Analysis of Regeneration Tissue Formed in the Treatment of Cartilage Defects
Tolgay Satana*, O Uzumcugil, OS Atik, S Omeroglu and D Erdogan
Orthopedic Surgeon, Clinic of Orthopaedic, Turkey
Submission:December 19, 2023; Published: February 10, 2023
*Corresponding author: Tolgay Satana, Orthopedic Surgeon, Clinic of Orthopaedic, Turkey
How to cite this article: Tolgay Satana, O Uzumcugil, OS Atik, S Omeroglu and D Erdogan. Analysis of Regeneration Tissue Formed in the Treatment of Cartilage Defects. Ortho & Rheum Open Access J. 2023; 21(2): 556058. DOI: 10.19080/OROAJ.2023.21.556058
Abstract
Joint function losses that occur as a result of injury, healing and scarring of the cartilage tissue are varied. In this study, the contralateral joints of rabbits with complete cartilage loss were defined as the control group. Self-healing of similar cartilage losses and popular surgical methods were compared, and the results were examined histopathologically. As a result, it was determined that while autogenous full-thickness osteochondral cartilage transfer resulted in the best cartilage healing, the healed cartilage tissue without any intervention was superior to methods such as drilling and carbon fiber. We concluded that cartilage tissue must be “no men land” if lower than full thickness cartilage lesions. Cartilage must be protected any surgical impact instead of main mechanical articular problems must be focused.
Keywords: Cartilage Defects; Mosaicplasty; Cartilage debridement
Introduction
Cartilage tissue is metabolically an active tissue. It does not have self-repair capability except superficial losses. As cartilage cells do not have regeneration ability, losses can be closed up to 1 mm through cellular migration and matrix production. No cellular migration occurs if injury does not reach subchondral tissue in superficial losses. In full-thickness injuries, we observe that fibrin and mesenchymal cells play a role in remodeling after hemorrhage. In this case, repair tissue is a scar tissue in the form of fibrocartilage. Hyaline cartilage provides a regular and smooth surface in addition to providing viscosity of cartilage, which is one of its basic functions. Fibrocartilage functions like filler as a scar tissue and paves the way for inflammatory response with PDGF, TGF-β mediators secreted from mesenchymal cells, which play an important role in the physiopathology of arthritis. Closure of a cartilage defect can be in the form of excessive scar tissue, which may constitute a mechanical barrier. Similarly, osteophyte formations are the hard osseous healing tissue of mesenchymal cells featuring as stem cells that are involved in the formation of fibrocartilage tissue [1,2]. Therefore, in addition to acute inflammatory response, late complications will occur in the form of healing tissue that constitutes a mechanical obstacle and impairs viscosity. Similar mechanical problems may occur if the tissue forming after cartilage transfer performed considering the advantages of hyaline cartilage healing is not aligned with surface.
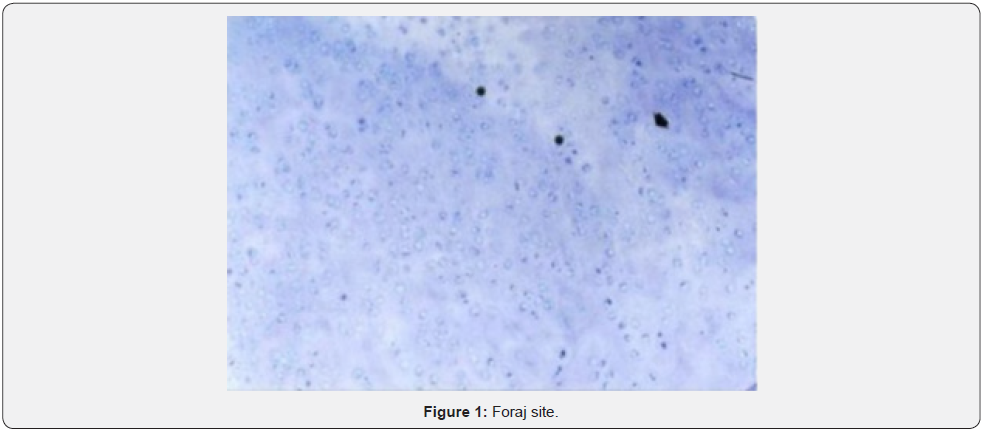
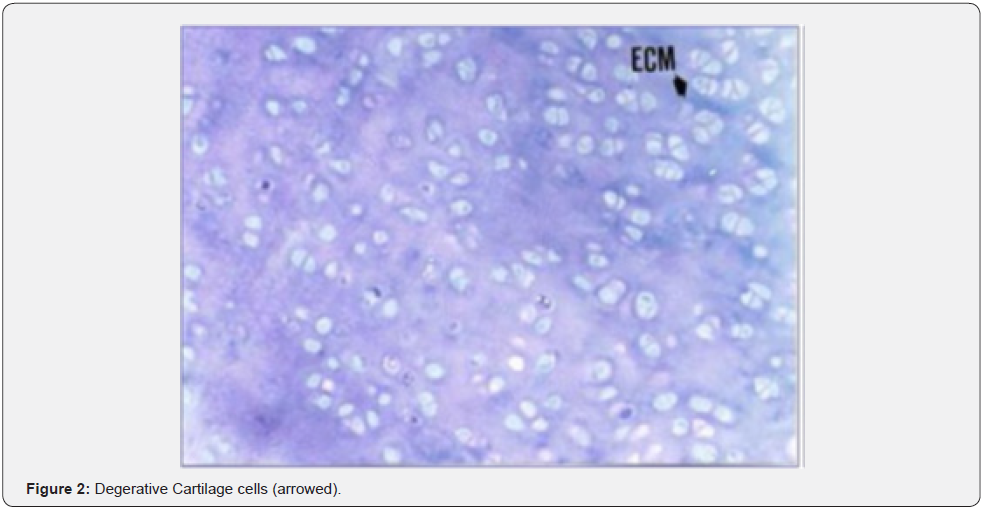
Hyaline cartilage implanted with an appropriate technique enables the defect to start superficial healing and allows remodeling. Carbon fibre filler which is another method used to close cartilage defect aims at stimulating the formation of matrix with a smooth surface while preventing excessive development of fibrocartilage tissue. Viscosuplementation not only creates a mechanical effect in closing superficial defects but also ensures migration of chondral cells and accelerates new cartilage formation. In histological examination, degenerative changes were marked in cartilage cells in forage group especially around forage, (Figures 1 & 2) while loosely localized cartilage cells were observed to form in the regeneration region, which were similar to fibrous cartilage [3-12].
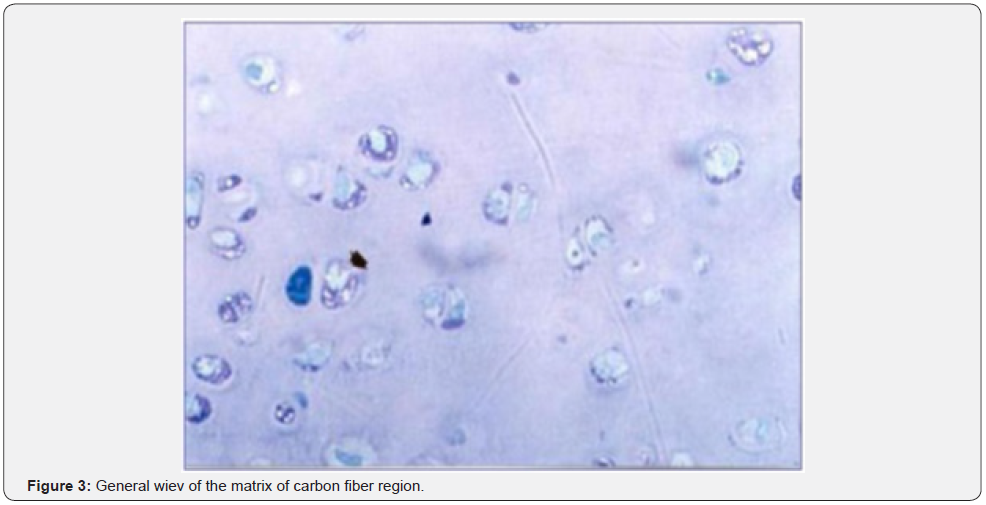
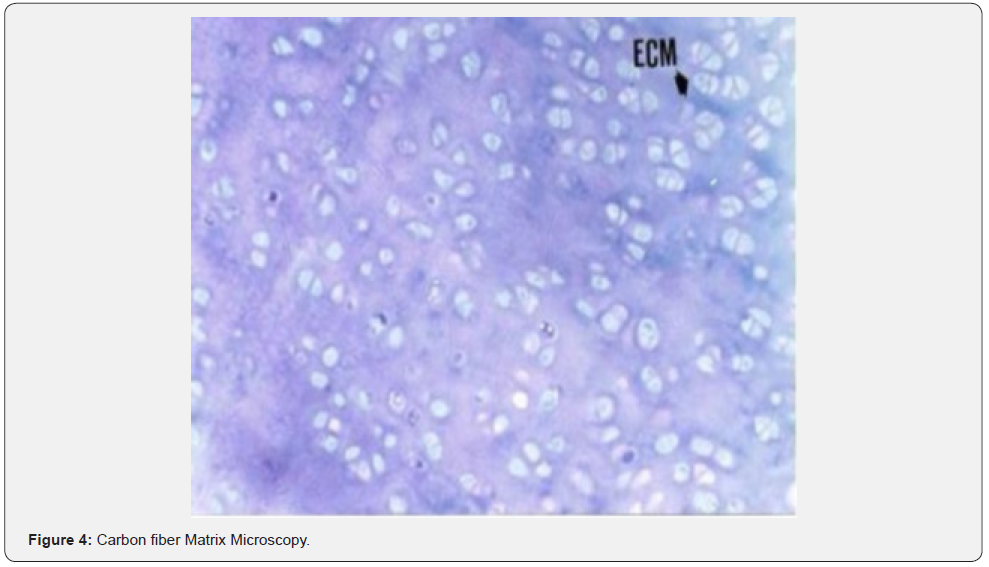
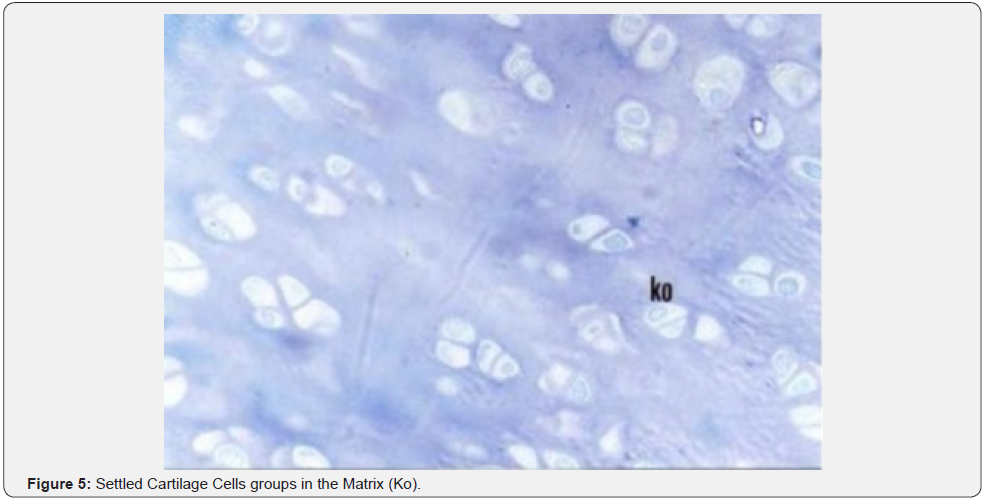
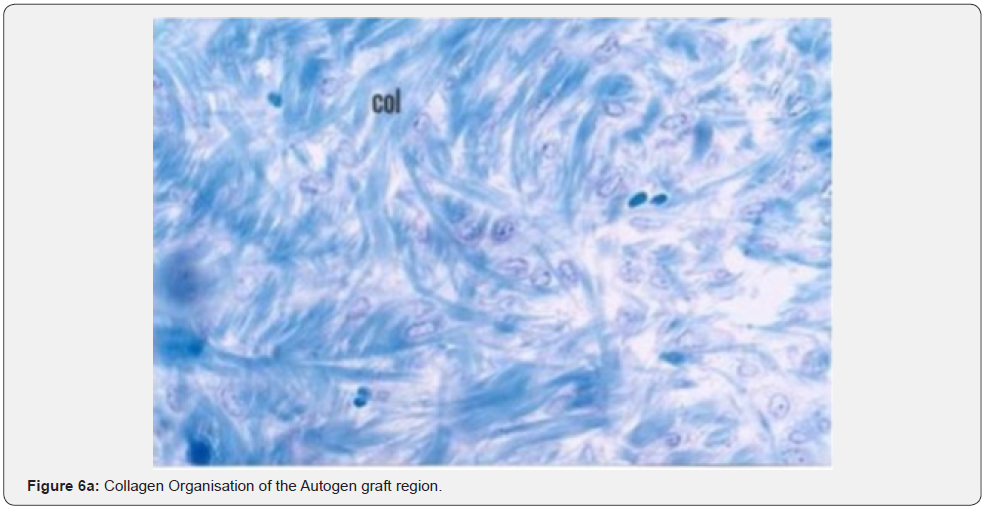
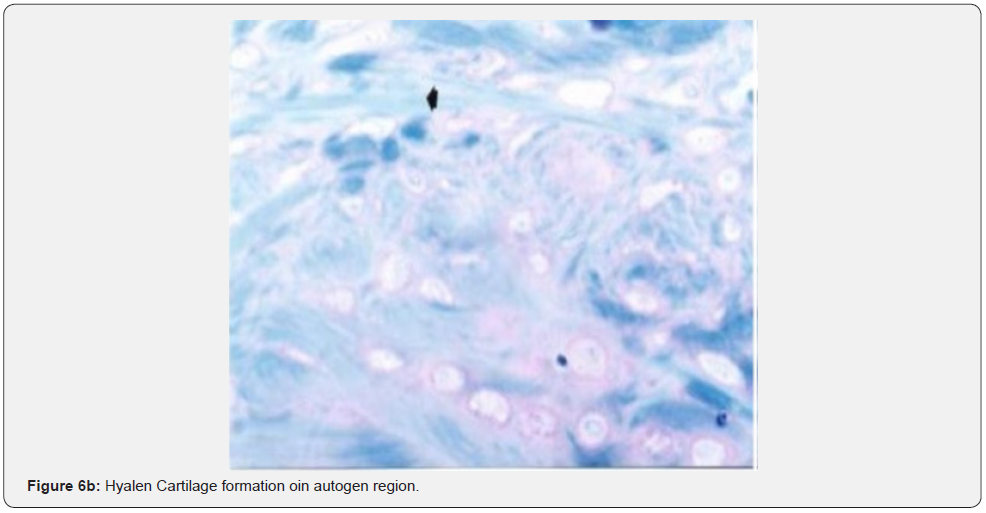
The histological examination of the group where Carbon Fibre was used revealed that single fibrous cartilage or those in isogeneic groups were formed between the extracellular matrix (ECM) (Figures 3 & 4). The magnified image of the same group demonstrated fibrous cartilage cells in isolation or in groups. The histological examination of the regions where autogenous osteochondral graft was placed showed that cartilage cells were highly marked within the lacunas between the thick and irregularly distributed collagen fibres within the scar tissue (Figure 5). Cartilage cells are more prominent towards the edge of the defect area and formed isogeneic groups. Collagen fibres were observed to be distributed irregularly within the cartilage matrix (Figure 6).
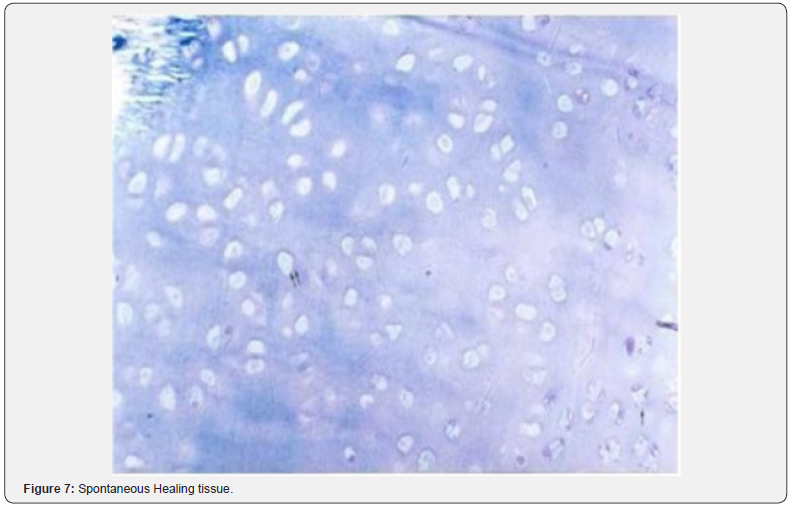
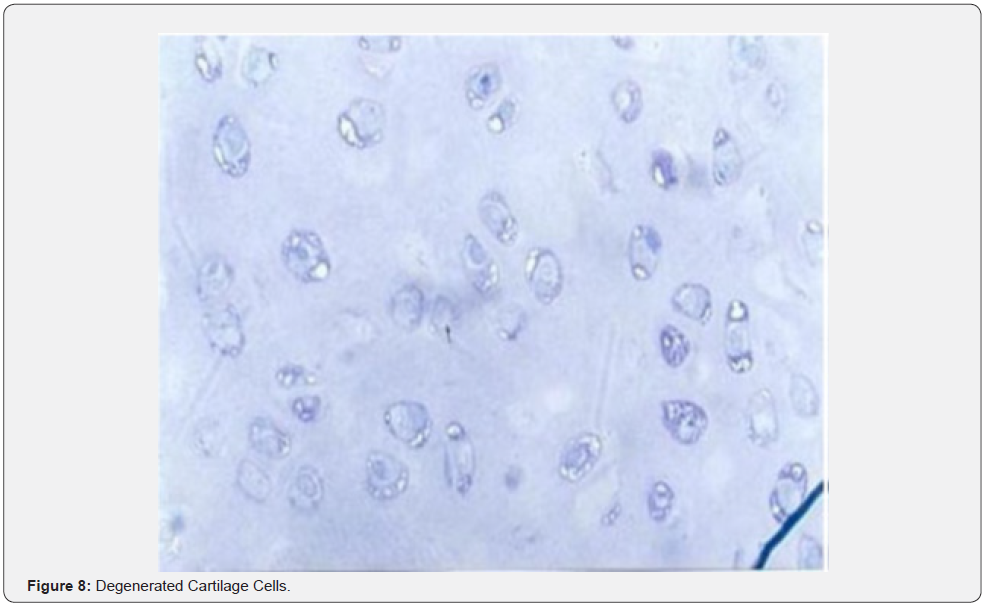
In other words, cartilage tissue was hyalene cartilage tissue with these features. In the group where the defect was formed and spontaneous healing was expected, healing tissue was observed to lose its normal structure and degenerated cartilage cells were formed (Figure 7). In magnified image, degenerated and intact cartilage cells were observed (Figure 8).
Materials and Method
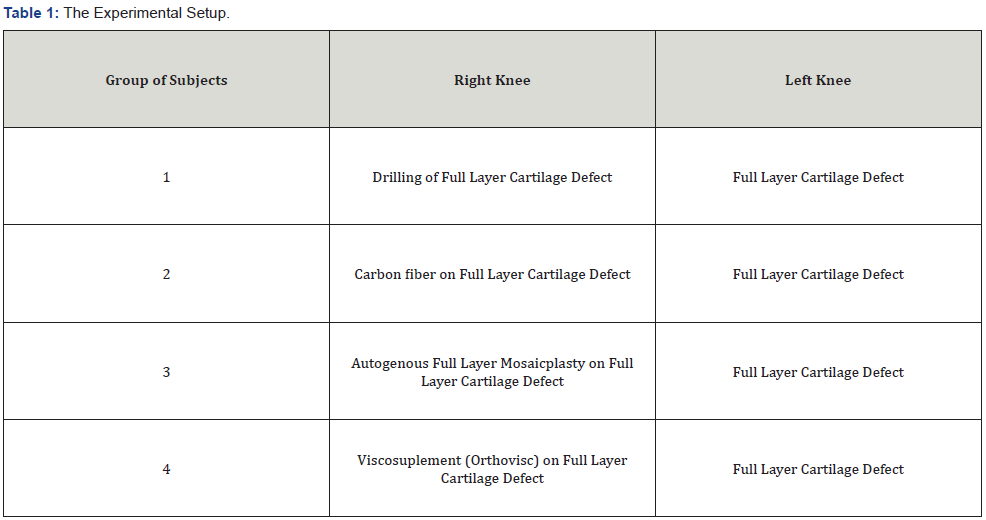
16 New Zealand rabbits were used. The subjects were divided into four groups, and both knees of the groups were included in the study (Table 1). All rabbits were fed under the same conditions and with the same type of feed. None of the rabbits developed tissue infection and extremity fixation was not performed.
Group of Subjects Right Knee Left knees
i. Drilling of Full Layer Cartilage Defect Full Layer Cartilage Defect
ii. Carbon fiber on Full Layer Cartilage Defect Full Layer Cartilage Defect
iii. Autogenous Full Layer Mosaicplasty on Full Layer Cartilage Defect Full Layer Cartilage Defect
iv. Viscosuplement (Orthovisc) on Full Layer Cartilage Defect Full Layer Cartilage Defect (Table 1).
Following the cleaning of the incision site under IV anaesthesia with Dormicum (Midazolam5mg/ml), anteromedial parapatellar incision was performed in a sterile setting. Patella was brought to the lateral position and femur condyles were exposed. Then full thickness cartilage defect was created in the medial femoral condyle in the same region and size using spinal needle nr. 17 in the right and left knees. Forage was performed on the defect area in the right knees using 1 mm drill in the first group. The defect was filled with carbon fibre in the second group. In the third group, osteochondral cylindrical grafts harvested with spinal needle retractor from the lateral side of femoral condyle were placed in the defect area with press-fit method after preparing the place with a drill. In the last group, 0.2 cc orthovisc (sodium hyaluronate 15mg/ml) was injected into the knees. All knees were closed with the due procedure, stitches were removed ten days later and there was no wound problem. Twenty-four weeks later, the knees were opened, and tissue samples were collected. Under macroscopic examination, tissues with rough surfaces similar to fibrocartilage that went beyond the cartilage surface were observed to be formed in the defect groups. In the defect area, osteophyte-like cornification’s were observed. The new tissue that was formed in the forage area passed over the cartilage surface but formed a more regular surface. In the group receiving carbon fibre, cartilage surface was not passed over with the regeneration tissue, but the defect area was uneven. Severe synovial reaction was observed. In the autogenous osteochondral graft area, cartilage with smooth surface that did not pass over the cartilage surface was observed to be formed. It was difficult to find the defect area in some of the knees. Regeneration was observed in the donor sites. The regeneration tissue appeared to be similar to solid cartilage [13-18].
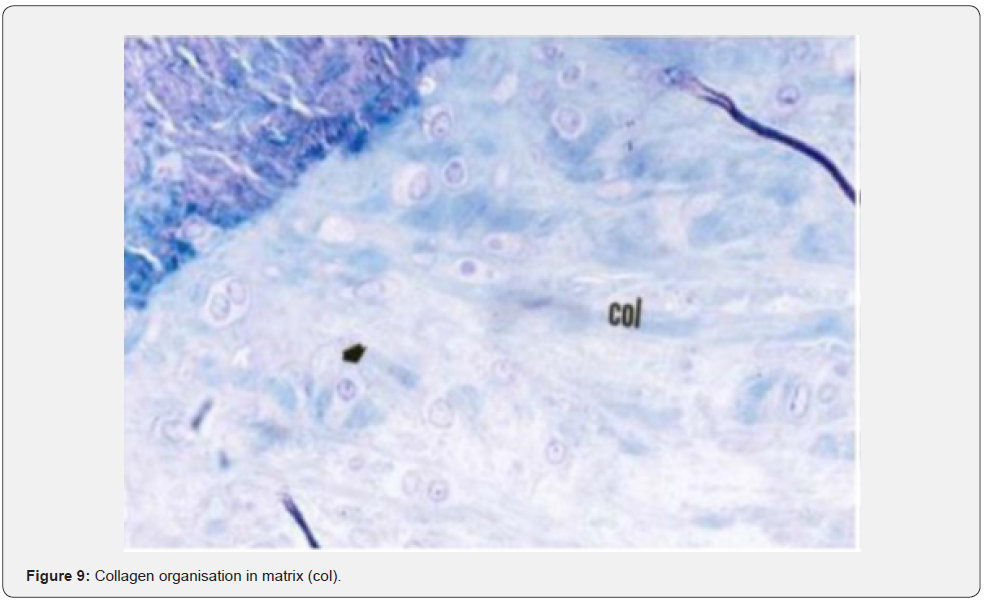
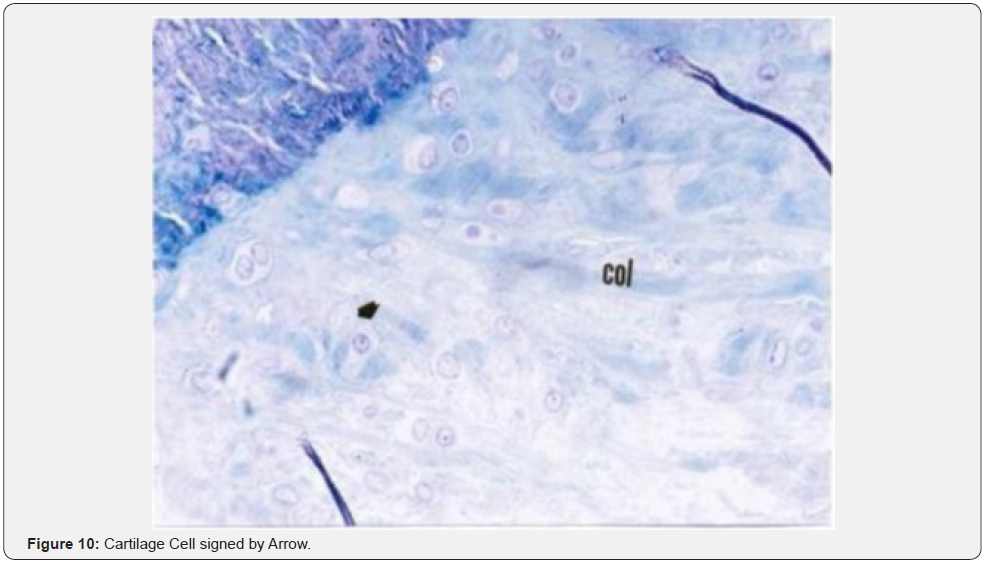
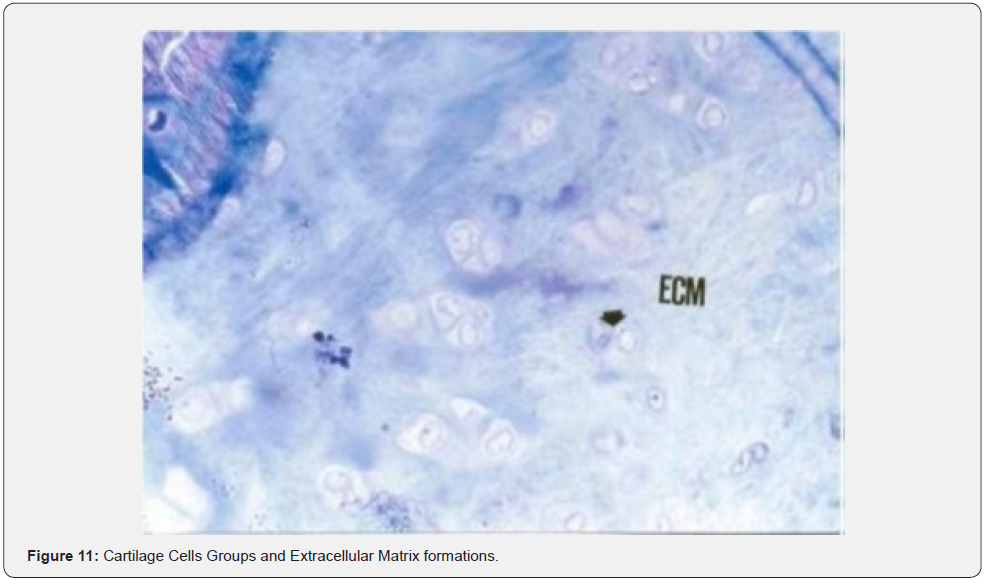
The histological examination of the group where a defect was created, and spontaneous healing was expected after Orthovisc injection showed that the connective tissue fibers (col) and extracellular matrix (ECM) within the scar tissue looked similar to the cartilage tissue (Figures 9-11). The irregular cartilage cell groups in the healing tissue were matured cartilage cells and formed isogeneic groups (Figure 11).
Discussion
In our study, we did not find any advantage of forage for spontaneous healing as it formed regeneration tissue passing over the cartilage tissue under macroscopic view and it was similar to spontaneous healing tissue under histological examination. The goal of the treatment of traumatic and degenerative focal cartilage losses is to relieve pain and create a viscous joint surface. Hunt et al. does not recommend forage and mechanical debridement for focal cartilage losses that do not constitute a mechanical obstacle. Arthroscopic debridement is similar with clinical outcomes of full thickness cartilage lesions and treatment outcomes of spontaneous healing and placebo groups. Cain et al. did not recommend microfracture technique like forage as it does not form hyaline cartilage. Cartilage debridement leads to regeneration with fibrocartilage in full thickness lesions and will result in degenerative arthritis in the long-term. Carbon fibre functions as a connective tissue that enhances the formation of matrix in the cartilage tissue. Its chondroprotective effect was demonstrated in our study with the migration of cartilage cells. The defect area where carbon filler was injected did not pass over the cartilage surface and allowed for a relatively viscous surface that did not create a mechanical obstacle. Foreign body reaction was reported in the literature by some authors such as Mortier et al. whereas Allcock et al. and Brittberg et al. reported successful outcomes. Like in the experimental study performed by Yonsei et al. with rabbits, it may allow hyaline cartilage in cartilage losses that do not reach the osteochondral region. In our study, regeneration in the form of fibrocartilage was detected as it was injected to the area of full-thickness focal cartilage loss. In the treatment of full-thickness and focal cartilage defects, osteochondral cartilage transfer forms hyaline cartilage and results in viscous joint with a scar tissue that does not go beyond cartilage surface. Cain et al. recommended it as the ideal treatment in their treatment algorithm for focal cartilage losses except kissing lesions.
Atik et al. developed the AMOT (Autogenous Multiple Osteochondral Graft Transfer) technique that resulted in hyaline cartilage by using press-fit osteochondral grafts in the treatment of focal full thickness lesions. Attmanspacher et al. also treated cartilage defects with a similar technique and published successful results. Makino et al. examined the regeneration in the implanted tissue histologically and suggested that scar tissue with a regular surface under macroscope was different from histologically intact tissue, which required mechanical examination. Sanders measured the thickness of the cartilage scar tissue that was formed and tried to assess long-term mechanical difference. In his study, cartilage thickness was preserved by more than 50% of the normal value in 15 of 21 cases. In the same study, they found that there was no adequate fusion in the graft area in the first two weeks, but osseous connection was found in 4-6 weeks. In an experimental study conducted by Duchow et al. on graft stability, they found a marked mechanical difference when the press-fit graft was 10 to 15mm long. That study highlighted the importance of a large contact surface in the defect area for stability in addition to the preparation of the graft. Graft must contain adequate osseous component and be implanted at an appropriate depth with press technique. Shorter length will decrease stability. Although the result of Orthovisc may look like spontaneous healing scar under macroscope, the presence of cartilage cell migration in histological examination may explain the success of the clinical outcomes. Adams et al. found that orthovisc was advantageous as an effective treatment of a healthy synovial tissue. Brandt et al. reported a marked healing in their clinical series compared to placebo.
Conclusion
Treatment of focal cartilage losses, either traumatic or degenerative, should relieve pain and increase functional capacity by creating a viscous joint surface. Osteochondral autogenous cartilage transfer technique is more advantageous that other treatment options if performed properly as it forms hyaline scar tissue that does not pass over joint surface and increases viscosity. When the disadvantages associated with donor site are eliminated through new materials to be developed via tissue engineering, cartilage transfer will be considered as the first option to treat focal cartilage losses. Orthovisc provides viscosupplementation in case of generalized cartilage losses and also increases cartilage repair in superficial cartilage lesions. In addition to this effect, it may also have chondroprotective effect.
References
- Adams ME, Lussier AJ, Peyron JG (2000) A risk-benefit assessment of injections of hyaluronan and its derivatives in the treatment of osteoarthritis of the knee. Drud Saf 23(2): 115-130.
- Allcock S, Ali MA (1997) Early failure of a carbon fiber composite femoral component. J Arthroplasty 12(3): 356-358.
- Atik OS, et al. (1996) Osteochondral multiple autograft transfer, a new surgical method in chondroplasty”. Transfer Arthroplasty Arthroscopic Surgery 7(2): 22-23.
- Waste OS, et al. (1997) Arthroscopic Osteochondral Multiple Autograft Transfer Arthroplasty Arthroscopic Surgery 8.
- Atik OS (1990) Biological Repair of Osteochondral Defects Using Carbon Implants. Artoplasty Arthroscopy 2.
- Atik OS (1991) Knee Replacement and Patella Problem. Arthroplasty Arthroscopic Surgery 2(1-2).
- Attmanspacher W, Dittrich V, Stedtfeld HW (2000) Experiences with arthroscopic therapy of chondral defects of the knee joint with osteochondral autograft system. Zentralbl Chir 125(6): 494-499.
- Brandt KD, J A Block, J P Michalski, L W Moreland, J R Caldwell, et al. (2001) Efficacy and safety of intraarticular sodium hyaluronate in knee osteoarthritis. Clin Orthop 385: 130-143.
- Britberg M, Faxen E, Peterson L (1994) Carbon fiber scaffolds in the treatment of early knee osteoarthritis. Clin Orthop 307: 155-164.
- Cain EL and Clancy WG (2001) Treatment algorithm for osteochondral injuries of the knee. Clin Sports Med 20(2): 321-342.
- Chen FS, Frenkel SR, Di Cesare PE (1999) Repair of articular cartilage defects: part II. Treatment options. Am J Orthop 28(2): 88-99.
- Duchow J, Hess T, Kohn D (2000) Primary stability of press-fit implanted osteochondral grafts influence of graft size, repeated insertion and harvesting technique”. Am J Sports Med 28(1): 24-27.
- H J Kang, C D Han, E S Kang, N H Kim, W I Yang (1991) An experimental implantation of wowen carbon fiber pad into osteochondral defect of the femoral condyle in rabbit. Yonsei Med J 32(2): 108-116.
- W M Kus, A Gorecki, P Strzelczyk, P Swiader (1999) Carbon fiber scaffolds in the surgical treatment of cartilage lesions. Ann Transplant 4(3-4): 101-102.
- Makino T, H Fujioka, M Kurosaka, N Matsui, H Yoshihara, et al. (2001) Histologic analysis of the implanted cartilage in an exact-fit osteochondral transplantation model. Arthroscopy 17(7): 747-751.
- Morelli M, Nagamori J, Miniaci A (2002) Management of chondral injuries of the knee by osteochondral autogeneous transfer. J Knee Surg 15(3): 185-190.
- Sanders TG, K D Mentzer, M D Miller, W B Morrison, S E Campbell, et al. (2001) Autogenous osteochondral plug transfer for the treatment of focal chondral defects. Skeletal Radiol 30(10): 570-578.
- Takahashi S, M Oka, Y Kotoura, T Yamamuro (1995) Autogeneous callo-osseous grafts for the repair of osteochondral defects. J Bone Joint Surg Br 77(2): 194-204.






























