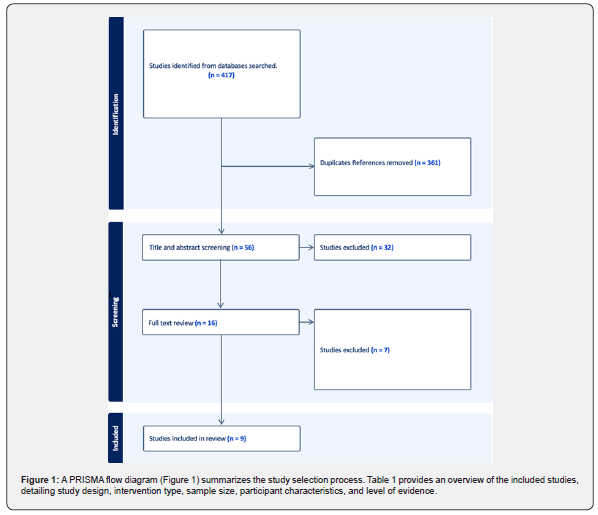
Three independent reviewers conducted a comprehensive literature search of studies published over the past 10 years, up to January 23, 2026, using the PubMed, MEDLINE, and EMBASE databases. The search strategy focused on studies comparing intra-articular (IA) and subacromial (SA) corticosteroid injections for the treatment of adhesive capsulitis. Eligible studies evaluated the efficacy and/or safety of these injection techniques. The primary outcomes of interest were changes in pain and range of motion (ROM). All retrieved articles were screened in multiple stages. Duplicate records were removed, followed by title and abstract screening to exclude irrelevant studies. Full-text screening was then performed to determine final eligibility. Any disagreements among reviewers were resolved through discussion until consensus was reached.
Inclusion criteria: studies were included if they met the
following criteria:
• Level I or Level II evidence
• Published in English
• Human participants
• Interventions involving corticosteroid injections (IA or
SA)
• Reported functional outcomes related to pain and/or
ROM
Exclusion criteria: studies were excluded if they were:
• Nonclinical
• Focused on corticosteroid dosage rather than injection
site
• Evaluating interventions other than corticosteroid
injections (Figure 1, Table 1).
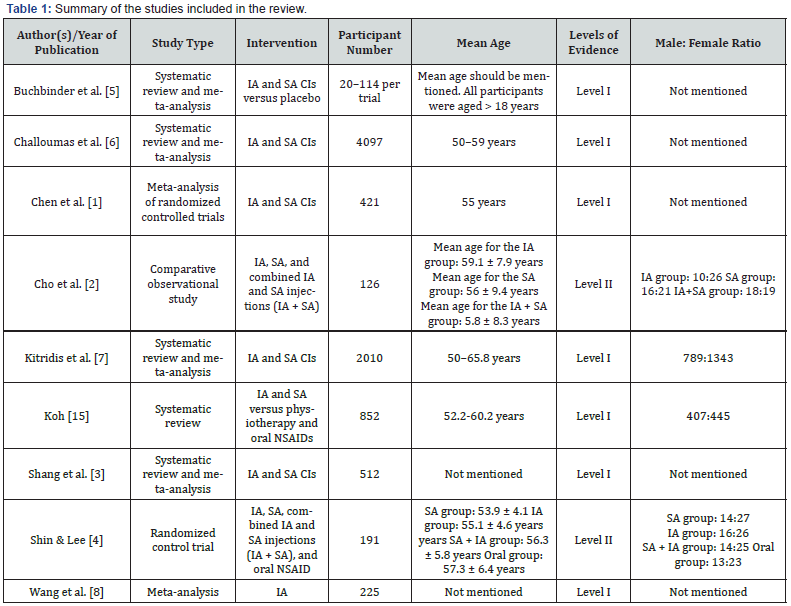
Abbreviations: IA: Intra-Articular; SA: Subacromial; CI: Corticosteroid Injection; NSAID: Nonsteroidal Anti-Inflammatory Drugs.
Results
Characteristics of the Studies All included studies examined the use of corticosteroid injections (CIs) for the management of adhesive capsulitis, with a focus on comparing intra-articular (IA) and subacromial (SA) approaches. Two studies, Chen et al. [1] and Shang et al. [3]-performed indirect comparisons between IA and SA injections through pooled analyses of randomized controlled trials. In contrast, Cho et al. [2] and Shin & Lee [4] directly compared IA and SA injections within their study populations, providing head-tohead clinical outcome data. The remaining studies-Kitridis et al. [7], Wang et al. [8], Koh [15], and Challoumas et al. [6]-evaluated the overall effectiveness of corticosteroid injections without isolating the injection site as the primary comparison.
The studies were conducted across diverse geographic regions, including Iran, Turkey, China, Korea, and the United States, reflecting a broad international interest in optimizing treatment for adhesive capsulitis. Sample sizes varied widely, ranging from small cohorts of 28 participants to large meta-analyses including over 1000 patients. The mean age of participants across studies ranged from 46 to 56 years, consistent with the typical age distribution of adhesive capsulitis. Symptom duration prior to intervention ranged from 4 to 18 months, indicating that patients were treated at various stages of the disease. Follow-up periods also varied, with some studies assessing short-term outcomes at 4 weeks and others extending follow-up to 12 months, allowing for evaluation of both early and sustained treatment effects.
Quality Assessment and Risk of Bias All studies underwent evaluation using the Cochrane Risk-of- Bias tool, which assesses methodological rigor across domains such as randomization, allocation concealment, blinding, incomplete outcome data, and selective reporting. The findings revealed notable variability in study quality. Chen et al. [1], Kitridis et al. [7], and Shang et al. [3] identified a high risk of bias in several included trials, particularly related to inadequate blinding of participants, clinicians, and outcome assessors. This limitation is common in injection-based studies, where blinding is inherently challenging due to differences in injection technique and anatomical landmarks.
These studies also noted concerns regarding incomplete reporting of outcomes and heterogeneity in intervention protocols. In contrast, Cho et al. [2], Wang et al. [8], and Buchbinder et al. [5] reported low risk of bias across most domains, indicating stronger methodological quality. These studies demonstrated clearer randomization procedures, more consistent outcome reporting, and better control of confounding variables. Shin and Lee [14] also reported low risk of bias in most domains, although blinding of outcome assessors remained a limitation. Overall, the quality of evidence varied across the literature.
Chen et al. [1] and Kitridis et al. [7] rated the evidence as moderate to low quality due to methodological inconsistencies and heterogeneity among included studies. In contrast, Wang et al. [8] and Buchbinder et al. [5] rated the evidence as moderate to high quality, reflecting stronger study designs and more robust outcome measures. Koh [15] did not provide a definitive overall quality rating but noted variability in study rigor. Differences in bias assessments were primarily attributed to challenges in blinding, inconsistent reporting of functional outcomes, and variations in corticosteroid type, dosage, and injection technique.
Findings and Synthesis
Chen et al. [1] conducted a meta-analysis comparing IA and SA corticosteroid injections and found no significant difference between the two techniques in improving Constant–Murley scores (mean difference: −0.21; 95% CI: −1.58 to 1.16; p = 0.77; I² = 86%). Their subgroup analyses, which examined outcomes based on corticosteroid type (triamcinolone vs. methylprednisolone) and symptom duration (<6 months vs. >6 months), also demonstrated no meaningful differences between IA and SA injections. These findings suggest that the injection site alone does not significantly influence short-term functional outcomes.
Similarly, Cho et al. [2] reported no significant difference in VAS pain scores between IA and SA groups four weeks after injection (p = 0.213). Although both groups showed substantial improvement from baseline, neither demonstrated superiority in pain reduction or range-of-motion (ROM) recovery at the fourweek follow-up. This reinforces the notion that both injection sites provide comparable early clinical benefits. In contrast, Kitridis et al. [7] and Wang et al. [8] reported findings favoring IA injections. Kitridis et al. [7], through a network meta-analysis, concluded that both IA and SA injections significantly improved pain and function compared with other pharmacologic interventions, but IA injections ranked as the most effective overall.
Wang et al. [8] similarly found that corticosteroid injections were superior to placebo and other treatments in both shortterm (≤4 weeks) and longer-term (>4 weeks) follow-up, with IA injections demonstrating the largest effect sizes for pain relief and functional improvement. Koh [15] also found corticosteroid injections to be more effective than placebo in improving pain and function. Interestingly, their analysis suggested that SA injections may offer greater short-term benefit (up to 6 weeks) compared with IA injections. However, this difference disappeared at longer follow-up intervals (up to 12 months), where IA and SA injections demonstrated equivalent long-term outcomes.
Shang et al. [3] conducted a systematic review and metaanalysis and found no significant differences between IA and SA injections in terms of pain reduction, ROM improvement, or functional recovery. Both injection types were effective for shortterm management of adhesive capsulitis. However, IA injections were associated with a higher risk of adverse events, including post-injection pain and rare cases of joint infection, suggesting that SA injections may offer a slightly safer profile. Shin & Lee [4], Buchbinder et al. [75], and Challoumas et al. [6] all reported similar findings: both IA and SA injections effectively reduced pain and improved ROM, with no statistically significant differences between the two techniques. Shin and Lee [4] observed nearly identical improvements in pain scores (4.7 vs. 4.8) and ROM (32.7° vs. 33.2°) between IA and SA groups.
Buchbinder et al. [5] found no significant pooled differences in pain reduction (SMD −0.06; 95% CI: −0.23 to 0.11; p = 0.50) or ROM improvement (SMD 0.02; 95% CI: −0.21 to 0.25; p = 0.88). Challoumas et al. [6] also reported small effect sizes and no significant differences in pain (SMD −0.29; p = 0.10) or ROM (SMD 0.16; p = 0.25) between IA and SA injections. Overall, the collective evidence demonstrates that both IA and SA corticosteroid injections are effective in reducing pain and improving shoulder function in patients with adhesive capsulitis. While some analyses suggest a slight advantage for IA injections-particularly when combined with physiotherapy-most studies show no clinically meaningful difference between the two injection sites. SA injections may offer a marginally safer profile due to fewer reported adverse events, whereas IA injections may provide slightly greater early functional gains in some populations.
Findings and Synthesis
Chen et al. [1] conducted a meta-analysis comparing IA and SA corticosteroid injections and found no significant difference between the two techniques in improving Constant–Murley scores (mean difference: −0.21; 95% CI: −1.58 to 1.16; p = 0.77; I² = 86%). Their subgroup analyses, which examined outcomes based on corticosteroid type (triamcinolone vs. methylprednisolone) and symptom duration (<6 months vs. >6 months), also demonstrated no meaningful differences between IA and SA injections. These findings suggest that the injection site alone does not significantly influence short-term functional outcomes.
Similarly, Cho et al. [2] reported no significant difference in VAS pain scores between IA and SA groups four weeks after injection (p = 0.213). Although both groups showed substantial improvement from baseline, neither demonstrated superiority in pain reduction nor range-of-motion (ROM) recovery at the fourweek follow-up. This reinforces the notion that both injection sites provide comparable early clinical benefits. In contrast, Kitridis et al. [7] and Wang et al. [8] reported findings favoring IA injections. Kitridis et al. [7], through a network meta-analysis, concluded that both IA and SA injections significantly improved pain and function compared with other pharmacologic interventions, but IA injections ranked as the most effective overall.
Wang et al. [8] similarly found that corticosteroid injections were superior to placebo and other treatments in both shortterm (≤4 weeks) and longer-term (>4 weeks) follow-up, with IA injections demonstrating the largest effect sizes for pain relief and functional improvement. Koh [15] also found corticosteroid injections to be more effective than placebo in improving pain and function. Interestingly, their analysis suggested that SA injections may offer greater short-term benefit (up to 6 weeks) compared with IA injections. However, this difference disappeared at longer follow-up intervals (up to 12 months), where IA and SA injections demonstrated equivalent long-term outcomes.
Shang et al. [3] conducted a systematic review and metaanalysis and found no significant differences between IA and SA injections in terms of pain reduction, ROM improvement, or functional recovery. Both injection types were effective for shortterm management of adhesive capsulitis. However, IA injections were associated with a higher risk of adverse events, including post-injection pain and rare cases of joint infection, suggesting that SA injections may offer a slightly safer profile. Shin & Lee [4], Buchbinder et al. [5], and Challoumas et al. [6] all reported similar findings: both IA and SA injections effectively reduced pain and improved ROM, with no statistically significant differences between the two techniques. Shin & Lee [4] observed nearly identical improvements in pain scores (4.7 vs. 4.8) and ROM (32.7° vs. 33.2°) between IA and SA groups.
Buchbinder et al. [5] found no significant pooled differences in pain reduction (SMD −0.06; 95% CI: −0.23 to 0.11; p = 0.50) or ROM improvement (SMD 0.02; 95% CI: −0.21 to 0.25; p = 0.88). Challoumas et al. [6] also reported small effect sizes and no significant differences in pain (SMD −0.29; p = 0.10) or ROM (SMD 0.16; p = 0.25) between IA and SA injections. Overall, the collective evidence demonstrates that both IA and SA corticosteroid injections are effective in reducing pain and improving shoulder function in patients with adhesive capsulitis. While some analyses suggest a slight advantage for IA injections-particularly when combined with physiotherapy, most studies show no clinically meaningful difference between the two injection sites. SA injections may offer a marginally safer profile due to fewer reported adverse events, whereas IA injections may provide slightly greater early functional gains in some populations.
Discussion
Summary of Evidence and Key Findings This systematic review synthesized evidence from nine studies evaluating the comparative effectiveness of intra-articular (IA) and subacromial (SA) corticosteroid injections (CIs) for the treatment of adhesive capsulitis. Across the included literature—Chen et al. [1], Cho et al. [2], Kitridis et al. [7], Wang et al. [8], Koh [15], Shang et al. [3], and Shin & Lee [4]-both IA and SA injections consistently demonstrated meaningful improvements in pain, range of motion (ROM), and overall shoulder function. These findings reinforce the role of corticosteroid injections as an effective early intervention for adhesive capsulitis. Despite the general agreement that both injection sites are beneficial, the evidence regarding superiority of one technique over the other remains inconsistent.
Several studies, including Chen et al. [1], Cho et al. [2], Shang et al. [3], and Challoumas et al. [6], reported no statistically significant differences between IA and SA injections in terms of pain reduction or functional improvement. These results suggest that the therapeutic benefit of corticosteroids may be largely independent of the injection site. However, some studies provided contrasting findings. Kitridis et al. [7] reported that IA injections were more effective than SA injections, particularly when combined with physiotherapy, which ranked as the most effective treatment strategy in their network meta-analysis. Wang et al. [8] also found that IA injections produced larger effect sizes compared with SA injections, although both were superior to placebo and other conservative treatments. Conversely, Koh [15] observed that SA injections offered greater short-term benefits (up to six weeks), although long-term outcomes were similar between the two approaches.
Adverse events were infrequently reported across studies. When mentioned, SA injections were associated with fewer minor complications compared with IA injections, which carried a slightly higher risk of post-injection pain and rare joint-related adverse events. However, the overall incidence of complications remained low for both techniques. Collectively, the evidence indicates that both IA and SA corticosteroid injections are effective shortterm treatments for adhesive capsulitis. However, the literature remains insufficient to determine whether one approach provides superior long-term outcomes. Given the variability in study designs, corticosteroid types, injection techniques, and followup durations, clinicians should individualize treatment decisions based on patient characteristics, anatomical considerations, and potential risk profiles.
Implications and Conclusion
Despite variability in methodological quality and differences
in publication bias assessments, the overall body of evidence
supports corticosteroid injections as an effective intervention for
adhesive capsulitis. This review demonstrates that both IA and SA
injections provide comparable short-term improvements in pain
and shoulder function, with no consistent evidence favoring one
injection site over the other. Given the absence of clear superiority,
the choice between IA and SA injections should be guided by:
• Patient-specific factors (e.g., comorbidities, pain
location, tolerance for injection technique)
• Clinician expertise and preference
• Risk of adverse events
• Availability of imaging guidance, if needed
Combining corticosteroid injections with physiotherapy may further enhance outcomes, as suggested by network metaanalysis findings. However, clinicians should exercise caution with repeated or long-term corticosteroid use due to potential risks such as cartilage damage, tendon weakening, and diminished shoulder function. In summary, IA and SA corticosteroid injections remain valuable components of the multimodal management of adhesive capsulitis, offering meaningful short-term relief and functional improvement.
Recommendations
• Both IA and SA corticosteroid injections are safe and
effective for short-term management of adhesive capsulitis, with
no consistent evidence supporting the superiority of one injection
site.
• Combining corticosteroid injections with structured
physiotherapy may yield greater functional improvement than
either intervention alone.
• Patient counseling is essential, ensuring that individuals
understand the expected benefits, the importance of adherence
to rehabilitation, and the potential risks associated with repeated
corticosteroid use.
• Clinicians should tailor injection site selection based
on clinical presentation, anatomical considerations, and patient
preference. Strengths and Limitations
This systematic review has several strengths, including a comprehensive search strategy, inclusion of Level I and II evidence, and the use of standardized tools for quality assessment. The inclusion of multiple study designs and international populations enhances the generalizability of the findings. However, several limitations must be acknowledged. The number of eligible studies was relatively small, and methodological quality varied considerably, with some studies demonstrating a high risk of bias. Significant heterogeneity existed in corticosteroid dosage, injection technique, outcome measures, and follow-up duration. Some studies had small sample sizes, limiting statistical power.
Additionally, long-term outcomes were insufficiently reported, making it difficult to determine the sustained effectiveness of IA versus SA injections. Finally, restricting the search to Englishlanguage studies may have introduced publication bias. Despite these limitations, this review provides a comprehensive synthesis of the current evidence regarding IA and SA corticosteroid injections for adhesive capsulitis and highlights the need for further high-quality randomized controlled trials with standardized protocols and long-term follow-up.
Future Research
Future research should aim to address the methodological limitations and evidence gaps identified in the current literature. High-quality randomized controlled trials with larger sample sizes, rigorous study designs, and standardized methodologies are needed to more definitively compare the effectiveness of intra-articular (IA) and subacromial (SA) corticosteroid injections for adhesive capsulitis. Future studies should employ consistent injection techniques, standardized corticosteroid formulations, and uniform outcome measures to reduce heterogeneity and improve comparability across trials. Long-term follow-up is critically needed.
Most existing studies focus on short-term outcomes (4–12 weeks), leaving uncertainty regarding the sustained effects of IA and SA injections on pain, range of motion, and functional recovery. Future trials should evaluate outcomes over extended periods to determine whether one injection site offers superior long-term benefits or reduced recurrence rates. Further research is also required to determine the optimal corticosteroid dose, injection frequency, and timing of intervention. Variability in doing protocols across studies limits the ability to draw firm conclusions about the most effective and safest regimen. Standardizing these parameters would enhance the quality of evidence and guide clinical practice more effectively.
Safety outcomes should be more thoroughly investigated. Although adverse events were infrequently reported in the included studies, future research should systematically evaluate the potential risks associated with corticosteroid injections, including infection, tendon rupture, chondrotoxicity, and osteonecrosis. Understanding these risks is essential for identifying patient populations that may benefit from alternative treatments. Additionally, future studies should address the limitations observed in the current evidence base, such as inconsistent outcome measures, small sample sizes, and lack of blinding of outcome assessors.
Incorporating robust blinding procedures, when feasible, would reduce performance and detection bias. Finally, emerging treatment strategies warrant exploration. Future research should investigate whether combining IA and SA injections offers additive or synergistic benefits compared with either technique alone. Evaluating multimodal approaches—such as combining corticosteroid injections with structured physiotherapy—may also provide insight into optimizing treatment pathways for adhesive capsulitis.
References
- Chen R, Jiang C, Huang G (2019) Comparison of intra-articular and subacromial corticosteroid injection in frozen shoulder: a meta-analysis of randomized controlled trials. Int J Surg 68: 92-103.
- Cho CH, Jin HJ, Kim DH (2020) Comparison of clinical outcomes between idiopathic frozen shoulder and diabetic frozen shoulder after a single ultrasound-guided intra-articular corticosteroid injection. Diagnostics (Basel) 10(6): 370.
- Shang X, Zhang Z, Pan X, Li J, Li Q (2019) Intra-articular versus subacromial corticosteroid injection for the treatment of adhesive capsulitis: a meta-analysis and systematic review. Biomed Res Int 2019: 1274790.
- Shin SJ, Lee SY (2013) Efficacies of corticosteroid injection at different sites of the shoulder for the treatment of adhesive capsulitis. J Shoulder Elbow Surg 22(4): 521-527.
- Buchbinder R, Green S, Youd JM (2003) Corticosteroid injections for shoulder pain. Cochrane Database Syst Rev 2003(1): CD004016.
- Challoumas D, Biddle M, McLean M, Millar NL (2020) Comparison of treatments for frozen shoulder: a systematic review and meta-analysis. JAMA Netw Open 3(12): e2029581.
- Kitridis D, Tsikopoulos K, Bisbinas I, Papaioannidou P, Givissis P (2019) Efficacy of pharmacological therapies for shoulder adhesive capsulitis: a systematic review and network meta-analysis. Am J Sports Med 47(14): 3552-3560.
- Wei Wang, Mingmin Shi, Chenhe Zhou, Zhongli Shi, Xunzi Cai, et al. (2017) Effectiveness of corticosteroid injections in adhesive capsulitis of the shoulder: a meta-analysis. Medicine (Baltimore) 96(28): e7529.
- Le HV, Lee SJ, Nazarian A, Rodriguez EK (2017) Adhesive capsulitis of the shoulder: review of pathophysiology and current clinical treatments. Shoulder Elbow 9(2): 75-84.
- Lee LC, Lieu FK, Lee HL, Tung TH (2105) Effectiveness of hyaluronic acid administration in treating adhesive capsulitis of the shoulder: a systematic review of randomized controlled trials. Biomed Res Int 2015: 314120.
- Georgiannos D, Markopoulos G, Devetzi E, Bisbinas I (2017) Adhesive capsulitis of the shoulder: is there consensus regarding the tr eatment? A comprehensive review. Open Orthop J 11(Suppl 1 M2): 65-76.
- Hsu JE, Anakwenze OA, Warrender WJ, Abboud JA (2011) Current review of adhesive capsulitis. J Shoulder Elbow Surg 20(3): 502-514.
- Jones DS, Chattopadhyay C (1999) Suprascapular nerve block for the treatment of frozen shoulder in primary care: a randomized trial. Br J Gen Pract 49(438): 39-41.
- Griggs SM, Ahn A, Green A (2000) Idiopathic adhesive capsulitis: a prospective functional outcome study of nonoperative treatment. J Bone Joint Surg Am 82(10): 1398-1407.
- Koh KH (2016) Corticosteroid injection for adhesive capsulitis in primary care: a systematic review of randomized clinical trials. Singapore Med J 57(12): 646-657.






























