Abstract
Introduction: Delayed carpal tunnel syndrome (DCTS) in patients with malunited distal radius fracture (DRF) develops several weeks to months after injury. The main treatment method for these patients is corrective osteotomy and fixation of the radius bone. However, the necessity and methods of median nerve decompression still remain controversial.
Purpose: To evaluate the long-term results of surgical treatment of patients with a malunited distal radius fracture and concurrent delayed carpal tunnel syndrome, depending on the method of median nerve decompression, and to develop a treatment concept.
Methods: The results of treatment were studied in 33 patients (30 women and three men, average age 54.6 years) with malunited DRF complicated by DCTS. All patients underwent corrective osteotomy of the distal radius and osteosynthesis with a volar locking plate. In addition, open carpal tunnel release was performed in the first group of patients through a separate limited surgical approach (OCTR, n=19), while decompression of the median nerve was carried out through an extended flexor carpi radialis approach in the second group (EFCR, n=14). Patients were evaluated clinically (wrist range of motion, hand strength, pain level VAS, DASH score), radiographically, and electromyographically before surgery and one year thereafter. Different degrees of severity of DCTS and deformation of the DRF were compared.
Results: Both groups of patients showed improvements in clinical, radiological, and EMG parameters after surgery (p<0.001). The average time for bone healing was 12 weeks. Patients in the first group (OCTR) achieved more favorable results: significantly increased hand grip strength (p = 0.010), compound muscle action potential (CMAP, p < 0.001), and an improved DASH score (p = 0.038). Depending on the severity of DCTS and the severity of deformity of the DRF, it was found that the most significant favorable changes occurred in moderate and severe degrees of DCTS, as well as with “intermediate” and “predominantly dorsal deformation” of the DRF.
Conclusion: Corrective osteotomy and volar locking plate osteosynthesis with carpal tunnel release are reliable and effective treatments for malunited distal radius fractures with concurrent delayed carpal tunnel syndrome. The best results were obtained after open carpal tunnel release (OCTR) in patients with moderate and severe degrees of DCLS, combined with “intermediate” and “predominantly dorsal deformation” of the DRF. With a mild degree of DCTS as well as a “predominantly palmar deformity” of DRF, decompression of the median nerve can be performed through the main EFCR approach.
Keywords:Distal radius fracture; Malunion; Osteotomy, Delayed carpal tunnel syndrome; Carpal tunnel release; Limited open approach; Extended flexor carpi radialis approach; Median nerve
Introduction
Malunion of distal radius fractures (DRF) can involve the soft tissue structures passing through the carpal tunnel during the regeneration process (Cognet et al., 2020) [1]. This often affects the median nerve, which, when compressed, predisposes the development of carpal tunnel syndrome (CTS). CTS can be classified by onset as acute (occurring within hours or days after the fracture, with a frequency of 5.4-8.6%), subacute or transient (4% of cases), and delayed or chronic, appearing weeks or months post-injury (0.5-22%) [2, 3]. The formation of delayed CTS (DCTS) typically occurs during the consolidation of radial bone fragments, most often in cases of malunion [4-10]. According to Stewart et al. (1985), the incidence of DCTS reaches 17% three months postfracture and 12% after six months [11]. Direct causes of DCTS include a reduction in carpal tunnel volume, residual dorsal or palmar displacement of the distal fragment, edema, tenosynovitis, prolonged wrist immobilization in the Cotton-Loder position, and excessive callus formation [12-17].
The questions surrounding the indications for median nerve decompression in DRF malunions and the preferred surgical techniques remain controversial [18,19]. From a systematic perspective, two main types of surgical approaches for treatment of DCTS can be distinguished. The first type is a single longitudinal incision, approximately 3cm long, made on the anterior surface of the lower third of the forearm, with a radial deviation at the palmar wrist creases through the tendon of the flexor carpi radialis and excision of the outer layers of the flexor retinaculum. This approach was first described by R. Weber et al. (1997) [20] and was initially used solely for median nerve decompression. Later, this method was adapted by F.W. Gwathmey et al. (2010) for prophylactic median nerve decompression combined with DRF osteosynthesis by extending the incision to 7-8cm [21]. A similar approach, 8-10 cm in length and used exclusively for DRF surgery, was applied by J.L. Orbay et al. (2001) [22].
We used the term EFCR (Extended Flexor Carpi Radialis Approach) [23] to describe approaches primarily for median nerve decompression. Approaches of this type involve one incision serving two purposes: median nerve release and reconstructive DRF intervention. The second type involves combined approaches, with two incisions: one for median nerve decompression on the palmar wrist (OCTR - Open Carpal Tunnel Release) [24], and a second for corrective osteotomy and DRF osteosynthesis in the lower third of the forearm. This approach entails two distinct incisions, each with a specific purpose. This study evaluated the efficacy of both approach types to determine the therapeutic potential and feasibility of their differentiated application depending on the severity of DCTS and the degree of palmar tilt in malunited DRF.
Objective of the Study
To evaluate the long-term outcomes of surgical treatment in patients with DCTS due to malunion of DRF fractures, depending on the method of median nerve decompression, and to develop recommendations for a differentiated surgical strategy.
Materials and Methods
From January 1, 2006, to December 31, 2022, we monitored 33 patients (30 women and 3 men), aged 36 to 71 years (mean age 54.6 years), with malunited distal radius fractures complicated by DCTS. The time from injury to surgery ranged from three to 16 months (mean 4.8 months). Patients were divided into two clinical groups through randomization. The first group included 19 patients who underwent a combined approach: a mini incision on the palmar wrist for median nerve decompression through transverse carpal ligament incision, epineurotomy, and neurolysis (OCTR approach), and a second incision on the lower third of the forearm for corrective osteotomy and DRF osteosynthesis. The second group consisted of 14 patients who had a single incision on the lower forearm for corrective osteotomy, DRF osteosynthesis, and median nerve decompression (EFCR approach) without epineurotomy or neurolysis.
Both groups were comparable in terms of age, DRF type, DCTS severity, degree of palmar tilt, and timing of reconstructive surgery (Table 1). Indications for surgery were primarily based on complaints of wrist pain (mostly nocturnal), numbness in the first, second, and third fingers, wrist deformity and pain under load, reduced grip strength, and limited wrist mobility. Radiographic parameters considered included a deficiency of palmar inclination >20°, a radioulnar angle <10°, a radioulnar index >2 mm, and intra-articular displacement >2 mm. Contraindications for corrective osteotomy included uncompensated diabetes mellitus, severe osteoporosis, and fixed wrist dislocation.
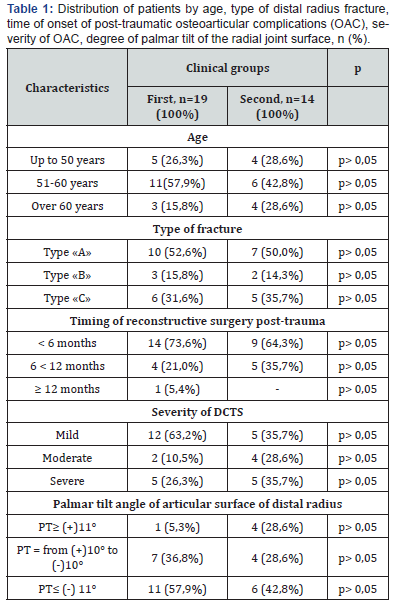
Preoperative Planning
Preoperative planning was based on wrist joint radiographs in standard projections—anteroposterior and sagittal views. Computed tomography (CT) was performed using “Toshiba Aquillion 32” and “Philips Brilliance 190 P” CT scanners (Netherlands) in spiral scanning mode with a reconstructed slice thickness of 0.5 mm and a reconstruction step of 0.3 mm. The degree of displacement of the distal radius fragments was measured in millimeters and degrees, and the level of osteotomy, shape, and size of the bone defect were determined.
Surgical Treatment Evaluation
The effectiveness of the surgical treatment was evaluated using 11 criteria, including clinical (pain intensity during load [VAS], total active range of motion in the wrist [TRAM], grip strength [GS], and daily activity [DASH-score]), radiographic (radial inclination [RI], palmar tilt [PT], ulnar variance [UV]), and electroneuromyographic (ENMG) indicators (CMAP amplitude of the short abductor muscle [mV], distal motor latency of the median nerve motor fibers [DML, ms], motor nerve conduction velocity [MCV, m/s], and sensory nerve conduction velocity [SCV, m/s]). All these parameters were assessed preoperatively and one year postoperatively.
Assessment of Deformity Severity
The severity of carpal tunnel syndrome (CTS) was determined using the A. Żyluk et al. (2014) classification. Based on the CMAP amplitude of the short abductor muscle (greater than 4 mV for mild, 4-2 mV for moderate, and less than 2 mV for severe CTS), patients were classified accordingly. Additionally, based on the palmar tilt of the radial joint surface (PT), patients were conditionally divided into those with “predominantly palmar deformity” (PT ≥ 11°), “intermediate deformity” (PT between +10° and -10°), and “predominantly dorsal deformity” (PT ≤ -11°) of the distal radius.
Statistical Analysis
Statistical analysis was performed using the IBM SPSS Statistics software (USA, license no. 5725-A54). Descriptive statistics are presented as means and standard deviations (M±SD). The analysis used Kruskal-Wallis ANOVA, Mann-Whitney tests, and the Wilcoxon paired test.
Study Design
This is a prospective, randomized, controlled, multi-center study. The subject of the study is DCTS developed due to malunion of the distal radius fracture. The object of the study is patients suffering from DCTS due to the malunion of the distal radius fracture. Inclusion criteria included the presence of malunion of the distal radius with clinical signs of DCTS. Exclusion criteria included concomitant diabetes, severe osteoporosis, and fixed wrist displacement.
Primary Endpoint
The primary endpoint of the study was the assessment of the effectiveness of surgical treatment for DCTS through a combined surgical approach, including an incision in the distal forearm for correcting the malunion of the fracture and distal radius osteosynthesis, and a mini-incision on the palmar surface of the wrist for median nerve decompression, epineurotomy, and neurolysis (OCTR access), with recommendations for differentiated surgical treatment depending on the severity of DCTS and the degree of palmar tilt of the radial joint surface.
Secondary Endpoints
Secondary endpoints included clinical outcomes (pain intensity during load, total active wrist motion, grip strength, daily activity), radiographic outcomes (radial inclination, palmar tilt, ulnar variance), and ENMG indicators (CMAP amplitude, distal latency of motor fibers of the median nerve, motor and sensory conduction velocities of the median nerve).
Surgical Technique
The surgery was performed under general anesthesia and arm tourniquet, with the upper limb placed in a supine position on a fluoroscopy table. In the first group of patients, open decompression of the median nerve was performed using a limited open approach (OCTR). A Z-shaped incision, 3.5-4 cm in length, was made on the palmar surface of the wrist. The skin and subcutaneous tissue were dissected at the level of the ulnar part of the carpal tunnel, and palmar aponeurosis was incised on its radial side.
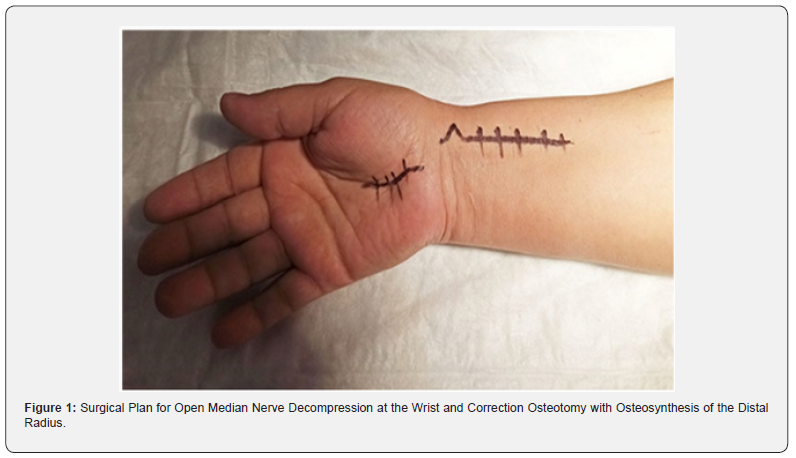
The incision line of the transverse palmar ligament was made obliquely from the dorsal-radial to the palmar-ulnar side of the tunnel. The median nerve and its motor branch were mobilized, and epineurotomy and neurolysis were performed. The palmar aponeurosis and skin were sutured. Following this, a corrective osteotomy and osteosynthesis of the distal radius were performed through an incision in the distal forearm along its palmar-radial surface, as described by J.L. Orbay et al. [22] (Figure 1). In patients of the second group, median nerve decompression was performed using an approach to the forearm, simultaneously performing corrective osteotomy and osteosynthesis, as described by F.W. Gwathmay et al. [21]. A linear incision of approximately 7-8 cm in length was made along the palmar-radial surface of the lower third of the forearm, at the level of the flexor carpi radialis tendon, and extended distally in a Z-shaped curve for 7-10 mm (EFCR approach) (Figure 2a).
The flexor retinaculum was mobilized and dissected along the tendon of the flexor carpi radialis. The palmar branch of the radial artery was ligated and transected or displaced aside. The tendon of the flexor carpi radialis was retracted radially, and the deep layer of the ligament was dissected. After ulnar retraction of the tendon of the long flexor of the thumb, both the superficial and deep layers of the transverse carpal ligament were finally dissected from the tubercle of the scaphoid and trapezium bones (Figure 2b). For access to the radial bone, the pronator quadratus muscle was cut, separated from the bone in the outer and distal sections and displaced towards the ulna.
The brachioradialis tendon was Z-shaped dissected, providing access to the deformed surface of the distal radius. In patients with “predominantly dorsal deviation” of the distal fragment of the radius (n=17), an additional dorsal mini incision of up to four cm was made. This was necessary for full mobilization and stabilization of the distal radial fragment [26]. The proximal part of the extensor retinaculum was Z-shaped dissected, and the flaps were spread apart. Using precision surgical techniques, the periosteal bone callus was identified, and bone-fibrous channels of the extensor tendons were dissected. The first, second, third, and fourth channels were dissected, and the extensor tendons were displaced laterally. Special attention was paid to preserving the tendon of the long extensor of the thumb.
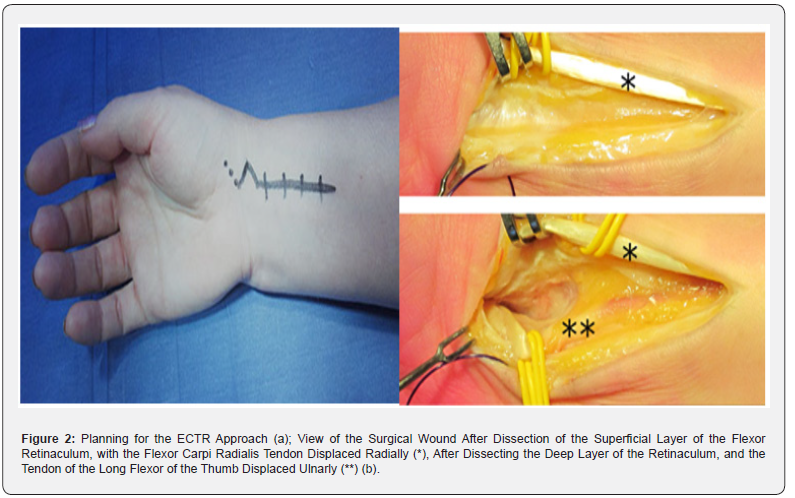
According to the preoperative plan and using fluoroscopy, corrective osteotomy of the distal radius was performed. Open reduction of bone fragments was carried out, restoring the normal anatomical relationships of the distal radius. The bone defect was filled with autologous bone or synthetic osteoplastic material based on β-3 calcium phosphate. A custom-sized plate was placed on the palmar surface of the radius so that it did not extend beyond the distal and palmar edges of the “watershed line” of the distal radius. Under endoscopic guidance, locking and cortical screws were placed (Figure 3). Continuity of the brachioradialis tendon, the integrity of the pronator quadratus, and the extensor retinaculum were restored.
The tendon of the long extensor of the thumb was left in the subcutaneous tissue. The wounds were closed in layers with active drainage placed. For the first two to three weeks, immobilization was performed using a removable palmar cast splint in a functional wrist position. For the next four weeks, a removable orthosis was used. Finger rehabilitation exercises began on the second postoperative day. Active rehabilitation started five to six weeks after the surgery. Full load bearing was permitted only after the consolidation of bone fragments, but not earlier than 12 weeks postoperatively.
Results
During the treatment, all patients experienced complete or significant correction of distal radius fracture (DRF) deformity, with bone fragment union observed on average by the 12th week. Clinical outcomes also showed substantial improvement. Pain intensity during load (VAS) decreased by 66%, from 4.97±1.66 to 1.68±0.80 points. The total range of wrist motion (TRAM) increased on average by 43%, from 57.05±12.15° to 81.76±7.46°, and grip strength (GS) rose by 82%, from 37.58±16.52% to 68.45±16.05%. Daily activity (DASH score) improved by 58%, from 41.83±8.74 to 17.50±7.22 points (Table 2). Radiographic data changes were similarly positive. The radial inclination angle (RI) increased by 75%, from 12.26±5.15° to 21.50±4.36°.
Palmar tilt (PT) improved by 144%, shifting from -9.49±18.58° to 4.19±5.06°, and the ulnar variance (UV) reduced by 73%, from 4.38±2.34 mm to 1.20±1.98 mm. Electroneuromyographic (ENMG) parameters also demonstrated positive dynamics. The compound muscle action potential (CMAP) amplitude increased by 78%, from 3.75±2.37 mV to 6.66±2.49 mV. Distal motor latency (DML) decreased by 27%, from 5.66±1.86 ms to 4.10±0.73 ms. Motor nerve conduction velocity (MCV) increased by 19%, from 45.53±10.08 m/s to 54.28±6.76 m/s, and sensory nerve conduction velocity (SCV) rose by 47%, from 30.19±15.01 m/s to 44.29±10.83 m/s (p<0.001).
As a result, all 11 indicators showed statistically significant improvement in all patients one year after surgery. The values of these indicators in the clinical groups before surgery were almost identical. However, after one year, all indicators in the first group changed more significantly than in the second group. For instance, grip strength (GS) in the first group increased to 74.37±13.52%, while in the second group, it only increased to 60.43±16.15%. Daily activity (DASH score) in the first group reached 15.06±6.38 points, whereas in the second group, it was 20.82±7.17 points.
The amplitude of the compound muscle action potential (CMAP) of the abductor pollicis brevis increased to 7.53±2.08 mV in the first group, while in the second group, it only increased to 5.48±2.58 mV (Table 3). These changes were statistically significant. When comparing all 11 parameters based on the severity of CRPS, it was found that one year after surgery, no significant changes occurred in the groups with mild CRPS (n=17). In contrast, for moderate and severe degrees of CRPS (n=16), statistically significant changes were observed in several indicators. One-year post-surgery, grip strength (GS) in the first group increased to 70.29±18.78%, while in the second group, it only increased to 51.89±10.86%. Daily activity (DASH score) showed an advantage in the first group compared to the second, amounting to 15.51±8.45 points and 25.00±4.45 points, respectively. The amplitude of the compound muscle action potential (CMAP) in the first group reached 5.53±1.98 mV, while in the second group, it was only 3.86±1.11 mV. The sensory conduction velocity (SCV) in the first group also exceeded that of the second group, measuring 43.43±7.39 m/s and 32.89±8.82 m/s, respectively (Table 4). These changes were statistically significant.
Our data indicate that for mild CRPS, performing median nerve decompression with epineurotomy and neurolysis through the OCTR approach is not advisable. This approach yields substantial results only for moderate and severe CRPS. For mild CRPS, positive outcomes can be achieved through the EFCR approach.
When comparing changes in indicators between the groups based on the severity of DMLK deformation, it was noted that for “predominantly palmar deformation” (n=5), there were no significant differences between the groups. However, in cases of “intermediate deformation” (n=11), significant differences were found in the values of several indicators. For example, the amplitude of the compound muscle action potential (CMAP) in the first group increased to 8.80±1.17 mV, while in the second group it only increased to 4.40±2.13 mV. The distal motor latency (DML) of the median nerve decreased to 3.87±0.39 ms in the first group and to 4.53±0.26 ms in the second group. The motor response speed also improved, rising to 57.16±1.68 m/s in the first group and to 43.63±9.59 m/s in the second (Table 5).
Similarly, for “predominantly dorsal deformation” (n=17), significant improvements were observed in several indicators. The total range of active motion in the wrist joint (TRAM) increased to 29.64±9.74% in the first group and only to 14.17±5.95% in the second. Grip strength (GS) values reached 37.60±10.96% and 18.47±6.91%, respectively. Daily activity (DASH score) also improved, amounting to 18.27±4.44 points in the first group and 29.67±7.26 points in the second. The data above indicate that for patients with “predominantly palmar deformity” of the distal radius, it is not advisable to perform median nerve decompression with epineurotomy and neurolysis through the OCTR approach. For these patients, decompression through the EFCR approach without epineurotomy and neurolysis is sufficient.
Complications
In two patients from the first group, a dense and painful postoperative scar developed within two to three months after surgery, and three patients reported pain in the hypothenar and thenar areas (pillar pain). These symptoms resolved during postoperative treatment and were not bothersome by the oneyear follow-up. One patient in the second group developed transient irritation of the superficial branch of the median nerve. No wound infections or screw migrations were observed. No reoperations were required.
Clinical Case
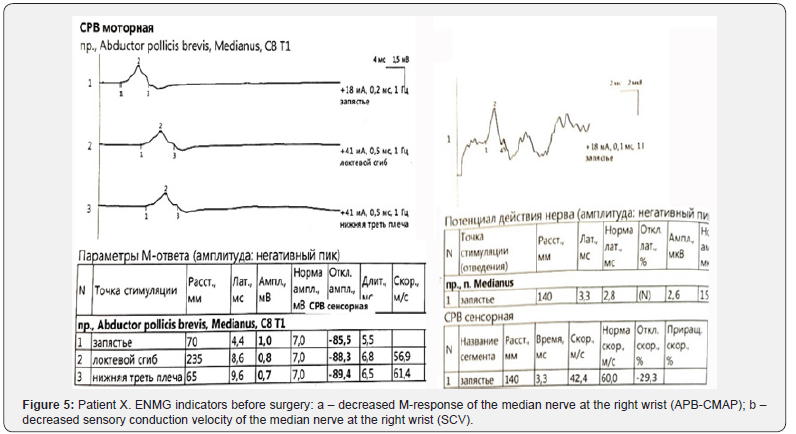
A clinical case. Patient X, a 58-year-old female, presented two months after injury with the diagnosis of a malunited distal radius fracture on the right (AO/ASIF type A 3.2) with delayed carpal tunnel syndrome (DCTS). Upon admission, she complained of wrist deformity, pain, limited range of motion in the right wrist, decreased sensation in fingers 1, 2, and 3, and reduced grip strength. Objectively, the total range of active movements in the right wrist compared to the contralateral limb was 64%, and the gross grip strength was 35%. Pain intensity on the VAS scale reached 4.7 points under load.
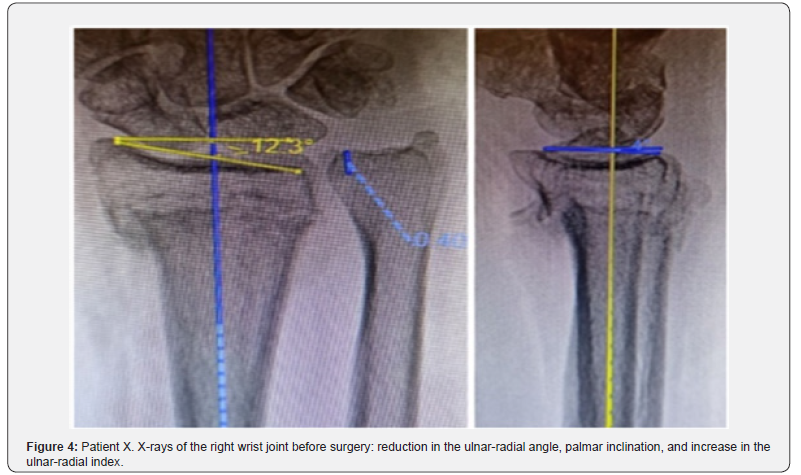
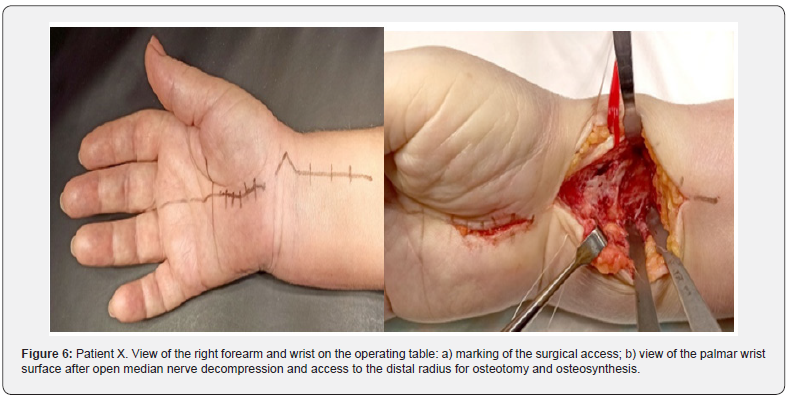
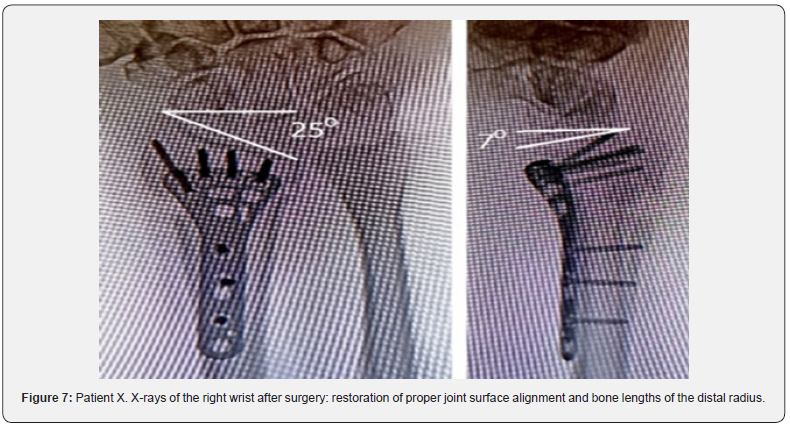
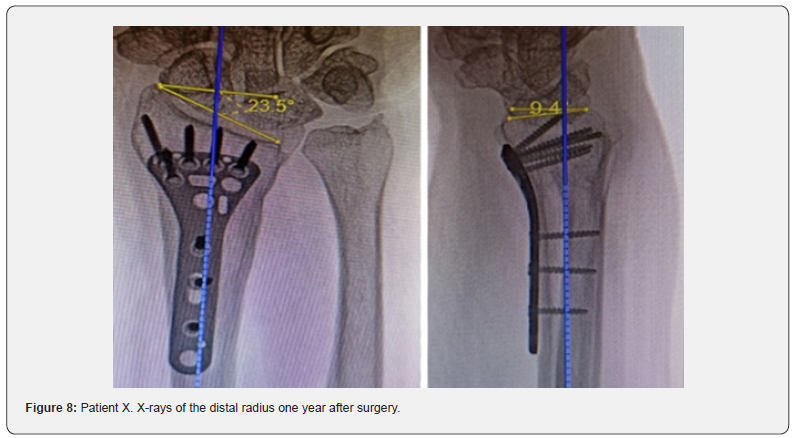
X-rays showed angular palmar displacement of the distal radial cortical plate, a reduction in the ulnar-radial angle to 12.3°, an increase in dorsal tilt of the radial joint surface in the sagittal plane to -4° and shortening of the radial bone by 4 mm (Figure 4). Electroneuromyography (ENMG) of the upper extremities revealed a decrease in the M-response of the median nerve at the right wrist (APB-CMAP) and a decrease in sensory conduction velocity of the median nerve (SCV) (Figure 5). According to the classification by A. Żyluk et al. (2014) [25], a severe grade of CTS was diagnosed. The function of the right upper limb on the DASH score was 45.8 points (unsatisfactory). In a planned surgery, from a limited palmar approach to the right wrist, open decompression of the median nerve with epineurotomy and external neurolysis was performed (Figure 6a). Subsequently, through a palmar approach to the forearm, extra-articular open-angle corrective osteotomy and osteosynthesis of the distal radius were performed (Figure 6b).
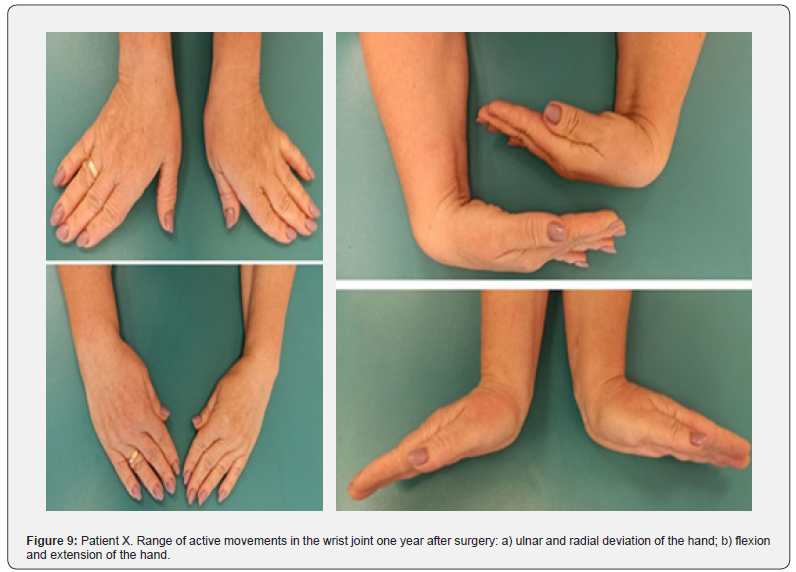
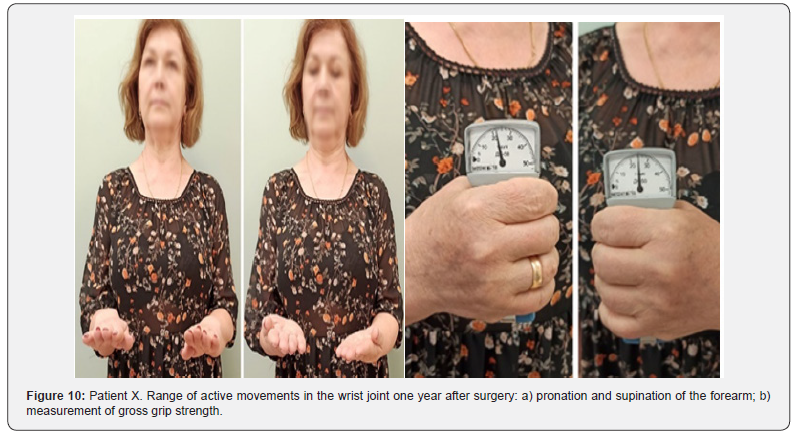
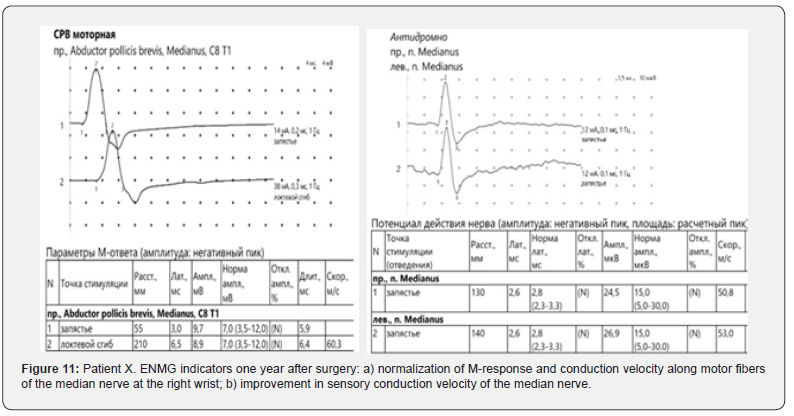
Discussion
The assertion by most hand surgery specialists that malunion of distal radius fractures (DRF) contributes to the development of carpal tunnel syndrome (CTS), and that only surgical intervention can correct or reduce the symptoms of this complication, is widely accepted. However, the preferred approaches for corrective osteotomy and median nerve decompression are still debated. For example, K. Watanabe and colleagues (2019) demonstrated that displacement of the distal fragment along with the wrist in DRF substantially alters the anatomical relationships of the carpal tunnel structures, resulting in both morphological and functional abnormalities in the median nerve. According to their findings, in cases where the center of the capitate bone was displaced dorsally by one centimeter or more from the palmar cortical plate of the radius, CTS developed almost invariably within six weeks to six months. Based on this, the authors recommended paying particular attention to these patients in the acute phase of injury and taking appropriate preventive measures [26].
K. Kim et al. (2020) examined 216 elderly patients with malunion of DRF and diagnosed CTS in only 12% of cases at least six weeks post-fracture. Independent predictors of this complication were radiographic criteria such as the degree of volar tilt (VT) and the tear drop angle (TDA). Their multivariate logistic regression analysis revealed that each degree of reduced volar tilt and TDA increased the likelihood of CTS development by 12% and 24%, respectively [27]. Among our patients, the volar tilt angle ranged from (+)28° to (-)45°, with all patients exhibiting both clinical and EMG signs of CTS. P. Bourque and colleagues (2020) observed that after cast removal six to eight weeks postinjury, paresthesia in the area innervated by the median nerve was present in 20% of patients, increasing to 26% a week later. However, EMG performed at the same time showed no signs of focal demyelination or axonal loss in any of the patients [28].
In our study of 33 patients, nerve conduction abnormalities on EMG at the time of admission for surgery were observed in all patients. In ten individuals (30.3%), EMG changes indicated severe CTS, in six (18.2%) moderate CTS, and in the remaining 17 individuals (51.5%) mild CTS. Stewart et al. (1985) found that conservative treatment in 235 patients with DRF (mean age 66 years) resulted in a CTS rate of 17% at three months and 12% at six months post-injury. The relatively low CTS rate was attributed to avoiding excessive wrist flexion when applying a cast during the acute injury phase. Due to CTS, eight patients (3.4%) required surgery, including standard decompression with an incision at the wrist (n=7) and an extended decompression with an additional incision at the fracture site (n=1). In these patients, the articular surface inclination angle was ≥ (+)12.6°. In contrast, patients with an inclination angle of ≤ (+)7° showed no CTS symptoms [11].
Kwasny et al. (1994) suggested, that for malunion of DRF complicated by CTS, an isolated open-angle osteotomy with osteosynthesis, without additional median nerve decompression, is sufficient to relieve nerve tension and compression. Among 12 patients who underwent this surgery, nighttime pain subsided within two days, and tactile and pain sensitivity on the fingers returned within two months. One patient required an additional open median nerve decompression six months after the initial procedure [18]. In our study, no patients had CTS that predated the DRF. All cases of CTS developed in the later period, with reconstructive surgeries performed between three- and thirteenmonth post-injury.
We believe that in the presence of clinical signs of CTS, median nerve decompression, along with corrective osteotomy of the radius, is indicated and should be mandatory regardless of CTS severity. Furthermore, chronic compression of the median nerve, in our view, necessitates not only distal radius deformity correction and nerve decompression, but also nerve mobilization and, in cases of moderate to severe CTS, epineurotomy and neurolysis. This study demonstrated significant improvement in clinical, radiological, and EMG characteristics in patients from both clinical groups, with a more pronounced improvement in the first group, who additionally underwent epineurotomy and neurolysis using a limited palmar wrist incision (OCTR approach).
O. Odumala et al. (2001) reported that in their study of 69 patients with DRF without concurrent CTS symptoms, simultaneous prophylactic median nerve decompression resulted in nerve dysfunction complications in 38% of cases. In contrast, patients who did not undergo prophylactic decompression had a nearly twofold lower rate of such complications at 18%, suggesting that prophylactic decompression of the median nerve is not advisable [29].
In summary, based on our findings, the EFCR approach should be recommended for patients with mild DCTS and “predominantly palmar deformity” of DRF, where nerve mobilization, epineurotomy, and neurolysis are not needed. In cases with moderate to severe DCTS, as well as for “intermediate” and “predominantly dorsal deformities” of distal radius, the OCTR approach is more appropriate. This limited incision on the palmar wrist surface enables effective decompression, epineurotomy, and neurolysis with minimal trauma, preventing the formation of a linear constricting scar in the palmar crease and minimizing the risk of damage to the superficial palmar branch of the median nerve.
Conclusion
1. Corrective osteotomy and simultaneous decompression
of the median nerve in patients with malunited distal radius
fractures and concurrent delayed carpal tunnel syndrome (DCTS)
is an effective and reliable method for surgical correction of this
complication.
2. For mild DCTS and cases with “primarily volar deformity”
of DRF, it is advisable to perform median nerve decompression
through the extended flexor carpi radialis (EFCR) approach,
involving an incision in the distal third of the forearm.
3. For moderate to severe DCTS, as well as cases with
“intermediate” and “primarily dorsal deformity” of DRF, median
nerve decompression should be performed via the open carpal
tunnel release (OCTR) approach, involving a separate, limited
incision on the palmar surface of the wrist.
4. In most cases, regardless of DCTS severity, it is optimal to
perform corrective osteotomy and osteosynthesis of DRF through
an incision in the distal third of the forearm along its anterior
surface. However, for patients with “primarily dorsal deformity,”
an additional mini-incision on the dorsal surface of the forearm
is recommended for accurate alignment and stabilization of bone
fragments.
Conflict of Interest
The authors of this article confirm the absence of any conflict of interest.
Funding Sources
The study was conducted with the authors’ personal funds.
References
- Cognet JM, Mares O (2021) Distal radius malunion in adults. Orthop Traumatol Surg Res107(1S): 102755.
- Dyer G, Lozano-Calderon S, Gannon C, Baratz M, Ring D (2008) Predictors of acute carpal tunnel syndrome associated with fracture of the distal radius. J Hand Surg 33(8): 1309-1313.
- Itsubo T, Hayashi M, Uchiyama S, Hirachi K, Minami A, Kato H (2010) Differential onset patterns and causes of carpal tunnel syndrome after distal radius fracture: a retrospective study of 105 wrists. J Orthop Sci 15(4): 518-523.
- Aro H, Koivunen T, Katevuo K, Nieminen S, Aho AJ (1988) Late compression neuropathies after Colles’ fractures. Clin Orthop Relat Res 233: 217-225.
- Jupiter JB, Fernandez DL, Toh CL, Fellman T, Ring D (1996) Operative treatment of volar intra-articular fractures of the distal end of the radius. J Bone Joint Surg 78(12): 1817-1828.
- Young BT, Rayan GM (2000) Outcome following nonoperative treatment of displaced distal radius fractures in low-demand patients older than 60 years. J Hand Surg Am 25(1):19-28.
- Bienek T, Kusz D, Cielinski L (2006) Peripheral nerve compression neuropathy after fractures of the distal radius. J Hand Surg 31(3): 256-260.
- Finsen V, Rod O, Rød K, Rajabi B (2013) Alm-Paulsen P. S., Russwurm H. The relationship between displacement and clinical outcome after distal radius (Colles’) fracture. J Hand Surg Eur 38(2): 116-126.
- Голубев ИО, Крупаткин АИ, Максимов АА, Меркулов МВ и др. Хирургическое лечение неправильно сросшихся переломов дистального метаэпифиза лучевой кости // Вестник травматологии и ортопедии им. Н.Н.Приорова. 2013, №3. - С.51-58
- Pope D, Tang P (2018) Carpal tunnel syndrome and distal radius fractures. Hand Clin 2018: 34: 27-32.
- Stewart HD, Innes AR, Burke FD (1985) The hand complications of Colles` fractures. J Hand Surg Br 10(1): 103-106.
- Lynch AC, Lipscomb PR (1963) The carpal tunnel syndrome and Colles’ fractures. JAMA 185: 363-366.
- Cooney WP, Dobyns JH, Linscheid RL (1980) Complications of Colles’ fractures. J Bone Joint Surg Am 62(4): 613-619.
- Kamath J, Jayasheelan N, Mathews R (2016) Compressive neuropathy of the palmar cutaneous branch of the median nerve after a malunited fracture of the distal radius J Hand Surg Eur 41(2): 231-232.
- Stark WA (1987) Neural involvement in fractures of the distal radius. Orthopedics 10(2): 333-335.
- Watanabe K, Ota H (2019) Carpal Malalignment as a Predictor of Delayed Carpal Tunnel Syndrome after Colles’ Fracture. Plast Reconstr Surg Glob Open 7(3): e2165.
- Rothman A, Samineni AV, Sing DC, Zhang JY, Stein AB (2022) Carpal tunnel release performed during distal radius fracture surgery. J Wrist Surg 12(3): 211-217.
- Kwasny O, Fuchs M, Schabus R (1994) Opening wedge osteotomy for malunion of the distal radius with neuropathy. Thirteen cases followed for 6 (1-11) years. Acta Orthop Scand 65(2)a: 207-208.
- Megerle K, Baumgarten A, Schmitt R, van Schoonhoven J, Prommersberger KJ (2013) Median neuropathy in malunited fractures of the distal radius Arch Orthop Trauma Surg 133(9):1321-1327.
- Weber RA, Sanders WE (1997) Flexor carpi radialis approach for carpal tunnel release. J Hand Surg 22(1): 120-126.
- F Winston Gwathmey Jr, Lance M Brunton, Raymond A Pensy, A Bobby Chhabra (2010) Volar plate osteosynthesis of distal radius fractures with concurrent prophylactic carpal tunnel release using a hybrid flexor carpi radialis approach. J Hand Surg Am 35(7): 1082-1088.e4.
- Orbay JL, Badia A, Indriago IR, Infante A, Khouri RK, Gonzalez E, Fernandez DL (2001) The extended flexor carpi radialis approach: a new perspective for the distal radius fracture. Tech Hand Up Extrem Surg 5(4): 204-211.
- Tannan SC, Pappou IP, Gwathmey FW, Freilich AM, Chhabra AB (2015) The extended flexor carpi radialis approach for concurrent carpal tunnel release and volar plate osteosynthesis for distal radius fracture. J Hand Surg Am 40(10): 2026-2031.
- Tulipan JE, Ilyas AM (2020) Carpal tunnel syndrome surgery: what you should know. Plast Reconstr Surg.Global Open 8(3): e2692.
- Żyluk A, Walaszek I, Szlosser Z (2014) No correlation between sonographic and electrophysiological parameters in carpal tunnel syndrome. J Hand Surg 39(2): 161-166.
- 2312631 Russian Federation, MPK A61B 17/56. Method of surgical treatment of fractures of the distal metaepiphysis of the radius / Izmalkov S.N., Semenkin O.M.; applicant and patent holder of the State Educational Institution of Higher Professional Education “SamSMU Roszdrav”. – No. 2005135390/14; appl. 11/14/2005; publ. 12/20/2007, Bulletin. No. 35.
- Odumala O, Ayekoloye C, Packer G (2001) Prophylactic carpal tunnel decompression during buttress plating of the distal radius--is it justified? Injury 32(07):577-579.
- Niver GE, Ilyas AM (2012) Carpal tunnel syndrome after distal radius fracture. Orthop Clin North Am 43(4): 521-527.
- Pensy R, Brunton LM, Parks BG, Higgins JP, Chhabra AB (2010) Singleincision extensile volar approach to the distal radius and concurrent carpal tunnel release: cadaveric study. J Hand Surg Am 35(2): 217-222.






























