- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Novel Therapies for Osteoarthritis Treatment: An Overview of Recent Developments
Rajesh Itha1*, Sandhya Bhavani Parabathula2 and Abhinav Aggarwal3
1Department of Orthopaedics, ESIC Model Hospital, Noida, Uttar Pradesh, India
2Department of Rheumatology, Max Super Specialty Hospital, New Delhi, India
3Department of Orthopaedics and Joint Replacement Surgery, Indraprastha Apollo Hospital, New Delhi, India
Submission:August 14, 2023; Published:September 05, 2023
*Corresponding author: Dr. Rajesh Itha, Department of Orthopaedics, ESIC Model Hospital, A-3, Tulsi Marg, Sector 24, Noida, Uttar Pradesh, India
How to cite this article: Rajesh I, Sandhya Bhavani P, Abhinav A. Novel Therapies for Osteoarthritis Treatment: An Overview of Recent Developments. Ortho & Rheum Open Access J. 2023; 22(2): 556081. DOI: 10.19080/OROAJ.2023.22.556081
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Abstract
Background and Aim: Current treatment options can be effective in reducing pain and improving function, but they do not address the disease process of Osteoarthritis (OA). Hence, there is a pressing need for novel therapies that can modify the disease process and slow or halt the progression of OA. We aimed to analyze and synthesize the information from the selected articles to provide an in-depth and comprehensive overview of the latest developments in the field of osteoarthritis treatment.
Purpose and Methods: We conducted a thorough literature search using relevant keywords such as “Osteoarthritis,” “DMOADS,” “novel therapies,” “regenerative medicine,” and “IADDS” on databases including Pubmed, Scopus, and Google Scholar. Our search involved a review of a significant number of peer-reviewed articles, ensuring that the information we gathered was reliable and up to date. To create a summary that truly reflects the current state of knowledge in the field of osteoarthritis treatment, we investigated and compiled the data from the most relevant articles.
Conclusion: Novel therapies, such as DMOADs, gene therapy, and regenerative medicine, are being investigated to modify disease progression. Biomechanical factors are critical in the development of OA, and therapies targeting abnormal joint loading and kinematics are emerging. Furthermore, intra-articular drug delivery systems (IADDS) have emerged as a promising strategy for the targeted and sustained delivery of drugs to the affected joint. Further research is needed to optimize these treatments, and clinical trials have shown moderate evidence of their efficacy. The variability of the illness, the absence of reliable biomarkers, and the requirement for suitable animal models are all challenges. Despite these challenges, the development of novel therapies offers hope for patients with OA.
Keywords: Osteoarthritis; DMOADs; Glucosamine; Chondroitin sulfate; Stem cell; IADDS
Abbreviations: BMPs: Bone Morphogenetic Proteins; DMOADs: Disease-Modifying Osteoarthritis Drugs; FDA: Food and Drug Authority; FGF: Fibroblast Growth Factor; IGF-1: Insulin-Like Growth Factor-1; IL-1Ra: Interleukin-1 Rheumatoid Arthritis; IADDS: Intra-Articular Drug Delivery Systems; MSCs: Mesenchymal Stem Cells NGF: Nerve Growth Factor; NSAIDs: NonSteroidal Anti-Inflammatory Drugs; PRP: Platelet-Rich Plasma RCTs: Randomized Controlled Trials; rhIGF: Recombinant Human Insulin-Like Growth Factor; rhTGF: Recombinant Human Transforming Growth Factor; ROS: Reactive Oxygen Species; TGF: Transforming Growth Factor; TGF-β: Transforming Growth Factor-Beta; TNF: Tumour Necrosis Factor; TJR: Total Joint Replacement; WHO: World Health Organization
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Introduction
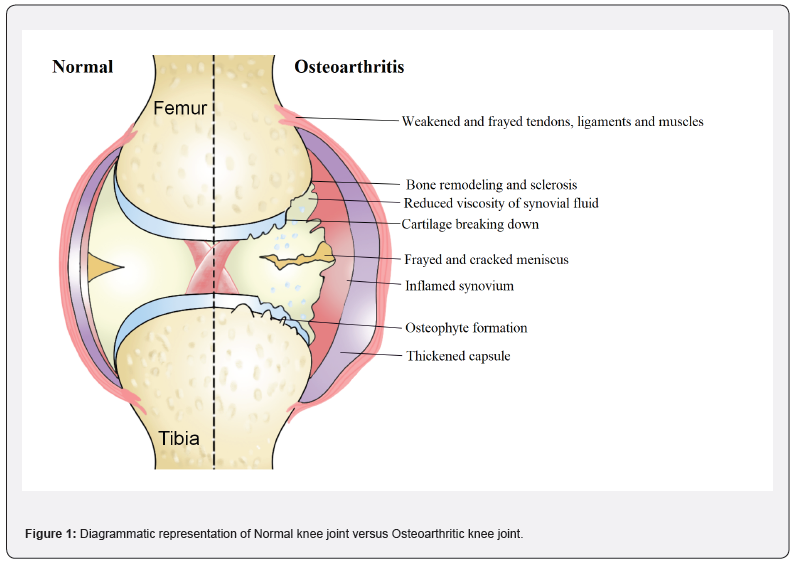
Osteoarthritis (OA) is a prevalent and debilitating condition that affects people worldwide. According to the World Health Organization, OA is one of the ten most disabling diseases in developed countries (WHO, 2021). The disease is characterized by the gradual loss of articular cartilage, subchondral bone sclerosis, synovial inflammation, and hypertrophy [1]. These changes lead to joint pain, stiffness, and decreased mobility, which can significantly impact the quality of life of affected individuals [2]. Although there is no cure for OA, there are several treatment options available that can help manage the symptoms and improve quality of life for patients.
A multidisciplinary approach is typically used to manage OA, which includes nonpharmacological interventions such as exercise, weight loss, and physical therapy, medications such as nonsteroidal anti-inflammatory drugs (NSAIDs), and surgery such as joint replacement [3]. While these treatment options can be effective in reducing pain and improving function, they do not address the disease process and, therefore, do not slow or halt the progression of joint damage [4]. Therefore, there is a crucial requirement for novel therapies that can alter the course of the illness and stop or slow the development of OA. These therapies may include disease-modifying drugs that target the underlying pathophysiology of OA [5]. Several potential targets for disease-modifying drugs have been identified, including enzymes that degrade cartilage and proteins that contribute to joint inflammation [6].
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Current treatment options
OA management often involves non-pharmacological interventions like exercise and weight loss to reduce joint stress and improve function [7]. The American College of Rheumatology recommends aerobic and resistance exercise for all OA patients and weight loss for those who are overweight or obese [8]. Exercise has been shown to reduce pain, improve physical function, and enhance mental health outcomes [9]. Weight loss is also crucial since excess weight can lead to additional joint stress, increased pain, and functional impairment [10]. Physical therapy with individualized exercise programs and assistive devices can also help [11].
If non-pharmacological interventions are insufficient, pharmacological interventions like NSAIDs and analgesics can be used to alleviate pain [12]. NSAIDs reduce inflammation, but they have side effects such as gastrointestinal bleeding and cardiovascular risks [13]. Acetaminophen is an option for OA pain relief, but it may not be as effective as NSAIDs and can cause liver damage [14]. Topical agents like capsaicin cream or topical NSAIDs can be used for localized pain relief [4]. Another class of medications used for OA management is disease-modifying osteoarthritis drugs (DMOADs). These medications are thought to slow the progression of joint damage and improve symptoms. The most used DMOADs are glucosamine and chondroitin sulfate [12]. Glucosamine is a natural compound found in joint cartilage, and chondroitin sulfate is a component of cartilage and other connective tissues [15]. Several studies have investigated the effectiveness of glucosamine and chondroitin sulfate for OA management. A meta-analysis of 10 randomized controlled trials found that glucosamine reduced pain and improved function in patients with knee OA [16]. However, the evidence for chondroitin sulfate was less clear, with some studies showing a benefit and others showing no effect [17,18]. More recently, a randomized, double-blind, non-inferiority trial found that the combination of chondroitin sulfate and glucosamine was as effective as the NSAID celecoxib for treating painful knee OA [19].
Surgery is sometimes necessary for OA management, alongside pharmacological and nonpharmacological interventions. Total joint replacement (TJR) is a common surgical option for advanced OA and can significantly reduce pain and improve function. In the United States, about 700,000 total knee replacements and 400,000 total hip replacements are performed annually [20]. However, TJR is a major surgical procedure that carries risks of complications like infection, blood clots, and implant failure. Furthermore, the procedure is expensive, with a cost of around $28,000 to $30,000 per surgery [21]. Therefore, it is generally recommended for patients who have not responded to other treatments.
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
The Need for Novel Therapies, Including Disease-Modifying Drugs
Currently, the main treatments for OA aim to alleviate pain and reduce inflammation, but they do not modify the underlying degenerative changes in the joint, thus they don’t alter the disease’s course. While no FDA-approved diseasemodifying drugs for OA exist, some classes of drugs such as disease-modifying osteoarthritis drugs (DMOADs) have shown potential in preclinical and clinical studies. These drugs target the pathophysiological processes underlying OA and may slow or stop joint degradation [22]. There are various DMOADs under investigation, including those targeting bone metabolism, cartilage metabolism, and inflammation, with some showing promising results in preclinical and early clinical studies. For instance, bisphosphonates used in treating osteoporosis have been shown to decrease cartilage degradation and enhance subchondral bone density in animal models of OA [23]. Another potential class of drugs is growth factors like insulin-like growth factor-1 (IGF-1), which stimulate chondrocyte proliferation and increase the synthesis of extracellular matrix components in vitro [24]. Gene therapy is another innovative treatment approach for OA, as several genes that participate in the disease’s development and progression have been identified, and manipulating their expression using gene therapy could help slow the disease’s course [25].
The potential of employing mesenchymal stem cells (MSCs) to treat OA is still being researched. MSCs have the ability to differentiate into chondrocytes and potentially regenerate damaged cartilage, making them a promising therapy for OA [26]. Currently, there are several clinical trials being conducted to assess the safety and efficacy of MSCs for OA treatment [27].
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
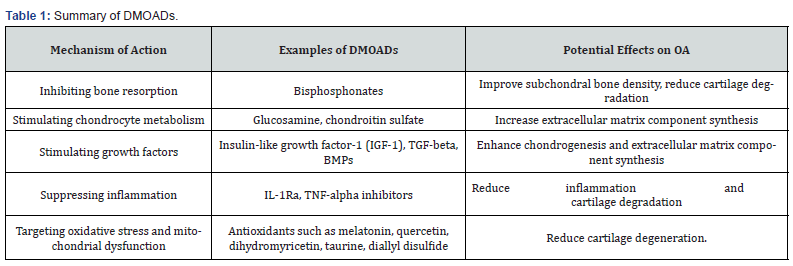
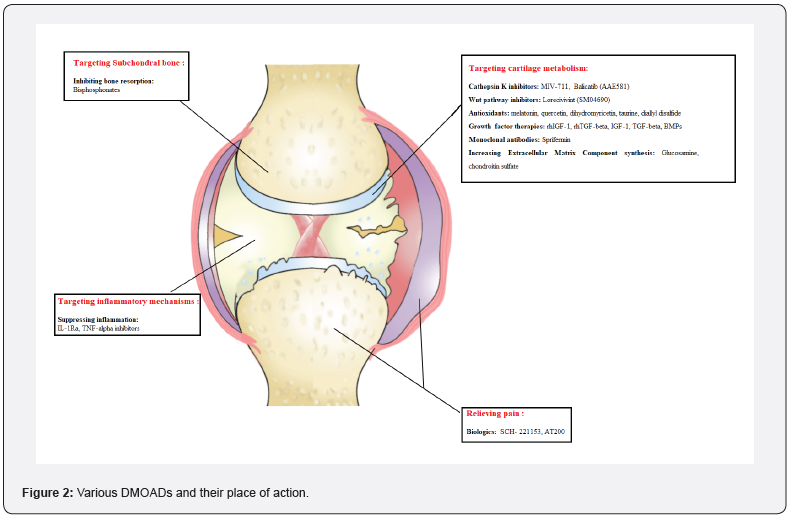
Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
DMOADs can have various mechanisms of action that target multiple aspects of the OA disease process, such as bone metabolism. OA induces changes in the subchondral bone, such as increased turnover, sclerosis, and bone mineral density alterations [28].
Bisphosphonates, which are commonly used to treat osteoporosis, have demonstrated potential as DMOADs as well.
They can inhibit bone resorption by osteoclasts, increase bone mineral density, and reduce subchondral bone turnover [29]. In animal models of OA, bisphosphonates have been shown to improve subchondral bone density and reduce cartilage degradation [30]. Another potential area for DMOADs is cartilage metabolism, as OA causes the progressive loss of articular cartilage, which mainly consists of proteoglycans and type II collagen. Chondrocytes, which produce and maintain cartilage, undergo changes in response to OA, including altered matrix synthesis, increased apoptosis, and decreased proliferation [31].
Several agents have been studied for their potential to modify cartilage metabolism in OA, including widely used agents such as glucosamine and chondroitin sulfate, which are believed to stimulate chondrocyte metabolism and increase the synthesis of extracellular matrix components, such as type II collagen and proteoglycans [32]. While evidence supporting their efficacy for managing OA symptoms is mixed, there is some evidence to suggest that they may have disease-modifying effects [33].
Growth factors, particularly insulin-like growth factor-1 (IGF- 1), have shown promise in stimulating chondrocyte proliferation and increasing extracellular matrix component synthesis in vitro [34]. Multiple studies have examined the effectiveness of IGF-1 for OA treatment, yielding mixed results [35]. Other growth factors, including TGF-beta and BMPs, have also demonstrated potential as DMOADs by enhancing chondrogenesis and extracellular matrix component synthesis [36]. Inflammation is a crucial target for DMOADs, as synovitis is a common feature of OA and associated with disease severity [37]. Although NSAIDs are commonly used to manage OA-associated pain and inflammation, there is limited evidence to support their disease-modifying effects [33]. Agents such as IL-1Ra and TNF-alpha inhibitors have exhibited potential as DMOADs by suppressing inflammation and cartilage degradation. In multiple trials, IL1Ra, a protein that naturally inhibits the pro-inflammatory cytokine IL-1 implicated in the pathophysiology of OA, has shown to reduce pain and enhance function TNF-alpha inhibitors, which target another proinflammatory cytokine involved in OA pathogenesis, have also shown potential as DMOADs [39].
Mixed results have been reported from several studies investigating the use of TNF-alpha inhibitors as DMOADs for the treatment of OA [22]. In addition, targeting oxidative stress and mitochondrial dysfunction may also hold promise as an approach for developing DMOADs. Reactive oxygen species (ROS) and mitochondrial dysfunction have been implicated in the pathogenesis of OA, and the use of antioxidants and mitochondrial-targeted agents has been investigated in several studies [40]. These studies have demonstrated that antioxidants like melatonin, quercetin, dihydromyricetin, taurine, and diallyl disulfide might potentially be used to treat OA [41].
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
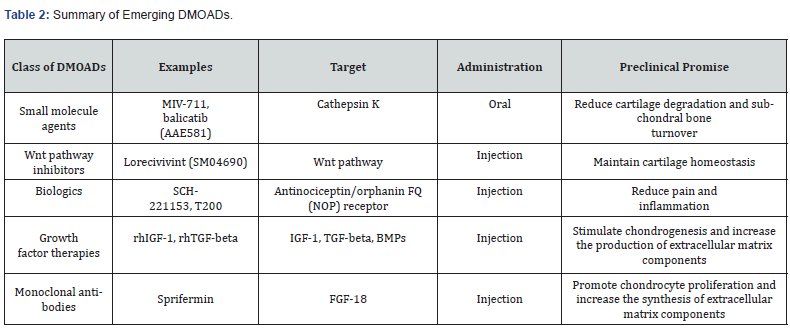
Emerging DMOADs: Small Molecule Agents and Biologics
Small molecule agents, which have a low molecular weight and can be orally administered, represent one class of small molecule DMOADs that has demonstrated promise in preclinical investigations. Among these small molecule DMOADs, cathepsin K inhibitors have demonstrated promise in preliminary research. Cathepsin K is a protease that is involved in the breakdown of type II collagen, a vital constituent of articular cartilage. Inhibition of cathepsin K has been shown to reduce cartilage degradation and subchondral bone turnover in animal models of OA [42]. Several cathepsin K inhibitors, including MIV-711 and balicatib (AAE581), are currently undergoing clinical development [43,44].
The Wnt pathway plays a crucial role in regulating chondrocyte differentiation and maintaining cartilage homeostasis, and its dysregulation has been linked to OA pathogenesis [45]. Lorecivivint (SM04690) is a Wnt pathway inhibitor currently in clinical development for the treatment of OA [46]. Biologics are drugs derived from living organisms and usually require injection. Antinociceptin/orphanin FQ (NOP) receptor agonists are a type of biologic DMOADs that have demonstrated potential in preclinical studies. The NOP receptor is a G protein-coupled receptor that participates in regulating pain and inflammation. Activation of the NOP receptor has been shown to reduce pain and inflammation in animal models of OA [47]. Several NOP receptor agonists, such as SCH-221153 and AT-200, are currently undergoing clinical development for the treatment of OA [43].
Growth factor therapies are another class of biologic DMOADs. As previously mentioned, growth factors such as insulin-like growth factor-1 (IGF-1), transforming growth factorbeta (TGF-beta), and bone morphogenetic proteins (BMPs) have shown promise as DMOADs by stimulating chondrogenesis and increasing the production of extracellular matrix components [45]. Currently, several growth factor therapies, including recombinant human IGF-1 (rhIGF1) and recombinant human TGF-beta (rhTGFbeta), are under investigation in clinical trials for the treatment of OA [43].
Monoclonal antibodies (mAbs) are biologics that can selectively target specific proteins involved in the pathogenesis of OA. An example of a mAb DMOAD is sprifermin, which is a recombinant human fibroblast growth factor 18 (FGF-18). FGF- 18 has been demonstrated to promote chondrocyte proliferation and increase the synthesis of extracellular matrix components in vitro and in animal models of OA [48]. In a Phase II clinical trial, sprifermin showed significant cartilage thickening and reduced cartilage loss in patients with knee OA [49].
Numerous clinical trials have examined the efficacy of mesenchymal stem cells (MSCs) and chondrocytes for treating OA. In a double-blind, placebo-controlled study, intra-articular injection of autologous adipose-derived MSCs led to noteworthy enhancements in pain, function, and quality of life in patients with knee OA compared to placebo treatment [50]. Another study found that the injection of autologous chondrocytes resulted in significant improvements in pain and function in knee OA patients [51]. These findings suggest that cells-based therapies have the potential to slow or halt the progression of OA and improve patient outcomes.
Regenerative medicine encompasses various approaches to encourage tissue repair and regeneration, such as tissue engineering and the use of growth factors and other biomolecules that promote tissue regeneration. Tissue engineering involves utilizing biomaterials, cells, and growth factors to create functional tissue constructs that can be implanted into the body to replace damaged or diseased tissues. Numerous preclinical studies have evaluated the use of tissue-engineered cartilage constructs for the treatment of OA, yielding promising results [52,53].
Promising biomolecules for the treatment of OA include growth factors like platelet-rich plasma (PRP) and growth hormone. PRP contains various cytokines and growth factors that can promote tissue regeneration, and multiple studies have explored its efficacy for OA treatment. A meta-analysis of 29 randomized controlled trials found PRP to significantly reduce pain and improve function in knee OA patients compared to control treatments [54]. Similarly, studies have suggested that growth hormone may have the potential to slow OA progression and promote cartilage regeneration [55].
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
Targeted and sustained drug delivery to the affected joint is a promising approach for the treatment of OA, and intra-articular drug delivery systems (IADDS) have emerged as an effective strategy to achieve this goal. There are two main types of IADDS: particulate and non-particulate. Particulate IADDS, including micro- and nanoparticles, liposomes, and dendrimers, are designed to encapsulate drugs and protect them from degradation and clearance. Non-particulate IADDS, such as hydrogels, are crosslinked networks of polymers that can absorb and retain large amounts of water, enabling sustained drug release [3].
Several drugs such as including nonsteroidal antiinflammatory drugs (NSAIDs), corticosteroids, and diseasemodifying osteoarthritis drugs (DMOADs) have been studied for the treatment of OA using IADDS. Although NSAIDs are frequently used to alleviate pain and inflammation in OA, their systemic administration can lead to undesirable effects. The use of IADDS can provide localized delivery of NSAIDs, resulting in lower systemic exposure and fewer side effects. A clinical study found that a diclofenac-loaded polymeric nanoparticle formulation delivered intra-articularly provided superior pain relief and functional improvement compared to conventional diclofenac injections [56].
Although corticosteroids are frequently utilized to alleviate pain and inflammation in individuals with osteoarthritis, their widespread use can produce negative effects that are comparable to those seen with NSAIDs. One possible solution to this problem is the use of intra-articular drug delivery systems (IADDS), which enable corticosteroids to be administered locally and for a longer period, reducing systemic exposure and minimizing adverse effects. In a randomized controlled study, patients who received an intraarticular injection of a sustained-release triamcinolone acetonide hydrogel exhibited significant improvements in pain and function compared to those who received a conventional triamcinolone acetonide injection [57].
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Biomechanics and the Development of Novel Therapies for Osteoarthritis
Biomechanical factors, including abnormal joint loading and altered joint kinematics, have been linked to the onset and progression of osteoarthritis (OA). Understanding the biomechanics of OA can lead to the development of novel therapies that target these factors and could potentially delay or stop the disease’s progression.
Studies have revealed that abnormal joint loading, like elevated compressive or shear forces on the joint, contributes to the development and advancement of osteoarthritis. One promising approach to mitigate this issue is to employ external braces or orthoses, which can change joint alignment and distribute forces across the joint. For example, in a randomized controlled trial [58], the use of a valgus knee brace showed a significant decrease in pain and improvement in function in patients suffering from medial compartment knee OA. Another effective technique to decrease abnormal joint loading is to utilize footwear with specialized insoles that can redistribute forces across the foot and lower extremity. In a randomized controlled trial [59], patients with knee OA who used a shoe with a contoured insole showed a significant reduction in pain and an improvement in function.
Altered joint kinematics, such as changes in joint range of motion or muscle activation patterns, have been identified as another factor that contributes to the development and progression of osteoarthritis. To restore normal joint kinematics, one effective strategy is to implement physical therapy or exercise programs that focus on specific muscle groups and joint movements. For example, a 12-week exercise program that targeted knee extensor strength and proprioception was found to significantly reduce pain and enhance function in patients with knee OA in a randomized controlled trial [60]. Another strategy for restoring normal joint kinematics is to consider surgical interventions, such as osteotomy or joint replacement, that can realign the joint and restore normal biomechanics. In a randomized controlled trial, patients who underwent high tibial osteotomy for medial compartment knee OA demonstrated significantly greater improvements in pain and function compared to those who received non-surgical treatment [61].
Apart from external braces, footwear, exercise, and surgical procedures, researchers are investigating other novel therapies that target biomechanical factors in osteoarthritis. One such therapy involves the use of injectable hydrogels that can restore the mechanical properties of articular cartilage and reduce friction in the joint. In a preclinical study, the injection of a hyaluronic acid hydrogel into the knee joint of rabbits with OA resulted in significant improvements in cartilage thickness and histology compared to saline injection [62]. Another promising therapy is the use of tissue engineering approaches that can regenerate damaged cartilage and restore normal joint biomechanics. In a preclinical study, the injection of a hydrogel scaffold seeded with chondrocytes into the knee joint of rats with OA led to significant improvements in cartilage structure and mechanics compared to saline injection [63].
Clinical Trials and Evidence Supporting the Use of Novel Therapies and DMOADs Clinical trials play a crucial role in assessing the safety and effectiveness of new therapies and disease-modifying drugs for osteoarthritis (OA). These trials generate evidence that is vital for obtaining regulatory approval and clinical acceptance of novel treatments.
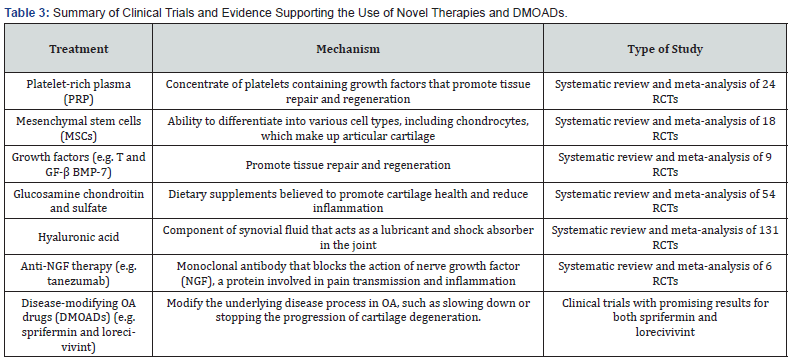
i. Platelet-rich plasma (PRP) is a concentrate of platelets that contains growth factors, which can promote tissue repair and regeneration. There have been several randomized controlled trials (RCTs) evaluating the efficacy of PRP for knee OA, with mixed results. A systematic review and meta-analysis of 24 RCTs found that PRP had a moderate effect on reducing pain and improving function in knee OA patients, but with a small effect size and low-quality evidence [54]. Another systematic review and meta-analysis of 14 RCTs found that PRP was more effective than hyaluronic acid injections for pain reduction and function improvement in knee OA patients [64].
ii. Mesenchymal stem cells (MSCs) can differentiate into various cell types, including chondrocytes, which make up articular cartilage. Several clinical trials have evaluated the safety and efficacy of MSCs for knee OA treatment. MSCs had a moderate impact on function and pain reduction in knee OA patients, according to a systematic review and meta-analysis of 18 RCTs, however the evidence was of low to moderate quality [65].
iii. Growth factors, such as transforming growth factorbeta (TGF-β) and bone morphogenetic protein-7 (BMP-7), can promote tissue repair and regeneration. Several clinical trials have evaluated the safety and efficacy of growth factors for knee OA treatment. A systematic review and meta-analysis of 9 RCTs found that growth factors had a moderate effect on reducing pain and improving function in knee OA patients, but with low to moderate-quality evidence [66].
iv. Glucosamine and chondroitin sulfate are dietary supplements that are believed to promote cartilage health and reduce inflammation. Several clinical trials have evaluated the efficacy of these supplements for knee OA treatment. A systematic review and meta-analysis of 54 RCTs found that glucosamine and chondroitin sulfate had a small but significant effect on reducing pain and improving function in knee OA patients, but with a small effect size and low to moderate-quality evidence [67].
v. Hyaluronic acid is a component of synovial fluid that acts as a lubricant and shock absorber in the joint. Several clinical trials have evaluated the efficacy of hyaluronic acid for knee OA treatment. A systematic review and meta-analysis of 131 RCTs found that hyaluronic acid had a moderate effect on reducing pain and improving function in knee OA patients, but with a small effect size and low to moderate-quality evidence [68].
vi. Anti-NGF therapy, such as tanezumab, is a monoclonal antibody that can block the action of nerve growth factor (NGF), a protein involved in pain transmission and inflammation. Several clinical trials have evaluated the safety and efficacy of tanezumab for knee OA treatment. A systematic review and meta-analysis of 6 RCTs found that tanezumab had a moderate effect on reducing pain and improving function in knee OA patients, but with low to moderate-quality evidence [69].
vii. Disease-modifying OA drugs (DMOADs) can modify the underlying disease process in OA, such as slowing down or stopping the progression of cartilage degeneration. Several DMOADs are currently being developed, including sprifermin, a recombinant human fibroblast growth factor-18 (FGF-18), and lorecivivint, a small molecule inhibitor of the Wnt signalling pathway. Clinical trials of sprifermin have shown promising results in terms of cartilage thickness and reduction in cartilage loss [70]. Clinical trials of lorecivivint have also shown promising results in terms of reducing cartilage loss and improving clinical outcomes [71].
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
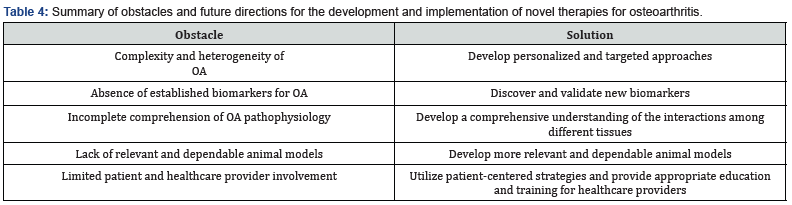
The development of novel therapies for osteoarthritis (OA) has faced significant challenges despite recent advances. One of the major obstacles is the complexity and heterogeneity of the disease, which presents in various phenotypes involving localized and systemic inflammation, synovitis, and cartilage damage [3]. This diversity poses difficulties in developing therapies that can effectively address all aspects of the disease. Thus, there is a need for more personalized and targeted approaches to address the unique requirements of individual patients.
In addition, the absence of established biomarkers for OA presents another obstacle in the development of effective therapies. Biomarkers can offer valuable insight into the pathogenesis of OA and can be utilized to monitor treatment efficacy and disease advancement. Nevertheless, currently available biomarkers do not comprehensively represent all aspects of OA [72]. Hence, it is imperative to discover and validate new biomarkers for OA to facilitate the development and successful implementation of effective therapies.
A more thorough comprehension of the underlying pathophysiology of OA is essential for the successful development and implementation of novel therapies. While cartilage degeneration is a characteristic feature of OA, recent research has revealed the significance of other tissues, including subchondral bone, synovium, and meniscus, in the onset and advancement of the disease [73]. Therefore, a comprehensive understanding of the intricate interactions among these tissues is necessary to devise effective therapies that can address all aspects of OA. Moreover, the use of appropriate animal models that can faithfully replicate the human disease is imperative for developing novel therapies for OA. However, at present, there is a paucity of animal models that can fully mimic the multifaceted and complex nature of human OA [74]. Consequently, it is essential to develop more relevant and dependable animal models that can facilitate the successful translation of novel therapies from preclinical investigations to clinical trials.
Finally, the effective implementation of new therapies for OA necessitates the active participation of patients and healthcare providers in the development process. Patient centered strategies that consider the distinctive requirements and choices of each patient are crucial for the creation of tailored and efficient therapies [75]. Moreover, healthcare providers must receive appropriate education and training on the application of novel therapies to guarantee their secure and effective utilization in clinical settings.
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Conclusion
Osteoarthritis (OA) is a prevalent and debilitating chronic degenerative joint disease that affects millions of people worldwide, and there is currently no cure for it. The need for innovative therapeutics that can alter the course of the disease and stop joint degeneration is urgent even though present treatments help manage symptoms. Promising approaches currently under investigation include disease-modifying osteoarthritis drugs (DMOADs), gene therapy, stem cell therapy, cell-based therapies, and regenerative medicine. Additionally, intraarticular drug delivery systems (IADDS) can provide targeted and sustained delivery of drugs to the affected joint, increasing their efficacy and reducing adverse effects. Biomechanical factors play a critical role in the development and progression of OA, and understanding these factors can lead to the development of novel therapies that target abnormal joint loading and altered joint kinematics. Current and emerging therapies include external braces and footwear, exercise programs, surgical procedures, injectable hydrogels, and tissue engineering approaches. However, further research is needed to optimize the design and formulation of these therapies and identify the most effective treatments for patients with OA. Additionally, while clinical trials and evidence support the use of certain novel therapies and DMOADs for the treatment of OA, the quality of evidence is often low to moderate, and more rigorous research is needed to fully understand the safety and efficacy of these therapies. Despite these challenges, the development of novel therapies and DMOADs offers hope for patients with OA who currently have limited treatment options.
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Acknowledgment
We are extremely thankful to Dr. Natasha Puthalath (Resident Doctor, ESIC model hospital, NOIDA, India) for drawing pictures for this review article.
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Authors’ contribution
RI: Conceptualization, writing, revision, and final approval; SP: Conceptualization, revision and final approval; AA: Literature search, revision and final approval.
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Ethical approval
Not required, being a narrative review and not involving any patient’s data or identity.
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Financial support and sponsorship
Nil.
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
Conflicts of interest
There are no conflicts of interest.
- Review Article
- Abstract
- Introduction
- Current treatment options
- The Need for Novel Therapies, Including Disease- Modifying Drugs
- Disease-Modifying Osteoarthritis Drugs (DMOADs): Overview and Mechanisms of Action
- Emerging DMOADs: Small Molecule Agents and Biologics
- Intra-articular Drug Delivery Systems for Osteoarthritis Therapy
- Biomechanics and the Development of Novel Therapies for Osteoarthritis
- Challenges and Future Directions for the Development and Implementation of Novel Therapies for Osteoarthritis
- Conclusion
- Acknowledgment
- Authors’ contribution
- Ethical approval
- Financial support and sponsorship
- Conflicts of interest
- References
References
- Goldring MB, Marcu KB (2009) Cartilage homeostasis in health and rheumatic diseases. Arthritis Res Ther 11(3): 224.
- Bijlsma JW, Berenbaum F, Lafeber FP (2011) Osteoarthritis: an update with relevance for clinical practice. Lancet 377(9783): 2115-2126.
- Hunter DJ, Bierma-Zeinstra S (2019) Osteoarthritis. Lancet 393(10182): 17451759.
- Zhang W, Moskowitz RW, Nuki G, Abramson S, Altman RD, et al. (2008) OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. Osteoarthritis Cartilage 16(2): 137-162.
- Loeser RF, Goldring SR, Scanzello CR, Goldring MB (2012) Osteoarthritis: a disease of the joint as an organ. Arthritis Rheum 64(6): 1697-707.
- Goldring MB, Berenbaum F (2015) Emerging targets in osteoarthritis therapy. Curr Opin Pharmacol 22: 51-63.
- Skou ST, Roos EM (2017) Good Life with osteoArthritis in Denmark (GLA:D™): evidencebased education and supervised neuromuscular exercise delivered by certified physiotherapists nationwide. BMC Musculoskelet Disord 18(1): 72.
- Kolasinski SL, Neogi T, Hochberg MC, Oatis C, Guyatt G (2020) 2019 American College of Rheumatology/Arthritis Foundation Guideline for the Management of Osteoarthritis of the Hand, Hip, and Knee. Arthritis Care Res (Hoboken) 72(2): 149-162.
- Bartels EM, Juhl CB, Christensen R, Hagen KB, Danneskiold-Samsoe B, et al. (2016) Aquatic exercise for the treatment of knee and hip osteoarthritis. Cochrane Database Syst Rev 3(3): CD005523.
- Vincent HK, Heywood K, Connelly J, Hurley RW (2012) Obesity and weight loss in the treatment and prevention of osteoarthritis. PM R 4(5 Suppl): S59-67.
- Deyle GD, Henderson NE, Matekel RL, Ryder MG, Garber MB, et al. (2000) Effectiveness of manual physical therapy and exercise in osteoarthritis of the knee. A randomized, controlled trial. Ann Intern Med 132(3): 173-181.
- Hochberg MC, Altman RD, April KT, Benkhalti M, Guyatt G, et al. (2012) American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res (Hoboken) 64(4): 465-474.
- Singh G (2000) Gastrointestinal complications of prescription and over-the-counter nonsteroidal anti-inflammatory drugs: a view from the ARAMIS database. Arthritis, Rheumatism, and Aging Medical Information System. Am J Ther 7(2): 115-121.
- Lee WM (2004) Acetaminophen and the U.S. Acute Liver Failure Study Group: lowering the risks of hepatic failure. Hepatology 40(1): 6-9.
- Reichenbach S, Sterchi R, Scherer M, Trelle S, Burgi E, et al. (2007) Meta-analysis: chondroitin for osteoarthritis of the knee or hip. Ann Intern Med 146(8): 580-590.
- de los Reyes GC, Koda RT, Lien EJ (2000) Glucosamine and chondroitin sulfates in the treatment of osteoarthritis: a survey. Prog Drug Res 55: 81-103.
- Bourgeois P, Chales G, Dehais J, Delcambre B, Kuntz JL, et al. (1998) Efficacy and tolerability of chondroitin sulfate 1200 mg/day vs chondroitin sulfate 3 x 400 mg/day vs placebo. Osteoarthritis Cartilage A: 25-30.
- Clegg DO, Reda DJ, Harris CL, Klein MA, O'Dell JR, et al. (2006) Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med 354(8): 795-808.
- Singh JA, Noorbaloochi S, MacDonald R, Maxwell LJ (2015) Chondroitin for osteoarthritis. Cochrane Database Syst Rev 1(1): CD005614.
- Shichman I, Roof M, Askew N, Nherera L, Rozell JC, et al. (2023) Projections and Epidemiology of Primary Hip and Knee Arthroplasty in Medicare Patients to 2040-2060. JB JS Open Access 8(1): e22.00112.
- Roche M, Law TY, Kurowicki J, Rosas S, Rush AJ 3rd (2018) Effect of Obesity on Total Knee Arthroplasty Costs and Revision Rate. J Knee Surg 31(1): 38-42.
- Kapoor M, Martel-Pelletier J, Lajeunesse D, Pelletier JP, Fahmi H (2011) Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol 7(1): 33-42.
- Lane NE, Schnitzer TJ, Birbara CA, Mokhtarani M, Shelton DL, et al. (2010) Tanezumab for the treatment of pain from osteoarthritis of the knee. N Engl J Med 363(16): 1521-131.
- Sanchez C, Deberg MA, Bellahcene A, Castronovo V, Msika P, et al. (2008) Phenotypic characterization of osteoblasts from the sclerotic zones of osteoarthritic subchondral bone. Arthritis Rheum 58(2): 442-455.
- Grassel S, Muschter D (2020) Recent advances in the treatment of osteoarthritis. F1000Res 9: F1000.
- Zhu C, Wu W, Qu X (2021) Mesenchymal stem cells in osteoarthritis therapy: a review. Am J Transl Res 13(2): 448-461.
- Molnar V, Pavelic E, Vrdoljak K, Cemerin M, Emil Klaric et al. (2022) Mesenchymal Stem Cell Mechanisms of Action and Clinical Effects in Osteoarthritis: A Narrative Review. Genes (Basel) 13(6): 949.
- Van Spil WE, Szilagyi IA (2020) Osteoarthritis year in review 2019: biomarkers (biochemical markers). Osteoarthritis Cartilage 28(3): 296-315.
- Li X, Qin L, Bergenstock M, Bevelock LM, Novack DV, et al. (2007) Parathyroid hormone stimulates osteoblastic expression of MCP-1 to recruit and increase the fusion of pre/osteoclasts. J Biol Chem 282(45): 33098-330106.
- Eriksen EF, Shabestari M, Ghouri A, Conaghan PG (2021) Bisphosphonates as a treatment modality in osteoarthritis. Bone 143: 115352.
- Loeser RF (2013) Aging processes and the development of osteoarthritis. Curr Opin Rheumatol 25(1): 108-113.
- Hochberg MC, Martel-Pelletier J, Monfort J, Moller I, Castillo JR, Arden N, et al. (2016) Combined chondroitin sulfate and glucosamine for painful knee osteoarthritis: a multicentre, randomised, double-blind, non-inferiority trial versus celecoxib. Ann Rheum Dis 75(1): 37-44.
- McAlindon TE, Bannuru RR, Sullivan MC, Arden NK, Berenbaum F, et al. (2014) OARSI guidelines for the non-surgical management of knee osteoarthritis. Osteoarthritis Cartilage 22(3): 363-388.
- Sanchez C, Deberg MA, Piccardi N, Msika P, Reginster JY, et al. (2005) Subchondral bone osteoblasts induce phenotypic changes in human osteoarthritic chondrocytes. Osteoarthritis Cartilage 13(11): 988-997.
- Fortier LA, Barker JU, Strauss EJ, McCarrel TM, Cole BJ (2011) The role of growth factors in cartilage repair. Clin Orthop Relat Res 469(10): 2706-2715.
- Thielen NGM, van der Kraan PM, van Caam APM (2019) TGFβ/BMP Signaling Pathway in Cartilage Homeostasis. Cells 8(9): 969.
- Scanzello CR, Goldring SR (2012) The role of synovitis in osteoarthritis pathogenesis. Bone 51(2): 249-257.
- Chevalier X, Goupille P, Beaulieu AD, Burch FX, Bensen WG, et al. (2009) Intraarticular injection of anakinra in osteoarthritis of the knee: a multicenter, randomized, double-blind, placebo-controlled study. Arthritis Rheum 61(3): 344-352.
- Berenbaum F, Meng QJ (2016) The brain-joint axis in osteoarthritis: nerves, circadian clocks and beyond. Nat Rev Rheumatol 12(9): 508-516.
- Henrotin Y, Kurz B, Aigner T (2005) Oxygen and reactive oxygen species in cartilage degradation: friends or foes? Osteoarthritis Cartilage 13(8): 643-654.
- Mao X, Fu P, Wang L, Xiang C (2020) Mitochondria: Potential Targets for Osteoarthritis. Front Med 7: 581402.
- Lindstrom E, Rizoska B, Tunblad K, Edenius C, Bendele AM, et al. (2018) The selective cathepsin K inhibitor MIV-711 attenuates joint pathology in experimental animal models of osteoarthritis. J Transl Med 16(1): 56.
- Conaghan PG, Bowes MA, Kingsbury SR, Brett A, Guillard G, et al. (2018) Disease-Modifying Effects of a Novel Cathepsin K Inhibitor in Osteoarthritis: A Randomized Controlled Trial. Ann Intern Med 172(2): 86-95.
- Alcaraz MJ, Guillen MI, Ferrandiz ML (2019) Emerging therapeutic agents in osteoarthritis. Biochem Pharmacol 165: 4-16.
- Monteagudo S, Lories RJ (2017) Cushioning the cartilage: a canonical Wnt restricting matter. Nat Rev Rheumatol 13(11): 670-681.
- Deshmukh V, Hu H, Barroga C, Bossard C, Kc S, et al. (2018) A small-molecule inhibitor of the Wnt pathway (SM04690) as a potential disease modifying agent for the treatment of osteoarthritis of the knee. Osteoarthritis Cartilage 26(1): 18-27.
- El Daibani A, Che T (2022) Spotlight on Nociceptin/Orphanin FQ Receptor in the Treatment of Pain. Molecules 27(3): 595.
- Yao X, Zhang J, Jing X, Ye Y, Guo J, et al. (2019) Fibroblast growth factor 18 exerts anti-osteoarthritic effects through PI3K-AKT signaling and mitochondrial fusion and fission. Pharmacol Res 139: 314-324.
- Lohmander LS, Hellot S, Dreher D, Krantz EF, Kruger DS, et al. (2014) Intraarticular sprifermin (recombinant human fibroblast growth factor 18) in knee osteoarthritis: a randomized, double-blind, placebo-controlled trial. Arthritis Rheumatol 66(7): 1820-1831.
- Jo CH, Lee YG, Shin WH, Kim H, Chai JW, et al. (2014) Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof-of-concept clinical trial. Stem Cells 32(5): 1254-1266.
- Brittberg M, Lindahl A, Nilsson A, Ohlsson C, Isaksson O, et al. (1994) Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med 331(14): 889-895.
- Chen G, Sato T, Ushida T, Ochiai N, Tateishi T (2004) Tissue engineering of cartilage using a hybrid scaffold of synthetic polymer and collagen. Tissue Eng 10(34): 323-330.
- Iyer SR, Xu S, Stains JP, Bennett CH, Lovering RM (2017) Superparamagnetic Iron Oxide Nanoparticles in Musculoskeletal Biology. Tissue Eng Part B Rev 23(4): 373385.
- Dai WL, Zhou AG, Zhang H, Zhang J (2017) Efficacy of Platelet-Rich Plasma in the Treatment of Knee Osteoarthritis: A Meta-analysis of Randomized Controlled Trials. Arthroscopy 33(3): 659-670.e1.
- Lubis AMT, Wonggokusuma E, Marsetio AF (2019) Intra-articular Recombinant Human Growth Hormone Injection Compared with Hyaluronic Acid and Placebo for an Osteoarthritis Model of New Zealand Rabbits. Knee Surg Relat Res 31(1): 44-53.
- Wen X, Huang X, Wu H (2021) Development of a novel intraarticular injection of diclofenac for the treatment of arthritis: a preclinical study in the rabbit model. Acta Biochim Pol 68(1): 71-76.
- Paik J, Duggan ST, Keam SJ (2019) Triamcinolone Acetonide Extended-Release: A Review in Osteoarthritis Pain of the Knee. Drugs 79(4): 455-462.
- Kirkley A, Webster-Bogaert S, Litchfield R, Amendola A, MacDonald S, et al. (1999) The effect of bracing on varus gonarthrosis. J Bone Joint Surg Am 81(4): 539-548.
- Shakoor N, Block JA (2006) Walking barefoot decreases loading on the lower extremity joints in knee osteoarthritis. Arthritis Rheum 54(9): 2923-2927.
- Bennell KL, Hinman RS, Metcalf BR, Buchbinder R, McConnell J, et al. (2005) Efficacy of physiotherapy management of knee joint osteoarthritis: a randomised, double blind, placebo-controlled trial. Ann Rheum Dis 64(6): 906-912.
- Brouwer RW, Bierma-Zeinstra SM, van Raaij TM, Verhaar JA (2006) Osteotomy for medial compartment arthritis of the knee using a closing wedge or an opening wedge controlled by a Puddu plate. A one-year randomised, controlled study. J Bone Joint Surg Br 88(11): 1454-1459.
- Wu J, Chen Q, Deng C, Xu B, Zhang Z, et al. (2020) Exquisite design of injectable Hydrogels in Cartilage Repair. Theranostics 10(21): 9843-9864.
- Zhou S, Bei Z, Wei J, Yan X, Wen H, et al. (2021) Mussel-inspired injectable chitosan hydrogel modified with catechol for cell adhesion and cartilage defect repair. J Mater Chem B 10(7): 1019-1030.
- Belk JW, Kraeutler MJ, Houck DA, Goodrich JA, Dragoo JL, et al. (2021) latelet-Rich Plasma Versus Hyaluronic Acid for Knee Osteoarthritis: A Systematic Review and Metaanalysis of Randomized Controlled Trials. Am J Sports Med 49(1): 249-260.
- Li S, Xing F, Yan T, Zhang S, Chen F (2023) Multiple Injections of Platelet-Rich Plasma Versus Hyaluronic Acid for Knee Osteoarthritis: A Systematic Review and MetaAnalysis of Current Evidence in Randomized Controlled Trials. J Pers Med 13(3): 429.
- Lamo-Espinosa JM, Mora G, Blanco JF, Granero-Molto F, Nunez-Cordoba JM, et al. (2016) Intra-articular injection of two different doses of autologous bone marrow mesenchymal stem cells versus hyaluronic acid in the treatment of knee osteoarthritis: multicenter randomized controlled clinical trial (phase I/II). J Transl Med 14(1): 246.
- Lee YH, Woo JH, Choi SJ, Ji JD, Song GG (2010) Effect of glucosamine or chondroitin sulfate on the osteoarthritis progression: a meta-analysis. Rheumatol Int 30(3): 357-363.
- Jevsevar DS, Brown GA, Jones DL, Matzkin EG, Manner PA, et al. (2013) The American Academy of Orthopaedic Surgeons evidence-based guideline on: treatment of osteoarthritis of the knee, (2nd edn), J Bone Joint Surg Am 95(20): 1885-1886.
- da Costa BR, Nuesch E, Kasteler R, Husni E, Welch V, et al. (2014) Oral or transdermal opioids for osteoarthritis of the knee or hip. Cochrane Database Syst Rev 17(9): CD003115.
- Hochberg MC, Guermazi A, Guehring H, Aydemir A, et al. (2019) Effect of Intra-Articular Sprifermin vs Placebo on Femorotibial Joint Cartilage Thickness in Patients with Osteoarthritis: The FORWARD Randomized Clinical Trial. JAMA 322(14): 1360-1370.
- Yazici Y, McAlindon TE, Gibofsky A, Lane NE, Lattermann C, et al. (2021) A Phase 2b randomized trial of lorecivivint, a novel intra-articular CLK2/DYRK1A inhibitor and Wnt pathway modulator for knee osteoarthritis. Osteoarthritis Cartilage 29(5): 654-666.
- Mobasheri A, Henrotin Y (2015) Biomarkers of (osteo)arthritis. Biomarkers 20(8): 513-518.
- Goldring MB, Otero M (2011) Inflammation in osteoarthritis. Curr Opin Rheumatol 23(5): 471-478.
- Malfait AM, Little CB (2015) On the predictive utility of animal models of osteoarthritis. Arthritis Res Ther 17(1): 225.
- El Miedany Y, Elwakil W (2023) Multidisciplinary patient-centred model of care for osteoarthritis: scoping review protocol—an initiative by the Egyptian Academy of Bone Health. Egypt Rheumatol Rehabil 50(3).






























