Transcriptomic Markers of Motor Neuron Diversity in Development, Aging and Disease
Akshiv Gulati* and Aditya Gulati
Astra Zeneca Pharma India Ltd, India
Submission: May 07, 2024; Published: May 21, 2024
*Corresponding author: Akshiv Gulati, Astra Zeneca Pharma India Ltd, India
How to cite this article: Akshiv G, Aditya G. Transcriptomic Markers of Motor Neuron Diversity in Development, Aging and Disease. Ortho & Rheum Open Access J. 2024; 23(2): 556108. DOI: 10.19080/OROAJ.2024.23.556108
Abstract
Amyotrophic Lateral Sclerosis (ALS) is a neurodegenerative and neuromuscular disease, the primary causes of which are yet to be discovered. It is a debilitating disease that results from the degeneration of the Brain and Spinal Motor Neurons, leading to muscular atrophy -- becoming fatal in 2-3 years from onset. One of the keen observations about the pathogenesis of ALS is the selective resistance some motor neurons (MN) possess to the disease. Notably, the Gamma MNs are resistant to the disease, while Alpha MNs are primarily implicated with the physiological changes that occur during the onset of the disease. To identify the molecular and genetic reasons for this selective vulnerability, various transcriptomic analyses have been conducted and successfully identified the diversity between these MN types, paving the way for a deeper understanding of how ALS pathogenesis occurs. While helpful, these markers provide little to no insight into their relationship to the pathogenesis of ALS. Our systematic meta-analysis of the currently identified transcriptomic markers could extrapolate exciting conclusions about the relationship between various MN markers and their function, expression in specific cell types, and intra-cellular location. Our study found that Alpha MN markers serve essential functions as membrane proteins and Enzymes, while Gamma MN markers serve as nuclear regulators. We also understood that various Alpha MN markers are present exclusively outside the cell, working with membrane proteins, while Gamma MN markers are localized mainly in the nucleus. We also found that various Alpha MN markers are broadly expressed throughout the body, while Gamma MN markers are relatively isolated. Through the study, we were also able to identify what could potentially be the elusive Beta MN. However, further studies are required to characterize the MN. Our primary goal with this study was to isolate currently identified transcriptomic markers and perform statistical and hierarchical clustering analyses on these markers to ascertain their functionality in the MNs and prevalence in specific organ systems to provide a robust classification of transcriptomic markers and their potential implication in the progression of ALS.
Introduction
Amyotrophic Lateral Sclerosis (ALS) is an irreversible adult-onset neurodegenerative disorder that results in the degeneration of the brain and the spinal motor neurons (MNs), leading to muscle atrophy, paralysis, and death. Progressive MN degeneration results in increasing failure of the neuromuscular system, most often becoming fatal due to respiratory failure 2-3 years after onset. ALS is touted to be a primarily sporadic disease, with up to 95% percent of cases resulting from unknown factors and the remaining 5% of cases showing autosomal dominant inheritance. Approximately 20% of the cases resulting in familial ALS inheritance result from a mutation caused by the Cu+/Zn+ superoxide dismutase (SOD1) gene. Mouse models expressing the SOD1 gene expressed similar pathogenesis as human ALS with MN degeneration leading to muscular weakness, ultimately resulting in death. As such, our understanding of ALS pathology has increased through the study of SOD1 ALS models, which have indicated potential mechanisms related to ALS, such as mitochondrial dysfunction, oxidative stress, protein misfolding, and axonal transport dysfunction [1-5].
Further investigations on the physiological impacts of ALS have also revealed the selective degeneration of the Upper Motor Neurons (UMNs) in the primary cortex. In the brainstem, hypoglossal MNs innervate the muscles of the tongue and trigeminal and facial MNs are also affected. However, MN subgroups within the oculomotor, trochlear, and abducens nuclei, which innervate the extraocular muscles, are relatively resistant to degeneration. Studies have revealed that these MNs can degenerate at late stages of the disease; however, that is primarily caused by confounding factors related to the disease [6-10].
In the spinal cord, MNs are organized into columns, which are organized by the innervated muscles. Within each column, MNs innervating each muscle are grouped into motor pools, which could innervate anywhere from 20-300 cells depending on the muscle. Alpha MNs is a subtype of motor neurons that innervate intrafusal muscle fibers, generating forces that control skeletal muscle movement. Gamma MNs innervate intrafusal muscle fibers, which modulate the sensitivity of muscle spindles to stretch. Beta MNs are ambiguous in their properties as they seem to mimic the functional and physiological characteristics of both Alpha and Gamma MNs -- innervating both intrafusal and extrafusal muscle fibers. There are also several physiological differences between Alpha and Gamma MNs, resulting in differences in their functional abilities. Whereas Alpha MNs have large axon sizes -- providing greater conductive effects -- Gamma MNs is typically half the size of Alpha MNs. Furthermore, Alpha MNs tend to have larger, more branched dendritic trees, whereas Gamma MNs is typically less extensive but larger in the size of the dendrites. Additionally, Alpha MNs receive direct IA proprioceptive sensory input, while Gamma MNs do not. Selective degeneration patterns in ALS are however limited to the UMNs [11-12].
In studies conducted on ALS models, it has been noted that selective degeneration of Alpha MNs is implicated with the loss of motor function, while Gamma MNs remains relatively unchanged in models. Furthermore, it has been noted that the degradation of the Fast-type Alpha MNs occurs in the early stages of the disease, while Slow-type Alpha MNs occur throughout the later stages. Functional differences between these MNs and their selective correlation with the onset and progression of ALS pose the question of whether fundamental distinctions in the MNs -- at the genetic level -- contribute to the onset of ALS. Specifically, can transcriptional differences of these MNs contribute to the progression of the disease? If so, can transcriptomic analyses of molecular markers that provide a distinction between these MNs also be implicated in the progression of ALS?
Materials and Methods
This comprehensive literature review assembles motor neuron transcriptomic markers that have been studied to mark and identify motor neuron subtypes. To maintain the fidelity of these experiments, several qualitative measures were ensured, which allowed for the streamlining of the review process. Considering that single-cell transcriptomic approaches have emerged in recent years, we focused on the 20+ years of advancement between 2000 and today. Three databases were used for the review: PubMed, JSTOR, and Google Scholar, and the references for each article were used to find more studies. Our inclusion criteria consisted of a quantitative evaluation of a given study (e.g., gene expression level and fluorescence intensity values) to ensure the identification of high-fidelity markers across studies. We further considered those markers that were identified in more than one study.
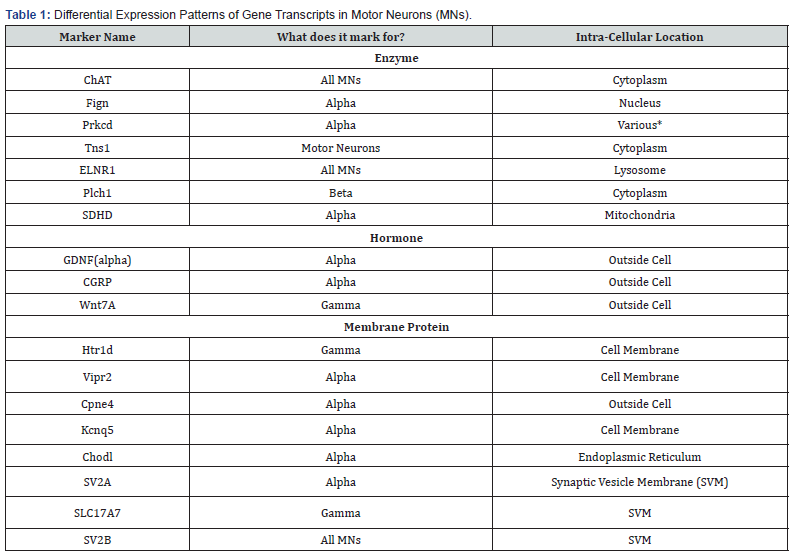
Results
Alpha and Gamma MNs can be distinguished through their expression of unique transcriptomic markers within the cell bodies. Through a systematic review of recent single-cell transcriptomic studies (see Bibliography), we identified 32 molecular markers of Alpha and Gamma MNs, and we further sought to determine commonalities and differences in their expression patterns. We grouped the 32 reliable markers based on the following criteria: 1) motor neuron subtypes, 2) cellular function of the protein which the marker encodes, 3) expression in cell types other than motor neurons, and 4) location within cells (e.g., plasma membrane, nucleus). All this information is presented in Table 1. In what follows, we present a detailed analysis using multi-dimensional data visualization and clustering approaches [12-14].
We first clustered the motor neuron subtypes and the 32 identified markers as presented in the dendrogram in Figure 1A). We found nine markers of Gamma motor neurons, six markers of motor neurons in general, and twelve of Alpha MNs. Within the Alpha motor neurons, five markers marked for the subtypes of Alpha motor neurons, including the fast and slow varieties, as shown in Figure 1B. Surprisingly, we found two putative markers for Beta motor neurons, whose existence and role are not precisely understood.
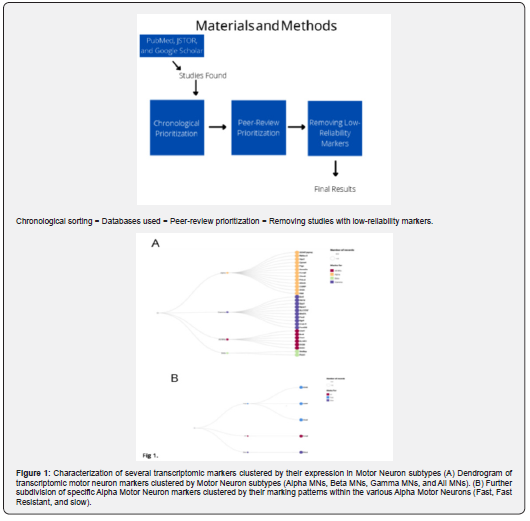
While most transcriptomic studies highlight specific markers that can be used to identify cell types, it is imperative to contextualize the identity of these markers further to further our understanding of their unique relationships. Therefore, we clustered the motor neuron subtype-specific markers based on their intracellular functions: Enzymes, Hormones, Membrane Proteins, Growth Factors ? or Regulators, Signaling Molecules, and Structural Proteins, as shown in the Figure 2A. Further, we created an alluvial diagram to visualize better the relationship between the subtypes of motor neurons and the intracellular functions, as illustrated in Figure 2B. Through a primarily qualitative analysis of the clustering data, we could ascertain a few general patterns of the diversity of function within the marker group and the relationships between MN subtypes and marker function. Upon investigation of the with the general outlay of the data, we can derive some clues regarding the broader function of these markers and study their changes in ALS. We can see that within the categories of function, the markers are most often categorized as membrane proteins, followed by Enzymes -- combined, accounting for more than half of all the markers. The other categories were prevalent but accounted for a smaller proportion of all the markers. Upon investigating the subtypes of motor neurons, we found gamma motor neurons to account for the highest diversity of function, having at least one marker in every function class except for the enzyme class. Additionally, gamma motor neurons account for most of the markers in the regulator class. Followed by gamma motor neurons, the Alpha subtype of markers had the highest diversity.
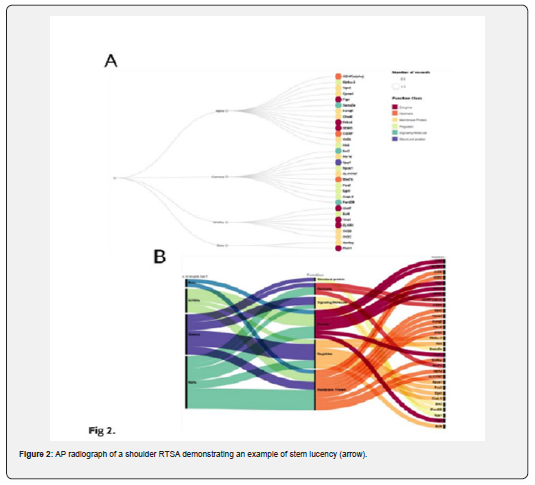
Notwithstanding, Alpha motor neurons were highly concentrated within the membrane protein and enzyme class of functions. Therein, we found that alpha motor neuron markers were most heavily concentrated in the membrane protein class of markers. After alpha motor neurons, we found that the All MNs protein class was the most prevalent. This subtype of marker had a relatively diverse function class and was concentrated within the enzyme class. Finally, beta motor neurons did not have any discernible class of function.
To further extend this analysis, we clustered the markers through their motor neuron markers and their intracellular location. In our analysis, we found various locations within which markers reside. From 17 different categories, we generalized the cellular locations of the markers to 11 to delineate the location into comparable clusters. The data found that beta motor neuron markers were solely present in the cytosol, their domains specifically near the cell membrane, as protein to support potassium channels in the cell. On the other hand, most cell markers had no discernible localization patterns within the cell. Nonetheless, gamma motor neuron markers were localized in the nucleus, which mainly functions as a transcription regulator and is heavily involved with gene expression. Alpha MN markers, the largest subgroup of markers in the review, had a high diversity of function class and were present throughout the cell. Four alpha MN markers were localized in functions outside the cells, either outside the cell when studied or in intracellular
capsules marked for outside secretion. Beta MN markers were also interesting, as their cellular functions were localized to the cytoplasm. Finally, all the MN markers had no discernible patterns of localization.
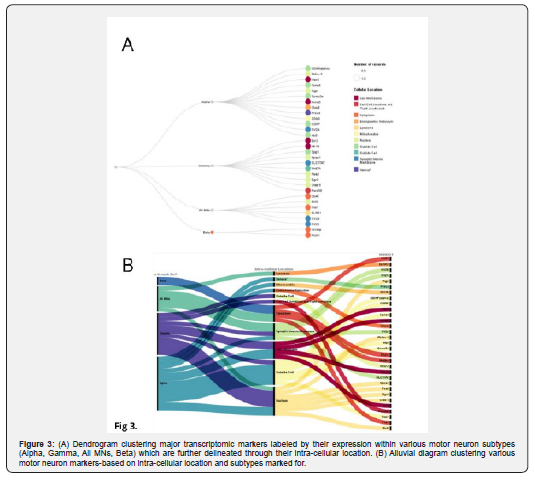
As a final consideration, we also separated the markers by their putative expression in different MN types and mapping onto other cell types in which these markers are most heavily expressed. To determine this expression, we chose a 10% prevalence as a qualifier for abundant expression. After that, we organized the data through a dendrogram and an alluvial diagram to compare the general clusters and subclusters based on cell type to motor neuron type. We also analysed the expression patterns, as shown in Figure 4. Through such visualization and analysis, we can underscore a clear distinction between the expression patterns of the Alpha and Gamma MNs, wherein most Gamma MN markers tend to be highly expressed in cells of the Central Nervous System -- which includes the Brain and the Spinal Cord -- and Skeletal Muscles. On the other hand, Alpha MN markers tend to be highly expressed in other cells throughout the body and thus could be potentially involved in high essential cell functions. Alpha MN markers are also the only ones expressed in all cells. Additionally, we found that most Gamma motor neurons marked for were highly expressed in the Retinal synapses, while Alpha motor neurons have some expression in glial cells. We found that most of the markers identifying general motor neurons were expressed in Skeletal muscles. Finally, the All MNs category had no discernible patterns of expression (Figures 1-4).
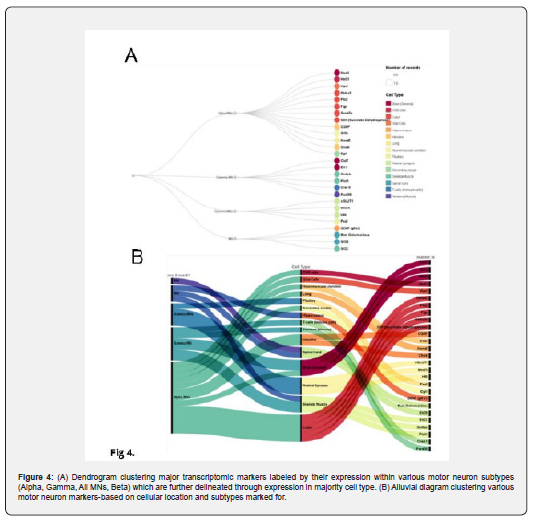
Discussion
Previous research has strongly indicated that various subtypes of motor neurons can be identified from their unique molecular expression through transcriptomic analysis. However, one of the critical goals of this study was to use a literature review and a qualitative meta-analysis of these markers to understand characteristic similarities in various criteria central to the function of the markers's protein products. In doing so, we ventured to provide a wide array of transcriptomic markers to distinguish between motor neuron subtypes and identify these markers as potential indicators that could be selectively vulnerable in neurodegenerative disease. Results from the study indicate that Gamma motor neurons are likely to be part of the nucleus's regulatory functions and, therefore, have a crucial role in gene expression and primary gene transcript regulation. Moreover, studies bolster the tangible electrophysiological changes caused by Gamma MN marker 5-Ht1d, a lack of which produces a tangible decrease in electrophysiological readings of monosynaptic reflexes, hinting at its vital role in proprioceptive regulation of sensitivity. It would be apt to consider whether direct nuclear regulation through 5-Ht1d is responsible for the recorded physiological changes. Additionally, further studies should consider the role of the 5-Ht1D marker in the neuroprotection of Gamma MNs during the progression of neurodegenerative disease.
Various studies have highlighted that Alpha MN possesses a selective vulnerability to degeneration in the pathogenesis of ALS. Another study discusses the selective vulnerability of Alpha MNs during the progression of ALS, mainly due to regulatory problems in maintaining the rate of Acetylcholinesterase (AChE), a molecule particularly active on Acetylcholine receptors on the cell membrane (PDB101: Molecule of the Month: Acetylcholinesterase, nd). In concurrence with these results, our studies indicate membrane proteins include the most significant proportion of Alpha motor neuron markers among all function classes. Further investigations could potentially consider the role of membrane protein class Alpha MN markers in their changes in the physiological regulation of membrane-type proteins during the onset of neurodegenerative disease.
Our results also established that Gamma motor neurons have a predominant intracellular localization in the cell's nucleus. While it was previously mentioned that this may suggest Gamma MN markers' involvement in gene regulation, it is also worth noting that Gamma MNs were present throughout the cell, which calls to question the validity of this localization study. Therefore, conducting a follow-up study to investigate the nuclear locations of Gamma MN markers could be beneficial. Furthermore, a plurality of Alpha MN markers displayed localization to regions outside the cells, particularly in the extracellular matrix, supporting cell structure and function or as additional proteins that play a role in cell membrane function. These results concur with the notion of Alpha MN markers performing similar functions in the cell membrane, suggesting commonalities between the function and location of the markers. Notably, a widely known marker in transcriptomic studies, GDNF, provides muscular trophic feedback to motor neurons, regulating their physiological properties and growth/death. Therein, a lack of GDNF is currently being investigated concerning its role in the death of Alpha MN, particularly during the onset of ALS (Gould et al., 2008). Therefore, for future investigations, it would be worth considering other Alpha MN markers, localized in the cell membrane or outside the cell, regarding changes in their function and expression patterns during the onset and pathogenesis of ALS and other neurodegenerative diseases that display selective vulnerability to motor neurons.
In our analysis of the expression of MN markers in specific cell types, we were able to extrapolate some interesting results regarding the expression of Alpha MN markers. Interestingly, many Alpha MN markers were said to be present in the Cells* category (which describes expression in all cell types throughout the body). Additionally, the Cells* category comprised entirely of Alpha MN markers. Therefore, it can be surmised that the Alpha MN markers tend to possess a function essential to cells throughout the body. Alternatively, they might tend to serve little to no purpose in the body. In further investigations, it would be interesting to explore the function of these cells within Alpha MNs and compare it with other cell types to expound on their potential functions.
Moreover, we found various markers in the Retinal Synapses, which comprised entirely of Gamma MN markers. Additionally, these markers served as ion channels throughout the Retinal synapse, maintaining the proper functioning of the cells involved in vision. Among these markers, we found the marker Wnt7A, a marker heralded as only of the only that can mark for Gamma MNs during the early embryonic period in mice. Additionally, markers such as vGlut1 are being further investigated for their relationship with the degeneration patterns shown during various neurodegenerative diseases. Therefore, it could be interesting to consider these markers and others throughout the Retinal Synapse in their relation to various neurodegenerative diseases. Our study delves into the intricate molecular signatures of motor neuron subtypes, identifying key markers and their potential implications in neurodegenerative diseases. Our findings highlight the importance of further investigating markers like 5-Ht1d in regulating motor neuron
function and neuroprotection. Moreover, the selective vulnerability of Alpha motor neurons in diseases like ALS underscores the significance of exploring membrane protein markers and extracellular matrix components as potential therapeutic targets. Additionally, our analysis suggests broader implications for motor neuron function beyond traditional boundaries, emphasizing the need for further research into the physiological roles of markers in diverse cell types. Furthermore, identifying specific markers within retinal synapses offers new avenues for understanding neurodegenerative diseases and vision-related disorders. Exploring these markers may provide valuable insights into disease mechanisms and therapeutic strategies.
In conclusion, our study provides a comprehensive overview of motor neuron subtype markers and their potential roles in neurodegenerative diseases, paving the way for future investigations and therapeutic interventions.
References
- Ashrafi S, Lalancette-Hebert M, Friese A, Sigrist M, Arber S, et al. (2012) Wnt7A identifies embryonic γ-Motor neurons and Reveals Early Postnatal dependence of γ-Motor neurons on a Muscle Spindle-Derived Signal. J NeuroSci 3(25): 8725-8731.
- Blum J, Klemm S, Shadrach J, Guttenplan K, Nakayama L, et al. (2021) Single-cell transcriptomic analysis of the adult mouse spinal cord reveals molecular diversity of autonomic and skeletal motor neurons. nature neuroscience 24: 572-583.
- Chakkalakal J, Nishimune H, Ruas J, Spiegelman B, Sanes J (2010) Retrograde influence of muscle fibers on their innervation revealed by a novel marker for slow motoneurons. The company of Biologists 137(20): 3489-3499.
- Conradi S, Ronnevi L (2003) Selective vulnerability of alpha motor neurons in ALS: Relation to autoantibodies toward acetylcholinesterase (ache) in als patients. Brain Research Bulletin 30(3): 369-371.
- Enjin A, Leao, K, Mikulovic S, Merre P, Tourtellotte W, et al. (2012) Sensorimotor function is modulated by the serotonin receptor 1d, a Novel marker for gamma motor neurons. 49(3): 322-332.
- Friese A, Kaltschmidt J, Ladle D, Sigrist M, Jessell T, et al. (2009) Gamma and ALPHA motor Neurons distinguished by expression of transcription FACTOR Err3. Proc Natl Acad Sci U S A 106(32): 13588-13593.
- Gould T, Yonemura S, Oppenheim R, Ohmori S, Enomoto H (2008) The neurotrophic effects of glial cell line-derived neurotrophic factor on spinal motoneurons are restricted to fusimotor subtypes. J Neurosci 28(9): 2131-2146.
- Lalancette-Hebert M, Sharma A, Lyashchenko A, Shneider N (2016) Gamma motor neurons survive and Exacerbate ALPHA motor neuron degeneration in ALS. PNAS 113(51): E8316-E8325.
- Landry M, Bouali-Benazzouz R, Mestikawy S, Ravassard P, Nagy F (2003) Expression of vesicular glutamate transporters in rat lumbar spinal cord, with a note on dorsal root ganglia. JCN Research in Systems Neuroscience 468(3): 380-394.
- Morisaki Y, Niikura M, Watanabe M, Onishi K, Tanabe S, et al. (2016) Selective expression of osteopontin in ALS-resistant motor neurons is a critical determinant of late phase Neurodegeneration mediated by Matrix metalloproteinase-9. Sci Rep 6: 27354.
- Nardo G, Iennaco R, Fusi N, Heath P, Marino M, et al. (2013) Transcriptomic indices of fast and slow disease progression in two mouse models of amyotrophic lateral sclerosis. Brain 136(11): 3305-3332.
- Oda Y (2002) Choline acetyltransferase: The structure, distribution and pathologic changes in the central nervous system. Pathology International 49(11): 921-937.
- PDB101: Molecule of the Month: Acetylcholinesterase. (n.d.) (2004).
- Simon A Dalla Torre di Sanguinetto, Dasen J, Arber S (2008) Transcriptional mechanisms Controlling motor NEURON diversity and connectivity. 18(1): 36-43.






























