Extracorporeal Shockwave Therapy for a Focal Musculoskeletal Complaint in Patients with Rheumatic Disease: A Cases Series
Phillip H Yun1*, Adam S Tenforde2, David Robinson2 and Minna J Kohler3
1Department of Family and Community Medicine, University of Illinois Chicago, USA
2Department of Physical Medicine and Rehabilitation, Spaulding Rehabilitation Hospital/Harvard Medical School, USA
3Division of Rheumatology, Allergy, and Immunology, Massachusetts General Hospital/Harvard Medical School, USA
Submission:January 14, 2023; Published: February 14, 2023
*Corresponding author: Phillip H Yun, Department of Family and Community Medicine, University of Illinois Chicago, USA
How to cite this article: Phillip H Yun, Adam S Tenforde, David Robinson and Minna J Kohler. Extracorporeal Shockwave Therapy for a Focal Musculoskeletal Complaint in Patients with Rheumatic Disease: A Cases Series. Ortho & Rheum Open Access J. 2023; 21(2): 556056. DOI: 10.19080/OROAJ.2023.21.556056
Abstract
Aim: The objectives of this retrospective case series were to assess the efficacy, safety, and potential for rheumatic disease flares in patients who were treated with extracorporeal shockwave therapy for a focal musculoskeletal complaint.
Methods: We retrospectively evaluated 17 patients with rheumatic disease who received extracorporeal shockwave therapy for a focal musculoskeletal complaint between August 2017 and April 2021. Patients were categorized into three groups: 1) those on a stable rheumatic medication regimen, 2) those not on a stable rheumatic medication regimen, and 3) those with a new diagnosis of rheumatic disease within six months of receiving extracorporeal shockwave therapy. Rheumatic disease flares, subjective reports of pain, and severe adverse events were obtained.
Results: Among patients on a stable rheumatic medication regimen, 100% did not experience a rheumatic disease flare, and 75% reported improved symptoms. Among patients not on a stable regimen, 62.5% did not experience a disease flare, and 50% reported improved symptoms. Among patients with a new diagnosis of rheumatic disease, 80% did not experience a disease flare, and 20% reported improvement of symptoms. No severe adverse events occurred.
Conclusion: In summary, our findings suggest that patients with rheumatic disease that are on a stable rheumatic medication regimen do not experience disease flares and can benefit from extracorporeal shockwave therapy for a focal musculoskeletal complaint.
Keywords: Autoimmune disease; Disease flare; ESWT; Pain; Safety
Introduction
Extracorporeal shockwave therapy (ESWT) is a non-invasive intervention that utilizes shockwaves to deliver energy to soft tissues and has been shown to be effective for a variety of musculoskeletal injuries including tendinopathies, plantar fasciitis, medial tibial stress syndrome, and bony non-unions or stress fractures [1–5]. It is theorized that ESWT promotes healing by affecting local regulation of metabolic and inflammatory pathways including increased expression of TGF-β1 and IGF-I, increased IL-6, IL-8, MMP-2, and MMP-9 levels, increased collagen (mainly Type 1) synthesis, increased glycosaminoglycan and protein synthesis, and increased tenocyte proliferation [6-10].
ESWT is delivered through two different forms: Focused Shockwave Therapy (F-SWT) and Radial Shockwave Therapy (R-SWT). F-SWT utilizes electrohydraulic, electromagnetic, or piezoelectric technology to deliver shockwaves that converge at a particular depth (or focus) within the body [11]. R-SWT utilizes ballistic technology to create pressure waves that exert maximal energy at the applicator-skin interface and attenuate at deeper structures [12]. Evidence suggests that combination treatment with both F-SWT and R-SWT, also known as Combined Shockwave Therapy (C-SWT), may be an effective treatment option as well [13]. A number of randomized controlled trials have shown the benefit of ESWT in rotator cuff tendinopathy, lateral epicondylitis, hamstring tendinopathy, patellar tendinopathy, Achilles tendinopathy, and plantar fasciitis [10,14-17]. However, only a limited number of studies have evaluated ESWT in patients with rheumatic disease [18-20].
While treatment of patients with rheumatic diseases are often focused on systemic medication therapy such as disease-modifying anti-rheumatic drugs (DMARDs), systemic glucocorticoids, and/or biologic agents to achieve remission or low disease activity [21], these patients may still experience significant levels of pain [22,23] and sporadic inflammatory flares that can affect tendon, joint, or muscle. Treatment of these flares depends on the severity and location. For example, if a single or a few joints are affected, intraarticular glucocorticoid injection may be considered to allow patients to treat focal inflammation, while avoiding increasing their baseline regimen or initiating a course of systemic glucocorticoids [24]. In addition, patients with rheumatic disease can suffer from other causes of musculoskeletal pain such as mechanical and overuse injuries leading to tendinopathies, as well as tendinopathies as a result of autoimmune mediated inflammatory damage.
Previous qualitative studies have shown discrepancies between patient and physician perceptions on how to manage pain for rheumatoid arthritis and other autoimmune conditions [25]. Pain relief has been noted to be a top priority among important concerns for patients with arthritis and can lead to alternative treatment seeking behavior [26]. Patients who have experienced adverse drug reactions (ADRs) from initiating or increasing their medication regimen and/or relying on high dose analgesics such as non-steroidal anti-inflammatory drugs (NSAIDs) or opiates may desire to try other therapeutic modalities. Therefore, there is growing interest in complementary or alternative therapeutic modalities that can provide pain relief and functional improvement in those with rheumatic disease. The objectives of this retrospective case series were to assess the safety, efficacy, and potential for disease flare in patients with any rheumatic disease who were treated with ESWT for a focal musculoskeletal complaint.
Material and Methods
This report is from the results of a quality improvement initiative approved by the Department of Physical Medicine and Rehabilitation with waiver of Institutional Review Board approval. SQUIRE-2 guidelines were used for reporting quality improvement data [27]. Patient characteristics, treatment measures, and functional outcomes were extracted through chart review from August 2017 to April 2021 by two authors (P.H.Y. and D.M.R.) in all patients with rheumatic disease receiving R-SWT, F-SWT, or C-SWT. All ESWT sessions occurred in a single outpatient sports medicine clinic by a single physician (A.S.T.).
The diagnosis of a rheumatic disease was confirmed through chart review of medical histories and documentation in rheumatology clinician notes. We evaluated patients in three categories: 1) patients with a stable rheumatic medication regimen, 2) patients without a stable rheumatic medication regimen, and 3) patients with a new diagnosis of a rheumatic disease within six months of receiving ESWT. A stable rheumatic medication regimen was defined as maintaining the same medication regimen and dose for three months or greater. According to the definition in prior studies, a rheumatic exacerbation (or flare) was defined as an increase in pain in two joints or more [28,29] and was counted if occurring during or within three months of receiving ESWT. An inclusion exception was made for monoarticular involvement in a gout flare. Disease flare was initially identified through chart review by a single author (P.H.Y.) and confirmed by another author (M.J.K.), a board-certified rheumatologist.
Rheumatic diseases included in the study were psoriatic arthritis, undifferentiated spondyloarthropathy, rheumatoid arthritis, gout, pseudogout, Sjogren’s disease, polymyalgia rheumatica, polyarteritis nodosa, eosinophilic granulomatosis with polyangiitis, HLA-B27 arthropathy, ANCA-associated vasculitis. Exclusion criteria included: 1) inpatient hospitalization during or within 3 months of ESWT, 2) absence of rheumatology notes within the electronic medical records, 3) surgery during or within 3 months of ESWT, 4) orthobiologic injections during or within three months of ESWT, 5) concomitant infections including Lyme disease, and 6) diagnosis of rheumatic disease greater than six months following ESWT treatment.
Treatment Procedure
Patients received C-SWT, F-SWT, or R-SWT once a week. Given that ESWT is not covered by insurance in the United States, patients paid a one-time fee for shockwave treatment sessions that was the same for R-SWT, F-SWT, and C-SWT. R-SWT treatments were provided using the Storz Extracorporeal pulse activation technology (EPAT®) device (Storz Medical, Tägerwilen, Switzerland) (Figure 1), and F-SWT treatments were performed using the Storz Duolith device (Storz Medical, Tägerwilen, Switzerland). C-SWT treatments involved consecutive application of R-SWT and F-SWT in each treatment session. All treatment sessions involved positioning the device over the affected area using the clinical focusing technique, and no local or regional anesthetic was used. Patients were allowed to resume or continue regular activities as tolerated. Physical therapy was prescribed to each patient with no prior treatment. Those who completed an extensive course of physical therapy were recommended to continue their home exercise program.
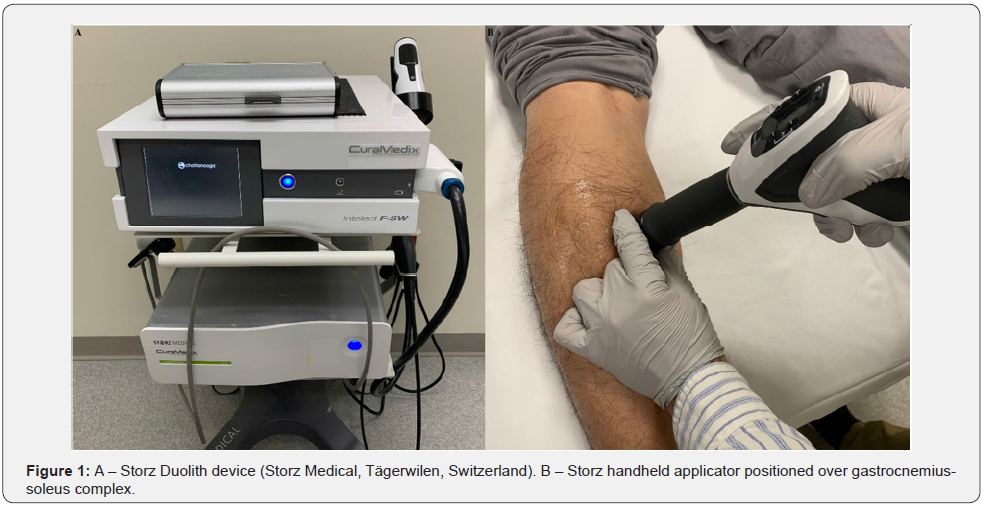
Outcome Assessment
Subjective reports of pain were obtained from the following: 1) discharge clinical notes written by a board-certified physiatrist (A.S.T.) who performed the shockwave treatment and the 2) follow up notes written by the corresponding rheumatologists. If the two reports were discordant, the less favorable clinical response was recorded. Clinical response of the presenting injury was defined as one of the following: 1) no improvement, 2) clinical improvement, or 3) clinical worsening. Adverse events were recorded based on chart review of medical documentation by the physiatrist who performed shockwave therapy and by the corresponding rheumatologists.
Results
Chart review of a single expert physiatrist (A.S.T.) who performs shockwave therapy in an outpatient clinic identified 23 eligible patients who had received ESWT and had a pre-existing or recent diagnosis of a rheumatic disease. Three were excluded given that the diagnosis of a rheumatic disease occurred more than six months after receiving the ESWT. One was excluded given lack of access to the patient’s outside rheumatology medical record. One was excluded due to the patient’s hospitalization for tracheitis within a week of receiving ESWT. One was excluded given an unconfirmed diagnosis of gout. This resulted in 17 eligible patients for inclusion. Demographic and baseline rheumatic disease characteristics are displayed in Table 1. Mean age and BMI were 53.9 years and 25.3kg/m2, respectively. A majority were female (64.7%), never smokers (58.8%), and carried a diagnosis of rheumatic disease longer than one year (64.7%) at time of initial evaluation for ESWT.
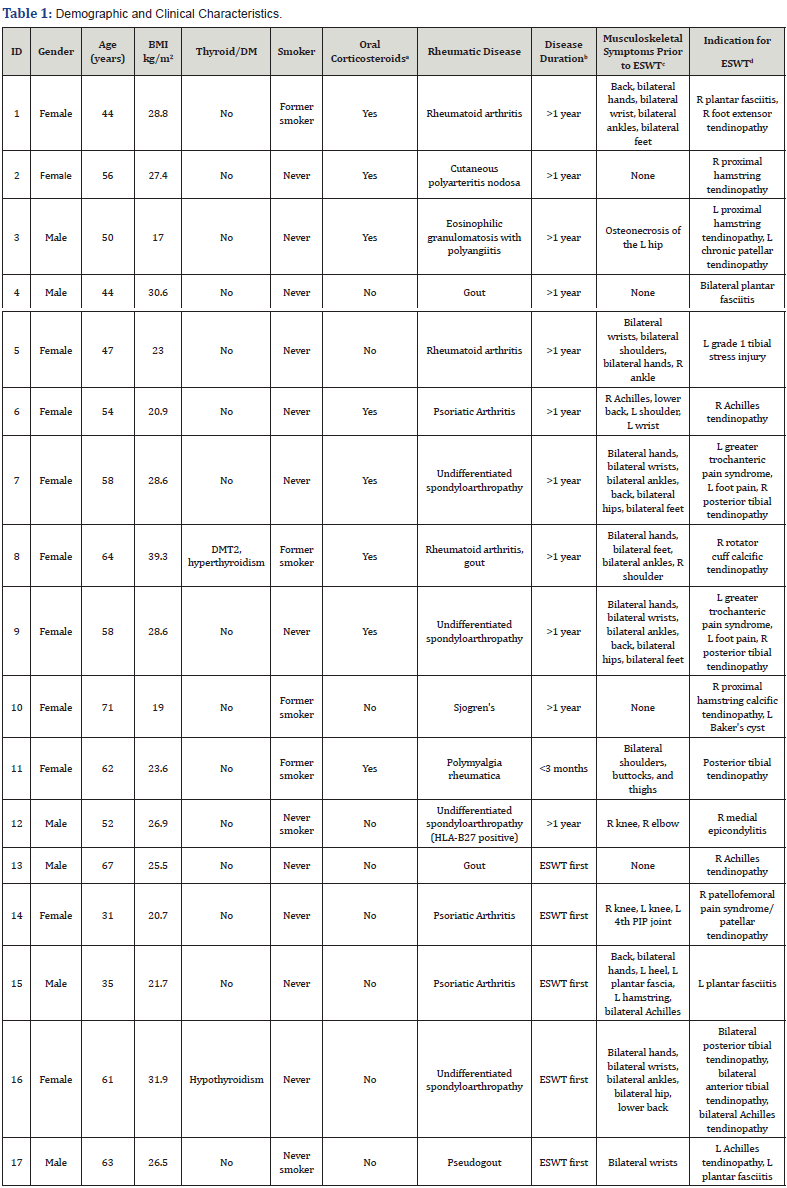
kg, kilogram; m, meter; DM, diabetes mellitus; DMT2, diabetes mellitus type 2; ESWT, extracorporeal shockwave therapy; R, right; L, left; ANCA, anti-neutrophil cytoplasmic antibody; ANA, antinuclear antibodies; PIP, proximal interphalangeal.
aDocumentation of oral glucocorticoid use prior to receiving ESWT.
bLength of time of known rheumatic disease prior to receiving ESWT. If ESWT first, rheumatic disease diagnosed within 6 months following ESWT.
Rheumatic disease exacerbation (Flare)
Four patients were on a stable rheumatic medication regimen prior to receiving ESWT (Table 2). Among these patients, all four patients (100%) patients did not experience a disease flare during or within three months of their last ESWT session and also maintained their rheumatic medication regimen and dose following ESWT. However, one patient did experience a vasculitic rash requiring a prednisone taper. Eight patients were not on a stable rheumatic medication regimen prior to receiving ESWT. Five patients (62.5%) did not experience a disease flare during or within three months of their last ESWT session. Three patients experienced a disease flare during or within three months of their last ESWT session requiring medication changes by their clinical rheumatologists. Five patients were diagnosed with a rheumatic disease within six months of their last ESWT session. Four patients (80%) did not experience a disease flare during or within three months of their last ESWT session. One patient had a gout flare (podagra, typical to prior experienced episode) that occurred during the weeks he was receiving ESWT treatment.
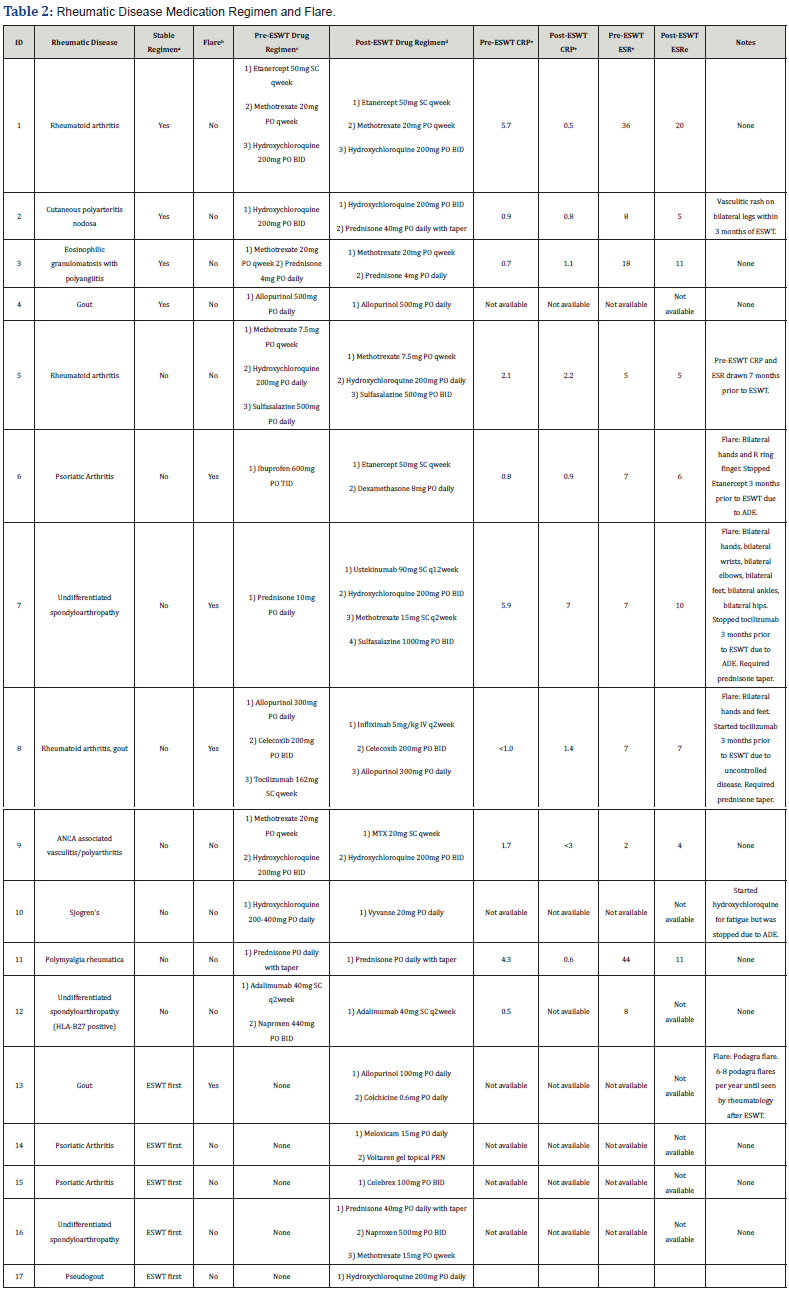
ESWT first, received ESWT prior to a diagnosis of a rheumatic disease; BID, twice a day; TID, three times a day; qweek, once a week; q2week, once every 2 weeks; PRN, as needed; ADE, adverse drug event; SC, subcutaneous; PO, by mouth.
a3 months or greater of the same rheumatic disease medication regimen and dose prior to receiving ESWT.
bTwo or more joints with increasing pain as reported by patient and documented in rheumatology notes during or within 3 months following ESWT.
cRheumatic medication regimen recorded in rheumatology note immediately preceding ESWT.
dRheumatic medication regimen recorded in rheumatology note immediately preceding ESWT.
ePre-ESWT CRP and ESR values within 6 months preceding ESWT, unless otherwise specified in notes. Post-ESWT CRP and ESR values within 6 months following ESWT, unless otherwise specified in notes.
Functional outcomes
Among those on a stable rheumatic medication regimen (n=4), three patients (75%) reported subjective improvement of their presenting injury (Table 3). One patient reported no improvement. Among those not on a stable rheumatic medication regimen (n=8), four patients (50%) reported subjective improvement of their presenting injury. One patient reported no change in symptoms. One patient reported worsening symptoms. There was no documentation of symptoms in two patients following ESWT treatment. Among those diagnosed with a rheumatic disease within six months of ESWT treatment (n=5), one patient (20%) reported subjective improvement of his presenting injury. Four patients reported no change in symptoms of their presenting injuries.
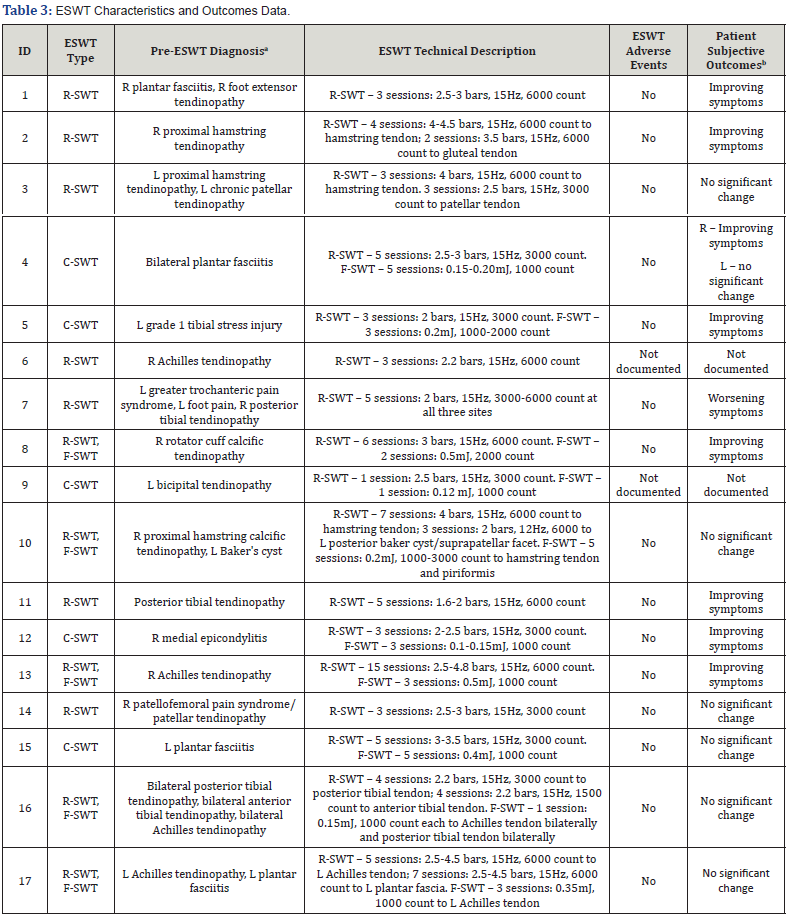
R-SWT, radial shockwave therapy; F-SWT, focused shockwave therapy; C-SWT, combined shockwave therapy; ESWT, extracorporeal shockwave therapy; R, right; L, left; Hz, hertz; mJ, millijoule.
aPatient presenting injury that was treated with ESWT.
bPatient reported symptoms of presenting injury after receiving ESWT, as documented by provider who performed ESWT and/or patient’s rheumatologist.
cPatient reported musculoskeletal symptoms documented in the rheumatology note immediately preceding the first session of ESWT.
dProvider performing ESWT established diagnosis through history, examination, and/or advanced imaging.
Adverse events
All patients reported pain while receiving shockwave therapy. No patient reported severe adverse events in relation to their presenting injury or ESWT treatment.
Discussion
Patients with rheumatic disease are commonly excluded from ESWT studies given the theoretical concern that ESWT may affect local metabolic and inflammatory pathways and stimulate systemic inflammation leading to an inflammatory flare. A corollary example would be how flares have been documented in patients with rheumatic disease after undergoing surgery [29]. In 2017, Liu et al. conducted a study on those with rheumatoid arthritis who received R-SWT alone and documented the following: 1) significant relief in arthralgia and morning stiffness, 2) no significant increase in C-reactive protein (CRP)/erythrocyte sedimentation rate (ESR) levels after ESWT, and 3) no severe adverse reactions related to R-SWT [19].
Although overall clinical status was assessed through measuring CRP/ESR levels, Health Assessment Questionnaire (HAQ) scores, and Disease Activity Score-28 for Rheumatoid Arthritis with ESR (DAS28-ESR) scores before and after ESWT, this study did not directly address the presence of rheumatic disease flares [19]. In addition, the current literature has only evaluated ESWT in select rheumatic diseases, such as systemic lupus erythematosus, rheumatoid arthritis, and systemic sclerosis [18-20]. Our retrospective study expands on the current literature by reviewing patients with a rheumatic disease who were treated with ESWT for a focal musculoskeletal complaint. To date, our case series is the largest study done on those with rheumatic disease who have received ESWT [18-20]. This study includes patients with rheumatoid arthritis, crystalline arthropathies (gout and pseudogout), seronegative spondyloarthritides (psoriatic arthritis, undifferentiated spondyloarthropathy), vasculitides, polymyalgia rheumatica, and Sjogren’s disease. In addition to evaluating for clinical improvement, this study also documented the safety profile and presence of rheumatic disease flares, as defined by two or more painful joints. We further classified patients as those on a stable rheumatic medication regimen, those not on a stable rheumatic medication regimen, and those who were diagnosed with rheumatic disease within six months of receiving ESWT. The latter two groups have not been previously studied in the current literature.
Among the four patients who were stable on a rheumatic medication regimen, all four patients (100%) did not experience a disease flare with three patients (75%) experiencing subjective improvement of their presenting injury. This is consistent with Liu et al. who demonstrated that R-SWT was effective in relieving arthralgias in rheumatoid arthritis with improvements seen in VAS, morning stiffness duration, DAS28-ESR, and HAQ scores [19]. This would suggest that despite being on immunomodulatory medications, the positive effects of ESWT are still seen in patients with rheumatic disease. Further studies are needed to quantify if there is a blunting effect of being on a DMARD or biologic medication given that ESWT is theorized to promote healing by affecting local inflammatory pathways.
This study evaluated patients who were not on a stable rheumatic medication regimen, which has not previously been reported. Three of eight patients (patients 6,7, and 8) experienced disease flares. As noted in Table 1, these three patients had poorly controlled underlying disease and were undergoing medication changes prior to receiving ESWT, which makes it difficult to determine whether disease flare was truly related to receiving ESWT or not. Interestingly, 50% of patients not stable on a baseline regimen still reported subjective improvement of their presenting injury after shockwave. This would suggest that ESWT can still be effective in relieving focal areas of pain that are not primarily driven by autoimmune mediated inflammation, which would generally require anti-inflammatory agents. The mixed results seen in those not on a stable rheumatic medication regimen should guide shared decision-making discussions in patients who may benefit from further medication optimization prior to receiving ESWT. Further studies are needed to elucidate whether there is a causal relationship between ESWT and disease flares in patients with uncontrolled rheumatic disease.
Five of seventeen (29.4%) patients who received ESWT were later identified as having rheumatic disease within six months of treatment. One of five (20%) patients experienced a disease flare, and one of five (20%) noted improvement following ESWT. The four patients who did not respond to ESWT had presenting injuries (patellar tendinopathy, Achilles tendinopathy, plantar fasciitis, posterior tibial tendinopathy) that previous studies have shown to respond favorably to ESWT [10]. These results suggest that providers should consider an undiagnosed rheumatic disease when patients are refractory to shockwave therapy who present with injuries that typically respond to ESWT. For example, enthesitis has been identified as a cardinal pathologic process in patients with spondyloarthropathies and should be considered in the differential diagnosis in patients who present with point tenderness at entheses [30]. Studies have shown that ultrasound can detect subclinical enthesitis in early psoriatic arthritis and spondyloarthropathies [31]. Therefore, providers with musculoskeletal ultrasound expertise can consider diagnostic ultrasound to identify findings of inflammatory enthesitis including hyperemia and bony changes of enthesophytes and erosions in patients suspected of early rheumatic disease.
While this study is first to evaluate for disease flares in patients with rheumatic disease who have received ESWT, we recognize the limitations in our study. Given the nature of a retrospective design, our study was limited by a small sample size and the use of non-validated outcomes measures. In addition, there was no randomization or control group due to retrospective design. Statistical comparisons between groups and between pre- and post-ESWT CRP/ESR levels were not calculated due to low power and lack of standardized timing of lab draws. Our definition of a disease flare involving two or more joints may not have adequately captured all clinical flares for certain rheumatic diseases. We attempted to mitigate this by including descriptive notes into Table 2 to mention whether a classic flare symptom was apparent, such as a vasculitic rash in polyarteritis nodosa.
Aside from immediate increased localized pain which resolved spontaneously, no severe adverse effects related to ESWT were observed in all 17 patients in our cohort. This safety profile is consistent with prior studies including one study evaluating ESWT for hip necrosis in systemic lupus erythematosus, which confirmed a favorable side effect profile with no systemic or neurovascular complications [18]. The overall safety of ESWT in patients with rheumatic disease has also been seen in two other studies evaluating arthralgias in rheumatoid arthritis patients and calcinosis cutis in systemic sclerosis patients [19,20].
Conclusion
In summary, our findings suggest that ESWT is feasible and safe in patients with rheumatic disease. Rheumatic patients that are on a stable medication regimen do not experience a disease flare from receiving ESWT. Providers and patients should consider delaying ESWT treatment until their rheumatic medication regimen has been stabilized. In addition, in patients who do not respond to ESWT with a musculoskeletal injury that typically responds well to ESWT, a new diagnosis of rheumatic disease should be considered. Further studies including prospective cohort studies and randomized controlled trials are needed to further elucidate the effects of ESWT in specific rheumatic diseases and musculoskeletal injuries.
Funding
None.
Conflict of Interest
None.
Acknowledgement
None.
References
- Smith WB, Melton W, Davies J (2017) Midsubstance Tendinopathy, Percutaneous Techniques (Platelet-Rich Plasma, Extracorporeal Shock Wave Therapy, Prolotherapy, Radiofrequency Ablation). Clin Podiatr Med Surg 34(2): 161-174.
- Thijs KM, Zwerver J, Backx FJG, Victor Steeneken, Stephan Rayer, et al. (2017) Effectiveness of shockwave treatment combined with eccentric training for patellar tendinopathy: A double-blinded randomized study. Clin J Sport Med 27(2): 89-96.
- Gollwitzer H, Saxena A, DiDomenico LA, Louis Galli, Richard T Bouche, et al. (2015) Clinically relevant effectiveness of focused extracorporeal shock wave therapy in the treatment of chronic plantar fasciitis: A Randomized, Controlled Multicenter Study. J Bone Jt Surg Am 97(9): 701-708.
- Moen MH, Rayer S, Schipper M, S Schmikli, A Weir, et al. (2012) Shockwave treatment for medial tibial stress syndrome in athletes; A prospective controlled study. Br J Sports Med 46(4): 253-257.
- Cacchio A, Giordano L, Colafarina O, Jan D Rompe, Emanuela Tavernese, et al. (2009) Extracorporeal shock-wave therapy compared with surgery for hypertrophic long-bone nonunions. J Bone Jt Surg - Ser A 91(11): 2589-2597.
- Chen YJ, Wang CJ, Yang KD, Yur-Ren Kuo, Hui-Chen Huang, et al. (2014) Extracorporeal shock waves promote healing of collagenase-induced Achilles tendinitis and increase TGF-β1 and IGF-I expression. J Orthop Res 22(4): 854-861.
- C M Waugh, D Morrissey, E Jones, G P Riley, H Langberg, H R C Screen (2015) In vivo biological response to extracorporeal shockwave therapy in human tendinopathy. Eur Cells Mater 29: 268-280.
- Vetrano M, d’Alessandro F, Torrisi MR, Ferretti A, Vulpiani MC, et al. (2011) Extracorporeal shock wave therapy promotes cell proliferation and collagen synthesis of primary cultured human tenocytes. Knee Surgery, Sport Traumatol Arthrosc 19(12): 2159-2168.
- Bosch G, Lin YL, Van Schie HTM, Van De Lest CHA, Barneveld A, et al. (2007) Effect of extracorporeal shock wave therapy on the biochemical composition and metabolic activity of tenocytes in normal tendinous structures in ponies. Equine Vet J 39(3): 226-231.
- Mitchkash M, Robinson D, Tenforde AS (2020) Efficacy of Extracorporeal Pulse-Activated Therapy in the Management of Lower-Extremity Running-Related Injuries: Findings from a Large Case Cohort. J Foot Ankle Surg 59(4): 795-800.
- Wang CJ (2012) Extracorporeal shockwave therapy in musculoskeletal disorders. J Orthop Surg Res 7: 11.
- Speed C (2014) A systematic review of shockwave therapies in soft tissue conditions: Focusing on the evidence. Br J Sports Med 48(21): 1538-1542.
- DeLuca S, Robinson DM, Yun PH, Rosenberg C, Tan CO, Tenforde AS (2021) Similar Functional Gains Using Radial Versus Combined Shockwave Therapy in Management of Plantar Fasciitis. J Foot Ankle Surg 60(6): 1098-1102.
- Cacchio A, Rompe JD, Furia JP, Susi P, Santilli V, et al. (2011) Shockwave therapy for the treatment of chronic proximal hamstring tendinopathy in professional athletes. Am J Sports Med 39(1): 146-153.
- Malay DS, Pressman MM, Assili A, Jason T Kline, Shane York, et al. (2006) Extracorporeal Shockwave Therapy Versus Placebo for the Treatment of Chronic Proximal Plantar Fasciitis: Results of a Randomized, Placebo-Controlled, Double-Blinded, Multicenter Intervention Trial. J Foot Ankle Surg 45(4): 196-210.
- Rompe JD, Nafe B, Furia JP, Maffulli N (2007) Eccentric loading, shock-wave treatment, or a wait- and-see policy for tendinopathy of the main body of tendo Achillis: A randomized controlled trial. Am J Sports Med 35(3): 374-383.
- Schroeder AN, Tenforde AS, Jelsing EJ (2021) Extracorporeal Shockwave Therapy in the Management of Sports Medicine Injuries. Curr Sports Med Rep 20(6): 298-305.
- Wang CJ, Ko JY, Chan YS, M S Lee, J M Chen, et al. (2009) Extracorporeal shockwave for hip necrosis in systemic lupus erythematosus. Lupus 18(12): 1082-1086.
- Liu Y, Zhang T, Feng Y (2018) Radial Extracorporeal Shock Wave Therapy for Relief of Arthralgia in Rheumatoid Arthritis. Pain Pract 18(3): 380-387.
- Sandra Blumhardt, Diana P Frey , Martin Toniolo , Hatem Alkadhi , Ulrike Held, et al. (2017) Safety and efficacy of extracorporeal shock wave therapy (ESWT) in calcinosis cutis associated with systemic sclerosis. Clin Exp Rheumatol 100(5): 177-180.
- Josef S Smolen, Gunter Steiner (2003) Therapeutic Strategies for Rheumatoid Arthritis. N Engl J Med 2(6): 473-488.
- Lee YC, Cui J, Lu B, Michelle L Frits, Christine K Iannaccone, et al. (2011) Pain persists in DAS28 rheumatoid arthritis remission but not in ACR/EULAR remission: A longitudinal observational study. Arthritis Res Ther 13(3): R83.
- Zamora-Legoff JA, Achenbach SJ, Crowson CS, Krause ML, Davis JM, et al. (2016) Opioid use in patients with rheumatoid arthritis 2005–2014: a population-based comparative study. Clin Rheumatol 35(5): 1137-1144.
- Weiss MM (1989) Corticosteroids in rheumatoid arthritis. Semin Arthritis Rheum 19(1): 9-21.
- Taylor P, Manger B, Alvaro-Gracia J, R Johnstone, J Gomez-Reino, et al. Patient perceptions concerning pain management in the treatment of rheumatoid arthritis. J Int Med Res 38(4): 1213-1224.
- Schlaeger JM, Roach K, Golas M, Takayama M, Wilkie DJ (2018) Treatment-Seeking Behaviors of Persons with Rheumatoid Arthritis. J Holist Nurs 36(2): 179-191.
- Ogrinc G, Davies L, Goodman D, Batalden P, Davidoff F, et al. (2016) SQUIRE 2.0 (Standards for QUality Improvement Reporting Excellence): Revised publication guidelines from a detailed consensus process. BMJ Qual Saf 25(12): 986-992.
- Jain A, Witbreuk M, Ball C, Nanchahal J (2002) Influence of steroids and methotrexate on wound complications after elective rheumatoid hand and wrist surgery. J Hand Surg Am 27(3): 449-455.
- Murata K, Yasuda T, Ito H, Yoshida M, Shimizu M, et al. (2006) Lack of increase in postoperative complications with low-dose methotrexate therapy in patients with rheumatoid arthritis undergoing elective orthopedic surgery. Mod Rheumatol 16(1): 14-19.
- Watad A, Cuthbert RJ, Amital H, McGonagle D (2018) Enthesitis: Much More Than Focal Insertion Point Inflammation. Curr Rheumatol Rep 20(7): 41.
- Bandinelli F, Prignano F, Bonciani D, Francesca Bartoli, Ledio Collaku, et al. (2013) Ultrasound detects occult entheseal involvement in early psoriatic arthritis independently of clinical features and psoriasis severity. Clin Exp Rheumatol 31(2): 219-224.






























