Intraoperative Traction in Scoliosis: A Safe and An Effective Tool to Achieve Better Correction
Kedar Prashant Padhye*, Jennifer Hurry, Ron EL-Hawary and Benjamin Orlik
Division of Orthopaedic Surgery, IWK Health Centre, Canada
Submission:September 12, 2022; Published: September 21, 2022
*Corresponding author: Kedar Prashant Padhye, Division of Orthopaedic Surgery, IWK Health Centre, Halifax, Nova, Scotia, Canada
How to cite this article: Kedar P P, Jennifer H, Ron EL-H, Benjamin O. Intraoperative Traction in Scoliosis: A Safe and An Effective Tool to Achieve Better Correction. Ortho & Rheum Open Access J. 2022; 20(3): 556040. DOI: 10.19080/OROAJ.2022.20.556040
Abstract
Purpose: We believe that intraoperative skull-femoral traction (IOT) may effectively assist with spinal deformity correction. The aim of this study is to find out the effect of IOT in single-stage posterior arthrodesis for AIS and NM.
Methods: A retrospective cohort study was performed after Institutional Review Board (IRB) approval. Inclusion criteria were Cobb’s angle >50degrees, single stage posterior spinal instrumented fusion, follow-up >6 months. Growth-friendly surgeries were excluded. Group I consisted of patients with IOT while group II was without IOT.
Results: Group I consisted of 35 patients with mean follow-up of 2.5 years (range 9 months to 6.3 years) and group II had 58 patients with a mean follow-up of 2.11 years (range 6 months to 6.6 years). Correction index was 11.1% more (p-value <0.05) in group I compared to group II. Mean blood loss and operative time were 662 ml (range 205 to 1513ml) and 7.14 hours (range 4.6 to 9.2 hours) in group I, while 647 ml (range 170 to 2200 ml) and 6.04 hours (range 4.1 to 10.2 hours) in group II. OR time was significantly more in group I. There was no statistical difference between the two groups in terms of flexibility index, complication rates, and blood loss. Neurophysiological changes were not seen in the traction group.
Conclusion: We found the use of IOT is a safe and an effective tool to achieve better correction without an increase in complication rates and blood loss.
Level of evidence: Level 3
Introduction
Axial traction has been used for centuries to treat spinal deformities either in the form of preoperative or postoperative traction [1]. The first mention of intra-operative traction (IOT) was made by Cotrel et al in 1988 [2]. Since then, there are a few studies on intra-operative traction by various authors [3–13]. The use of traction exploits the viscoelastic properties of musculoskeletal tissues and aids greater deformity corrections when combined with other procedures. The advantage being it requires lesser corrective maneuvers intra-operatively, with less stress on the spinal instrumentation along with a possible rotation of the apical vertebra [6,10], while disadvantages include neuromonitoring changes, and pin tract related complications [14,15]. We believe that intraoperative skull-femoral skeletal traction may effectively assist with spinal deformity correction in neuromuscular scoliosis (NM) and adolescent idiopathic scoliosis (AIS) patients. The primary aim of this study is to find out the effect of IOT in single- stage posterior arthrodesis for AIS and NM on curve correction and the secondary aim is to study its impact on operative time (OR time), intraoperative blood loss, intraoperative neurophysiological (IOM) changes and complications.
Methods
A retrospective case-control study was conducted at our center after the approval of the Review Board on scoliosis patients operated from the period of 2010 to 2017 and meeting the inclusion and exclusion criteria. Inclusion criteria of the study were cases of idiopathic scoliosis and neuromuscular scoliosis, preoperative major Cobb angle >50 degrees, and cases involving posterior instrumented stabilization and fusion of the spine (PSIF). Exclusion criteria of the study were preoperative Cobb’s angle <50 degrees, cases involving anterior spinal instrumentation, congenital and syndrome scoliosis, and cases of growth-friendly surgeries.
Group I included cases with the use of IOT, while group II included cases without the use of IOT. Radiographs along with the hospital charts were used to gather the necessary information. Radiographic parameters including preoperative Cobb’s angle based on anteroposterior (AP) and stretched/bending x-rays and postoperative Cobb’s angle based on AP x-rays was used to measure correction index and flexibility index. Correction index was defined as [magnitude of postoperative cobb’s angle - magnitude of preoperative upright coronal Cobb’s angle] / preoperative upright coronal Cobb’s angle while flexibility index was defined as [magnitude of the side bend/traction Cobb’s angle-magnitude of the preoperative upright coronal Cobb’s angle] /preoperative upright coronal Cobb’s angle. Demographic information along with surgery-related information including operative time (OR time), intraoperative blood loss, intraoperative neurophysiological (IOM) changes and complications were derived from the hospital charts.
During PSIF, intraoperative traction was applied with Gardner Wells tongs cranially and either skeletal or skin traction caudally. Weights applied in 5 lb increment every 5 minutes with maximum weight not more than 15% of the bodyweight at the head end and each limb ensuring no neuromonitoring changes after each increment. In cases with pelvic obliquity, maximum up to 20% of the bodyweight on the higher side, 10% on the lower side. To accommodate the time taken from the application of traction, OR time was calculated from the time when the patient was ready after anesthesia. Intraoperative neurophysiological monitoring (IOM) was conducted in the form of somatosensory and transcranial motor evoked potentials (SSEP and MEP) by a trained neurophysiologist in all cases. Apical facetectomy was performed in all cases to aid correction and fusion. All radiological and clinical parameters were measured by a trained clinical fellow and confirmed by a senior staff surgeon. A two-tailed student t-test of significance will be used for comparing means of each quantitative data between the two groups, while qualitative data was compared with help of Chi-Square test and Odds ratio was obtained with Mantel-Haenszel common odds estimate. Significance was defined as p< 0.05.
Results
Orthostatic headache is the characteristic feature of intracranial hypotension which was absent in our case. In the presence of preexisting hydrocephalus, MRI findings did not show the classical features of intracranial hypotension. High suspicion of dural tear should be kept in mind, especially in the presence of headache following spinal surgery with evidence of RCH. Persistently increased drain volume should alert the surgeon regarding this possibility. Clamping or removal of the suction drain often result in significant improvement. Early diagnosis could significantly reduce the morbidity in such cases. Our case is interesting as the associated arrested hydrocephalus acted as a red herring in delaying the diagnosis.
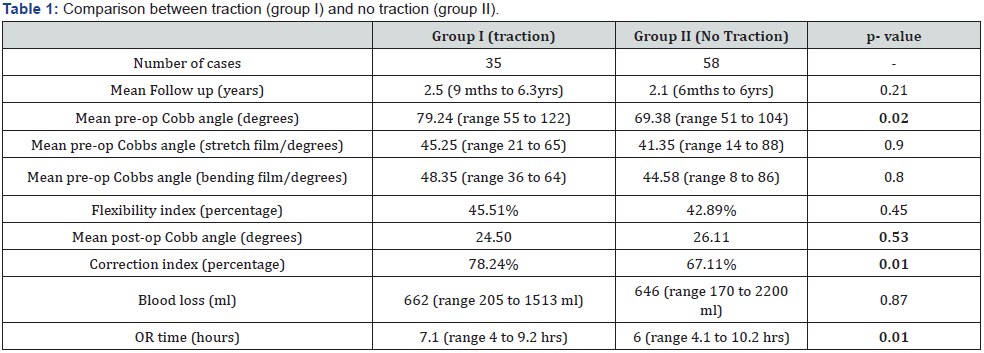
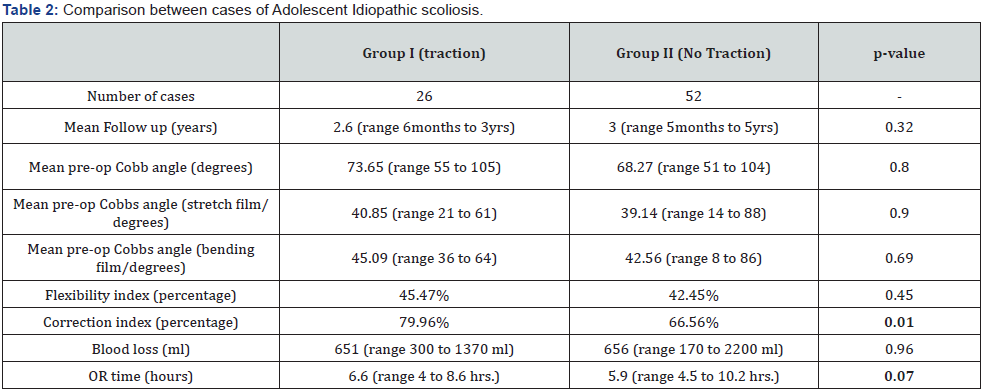
Group I consisted of 35 patients with a mean follow-up of 2.5 years (range 9 months to 6.3 years) and group II had 58 patients with a mean follow-up of 2.11 years (range 6 months to 6.6 years). On further subdivision based on etiology, group I had 26 patients with AIS and 9 with NM scoliosis, while group II had 52 patients with AIS and 5 NM scoliosis. Seven cases of AIS cases had a thoracoscopic release (2 in group I and 5 in group II). None of the NM cases had anterior release. As per the Lenke classification, group I had 15 cases classified as type I, 3 cases as type III, 2 cases as type V, 6 cases as type VI while group II 29 cases classified as type I, 7 cases as type II, 7 cases as type III, 7 cases as type IV, 2 cases as type V. The mean preoperative Cobb angle on standing x-rays, stretch x-rays and bending x-rays was 79.29° (range 55 to 122°), 45.25° (range 21 to 65°) and 48.35° (range 36 to 64°) respectively for group I while for group it was 69.38°(range 51 to 104)°, 45.35° (range 14 to 88°) and 44.58° (range 8 to 86°) respectively (Table 1). Although there was a statistically significant difference between the mean preoperative Cobb angle (p = 0.02), there was no statistically significant difference between the two groups in terms of flexibility index (group I - 45.51%, group II – 42.89%, p = 0.45), which suggests that the two groups were comparable in terms of preoperative flexibility. The groups were further subdivided based on the etiology into AIS and NM (Table 2).
In cases of AIS, the correction index was 79% in group I vs 66% in group II which was statistically significant better in favor of traction group (p = 0.01). In the case of NM scoliosis, correction index was 73% in group I vs 72% in group II, with no statistically significant difference between the groups (p = 0.93). We found that the correction index was 11.1% more (p-value <0.05) in group I compared to group II, seen mainly in the AIS group. Mean blood loss and operative time were 662 ml (range 205 to 1513ml) and 7.14 hours (range 4.6 to 9.2 hours) in group I, while 647 ml (range 170 to 2200 ml) and 6.04 hours (range 4.1 to 10.2 hours) in group II (Table 2). Operative time was significantly more in group I (p = 0.01). Group I had 1 case of a dural leak which was sealed. Group II had 1 case of screw migration which was revised, 2 cases of dural tears which were sealed, 1 case with intraoperative neuromonitoring changes which returned to baseline, and 1 case of implant prominence which was revised at 3-year follow-up. There was no statistical difference between the two groups in terms of complication rates, and blood loss (Table 1).
Discussion
Axial traction has been used for centuries to treat spinal deformities either in the form of preoperative or postoperative traction [1]. The first mention of intra-operative traction (IOT) was made by Cotrel et al in 1988 [2]. Since then, there are a few studies on intra-operative traction by various authors [3–13]. The aims to find out the effect of IOT in single-stage posterior arthrodesis for AIS and NM on perioperative outcomes and overall, on health resource utilization.
In this study, we found that the correction index was 11.1% more (p-value <0.05) in the traction group compared to the group with no intraoperative traction. Comparing based on the etiology, correction index was 13.4 % (p-value <0.05) more in idiopathic scoliosis in group I, while there was no significant difference in cases of neuromuscular scoliosis in both groups. This was considering that the preoperative flexibility index was comparable in both groups, suggesting that the traction benefitted in achieving better curve correction, especially in idiopathic scoliosis. Previous studies assessing the use of intraoperative skull femoral traction with AIS patients do not mention the flexibility index and therefore it cannot be concluded if the two groups were comparable in terms of their preoperative flexibility [5,10,11,14,16]. Few studies have reported better curve correction with traction [6,9,10], while others have reported no difference in the curve correction [4,8,11].
In terms of blood loss, our study did not show any significant difference between the two groups. The mean blood loss was 662 ml in the traction group compared to 647 ml in the group with no traction. In a study by Da Cunha et al. [4], reported blood loss of 1485 ml (range 483–3003 ml) in the traction group compared to 2083 ml (range 839–7130 ml). Although there was significantly less blood loss in the traction group in that study, it is much more than the blood loss reported in this study. Also, that study mentions >70% pedicle screw construct, while in this study a standard pedicle screw construct with transverse process hooks in the most cephalad level was performed in all cases. In this study, the mean OR time was significantly higher in the traction group compared to the no traction group (7.14 hours vs 6.04 hours), which is contrary to the finding in a similar study in the literature [4]. Although the study by Keeler et al. [8] reported less blood loss and OR time with traction, they have compared between posterior approach vs anterior and posterior approach, while the current study has all cases with a posterior approach.
Traction related complications are rare as reported in the literature [4,5]. In the study by Lewis et al. [5], the application of traction led to changes in motor evoked potentials (MEPs) in more severe and stiff AIS curves. In these patients, MEP changes responded immediately by decreasing or removing the traction weight and as a result, there were no long-term permanent neurologic damages. The traction protocol in that study was approximately 20% of body weight (to a maximum of 15 lb) through the Gardner-Wells tongs and 50% of body weight (to a maximum of 65 lb) evenly distributed between the bilateral femurs was used. In this study, there were no traction related complications and protocol used was an application of weight in 5 lb increment every 5 minutes with maximum weight not more than 15% of the bodyweight at head end and each limb ensuring no neuromonitoring changes after each increment. In the case of pelvic obliquity, maximum up to 20% of the bodyweight on the higher side, 10% on the lower side.
The strength of the study is use of a standard traction protocol for all patients. Secondly, both groups were matched in terms of preoperative flexibility and comparison was made with similar pedicle screw construct performed with a posterior approach making both groups comparable preoperatively. The weaknesses of this study include it is a retrospective analysis, a relative low number of patients with NM scoliosis and data includes surgeries by two surgeons with different correction techniques.
Conclusion
Intraoperative traction is a safe and an effective tool which gives better curve correction in cases of AIS and NM scoliosis without causing any significant impact on the blood loss or traction related complications like neuromonitoring changes or pin tract infections.
Conflict of Interest
Dr. Hurray, Dr. Padhye and Dr. Orlik have no conflicts of interest to declare.
References
- Kumar K (1996) Spinal deformity and axial traction. Spine 21(5): 653–655.
- Cotrel Y, Dubousset J, Guillaumat M (1988) New universal instrumentation in spinal surgery. Clin Orthop 227: 10–23.
- Al Sayegh, Samir JLaMotheML (2013) Intraoperative skull femoral traction (ISFT) in posterior instrumentation for adolescent idiopathic scoliosis: Safety and effect on perioperative care. Can J Surg 56: S54-S55.
- Da Cunha RJ, Al Sayegh S, LaMothe JM, Michael Letal, Herman Johal, et al. (2015) Intraoperative skull-femoral traction in posterior spinal arthrodesis for adolescent idiopathic scoliosis: the impact on perioperative outcomes and health resource utilization. Spine 40(3): E154-160.
- Lewis SJ, Gray R, Holmes LM, Samuel Strantzas, Subir Jhaveri, et al. (2011) Neurophysiological changes in deformity correction of adolescent idiopathic scoliosis with intraoperative skull-femoral traction. Spine 36(20): 1627-1638.
- Hamzaoglu A, Ozturk C, Aydogan M, Mehmet Tezer, Neslihan Aksu, et al. (2008) Posterior only pedicle screw instrumentation with intraoperative halo-femoral traction in the surgical treatment of severe scoliosis (>100 degrees) Spine 33(9): 979–983.
- Huang MJ, Lenke LG (2001) Scoliosis and severe pelvic obliquity in a patient with cerebral palsy: Surgical treatment utilizing halo-femoral traction. Spine 26(19): 2168–2170.
- Keeler KA, Lenke LG, Good CR, Keith H Bridwell, Brenda Sides, et al. (2010) Spinal fusion for spastic neuromuscular scoliosis: is anterior releasing necessary when intraoperative halo-femoral traction is used? Spine 35(10): E427–E433.
- Vialle R, Delecourt C, Morin C (2006) Surgical treatment of scoliosis with pelvic obliquity in cerebral palsy: The influence of intraoperative traction. Spine 31(13): 1461–1466.
- Jhaveri SN, Zeller R, Miller S, Lewis SJ (2009) The effect of intra-operative skeletal (skull femoral) traction on apical vertebral rotation. Eur Spine J 18(3): 352–356.
- Mac-Thiong J-M, Labelle H, Poitras B, Charles-Hilaire Rivard, Julie Joncas, et al. (2004) The effect of intraoperative traction during posterior spinal instrumentation and fusion for adolescent idiopathic scoliosis. Spine 29(14): 1549–1554.
- Sponseller PD, Takenaga RK, Newton P, Oheneba Boachie, Jack Flynn, et al. (2008) The use of traction in the treatment of severe spinal deformity. Spine 33(21): 2305–2309.
- Zhang HQ, Wang YX, Guo CF, Ming-xing Tang, Ling-qiang Chen, et al (2011) Posterior-only surgery with strong halo-femoral traction for the treatment of adolescent idiopathic scoliotic curves more than 100°. Int Orthop 35(7): 1037–1042.
- Da Cunha RJ, Al Sayegh S, LaMothe JM, Michael Letal, Herman Johal, et al. (2015) Intraoperative Skull-Femoral Traction in Posterior Spinal Arthrodesis for Adolescent Idiopathic Scoliosis: The Impact on Perioperative Outcomes and Health Resource Utilization. Spine 40(3): E154–E160.
- Sponseller PD, Takenaga RK, Newton P, Oheneba Boachie, Jack Flynn, et al. (2008) The Use of Traction in the Treatment of Severe Spinal Deformity. Spine 33(21): 2305–2309.
- Hamzaoglu A, Ozturk C, Aydogan M, Mehmet Tezer, Neslihan Aksu, et al. (2008) Posterior only pedicle screw instrumentation with intraoperative halo-femoral traction in the surgical treatment of severe scoliosis (>100°). Spine 33(9): 979–983.






























