Abstract
The presence of air inside the cranial cavity is called pneumocephalus (PNC). It is commonly caused by trauma but can also be a result of surgical procedures and other factors. This systematic review aims to discuss the case reports on PNC after spinal surgery, published in PubMed/Medline, Embase, Scopus, and Web of Science up to July 2023. Built-up precise audit strategies were utilized for screening and information extraction. Of 428 studies, thirty-six reports from 14 countries, comprising 37 patients, were included. We found out the most common symptoms in patients with PNC, in order of prevalence, were headache (up to 59% of patients), nausea, dizziness, and seizures. In 62% of the cases, a dural tear was clearly quoted. Additionally, in the studies that reported the time interval between surgery and clinical presentation of PNC, a large majority of patients displayed PNC after 24 hours (55%). The most used diagnostic method and treatment modality in our studies were computerized tomography (CT) scan (81%) and conservative treatment (70%), respectively. We propose that every patient who underwent spinal surgery, when they complained of a newly onset headache, especially after 24 hours of the operation, a brain CT scan to rule out the presence of PNC is beneficial
Keywords:Pneumocephalus; Spinal Surgery; PNC; Systematic Review
Introduction
Pneumocephalus (PNC) is a rare condition characterized by the accumulation of gas or air in the cranial cavity. It is typically caused by specific factors such as head trauma or cranial surgeries [1-3]. Other common causes include intracranial neoplasms, infections, neurosurgical or otolaryngological procedures, and diagnostic interventions [2,4,5]. Several factors, including anesthesia type, head position, surgery duration, and the presence of hydrocephalus, can influence the development of PNC [6,7]. While dural injury and invasive spinal procedures can contribute to PNC, they are less frequently observed in such cases [1,7]. Although PNC after spinal surgery is a rare phenomenon, if it occurs, it can cause multiple complications, including difficulty in awakening from anesthesia, the occurrence of tonic-clonic seizures, or even the induction of status epilepticus, possibly due to the significant amount of gas inside the cranium [8]. PNC is also regarded by its severity as simple and tension, ensuring proper treatment [6,7].
Simple PNC typically resolves spontaneously without significant complications, whereas tension PNC requires immediate medical attention due to symptoms of increased intracranial pressure [6,7,9]. Usually, there are three methods for the diagnosis of PNC. Although plain skull radiography can be used, it is generally considered unreliable in comparison with Computed Tomography (CT) scanning and Magnetic Resonance Imaging (MRI) [5,10,11]. From the therapeutic point of view, most cases of PNC are treated conservatively with bed rest, raising the head of the bed by 30, oxygen therapy, antipyretics, painkillers, and osmotic diuretics. Tension PNC is an exception and considered a neurosurgical emergency that usually needs immediate intracranial decompression through a variety of surgical interventions [5,10]. There is currently no consensus among experts on evidence-based guidelines for managing PNC in neurosurgery. Given the lack of systematic reviews specifically focusing on postoperative PNC after spinal surgery, conducting such a review would be highly valuable. It would provide a more accurate understanding of the complications and disadvantages associated with PNC after spine surgery and assess the quality of existing reports. This information could greatly assist in making clinical decisions when faced with such incidents. While large cohort studies and randomized controlled trials offer important statistical insights, they often fail to capture the in-depth clinical details needed to understand rare or atypical symptoms. In contrast, case reports and case series provide comprehensive clinical observations. Given the rarity of this condition, extensive studies have not been conducted on this topic. This systematic review evaluated PNC after spine surgeries (in terms of presentation, diagnosis, and treatment) to provide an appropriate guideline for the correct management of this rare occurrence.
Materials and Methods
This systematic review was reported following the guidelines and checklist of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [12] and registered in the International Prospective Register of Systematic Reviews; PROSPERO (CRD42023467213, https://www.crd.york.ac.uk/PROSPERO/). Search strategy: We searched the databases of PubMed, Embase, Scopus, and Web of Science up to July, 2023 using the following Mesh keywords which were combined first with “OR” then by “AND”: “Spine Surgery,” “Laminectomy,” “Spinal Decompression,” “Discectomy”, “Kyphoplasty”, “Vertebroplasty”, “Foraminotomy”, “Spinal Fusion”, “Herniated Disc Surgery”, “Spinal Operation”, “Scoliosis Surgery”, and “Pneumocephalus”, “Pneumatocele”, “Intracranial Aerocele”, and “Pneumoventricle”. Studies were considered eligible for inclusion in this systematic review if they had the following PICOS conditions: Population (P) and Intervention (I) for the patients undergoing spinal surgery, Comparison (C) for comparing the simple with the tension PNC, Outcomes (O) of the event, and Study design (S) stands for only case series and case reports. We excluded those studies that failed to report the outcome, had non-English language abstracts, and were unavailability in full text (Figure 1). Study Selection: In the initial step, any duplicate entries were eliminated. Subsequently, two reviewers (S.A.G. and A.O.K.) independently inspected references in two consecutive phases. At the first stage, reviewers appraised titles and abstracts and apart those that did not meet inclusion criteria or were not accessible but kept abstracts with inadequate information for full-text evaluation. Then, reviewers independently evaluated full-text articles and omitted those not meeting eligibility criteria or those that were inaccessible. Any incongruities between two independent reviewers were resolved by debate with a senior reviewer (F.O.K).
Risk of Bias Assessment
Bias in case report studies is unavoidable. In any case, standardized tools have been created to evaluate their methodological quality in systematic reviews. Subsequently, two Authors (Mah.S., Mas.S.) independently evaluated study quality employing a standard tool provided by the Joanna Briggs Institute (JBI) [13]. The JBI risk-of-bias assessment tool for case reports consists of eight questions that assess the presence or absence of various reported items. The possible answers to these items include yes, no, unclear, and not applicable. If each item was clearly depicted, it was evaluated as “Yes,” “No” if it was not presented, “Unclear” if it was not clear, and “Not applicable” in case it did not seem to be applied. The result of the risk of bias assessment using the JBI checklist was reported as a checklist instead of a cumulative score.
Data extraction
Two authors (Mah.S. and A.O.K.) independently extricated the following data: first author, publication date, sample size, patients’ characteristics (age and sex), surgery, sign and symptom, type of PNC, time until developing PNC, treatment, outcome, and design of each study. A third author (F.O.K.) interceded when the two authors opposed.
Aims & Objective
i. To decrease spasticity and prevent contractures and
deformities from occurring.
ii. The aim is to enhance gross motor function to achieve
maximum independence.
iii. To enhance the functional capacities of the children to
make them self-dependent.
iv. To improve the quality of life of children affected with
Spastic Cerebral Palsy.
v. To evaluate the therapeutic effect of Kalyanaka Avaleha
in the management of spastic cerebral palsy in children.
vi. To evaluate the therapeutic effect of Karma Basti in the
management of spastic cerebral palsy in children.
Iec Registration No
• After receiving institutional ethics committee approval
vide letter no DSRRAU/UCA/IEC/19-20/310, this study was
initiated.
• The study was also registered in CTRI with registration
number CTRI /2021/07/034864.
Study Design
1. Study Type: Interventional
2. Open labelled Study
3. Interventional Groups: Single Arm
4. Purpose: Treatment
5. Timing: 90 days
6. End Point: Efficacy
Material and Methods
Selection of cases
i. Source of patients: Children with cerebral palsy were
chosen from the outpatient and inpatient departments (OPD &
IPD) of the Bal Roga Department of the attached Ayurved Hospital
of the University Postgraduate Institute of Ayurved Studies and
Research, Dr. S. R. Rajasthan Ayurved University Jodhpur.
ii. Number of Patients: 35 patients were registered for the
present study but 5 patients were dropped out during the study.
Therefore, the present study was completed on 30 patients.
iii. Age of patients: Children suffering from spastic cerebral
palsy were of age groups one to sixteen years of either sex.
iv. Follow-up: Three monitoring times were done at
monthly intervals during the study and follow-up was done after
one month of completion of the study.
Diagnostic Criteria Inclusion Criteria
• Children of age groups one to sixteen years of either sex
were included which were stratified as 1-5 years, 5-10 years, and
above 10 years.
• Diagnosed cases of Spastic Cerebral palsy were
selected.
Exclusion Criteria
• Children below 1 year and above 16 years of age were
excluded from study.
• Children suffering from any progressive neurologic
disorders and any serious illness like meningitis, or encephalitis
were excluded from the study.
• Children suffering from various disorders like
myopathies, neuropathies, juvenile diabetes mellitus, essential
hypertension, and any acute respiratory distress were excluded
from the study.
Withdrawal Criteria
Patients who develop life-threatening complications during treatment.
Parents/guardians are not willing to continue treatment.
Untoward Evaluation Criteria
To record adverse reactions an untoward reaction assessment form was developed, and all such adverse reactions were recorded.
Protocol of Research
I. Consent of patient/attendant after making him/
her aware of merits/demerits of trial with the duration of the
proposed trial.
II. Fulfillment of inclusion criteria.
III. Registration of the patients.
IV. The investigations mentioned were advised to them
before presenting the formulation.
V. The data obtained was clinically deducted and
statistically analyzed.
Drugs
For the present study, two drugs were selected. Both drugs were Kalyanaka Avaleha (Table 1) & Prasarni Taila (Table 2) which are described under the chapter of Vata Vyadhi in Bhasijhaya Ratnavalli and Yogratanakar respectively. Preparation of Drug - Haridra, Vacha, Kustha, Pippali, Shunthi, Ajaji, Ajmoda, Yasthimadhu, and Saindhava lavana taken all the ingredients and grind these materials into a fine powder together and administered regularly with Goghrita or honey. Prepared Churna was packed in a sterile polybag of capacity 100 gm and labelled with the date of manufacturing, batch no., and drug license number.


Dose & Duration - Kalyanaka Avaleha was given in the form of Churna in a dose of 40 mg/kg/day in two divided doses as per Young Formula. (Adult dose was considered as 3 grams).
Preparation of Drug
The whole plant of Prasarni collected and coarsely powdered should be placed in a vessel and decocted by adding water (till one-fourth is left) to this, curd, amla kanji in double quantity, and Kalka (paste) made from the following should be added, Tila Taila and subjected to Taila paka procedure to obtain Prasarni Taila (the Kalka Dravyas are- Manjistha, Haridra, Triphala, and Nagarmotha). This Prasarni Taila is highly useful as Nasya and for application. It relieves Ekanga Vata, Apasmara, Unmada, Vidradhi, Mandagni, Tvak-Vata, Sira, Sandhivata, and other Vata Roga. It is also indicated in veterinary practice. It imparts strength and, virility, useful for Vrddha, Bala, Stree, and kings. It is especially indicated in ‘Pangu’ for internal use.
Presentation of Drugs: Prepared oil was packed in a sterile bottle of capacity 100 ml and labeled with the date of manufacturing, batch no., and drug license number.
Aasthapana Basti: The dose of Basti Dravya was calculated using the (Kashyap Samhita/Khilasthan/Bastivisesaniya/109), as shown in the table below. For Aasthapana Basti, doses are mentioned in the given table.
Anuvasana Basti: The dose of Basti Dravya is calculated using the (Kashyap Samhita/khilasthan/Bastivisesaniya/109), as shown in the table below. For Anuvasana Basti, these dosages are mentioned.
Assessment Criteria
• Gross motor function Classification Scale (GMFCS)- to
study gross motor function.
• Modified Ashworth scale- to measure the change in
spasticity.
• Modified Barthel’s Scale of Advance daily living- to see
changes in the Quality of life of Spastic cerebral palsy.
Clinical Study
30 patients satisfying the selection criteria were registered in the study, after following the standard informed consent procedure. Out of these two patients dropped out due to personal family problems and three patients due to acute respiratory distress and suspected pneumonia-like conditions.
Observations
The detailed observations, regarding demographic data, socio-economic status, parental history, family history, personal history, and complete history of the patient regarding the disease were collected as shown in given table below.
Result
Study selection
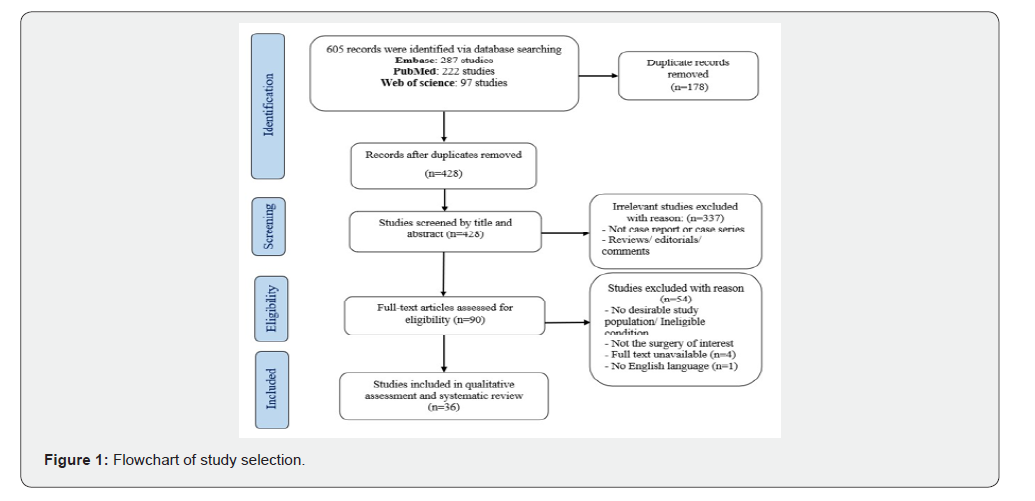
A total of 605 potentially related references were selected from the three electronic databases. Out of these, 178 were duplicates, and 337 articles were deemed irrelevant through a review of their titles and abstracts. After this initial screening, 90 articles underwent a full-text review for eligibility. Among these, 54 articles were further excluded. The main reasons for exclusion were (I) no desirable study population/ineligible condition, (II) lack of full-text availability, (III) non-English language, and (IV) the treatment method was not mentioned precisely. Finally, a total of 36 papers published between 2006 and 2023 met the eligibility conditions and were counted in the qualitative assessment and systematic review (Figure 1).
Risk of Bias Assessment
The critical appraisal checklist indicates that risk of bias or methodological quality is generally acceptable (Table 1) in most case reports. Although we do not set criteria for low risk of bias for a study based on cumulative scores, risk of bias assessments is reported for each study and separately for each question. Using JBI tool, a total of 18 (50%) cases did not clearly describe the patient’s demographic characteristics; the history of 5 patients (13.6%) was not clearly described and presented as a timeline, and 20 (55%) patients were not clearly described at the time of presentation, and finally explanation of treatment procedure(s) was not clearly reported in only one patient (2.7%). Study characteristics and case demographics: Thirty-six eligible studies reported a total of 37 cases. The retrieved case reports were published between 2006 and 2023, of which almost 70% were published in the last 10 years. Cases were from 14 countries: Turkey (n=8), India (n=6), USA (n=5), South Korea (n=3), Spain (n=3), Taiwan (n=3), France (n=2), and single case report from Greece, Poland, UK, Tunisia, Belgium, Jordan, and Japan (Table 2). Most reported cases were male (24 [65%] of 37), and the median age of study patients was 59±6 years (ranged 2 to 79). Details of bibliographic features of the regained studies, along with the demographic descriptions of the reported cases, are presented in Table 2.

Y, Yes; U, Unclear; N/A, Not applicable
Q1. Were the patient’s demographic characteristics clearly described?
Q2. Was the patient’s history clearly described and presented as a timeline?
Q3. Was the current clinical condition of the patient on presentation clearly described?
Q4. Were diagnostic tests or assessment methods used, and the results clearly described?
Q5. Were the intervention(s) or treatment procedure(s) clearly described?
Q6. Was the post-intervention clinical condition clearly described?
Q7. Were adverse events (harms) or unanticipated events identified and described?
Q8. Does the case report provide takeaway lessons?

Clinical Characteristics of the Reported Patients
Time until developing PNC (day)
The maximum and minimum time intervals between surgery and diagnosis of PNC in the studied patients were 186 and 6 hours, respectively. This time interval was not reported for 8 patients [8,14-19]. The diagnosis of PNC was primarily based on Computed Tomography (CT) scans in 34 (91%) cases. Most of the reported cases were diagnosed within 1 to 4 days (24-96 hours). Among 29-time interval-reported cases, 13 (45%) cases were diagnosed within 1 day, seven (24%) cases were diagnosed on the 2nd day, and five (17%) cases were diagnosed on the 3rd and 4th day. In four patients (14%), it took more than 4 days to diagnose PNC (Table 3).
Signs and Symptoms
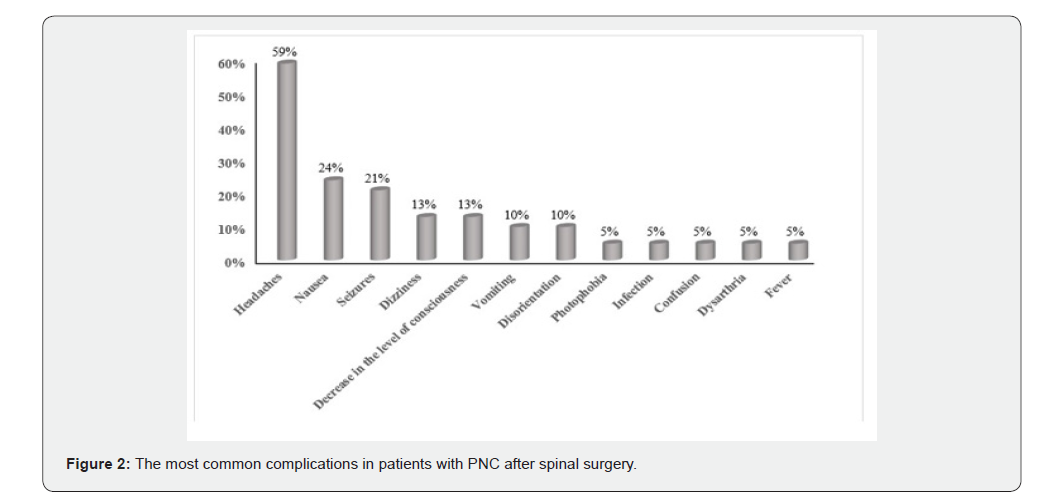
The most common complications reported are presented in Figure 2. Headaches were the most common complaint reported by the patients (22 of 37 patients; 59%). Other complaints were separately depicted in Figure 2.

Treatment and Outcome
Of the 37 cases, 26 (70%) underwent conservative treatment and 10 (27%) required surgery. Out of the total 37 patients, 32 (86%) experienced good results after treatment, while 5 (13%) patients had poor results. Among the poor outcomes, three cases were observed after conservative treatment, and two cases occurred after surgery.
Dural tear
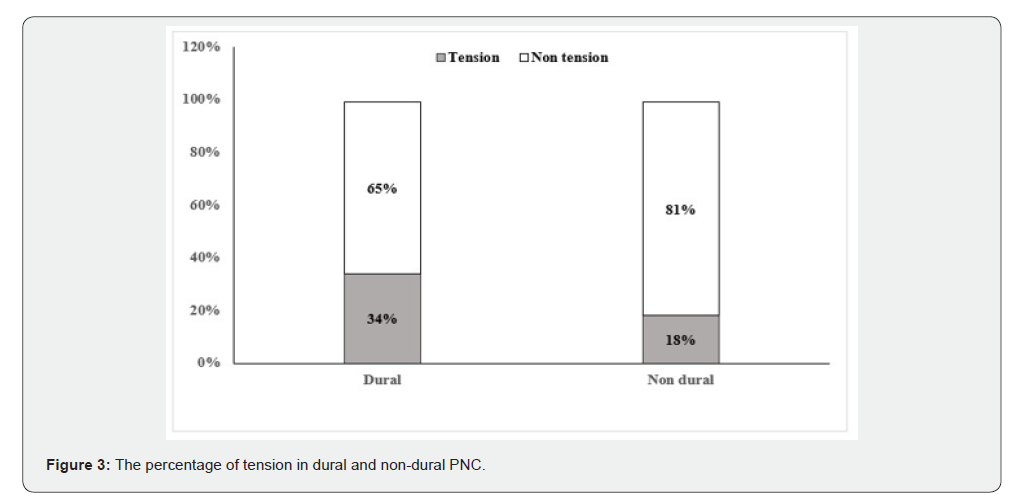
There were 23 (62%) reported cases of spinal surgery associated with dural tears which 6 (26%) were classified as tension PNC (Figure 3). Among the 23 cases, 8 (34%) underwent surgery for treatment, and out of these 8 cases, 6 (75%) showed good results after surgery.
Method of confirmation
Of the 37 cases, 30 (81%) underwent CT scan for confirmation, and 2 (5.4%) with MRI. Out of the total 37 patients, 5 (13.5%) underwent both a CT scan and an MRI for confirmation.
Discussion
Pathological accumulation of gas within the cranial space is commonly called PNC [41]. PNC generally has various causes, including trauma, infections by gas-forming bacteria, tumors of the brain or skull-based, cranial operations, along with iatrogenic and idiopathic causes [42-44]. PNC during spinal surgeries is rarely reported [5,29,32]. PNC can be allocated based on the time of intracranial air buildup into acute (<72 h) and delayed (>72 h) [45]. We systematically reviewed all the case reports that occurred after various types of spinal operations, summarized, explained, and compared them from different aspects, including age, sex, type of surgery, clinical symptoms, diagnostic technique, management, and prognosis. Although incidental dural tears, CSF leakage, and in fact any cranial surgery can lead to iatrogenic PNC, spinal operation- induced PNC is seldom reported [20,24,25,27-29]. These cases may have different presentations, including headache, nausea, vomiting, dizziness, altered consciousness or mental status, CSF rhinorrhea, or convulsions [43]. In the present review, 59% of cases complained of headaches. In an observational study on 20 patients with spinal intradural extramedullary (IDEM) tumors who underwent surgical resection, findings showed that the occurrence of PNC was common (90% of patients) after spinal surgeries associated with durotomy, and the reported rate of significant headache was 35% (seven of the 20 patients) [46]. Twenty-five percent of those patients had severe PNC, and the severity of the headache and PNC was positively correlated. Multiple studies have shown that PNC caused by extreme CSF loss usually leads to diminished consciousness, convulsions, and intracranial hematoma [28,47]. Due to the low density of air (about -1000 Hounsfield Units) on CT scans, this modality has become the most reliable imaging modality and highly sensitive technique for the diagnosis of PNC and can detect even 0.55 ml of air inside the skull, and this feature makes CT more accurate than MRI [48,20]. CT scan for diagnosis of PNC had been used for 34 patients of all the cases (92%) in the included reports. In most cases, PNC can be non-operatively treated with bed rest, elevating the head of the bed by 30 degrees, oxygen auxiliary therapy, and numerous other similar approaches [49,50]. Tension PNC is a neurosurgical emergency that mostly needs immediate intracranial decompression by a variety of operative techniques, such as twist-drill trephination or craniotomy [51,52]. In our review, 26 (70%) cases out of the 37 cases underwent conservative treatment, while 10 (27%) required surgery (craniotomy or reoperative spinal surgery and dura repair).
Many hypotheses were proposed to explain the pathophysiology of PNC. Initially, the inverted bottle theory was suggested, which refers to the idea of negative pressure development in the subarachnoid space due to CSF drip, leading to CSF substitution by air until equalization of the intracranial and atmospheric pressures [53]. Then, the ball valve theory was suggested. This theory claims that PNC is credited to a dural defect playing as a oneway valve, leading to air imprisonment in the intracranial cavity [54]. The third theory was the Nitrous Oxide (N2O) theory; N2O is frequently accompanied by anesthesia, leading to tension PNC [55,56]. Another hypothesis indicates that PNC can be developed by gas-forming bacteria [57,58]. In our review, there were 23 (62%) reported cases of spinal surgery associated with dural tears. Out of the 23 cases, 12 (52%) patients were classified as tension PNC. The remaining 14 (38%) reported cases of spinal surgery were not associated with any dural tears. Our study has some important limitations. Firstly, it relies exclusively on case reports and case series, which lack the statistical strength and broad applicability of cohort studies or randomized controlled trials. Secondly, there is a potential for publication bias, as reports tend to focus on severe or atypical cases, while milder or self-resolving instances may be underrepresented. Thirdly, as this condition is essentially a neurosurgical complication, it would have been much better if we had included an academic neurosurgeon among the authors and added her/him to the group of authors. Lastly, the absence of a control group limits the ability to directly compare different treatment approaches. Future studies with larger sample sizes and robust strategies are necessary to better define the signs, outcomes, and optimal treatment strategies for PNC following spinal surgery.
Conclusion
PNC could occur rarely after spinal surgery. The present report is the first systematic review on the signs and symptoms, diagnosis, treatment, and outcome of PNC after spine surgery. Spine surgeons should be aware of this rare complication after spinal surgeries and should not neglect to perform a brain CT scan in cases where unusual symptoms such as headache occur 24 hours after spinal surgery.
Declaration
Conflict of Interest
No potential conflict of interest relevant to this article was declared.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Funding
This research did not receive any specific grant from funding agencies.
Authors’ contributions
F.O.K. worked on the literature search, data extraction, manuscript preparation, and quality assessment. Mah.S.’s role was literature search, data extraction, quality assessment, and manuscript preparation. Mas.S. performed the statistical analysis and quality assessment of the research. S.A.G. worked on data extraction and revision, conceptualization, and supervision of the research. A.O.K.’s role was in data extraction and manuscript revision. E.R. and Maj S. had the idea for the manuscript and worked on the literature search. Maj. S. helped in drafting and critically revising the manuscript. All authors read and approved the final version.
Acknowledgement
Not applicable.
Trial registration
We registered the proposal in the International Prospective Register of Systematic Reviews (PROSPERO). Its registration number is CRD42023467213
References
- Yang CT, Chiu CD, Wu CY (2022) Diffuse symptomatic pneumocephalus after biportal endoscopic spinal surgery: illustrative case. J Neurosurg Case Lessons 3(26): case 22168.
- Katarzyna Śmiłowska, Katarzyna Sznajder-Stacha, Daniel Kocyłowski, Aleksandra Popek, Kamila Rozpondek, et al. (2021) Pneumocephalus as a rare complication: a systematic review plus clinical vignette. Neurol Neurochir Pol 55(3): 253-268.
- Ken Sasaki, Tomoyuki Matsumoto, Toshiyuki Mizuno, Shinichi Ikuta, Toshihiro Akisue, et al. (2010) Pne: aephalus associated with cerebrospinal fluid fistula as a complication of spinal surgery: a case report. Case Rep Med 2010: 328103.
- R Gauthé, C Latrobe, C Damade, E Foulongne, X Roussignol, et al. (2016) Symptomatic compressive pneumocephalus following lumbar decompression surgery. Orthop Traumatol Surg Res 102(2): 251-253.
- Abu-Hamdiyah OJ, Al Sharie S, Awadi S, Athamneh MJ (2021) Pneumocephalus secondary to a spinal surgery: a literature review and a case report. Int J Surg Case Rep 86: 106342.
- Andarcia-Bañuelos C, Cortés-García P, Herrera-Pérez M, Deniz-Rodríguez B (2015) Pneumocephalus: an unusual complication of lumbar arthrodesis. A clinical case and literature review. Rev Esp Cir Ortop Traumatol 59(4): 222-226.
- Maupin J, Burrow Z, Shirazi C, Vallurupalli S (2018) Tension pneumocephalus after cervical spine surgery: a case report with review of the literature. J Neurol Surg Rep 79(04): e88-e92.
- Roel CB, Etxebarria MG, Bendaña CD, Picallo AM, Lopez BV (2023) Pneumocephalus, coma and seizures following lumbar decompression surgery. Rev Esp Anestesiol Reanim (Engl Ed) 70(3): 160-164.
- Biju R, Wu J, Hussain Z (2020) Tension pneumocephalus after skull base surgery. A case report and review of literature. J Clin Neurosci 75: 218-20.
- Garcia-Garcia D, Gomez-Rice A, Vázquez-Vecilla I, López-Franco M (2021) Tension Pneumocephalus: A Case Report of a Rare Complication After Spinal Surgery. SN Compr Clin Med 3: 1610-1614.
- Orhan Akyüz, Deniz Gökpınar, Emsal Aydın, Sergülen Aydın, Mahmut Duymuş, et al. (2016) Pneumocephalus and pneumorrhachis after spinal surgery. Pol J Radiol 81: 34-5.
- David Moher, Larissa Shamseer, Mike Clarke, Davina Ghersi, Alessandro Liberati, et al. (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Systematic reviews 4(1): 1-9.
- Moola S (2020) Chapter 7: Systematic reviews of etiology and risk. IN: Aromataris E, Munn Z (Editors). JBI Evid Synth.
- Kim YD, Lee JH, Cheong YK (2012) Pneumocephalus in a patient with no cerebrospinal fluid leakage after lumbar epidural block-a case report. Korean J Pain 25(4): 262-266.
- Chang CY, Hung CC, Liu JM, Chiu CD (2022) Tension pneumocephalus following endoscopic resection of a mediastinal thoracic spinal tumor: A case report. World J Clin Cases 10(2): 725-732.
- Gupta M, Kumar Varma KK, Singh Chhabra H (2019) A rare case of concomitant pneumocephalus and pneumorachis after lumbar spine surgery with late presenting dural leak. Spinal Cord Ser Cases 5: 86.
- Lin CH, Lin S-M, Lan TY, Pao JL (2019) Pneumocephalus with conscious disturbance after full endoscopic lumbar Diskectomy. World Neurosurg 131: 112-115.
- Kumar A, Kishen TJ (2020) Delayed Onset Tension Pneumocephalus Following Lumbar Spine Surgery: A Report of Two Cases and Review of Literature. Annals of International Medical and Dental Research 6(5): 18-22.
- Kinthala S, Jiao K, Ankam A, Paramore CG (2020) Cerebellar hemorrhage and spinal fluid overdrainage with tonsillar herniation following spine surgery. Cureus 12(9): e10418.
- Ozturk E, Kantarci M, Karaman K, Cinar Basekim C, et al. (2006) Diffuse pneumocephalus associated with infratentorial and supratentorial hemorrhages as a complication of spinal surgery. Acta Radiol 47(5): 497-500.
- Rovlias A, Pavlakis E, Kotsou S (2006) Symptomatic pneumorachis associated with incidental durotomy during microscopic lumbar disc surgery: case report. J Neurosurg Spine 5(2): 165-167.
- Turgut M, Akyüz O (2007) Symptomatic tension pneumocephalus: an unusual post-operative complication of posterior spinal surgery. J Clin Neurosci. 14(7): 666-668.
- Song K-J, Lee K-B (2009) Spontaneous extradural pneumorrhachis causing cervical myelopathy. Spine J 9(2): e16-e18.
- Yun JH, Kim YJ, Yoo DS, Ko JH (2010) Diffuse pneumocephalus: a rare complication of spinal surgery. J Korean Neurosurg Soc 48(3): 288-290.
- Ayberk G, Yaman ME, Ozveren MF (2010) Symptomatic spontaneous pneumocephalus after spinal fusion for spondylolisthesis. J Clin Neurosci 17(7): 934-936.
- Dhamija B, Saxena A (2011) Pneumocephalus–a possible cause of post-spinal surgery confusion. J R Soc Med 104(2): 81-83.
- Nowak R, Maliszewski M, Krawczyk L (2011) Intracranial subdural hematoma and pneumocephalus after spinal instrumentation of myelodysplastic scoliosis. J Pediatr Orthop 20(1): 41-45.
- Pirris SM, Nottmeier EW (2013) Symptomatic pneumocephalus associated with lumbar dural tear and reverse trendelenburg positioning: a case report and review of the literature. Case Rep Neurol Med 2013: 792168.
- Karavelioglu E, Eser O, Haktanir A (2013) Pneumocephalus and pneumorrhachis after spinal surgery: case report and review of the literature. Neurol Med Chir (Tokyo) 54(5): 405-407.
- Baran Yılmaz, Semra Işık, Murat Şakir Ekşi, Emel Ece Özcan Ekşi, Akın Akakın, et al. (2015) Multiple hemorrhages in brain after spine surgery supra-and infra-tentorial components together. J Craniovertebr Junction Spine 6(4): 223-226.
- Çakmak MAÜ, Şahin Ş, Çınar N, Kelten B, Karşıdağ S (2014) Seizure After Laminectomy Associated with Pneumocephalus. Turk Noroloji Dergisi 20: 144.
- Kizilay Z, Yilmaz A, Ismailoglu O (2015) Symptomatic pneumocephalus after lumbar disc surgery: a case report. Open Access Maced J Med Sci 3(1): 143-145.
- Goodwin CR, Boone CE, Pendleton J, et al. (2016) Pneumocephalus leading to the diagnosis of cerebrospinal fluid leak and esophageal perforation after cervical spine surgery. J Clin Neurosci 26: 141-142.
- Kumar B, Banerjee C, Ghosh S (2017) Pneumocephalus following dorsal laminectomy: an unusual complication. Asian J Neurosurg 12(04): 772-773.
- Ozdemir U (2017) A rare complication of lumbar spinal surgery: pneumocephalus. Korean J Neurotrauma 13(2): 176-179.
- Kieser D, Cawley D, Tavolaro C, et al. (2018) Delayed post-operative tension pneumocephalus and pneumorrhachis. Eur Spine J 27(1): 231-235.
- Lamsal R, Mishra RK, Kumar N (2018) Pneumocephalus following Lumbar Spine Surgery: A Rare Cause of Delayed Emergence from Anesthesia. J Neuroanaesth Crit Care 5(01): 30-32.
- Gader G, Karmeni N, Ben Saïd I, Jemel H (2019) Symptomatic pneumocephalus: a rare complication of discal herniation's surgery. J Spinal Cord Med 42(6): 810-812.
- Denis B, Ledent M (2019) Pneumocephalus as a cause of seizure after lumbar spine surgery. A case report and review. Acta Anaesthesiol Belg 70(1): 43-47.
- Kundangar RS, Bhat SN, Mohanty SP (2021) Tension pneumocephalus following an implant removal from spine. BMJ Case Rep 14(2): e239694.
- Matei A Banu, Oszkar Szentirmai, Lino Mascarenhas, Al Amin Salek, Vijay K Anand, et al. (2014) Pneumocephalus patterns following endonasal endoscopic skull base surgery as predictors of postoperative CSF leaks. J Neurosurg Case Lessons 121(4): 961-975.
- Aguilar-Shea AL, Manas-Gallardo N, Romero-Pisonero E (2009) Post-traumatic pneumocephalus. Int J Emerg Med 2(2): 129-130.
- Promod Pillai, Rohit Sharma, Larami MacKenzie, Eugene F Reilly, Paul R Beery 2nd, et al. (2017) Traumatic tension pneumocephalus–Two cases and comprehensive review of literature. Int J Crit Illn Inj Sci 7(1): 58-64.
- Kim HS, Kim SW, Kim SH (2013) Spontaneous pneumocephalus caused by pneumococcal meningitis. J Korean Neurosurg Soc 53(4): 249-51.
- Kankane V, Jaiswal G, Gupta T (2016) Posttraumatic delayed tension pneumocephalus: Rare case with review of literature. Asian J Neurosurg 11(04): 343-347.
- Nam KH, Song Y, Kim DH, et al. (2019) Symptomatic Pneumocephalus After Spinal Intradural-Extramedullary Tumor Surgery. World neurosurg 130: e344-e349.
- Lam KK, Prasad A, Fehlings MG, Venkatraghavan L (2014) Pneumocephalus: an unusual case of postoperative seizure after intradural spine surgery. Can J Anaesth 61(10): 969-970.
- Chan YP, Yau CY, Lewis RR, Kinirons MT (2000) Acute confusion secondary to pneumocephalus in an elderly patient. Age ageing 29(4): 365-367.
- Anandpara KM, Aswani Y, Hira P (2015) The Mount Fuji sign. Clin Med (Lond) 15(6): 596.
- Rathore AS, Satyarthee GD, Mahapatra AK (2016) Post-traumatic tension pneumocephalus: series of four patients and review of the literature. Turk Neurosurg 26(2): 302-305.
- L'Hommedieu LM, Dingeldein MW, Tomei KL, Kilbane BJ (2018) Acute management of tension pneumocephalus in a pediatric patient: a case report. J Emerg Med 54(1): 112-115.
- Arbit E, Shah J, Bedford R, Carlon G (1991) Tension pneumocephalus: treatment with controlled decompression via a closed water-seal drainage system: case report. J Neurosurg Case Lessons 74(1): 139-142.
- Horowitz M (1964) Intracranial pneumocoele: An unusual complication following mastoid surgery. J Laryngol Otol 78(2): 128-134.
- Dandy WE (1926) Pneumocephalus (intracranial penumatocele or aerocele). Arch Surg 12(5): 949-982.
- Theilen HJ, Heller AR, Litz RJ (2008) Nitrous oxide-induced tension pneumocephalus after thoracic spinal cord surgery: a case report. J Neurosurg Anesthesiol 20(3): 211-212.
- Raggio JF, Fleischer AS, Sung YF, Hoffman JC (1979) Expanding pneumocephalus due to nitrous oxide anesthesia: case report. Neurosurgery 4(3): 261-263.
- Klein M, Kelly J, Jacobs I (1989) Diffuse pneumocephalus from Clostridium perfringens meningitis: CT findings. AJNR Am J Neuroradiol 10(2): 447.
- Penrose-Stevens A, Ibrahim A, Redfern R (1999) Localized pneumocephalus caused by Clostridium perfringens meningitis. Br J Neurosurg 13(1): 85-86.






























