Abstract
A 93-year-old female patient presented with acute-on-chronic right shoulder pain, accompanied by weakness and functional limitations. Multimodal imaging, including radiographs, computed tomography (CT), magnetic resonance imaging (MRI), and ultrasound, revealed extensive destruction of the glenohumeral joint, along with effusion containing calcific debris and rotator cuff tears, consistent with a diagnosis of Milwaukee Shoulder Syndrome (MSS). Previous imaging studies also indicated the presence of chondrocalcinosis, suggesting calcium pyrophosphate dihydrate crystal deposition disease (CPPD), thus reinforcing the diagnosis of MSS. However, it is crucial to note that aspiration of the synovial fluid from the glenohumeral joint yielded needle-shaped, negatively birefringent monosodium urate (MSU) crystals, thereby establishing a definitive diagnosis of gout complicating MSS.
Keywords:Milwaukee Shoulder syndrome; Rotator cuff tear; Calcium Pyrophosphate Dihydrate crystal deposition disease; Gout; Rheumatoid arthritis
Abbreviations:CPPD: Crystal Deposition Disease; MSS: Milwaukee Shoulder Syndrome; MSU: Monosodium Urate; CT: Computed Tomography; MRI: Magnetic Resonance Imaging; MSS: Milwaukee Shoulder Syndrome; CPPD: Calcium pyrophosphate dihydrate; RA: Rheumatoid Arthritis; DECT: Dual Energy Computed Tomography
Case Description
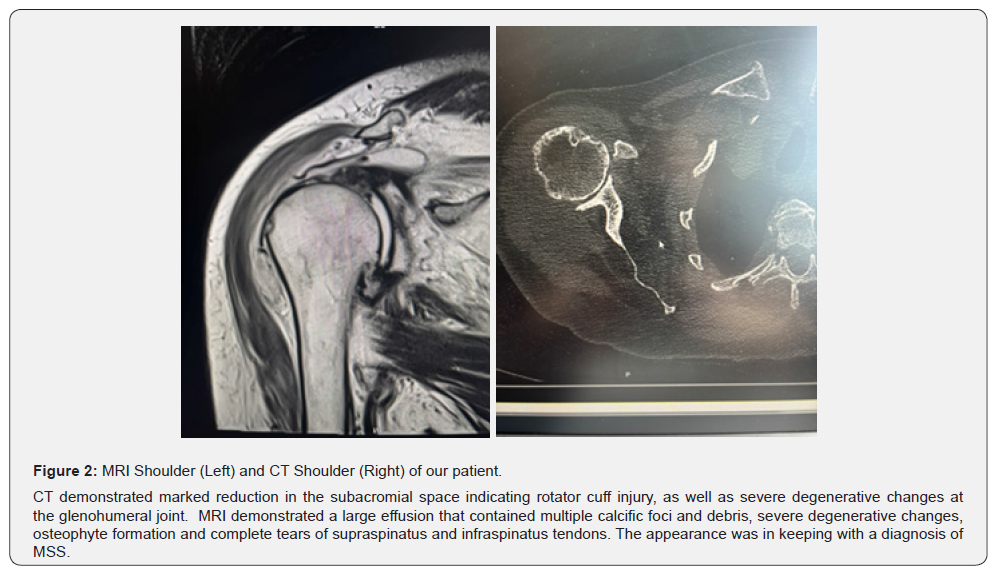
A 93-year-old woman with a history of “osteoarthritis “presented with acute-on-chronic right shoulder pain, significant weakness, and functional limitations in the absence of trauma. Examination revealed notable muscle waste and a large joint effusion. Multimodal imaging—including plain radiographs, CT, MRI, and ultrasonography-demonstrated extensive glenohumeral joint destruction, a large joint effusion with calcific debris, rotator cuff tears with tendon retraction, and intra-articular loose bodies, all consistent with Milwaukee Shoulder Syndrome (MSS) (Table 1).
Given the radiologic impression of MSS, a retrospective review of prior joint imaging revealed chondrocalcinosis of the wrists and knees. However, synovial fluid aspiration of the glenohumeral joint yielded needle-shaped, negatively birefringent monosodium urate (MSU) crystals, establishing a biochemical diagnosis of gout. This biochemical–radiologic discordance highlights a rare presentation of gout complicating MSS, in the absence of typical gouty peripheral joint involvement or tophaceous disease. The patient responded well to intra-articular corticosteroid therapy and was started on urate-lowering treatment, Allopurinol, in addition to a change in antihypertensive medication from valsartan to the uricosuric antihypertensive agent losartan (Figures 1-3).
Milwaukee Shoulder
Milwaukee Shoulder Syndrome (MSS) is an often under diagnosed destructive, crystalline arthropathy, primarily affecting females. Its associated classically with calcium hydroxyapatite crystals however, it can also arise from calcium pyrophosphate deposition or mixed crystal deposition and is increasingly appreciated as part of a spectrum of crystal induced arthropathies [1]. It may therefore either co-exist or be misdiagnosed as CPPD. A triad of chronic shoulder pain, massive rotator cuff tears, and noninflammatory joint effusions containing basic calcium phosphate crystals is characteristic of MSS. MSS was first described by McCarthy et al. in the 1980s in a series of elderly women from Milwaukee, Wisconsin. [2,3].

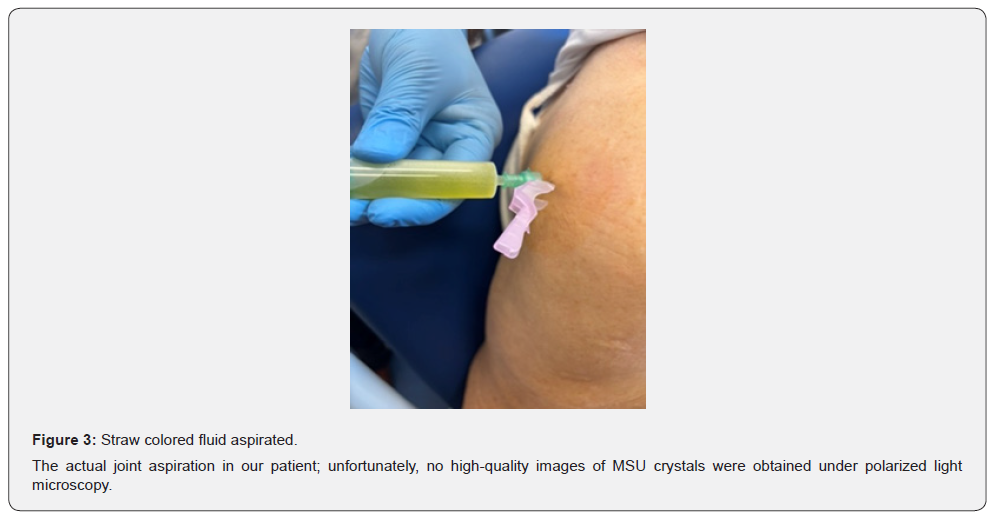
The inflammatory nature of the synovial fluid is consistent with Gout
Serum uric acid levels: 261 μmol/litre (Normal).
Subchondral bone and cartilage are rapidly destroyed in MSS, accompanied by subluxation of the humeral head and significant effusion. Complete rupture of the rotator cuff is common, resulting in severe functional impairment. Patients typically present with chronic shoulder pain, limited range of motion, and joint swelling. Unilateral shoulder involvement is far more common than bilateral, and the condition tends to affect the dominant side, even when there is bilateral involvement, the dominant side usually exhibits more advanced damage [4].
Calcium Pyrophosphate Dihydrate (CPPD) Crystal Deposition Disease
Calcium pyrophosphate dihydrate (CPPD) crystal deposition disease, commonly referred to as pseudogout, is a distinct yet related crystalline arthropathy characterized by the deposition of CPPD crystals in particular tissues. The disease is said to mimic other rheumatic disorders, hence the development of the wellknown term “pseudogout”. CPPD typically affects elderly patients and those with metabolic disorders. The inflammatory response in CPPD is usually episodic, but chronic CPPD arthropathy can mimic osteoarthritis, rheumatoid arthritis or, less commonly, destructive arthropathies like MSS [5,6]. CPPD typically presents as an acute arthritis that differs from gout mainly in the location of the affected joints. Similar to gout, CPPD can be monoarticular, oligoarticular, or polyarticular, but it most frequently impacts the knees and wrists.
In contrast to gout, CPPD less often involves the ankles, elbows, metacarpophalangeal joints, and rarely the shoulder [7]. Chronic CPPD typically presents in two main phenotypes: persistent inflammatory arthritis or recurrent acute flares [8]. Recurrent acute flares manifest identically to the acute phenotype of CPPD on a recurring basis, making them somewhat more recognizable. The persistent inflammatory phenotype, however, can be more diagnostically challenging, as it is often mistaken for other conditions. It may present as symmetrical polyarthritis involving the wrists and hands in elderly patients, frequently leading to a misdiagnosis of seronegative late-onset rheumatoid arthritis (RA) or polymyalgia rheumatica [9]. Unlike inflammatory arthropathies such as rheumatoid arthritis, MSS and CPPD typically exhibit minimal systemic symptoms and a non-inflammatory pattern. However, this may also lead clinicians to lean towards an impression of late-onset RA, as one might not expect such systemic symptoms in already damaged joints.
Diagnosis
Effusions in CPPD and MSS are often large and serosanguinous, a finding that can mislead initial diagnostic impressions, making both radiological imaging and synovial fluid analysis paramount to accurate diagnosis [10,11].
Radiological Features
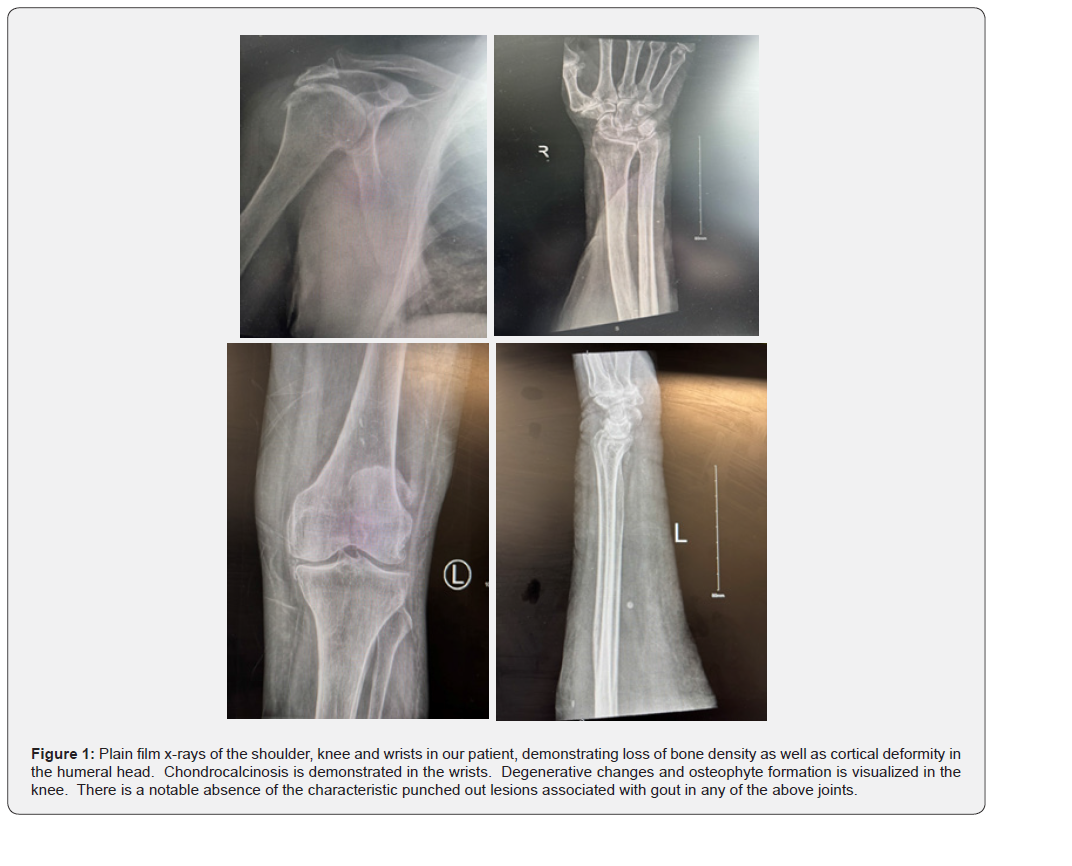
Radiological findings are pertinent in the diagnosis of MSS and CPPD. Plain radiographs generally serve as the first investigation and typically reveal subluxation of the humeral head, joint space narrowing, and intra-articular calcifications. There is usually a lack of osteophyte formation, which is a key differentiating factor from osteoarthritis. In CPPD, radiographs may also show chondrocalcinosis, most found in the fibrocartilage of the menisci, the pubic symphysis, or the wrist fibrocartilage [12], as demonstrated in early x-rays of the patient discussed above.
Chondrocalcinosis, defined as the presence of calcification in joint cartilage, was not previously considered specific to CPPD, as other crystals might also be present and causative of this finding. However, in recent years, radiographic definitions specific to CPPD have been proposed [13,14]. These definitions were validated against histological samples from the knees, and exhibited a specificity of 92% [15], highlighting the utility of plain film in accurate diagnosis of CPPD. CT and MRI are used to further assess bony destruction, characterize the extent of the rotator cuff tear, and detect intra-articular calcific material or capsular distension.
MRI is beneficial for demonstrating massive rotator cuff tears and joint effusions T2-weighted images often reveal high signal intensity in the glenohumeral joint, indicative of synovial fluid, with possible low signal areas corresponding to calcific deposits [16]. Ultrasound is a safe, readily available, and particularly valuable imaging modality for investigating CPPD. It has proven to be accurate for CPPD diagnosis compared to histological sampling, with a sensitivity of up to 87% and a specificity of up to 88%, varying depending on the joint in question [17]. Furthermore, ultrasound offers the benefit of distinguishing between CPPD and other crystals, although it has significant limitations, primarily concerning the need for trained sonographers and the limited acoustic windows of certain joints [6,18].
More advanced and less commonly available imaging modalities, such as Dual Energy Computed Tomography (DECT), can provide information on the composition of visualised joint deposits based on their respective dual energy indices, and this has sparked substantial interest in recent years [19]. However, thus far, its ability to identify the early stages of calcium deposition within the joints has been unimpressive, primarily due to potential artefacts and low spatial resolution. This means that crystal deposition is likely identifiable on conventional CT as chondrocalcinosis by the time it becomes apparent on DECT [20].
Synovial Fluid Analysis
Synovial fluid analysis is a crucial aspect of all crystalline arthropathies. The fluid is typically serosanguinous in MSS and CPPD with a low leukocyte count (<2,000 cells/mm³), reflecting the non-inflammatory nature of MSS.CPPD ranges from mild to severe inflammatory nature, not limited to a single joint like MSS, which is primarily affects the shoulder. Gouts are inflammatory and can be mono-articular, oligo-articular and polyarticular. Analysis of synovial fluid involves the use of both plain light and polarised light microscopy. If CPPD is present, microscopic analysis will reveal weakly positive birefringent rhomboid crystals identifiable under polarized light. In MSS the crystals are basic calcium hydroxyapaitite,often clumped of found in globules and are non-birefringent, appearing dark under light microscopy.
The crystals are typically tiny in size in both CPPD and MSS thus difficult to visualise, leading to a relatively high false negativity rate and significant inconsistency between observers [21,22]. Alizarin red stain can be used to help visualize both Calcium Hydroxyapatite and Calcium Pyrophosphate Dihydrate phosphate crystals found in MSS and CPPD respectively. In our case, the synovial fluid analysis revealed a diagnostic discordance in our patient’s investigations, when microscopy yielded needleshaped, negatively birefringent monosodium urate (MSU) crystals, furthermore the synovial fluid was inflammatory in nature establishing a diagnosis of gout, complicating MSS. Alizarin red stain, was requested for but was not available in our laboratory to ascertain if there were BCP/CPPD crystals missed by the observer during polarized light microscopy.
Gout in the Shoulder – A Rare Presentation
Gout is characterized by the deposition of monosodium urate crystals in joints and connective tissue. It is the most prevalent form of inflammatory arthritis and is well known to affect the feet, hands, knees, and elbows, typically [23]. The condition seldom affects the shoulder joint, and the bone tissue of the shoulder joint is rarely exposed to the characteristic osteolytic damage that gout imposes [24]. Radiological features of gout are well described, particularly on plain film x-rays, and classically involve the preservation of joint space, soft tissue nodules that are occasionally calcified, punched-out erosions with overhanging cortex and sclerotic edges [25]. T1 images on MRI may show homogeneous intermediate signal intensity, depending on the calcium concentration, with T2-weighted images displaying heterogeneous intermediate to low signal intensity.
Although radiological evidence is often a late manifestation of the condition, the diagnosis is usually made clinically and biochemically [25]. Reported cases of shoulder involvement are relatively uncommon, with one recent report describing a patient with nonspecific symptoms of unilateral shoulder pain. Radiological imaging of the patient was completely normal; however, arthrocentesis yielded yellow, needle-shaped monosodium urate crystals under polarized light microscopy [26], allowing for a diagnosis of gout and subsequent effective treatment with colchicine and allopurinol. Another case described a patient with MRI findings of supraspinatus tendonitis, but no other notable radiological findings were noted. The patient proceeded to arthroscopy, and the supraspinatus and subscapularis tendons were found to have tophaceous deposits, leading to a diagnosis of subacromial impingement secondary to tophaceous rotator cuff gout [27].
Another reported case of tophaceous gout involving the rotator cuff was described, which demonstrated some subtle radiological clues. In the case of rotator cuff tophaceous gout, a plain radiograph of the shoulder exhibited a small amorphous opacity above the head of the humerus, and urate crystal deposits were subsequently discovered by MRI and on the supraspinatus tendon’s articular surface [28]. These individual case examples highlight not only the rarity of shoulder involvement in gout but also the diagnostic challenges associated with this type of presentation. While shoulder gout is uncommon, clinicians need to consider its possibility when evaluating a painful arthritic shoulder to prevent the condition from going unnoticed and untreated.
Treatment
Treatment for CPPD focuses on treating the associated pain and inflammation, as no treatments have been found effective in dissolving the crystals to remove the underlying cause [6,29]. There are currently no randomized control trials analyzing treatment options for MSS. Management is primarily supportive, especially in advanced cases, and involves repeated aspiration of the effusion, NSAIDs for symptomatic relief and intra-articular corticosteroid injection [30]. Physiotherapy is central to the management of the MSS, to maintain residual range of motion and strengthen the surrounding musculature; however, this is often limited by pain and mechanical restriction.
In advanced cases and the absence of contraindications, surgical intervention can be considered, such as partial or complete shoulder arthroplasty, although outcomes are often suboptimal due to rotator cuff insufficiency. In cases with CPPD, colchicine is typically used for acute flares, and surgical intervention is generally reserved for refractory cases or those with severe functional impairment. The management of gout is divided into treating acute flares of the disease and preventing further flares. In the acute phase, treatment is typically initiated with one or more potent anti-inflammatory agents, such as NSAIDs, colchicine, or corticosteroids.
Despite common relative contraindications to these drugs, many patients are often treated out of necessity with medications that can have potentially harmful effects, due to the severity of the pain from an acute flare [31]. The treatment of established gout aims to prevent flares and focuses on urate-lowering therapy by xanthine oxidase inhibitors such as allopurinol or febuxostat [31,32]. This is based on the understanding that the primary problem in gout is hyperuricemia, which leads to crystal formation. The higher the serum urate level, the rapidly flares return. Patients who require urate-lowering therapy generally need to remain on some level of the treatment indefinitely; however, with adequate medication adherence and correct management, gout can be brought under effective control [31].
Summary and Clinical Learning Points
This case illustrates a rare presentation of gout affecting the shoulder joint and complicating Milwaukee Shoulder Syndrome. The diagnostic discordance between imaging findings suggestive of MSS and the identification of monosodium urate crystals in synovial fluid underscores the importance of joint aspiration and subsequent analysis of fluid in all cases. In addition, where discordance exist, Alizarin red stain should be used. Clinicians should remain alert to the possibility of gout as an underlying cause of destructive shoulder arthropathy, even in elderly patients without prior history of crystal arthritis or classic peripheral joint involvement. Early recognition and targeted therapy can significantly improve outcomes and prevent unnecessary diagnostic delays.
Conclusion
This case highlights a rare presentation of gout complicating MSS, in the absence of typical peripheral joint involvement or tophaceous disease. This diagnostic discordance emphasizes the critical importance of synovial fluid analysis in all cases of destructive shoulder arthropathy. The patient demonstrated a favorable response to intra-articular corticosteroid therapy and commenced urate-lowering treatment with Allopurinol, in addition to a change in antihypertensive medication from valsartan to the uricosuric antihypertensive agent losartan.
References
- Dieppe PA, Doyle DV, Huskisson EC, Willoughby DA, Crocker PR (1978) Mixed crystal deposition disease and osteoarthritis. Br Med J 1(6106): 150.
- Genta MS, Gabay C (2006) Milwaukee Shoulder. N Engl J Med 354(2): e2.
- Garancis JC, Cheung HS, Halverson PB, McCarty DJ (1981) “Milwaukee shoulder” --association of microspheroids containing hydroxyapatite crystals, active collagenase, ad neutral protease with rotator cuff defects. III. Morphologic and biochemical studies of an excised synovium showing chondromatosis. Arthritis Rheum 24(3): 484-491.
- Nguyen VD (1996) Rapid destructive arthritis of the shoulder. Skeletal Radiol 25(2): 107-112.
- Rosenthal AK, Ryan LM (2016) Calcium Pyrophosphate Deposition Disease. N Engl J Med 374(26): 2575-2584.
- Pascart T, Filippou G, Lioté F, Sirotti S, Jauffret C, Abhishek A (2024) Calcium pyrophosphate deposition disease. Lancet Rheumatol 6(11): e791-804.
- Abhishek A, Tedeschi SK, Pascart T, Latourte A, Dalbeth N, et al. (2023) The 2023 ACR/EULAR classification criteria for calcium pyrophosphate deposition disease. Ann Rheum Dis 82(10): 1248-1257.
- Damart J, Filippou G, Andrès M, Cipolletta E, Sirotti S, Carboni D, et al. (2024) Retention, safety and efficacy of off-label conventional treatments and biologics for chronic calcium pyrophosphate crystal inflammatory arthritis. Rheumatol Oxf Engl 63(2): 446-455.
- Paalanen K, Rannio K, Rannio T, Asikainen J, Hannonen P, et al. (2020) Prevalence of calcium pyrophosphate deposition disease in a cohort of patients diagnosed with seronegative rheumatoid arthritis. Clin Exp Rheumatol 38(1): 99-106.
- Nadarajah CV, Weichert I (2014) Milwaukee Shoulder Syndrome. Case Rep Rheumatol 2014: 458708.
- Becker JA, Daily JP, Pohlgeers KM (2016) Acute Monoarthritis: Diagnosis in Adults. Am Fam Physician 94(10): 810-816.
- Abhishek A, Doherty M (2016) Update on calcium pyrophosphate deposition. Clin Exp Rheumatol 34(4 Suppl 98): 32-38.
- Zhang W, Doherty M, Bardin T, Barskova V, Guerne PA, et al. (2011) European League Against Rheumatism recommendations for calcium pyrophosphate deposition. Part I: terminology and diagnosis. Ann Rheum Dis 70(4): 563-570.
- Tedeschi SK, Becce F, Pascart T, Guermazi A, Budzik JF, et al. (2023) Imaging Features of Calcium Pyrophosphate Deposition Disease: Consensus Definitions From an International Multidisciplinary Working Group. Arthritis Care Res 75(4): 825-834.
- Sirotti S, Becce F, Sconfienza LM, Terslev L, Naredo E, et al. (2023) Reliability and Diagnostic Accuracy of Radiography for the Diagnosis of Calcium Pyrophosphate Deposition: Performance of the Novel Definitions Developed by an International Multidisciplinary Working Group. Arthritis Rheumatol Hoboken NJ 75(4): 630-638.
- Llauger J, Palmer J, Rosón N, Bagué S, Camins À, Cremades R (2000) Nonseptic Monoarthritis: Imaging Features with Clinical and Histopathologic Correlation. RadioGraphics 20(suppl_1): S263-S278.
- Filippou G, Scanu A, Adinolfi A, Toscano C, Gambera D, et al. (2021) Criterion validity of ultrasound in the identification of calcium pyrophosphate crystal deposits at the knee: an OMERACT ultrasound study. Ann Rheum Dis 80(2): 261-267.
- Cipolletta E, Abhishek A, Matteo AD, Grassi W, Filippucci E (2023) Dynamic assessment of the double contour sign by ultrasonography helps to distinguish between gout and calcium pyrophosphate deposition disease. RMD Open 9(1): e002940.
- Pascart T, Norberciak L, Legrand J, Becce F, Budzik JF (2019) Dual-energy computed tomography in calcium pyrophosphate deposition: initial clinical experience. Osteoarthritis Cartilage 27(9): 1309-1314.
- Budzik JF, Marzin C, Legrand J, Norberciak L, Becce F, et al. (2021) Can Dual-Energy Computed Tomography Be Used to Identify Early Calcium Crystal Deposition in the Knees of Patients with Calcium Pyrophosphate Deposition? Arthritis Rheumatol Hoboken NJ 73(4): 687-692.
- Schumacher HR, Sieck MS, Rothfuss S, Clayburne GM, Baumgarten DF, et al. (1986) Reproducibility of synovial fluid analyses. A study among four laboratories. Arthritis Rheum 29(6): 770-774.
- Berendsen D, Neogi T, Taylor WJ, Dalbeth N, Jansen TL (2017) Crystal identification of synovial fluid aspiration by polarized light microscopy. An online test suggesting that our traditional rheumatologic competence needs renewed attention and training. Clin Rheumatol 36(3): 641-647.
- Punzi L, Scanu A, Galozzi P, Luisetto R, Spinella P, et al. (2020) One year in review 2020: gout. Clin Exp Rheumatol 38(5): 807-821.
- Shrateh ON, Jobran AWM, Zaid MA, Hamed HK, Asees MY, et al. (2023) Unusual location of gouty arthritis with shoulder joint involvement in an elderly male patient: a rare case report. Ann Med Surg 85(4): 1158-1161.
- Gentili A (2006) The advanced imaging of gouty tophi. Curr Rheumatol Rep 8(3): 231-235.
- Shrateh ON, Jobran AWM, Zaid MA, Hamed HK, Asees MY, et al. (2023) Unusual location of gouty arthritis with shoulder joint involvement in an elderly male patient: a rare case report. Ann Med Surg 85(4): 1158-1161.
- O’leary ST, Goldberg JA, Walsh WR (2003) Tophaceous gout of the rotator cuff: a case report. J Shoulder Elbow Surg 12(2): 200-201.
- Chang CH, Lu CH, Yu CW, Wu MZ, Hsu CY, Shih TTF (2008) Tophaceous gout of the rotator cuff. A case report. J Bone Joint Surg Am 90(1): 178-182.
- Parperis K, Papachristodoulou E, Kakoullis L, Rosenthal AK (2021) Management of calcium pyrophosphate crystal deposition disease: A systematic review. Semin Arthritis Rheum 51(1): 84-94.
- Zhang W, Doherty M, Pascual E, Barskova V, Guerne PA, et al. (2011) EULAR recommendations for calcium pyrophosphate deposition. Part II: management. Ann Rheum Dis 70(4): 571-575.
- Pillinger MH, Mandell BF (2020) Therapeutic approaches in the treatment of gout. Semin Arthritis Rheum 50(3, Supplement): S24-30.
- Richette P, Doherty M, Pascual E, Barskova V, Becce F, Castañeda-Sanabria J, et al. (2017) 2016 updated EULAR evidence-based recommendations for the management of gout. Ann Rheum Dis 76(1): 29-42.






























