Diagnostic strategies and Therapeutic Approach to Secondary Osteoporosis
João Lagoas Gomes1,2* and Jaime C Branco1,2
1Rheumatology Department, Hospital Egas Moniz (CHLO), Lisbon, Portugal
2CEDOC, NOVA Medical School, Nova University of Lisbon, Portugal
Submission: January 26, 2019;Published: February 27, 2019
*Corresponding author: João Lagoas Gomes, M.D. (JL Gomes), Rheumatology Department, Hospital Egas Moniz (CHLO), Lisbon, Portugal & CEDOC, NOVA Medical School, Nova University of Lisbon, Serviço de Reumatologia, Hospital Egas Moniz, Rua da Junqueira 126, 1349-019 Lisboa, Portugal
How to cite this article: João Lagoas Gomes, Jaime C Branco. Diagnostic strategies and Therapeutic Approach to Secondary Osteoporosis. Ortho & Rheum Open Access J 2019; 13(4): 555870. DOI: 10.19080/OROAJ.2019.13.555870
Abstract
Osteoporosis is characterized by low bone mass, microarchitectural disruption and skeletal fragility, resulting in decreased bone strength and increased risk of fragility fractures. In fact, fractures are the clinically relevant signs of Osteoporosis, an otherwise silent disease. Demographic changes in the European population are estimated to rise the number of men and women with osteoporosis from 27.5 million in 2010 to 33.9 million in 2025. It is thus important for every clinician to be capable of identifying at risk patients in order to diagnose and treat OP before fractures and disability take place. When starting a diagnostic approach to Osteoporosis, it is important to be aware of its possible secondary causes, which can be highly prevalent in some populations. Also, the clinician must be familiar with specific tests to screen and diagnose secondary Osteoporosis. In this review article, we will discuss in more detail some of the causes of secondary Osteoporosis, providing strategies for screening, diagnosis and treatment.
Keywords: Osteoporosis; Osteopenia; Fragility fracture
Abbrevations: AS: Ankylosing Spondylitis; BMD: Bone Mineral Density; CRP: C-Reactive Protein; DXA: Dual-Energy X-Ray Absorptiometry; ESR: Erythrocyte Sedimentation Rate; GFD: Gluten Free Diet; IGF: Insulin-Like Growth Factors; M-CSF: Macrophage-Colony Stimulating Factor; OP: Osteopososis; PTH: Parathyroid Hormone; PPI: Proton-Pump Inhibitors; PsA: Psoriatic Arthritis; RANKL: Receptor Activator of Nuclear Factor Κb; RA: Rheumatoid Arthritis; SOP: Secondary Osteoporosis; TNF Α: Tumor Necrosis Factor; WHO: World Health Organization
Introduction
Osteoporosis (OP) is characterized by low bone mass, microarchitectural disruption and skeletal fragility, resulting in decreased bone strength and an increased risk of fracture. In fact, fractures are the clinically relevant signs of OP, an otherwise silent disease. Despite being the most relevant determinant of bone strength, bone mineral density (BMD) is not the only factor contributing to the increased risk of fractures with aging. Other important factors include age itself, bone turnover rate, bone geometry and microarchitecture. OP is estimated to affect 200 million women worldwide, 75 million of which in Europe, USA and Japan [1]. In the year 2000, there were an estimated 9.0 million osteoporotic fractures worldwide, of which 1.6 million were at the hip, 1.7 million at the forearm and 1.4 million were clinical vertebral fractures. The greatest number of osteoporotic fractures occurred in Europe (34.8%) [2,3]. Based on the WHO diagnostic criteria (T-score less than or equal to -2.5 SD) approximately 22 million women and 5.5 million men aged 50 to 84 are estimated to have OP in the European Union (2010 figures). Due to changes in population demography the number of men and women with OP in the EU is estimated to rise to 33.9 million in 2025, corresponding to a 23% increase [4]. The remaining lifetime risk of fracture of the spine, hip, distal forearm and proximal humerus for women at 50 years of age is 46.4% [5].
Secondary causes of OP might be more frequent than once thought and should be suspected in the presence of: a) conditions known to induce osteoporosis (Table 1); b) in the presence of fragility fractures occurring before the age of 70 for men or before menopause for women; c) low Z scores (≤-2.0) in dual-energy X-Ray absorptiometry (DXA) studies [6]. In this review article, some the causes of SOP will be discussed in more detail, as well as their screening, diagnosis and treatment. Although widely considered a disease of postmenopausal women, OP is quite prevalent in elderly men [7], which represent 39% of all fragility fractures occurring in the year 2000 [2]. Also, men have greater morbidity and mortality following hip fractures than women [8]. In an observational study [9] taking place in an outpatient rheumatology department, causes of osteoporosis in 81 osteoporotic men were evaluated and secondary osteoporosis was diagnosed in 63 (78%). In men with vertebral crush fractures, some investigators have suggested that 55% have a secondary cause and 20% of these cases are due to hypogonadism. In a series of 214 women with vertebral crush fractures [10], 30.4% were found to have an underlying cause of osteoporosis or early menopause (36.4%) before the age of 45 years.
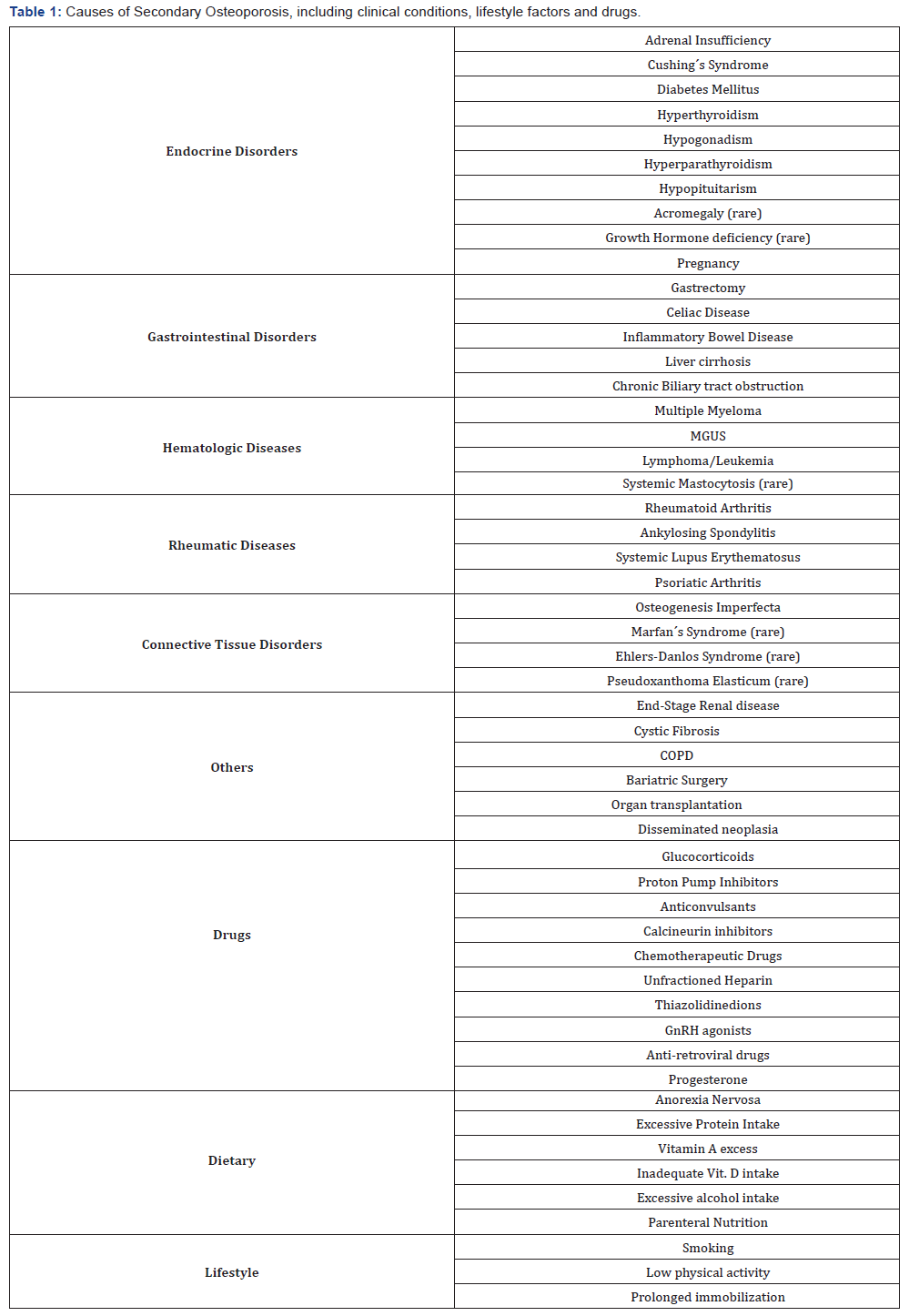
COPD-chronic obstructive pulmonary disease; MGUS-monoclonal gammopathy of undetermined significance
Other estimates of osteoporosis in women suggested that approximately 20% of women who appear to have postmenopausal osteoporosis have an identifiable secondary cause, whereas the incidence of men with a secondary cause has been estimated to be as high as 64%. A cross-sectional study [11] with 173 postmenopausal women with primary osteoporosis showed that after lab tests including complete blood count, renal and hepatic function, chemistry profile, 24-h urinary calcium, 25(OH)vitamin D and parathyroid hormone (PTH), 55 (32%) of them had undiagnosed disturbances of bone and mineral metabolism. The most frequent diagnosis were vitamin D deficiency (20.2%), hypercalciuria (9.8%), malabsorption (8.1%), hyperparathyroidism (6.9%) and exogenous hyperthyroidism (2.3%).
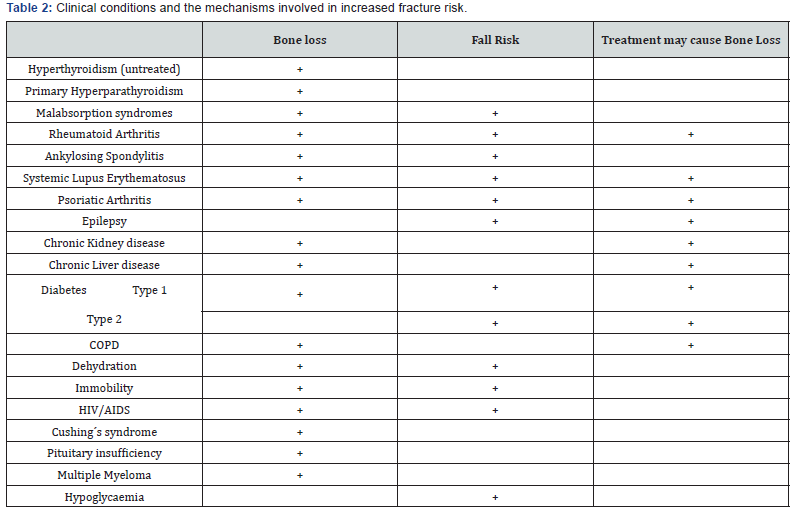
COPD-chronic obstructive pulmonary disease; HIV- human immunodeficiency virus; AIDS- acquired immunodeficiency syndrome.
The treatment, prognosis and monitoring of osteomalacia is different from osteoporosis, thus it is an important differential diagnosis to consider during the initial evaluation of SOP. A significant proportion of women with fragility fractures have osteomalacia and/or osteoporosis. In a retrospective study [12], secondary causes for reduced BMD were evaluated in 196 postmenopausal and 41 premenopausal women. Sixteen percent of these patients had 25-hydroxyvitamin D levels lower than 15 ng/ml. By using the World Health Organization (WHO) definition of osteopenia based on T score value (–1.0 to –2.5), 11% of osteopenic patients had 25-hydroxyvitamin D levels lower than 15 ng/ml. On the other hand, osteomalacia is present in 4% to 47% of men with femoral fractures, with most studies [13,14] reporting a rate of close to 20%. As shown in table 2, several clinical conditions are associated with increased fracture risk not only by directly decreasing bone density or deteriorating bone quality, but also by increasing fall risk. Also, pharmacological treatment of some diseases might lead to decreased bone quality and increased fracture risk due to drug induced SOP.
Drug-Induced Osteoporosis
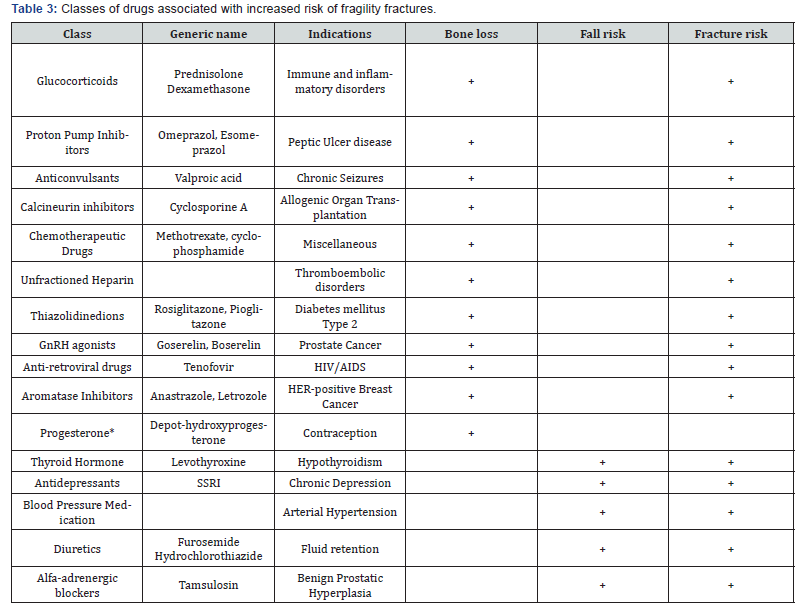
*reversible effect on bone loss; GnRH- gonadotrophin releasing hormone; HER- human epidermal growth factor receptor; HIV- Human Immunodeficiency Virus; AIDS- acquired immunodeficiency syndrome; SSRI- selective serotonin reuptake inhibitors.
Several drugs have been associated with increased fracture risk (Table 3) and glucocorticoids are the most common cause of drug-induced osteoporosis [15]. Bone loss is due to suppression of osteoblast function, inhibition of intestinal calcium absorption leading to secondary hyperparathyroidism and increased osteoclast-mediated bone resorption. Glucocorticoid excess results in diffuse bone loss and may affect trabecular bone more than cortical bone. Bone loss is also promoted by direct stimulation of renal excretion of calcium by glucocorticoids. BMD is reduced in 40% to 60% of patients with an endogenous glucocorticoid excess, and pathologic fractures have been observed in 16% to 67%. The risk of hip fractures is doubled in glucocorticoid-treated patients [16]. Studies evaluating shortterm exposures to glucocorticoid therapy have indicated that glucocorticoid-induced bone loss appears greater in the first 6 to 12 months of therapy [17].
Inhaled glucocorticoid therapy was associated with a doserelated decrease in BMD at the total hip and trochanter (0.00044 g/cm2 per puff per year of treatment) [18]. This finding, along with a retrospective study [10] revealing an association between doses of 2.5 mg daily prednisone and bone loss, suggest a low threshold at which glucocorticoids cause skeletal harm. Fracture risk increases with dose and duration of glucocorticoid use [19]. Serum and urine biochemical indices in patients with glucocorticoid-induced osteopenia are generally normal, but urinary markers of bone resorption may be increased. Serum PTH levels may be normal or mildly elevated (secondary hyperparathyroidism) and serum alkaline phosphatase activity and osteocalcin levels decline steadily after the initiation of glucocorticoid therapy, reflecting inhibition of osteoblast activity. Urinary calcium excretion may be increased during the first several months to years of glucocorticoid therapy because of the direct calciuric effect of glucocorticoids on the kidney. The first principle in the treatment of patients with glucocorticoidinduced OP is to use the lowest effective dose of glucocorticoid. General health measures that are applicable to patients with OP should be encouraged, such as weight-bearing exercise and good nutritional status.
According to the 2017 update of the American College of Rheumatology recommendations for the prevention and treatment of glucocorticoid-induced OP, patient taking glucocorticoids should optimize calcium (1,000–1,200 mg/ day) and vitamin D intake (600–800 IU/day) [19]. For postmenopausal women and men >40 years starting glucocorticoid therapy with an anticipated duration of more than 3 months, risk stratification using FRAX and the dose of glucocorticoids are key for an adequate therapeutic decision. For low risk patients, no further treatment is recommended and patients should be monitored yearly for clinical features and with DXA every 2-3 years. For moderate to high risk patients, oral bisphosphonates should be started and are still the first-line of treatment [19]. Bone diseases associated with anticonvulsant therapy is a form of osteomalacia. In this condition, high-turnover osteoporosis is often present. Phenobarbital, diphenylhydantoin, and carbamazepine, 3 commonly used anticonvulsants, increase the metabolism and clearance of vitamin D. Thus, Rickets has been observed in children taking anticonvulsant medication. In some reports [20,21], rates were as high as 20% to 65%, with patients being at particularly increased risk of fracture during seizures.
In the outpatient setting, abnormalities on bone biopsy specimens, such as increased osteoid, are observed in 10% to 40% of patients receiving long-term anticonvulsant therapy. However, if the patient is well nourished and exposed to adequate amounts of sunlight, clinically significant bone disease is less likely to occur. Many retrospective studies have showed that long-term use of proton-pump-inhibitors (PPI) is associated with an increased risk of fragility fractures [22], but the underlying mechanisms have not been clarified yet. Chronic acid suppression caused by long-term proton pump inhibitor therapy may play a crucial role in decreased absorption of calcium and vitamin B12 and, therefore, indirectly affecting the bones resulting in decreased BMD. The available data suggest that proton pump inhibitors should be used with caution in patients with increased risk of osteoporosis.
Inflammatory Rheumatic Diseases
In inflammatory rheumatic diseases such as rheumatoid arthritis (RA), psoriatic arthritis (PsA) and ankylosing spondylitis (AS) the systemic burden caused by inflammation, glucocorticoid treatment, immobilization and reduced physical activity due to painful joints and muscle weakness are associated with decreased bone mass and deterioration of bone quality leading to increased risk of falls and fragility fractures [23]. The reason for this effect on bone is thought to come from the local and systemic action of pro-inflammatory cytokines, such as an increased expression of receptor activator of nuclear factor κB (RANKL), macrophage-colony stimulating factor (M-CSF) and the presence of Tumor Necrosis Factor (TNF α), IL-1, IL-6, IL-7, and IL-17. Other factors probably contributing to the increased risk of fracture in this population include older age, lack of physical exercise, long-term use of corticosteroids and high disability index [24,25].
The frequency of occurrence of osteoporosis in patients with inflammatory rheumatic diseases, including RA, is about 50% and the course of osteoporosis is closely connected with the activity of the underlying disease [26]. With regards to RA, local and systemic phenomena of bone loss occur, and they seem to be intimately related. Early in the course of the disease reduced BMD within joints occurs, called periarticular osteoporosis, which is associated with the destructive articular processes of bone erosion and joint space narrowing. In the course of RA, especially with longstanding active disease, secondary osteoporosis occurs. Currently, it is thought that the mechanisms causing periarticular and secondary osteoporosis are at least partly the same [27]. Unlike postmenopausal osteoporosis, osteoporosis in RA is characterised by a marked loss of cortical bone (hip and the radius), while the axial bone is relatively preserved [25], except when high cumulative doses of glucocorticoids are used.
In contrast with RA, AS is associated with both osteopenic changes and bone forming phenomena. The reported prevalence of osteoporosis in AS patients varies largely and this variation reflects the difficulties in assessing BMD in AS due to new bone formation. There are also inflammation-induced structural changes in the spine predisposing to vertebral wedge and fracture. In a prospective cohort study [28] involving 504 AS patients there was a significantly higher prevalence of OP (9,7% vs 0%) and osteopenia among AS patients (57,5% vs. 34,9%) comparing with healthy controls. The BMD was significantly lower in the patients with higher elevated erythrocyte sedimentation rate (ESR) or C-reactive protein (CRP). Risk factors for lumbar spine bone loss were Juvenile onset, morning stiffness lasting over 30 minutes and elevated ESR levels, which correlate with the inflammatory burden of the disease.
A case-control study to assess the prevalence of fragility fractures in patients with PsA [29] found that disease duration, age and femoral neck BMD were associated with increased rate of non-vertebral fractures and a large population-based cohort study [30] including 9788 PsA patients, 158,323 psoriasis patients and 821,834 matched controls reported that PsA and psoriasis patients had a 7–26% increased incidence of fracture when compared to the general population.
Endocrine Dysfunction
Primary hyperparathyroidism is a common disorder, with an incidence of 1 in 500 to 1 in 1000 and is usually asymptomatic [31]. Classically, primary hyperparathyroidism is associated with osteitis fibrosa cystica characterized by subperiosteal resorption of the distal phalanges, tapering of the distal clavicles, a “salt and pepper” appearance of the skull, brown tumors, and bone cysts. With the widespread availability of screening blood tests for asymptomatic hypercalcemia, the incidence of this manifestations has decreased dramatically over the years. Osteopenia and osteoporosis are also recognized as bone diseases associated with excess PTH. The increase in circulating levels of PTH in primary hyperparathyroidism is associated with increased bone turnover, both in osteoclast-mediated bone resorption and osteoblast activity, leading to the loss of cortical and trabecular bone. In mild hyperparathyroidism, however, BMD may be increased in areas that are primarily trabecular, whereas bone is lost in the cortical areas [32]. This anabolic effect is the basis for the treatment with PTH analogues. Nevertheless, bone resorption is enhanced by sustained elevations in the levels of PTH.
In a longitudinal cohort of patients with primary hyperparathyroidism [33], bone density has been measured at 3 sites to evaluate cortical bone, trabecular bone, and a mixture of both. At the distal radius, an area rich in cortical bone, BMD was less than 80% of age- and sex-matched controls. In contrast, at the lumbar spine, BMD was relatively well preserved. The values for the hip region, which is made of mixed trabecular and cortical bone, were midway between the data obtained for the spine and the distal radius. This finding is consistent with the observation that PTH mobilizes calcium from cortical sites before it has a negative impact on trabecular skeleton. Quantitative histomorphometric analyses of bone biopsy specimens are consistent with the loss of cortical bone and preservation of cancellous bone [32]. Cortical thinning is noted on biopsy specimens, and PTH levels correlate with cortical porosity [33].
After surgical cure of primary hyperparathyroidism, BMD increases in the forearm and lumbar spine. In a longitudinal cohort [34] of patients with primary hyperparathyroidism followed up for 10 years, parathyroidectomy resulted in normalization of biochemical values and increased BMD. The increase in bone density was prompt and sustained, but a trend toward further increase after 1 year was significant only for femoral neck values. In a subset of patients who did not undergo surgery, there was no progression of bone disease if they were asymptomatic, but one quarter had some progression with bone loss. Regarding the thyroid gland, both thyroid hormone insufficiency and excess can lead to alterations in bone mass. Thyroid hormone increases the creation of new bone remodeling units with an enhancement of remodeling activity. Thyroid hormones directly stimulate production of osteocalcin, alkaline phosphatase, and insulin-like growth factors (IGF).
In patients with thyrotoxicosis, increased serum levels of osteocalcin and alkaline phosphatase may be seen. Despite the increase in osteoblast activity, there are also thyroid hormone– induced increases in bone resorption. In the thyrotoxic patient, the bone remodeling cycle is shortened because of a decrease in the length of the bone formation and, overall, there is failure to replace resorbed bone completely, leading to bone loss. In patients with thyrotoxicosis, BMD is reduced [35,36]. Several studies indicated that individuals with a history of thyrotoxicosis have an increased risk of fracture and may sustain fracture at an earlier age compared with patients who have never had an increase in thyroid hormone levels. After effective treatment of the thyrotoxic patient, the decrease in BMD may be reversible. Normalization of the results of thyroid function tests results in increased BMD comparing with pre-treatment values [37].
In a meta-analysis [38], BMD was assessed in women receiving thyrotropin-suppressive doses of thyroxin. The study concluded that there was a 1% increase in annual bone loss in postmenopausal women. A large prospective study, the Study of Osteoporotic Fractures [39], examined the relationship between thyroid disease and fractures. In this study, postmenopausal women with a history of hyperthyroidism had an 80% increased risk of subsequent hip fracture. Thyroid hormone use itself was associated with a 60% increase in fracture risk.
Eating Disorders
Anorexia nervosa and bulimia are associated with significant morbidity and mortality and are chronic in nature. They affect 5% to 10% of women and the onset may be at any time from adolescence through the fourth decade of life. Anorexia nervosa has been associated with osteoporosis. There are several metabolic disorders associated with anorexia nervosa that may adversely affect bone metabolism. These include estrogen deficiency, endogenous cortisol excess, reduced IGF-1 levels, protein-energy malnutrition, and secondary hyperparathyroidism due to low dietary calcium intake or vitamin D deficiency. It has been estimated that 50% of anorexic patients have BMD values at the lumbar spine that are more than 2 SDs below those of age-matched, healthy controls [40]. Total alkaline phosphatase activity may be elevated, but liver enzyme levels are also elevated. Osteocalcin has been noted to be very low in women with anorexia nervosa and may be due to the excess endogenous cortisol levels. Markers of bone resorption, such as pyridinoline and N-telopeptide excretion are usually increased.
Celiac Disease
Regarding bone involvement in celiac disease, it can present as osteoporosis, osteomalacia, or both. Untreated adults usually present with reduced bone mineral at the time of diagnosis, whereas children may present with growth retardation. Chronic inflammatory intestinal diseases, including celiac disease, can affect bone and mineral metabolism due to changes in both systemic and local regulatory factors. The pathogenetic processes are still controversial, but two main mechanisms seem to be involved: intestinal malabsorption and the presence of chronic inflammation [41]. A strict and lifelong gluten free diet can help recover normal bone density when a diagnosis of Celiac disease is made in children and adolescents [42, 43]. In the case of adult celiac disease with associated bone disease, a gluten free diet rarely normalizes BMD in adulthood [41,44]. A cross-sectional study [45] as shown that despite long-term strict adherence to gluten free diet (GFD), 74% of patients displayed low BMD. Among these, 24% had osteoporosis and 76% osteopenia. Therefore, nutritional supplementation should be considered for all patients with celiac disease. Patients may present with normal serum biochemical analysis results or with reduced serum and urine calcium levels and elevated alkaline phosphatase levels. With GFD, biochemical abnormalities and BMD measurements may improve [45].
Conclusion
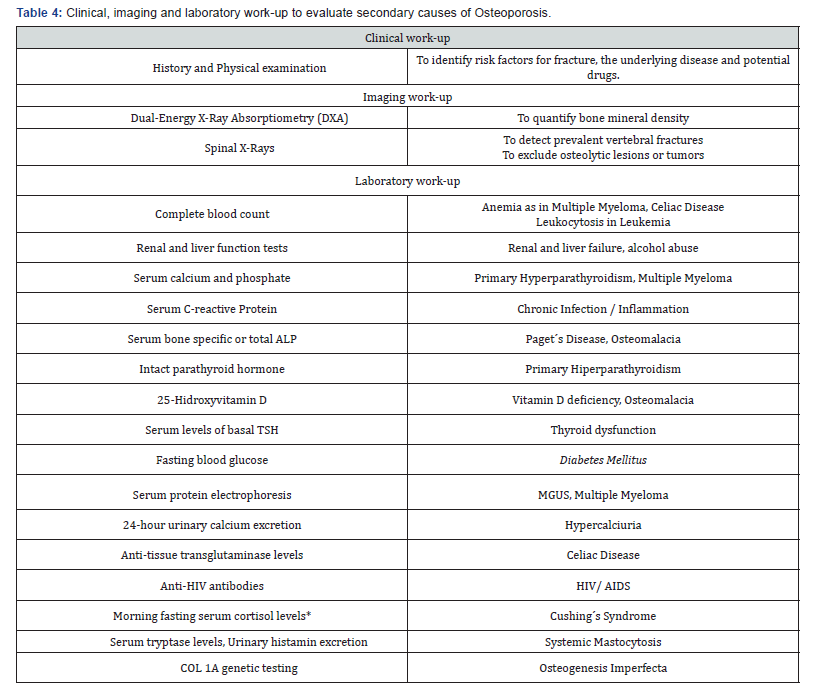
*After dexamethasone suppression; ALP- alkaline phosphatase; COL 1A- collagen type I alpha 1; COPD-chronic obstructive pulmonary disease; HIV- human immunodeficiency virus; MGUS-monoclonal gammopathy of undetermined significance; TSH- thyroid stimulating hormone
In conclusion, an appropriate diagnostic workup should be performed for patients presenting with fragility fractures or those presenting with risk factors for secondary osteoporosis. As shown in table 4, this should include a careful clinical history and physical examination, imaging work-up with DXA to quantify BMD and spinal X-rays to detect prevalent vertebral fractures and exclude osteolytic lesions or tumours. In terms of laboratory work-up, a complete blood count should be performed to exclude anaemia (as in multiple myeloma, celiac disease or leukaemia), renal and liver function tests to exclude renal and liver impairment as well as alcohol abuse, serum calcium and phosphate as well as intact parathyroid hormone to exclude hyperparathyroidism, ESR and CRP to exclude chronic infection/inflammation, 25-hydroxyvitamin D to exclude vitamin D deficiency and osteomalacia, serum bone specific or total alkaline phosphatase for Paget´s disease of the bone, serum protein electrophoresis for monoclonal gammopathy of undetermined significance (MGUS)/multiple myeloma and a 24 hour urinary calcium excretion to check for hypercalciuria.
References
- Kanis JA (2007) on behalf of the World Health Organization Scientific Group (2007) Assessment of osteoporosis at the primary health-care level. Technical Report. World Health Organization Collaborating Centre for Metabolic Bone Diseases, University of Sheffield, UK. Printed by the University of Sheffield, England.
- Johnell O, Kanis JA (2006) An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int 17: 1726–1733.
- Cummings SR, Melton LJ (2002) Epidemiology and outcomes of osteoporotic fractures. Lancet 359: 1761-1767.
- Hernlund E, Svedbom A, Ivergard M, Compston J, Cooper C, et. Al (2013) Osteoporosis in the European Union: Medical Management, Epidemiology and Economic Burden. A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch Osteoporos 8: 136
- Kanis JA, Johnell O, Oden A, Sembo I, Redlund-Johnell I, et al. (2006) Long-term risk of osteoporotic fracture in Malmo. Osteoporos Int 11(8): 669–674.
- Rodrigues AM, Canhão H, Marques A, Ambrósio C, Borges J, et al (2018) Portuguese recommendations for the prevention, diagnosis and management of primary osteoporosis – 2018 update. Acta Reumatol Port 43(1): 10-31.
- Kaufman JM, Reginster JY, Boonen S, Brandi ML, Cooper C, et al. (2013) Treatment of osteoporosis in men. Bone 53(1): 134-144.
- Seeman E (2002) Pathogenesis of bone fragility in women and men. Lancet 359(9320): 1841-1850.
- Peris P, Guanabens N, Monegal A, Suris X, Alvarez L, et al. (1995) Aetiology and presenting symptoms in male osteoporosis. Br J Rheumatol 34(10): 936-941.
- Caplan GA, Scane AC, Francis RM (1994) Pathogenesis of vertebral crush fractures in women. J R Soc Med 87(4): 200-202.
- Tannenbaum C, Clark J, Schwartzman K, Wallenstein S, Lapinski R, et al. (2002) Yield of laboratory testing to identify secondary contributors to osteoporosis in otherwise healthy women. J Clin Endocrinol Metab 87(10): 4431-4437.
- Haden ST, Fuleihan GE, Angell JE, Cotran NM, LeBoff MS (1999) Calcidiol and PTH levels in women attending an osteoporosis program. Calcif Tissue Int 64: 275-279.
- Fitzpatrick LA (2002) Secondary Causes of Osteoporosis. Mayo Clin Proc Vol 77(5): 453-468.
- Orwoll ES (1998) Osteoporosis in men. Endocrinol Metab Clin North Am 27: 349-367.
- Panday K, Gona A, Humphrey MD (2014) Medication-induced osteoporosis: screening and treatment strategies. Ther Adv Musculoskelet Dis 6(5): 185-202.
- Van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C (2000) Use of oral corticosteroids and risk of fractures. J Bone Miner Res 15(6): 993-1000.
- Michel GA, Bloch DA, Wolfe F, Fried JF (1993) Fractures in rheumatoid arthritis: an evaluation of associated risk factors. J Rheumatol 20(10): 1666-1669.
- Israel E, Banerjee TR, Fitzmaurice GM, Kotlov TV, LaHive K, et al (2001) Effects of inhaled glucocorticoids on bone density in premenopausal women. N Engl J Med 345: 941-947.
- Buckley L, Guyatt G, Fink HA, Cannon M, Grossman J, et al. (2017) American College of Rheumatology guideline for the prevention and treatment of glucocorticoid-induced Osteoporosis. Arthritis Care Res 69(8): 1095-1110.
- Dent CE, Richens A, Rowe DJ, Stamp TC (1970) Osteomalacia with long-term anticonvulsant therapy in epilepsy. Br Med J 4(5727): 69-72.
- Hunter J, Maxwell JD, Stewart DA, Parsons V, Williams R, et al. (1971) Altered calcium metabolism in epileptic children on anticonvulsants. Br Med J 4(5781): 202-204.
- Fraser LA, Leslie WD, Targownik LE, Papaioannou A, Adachi JD, et al. (2013) The effect of proton pump inhibitors on fracture risk: report from the Canadian Multicenter Osteoporosis Study. Osteoporos Int 24(4): 1161-1168.
- Maruotti N, Corrado A, Cantatore FP (2014) Osteoporosis and rheumatic diseases. Reumatismo 66(2): 125-135.
- Matuszewska A, Szechiński J (2014) Mechanisms of osteoporosis development in patients with rheumatoid arthritis. Postepy Hig Med Dosw 68:145-152.
- Vosse D, de Vlam K (2009) Osteoporosis in rheumatoid arthritis and ankylosing spondylitis. Clin Exp Rheumatol 27(4 Suppl 55): S62-S67.
- Węgierska M, Dura M, Blumfield E, Żuchowski P, Waszczak M, et al. (2016) Osteoporosis diagnostics in patients with rheumatoid arthritis, Reumatologia 54(1): 29-34.
- Vis M, Havaardsholm EA, Haugeberg G, Uhlig T, Voskuyl AE, et al. (2006) Evaluation of bone mineral density, bone metabolism, osteoprotegerin and receptor activator of the NFkappaB ligand serum levels during treatment with infliximab in patients with rheumatoid arthritis. Ann Rheum Dis 65(11): 1495-1499.
- Wang DM, Zeng QY, Chen SB, Gong Y (2015) Prevalence and risk factors of osteoporosis in patients with ankylosing spondylitis: a 5-year follow-up study of 504 cases. Clin Exp Rheumatol 33(4): 465-470.
- Del Puente A, Esposito A, Costa L, Benigno C, Del Puente A, et al. (2015) Fragility Fractures in Patients with Psoriatic Arthritis. J Rheumatol Suppl 93: 36-39.
- Ogdie A, Harter L, Shin D, Baker J, Takeshita J, et al. (2016) The risk of fracture among patients with psoriatic arthritis and psoriasis: a population-based study. Ann Rheum Dis 76(5): 882-885.
- Bilezikian JP, Silverberg SJ (2000) Clinical spectrum of primary hyperparathyroidism. Rev Endocr Metab Disord 1(4): 237-245.
- Dempster DW, Parisien M, Silverberg SJ, Liang XG, Schnitzer M, et al. (1999) On the mechanism of trabecular bone preservation in postmenopausal women with mild primary hyperparathyroidism. J Clin Endocrinol Metab 84(5): 1562-1526.
- Silverberg SJ, Shane E, Jacobs TP, Siris E, Bilezikian JP (1999) A 10-year prospective study of primary hyperparathyroidism with or without parathyroid surgery. N Engl J Med 341(17): 1249-1255.
- Parisien M, Silverberg SJ, Shane E, de la Cruz L, Lindsay R, et al. (1990) The histomorphometry of bone in primary hyperparathyroidism: preservation of cancellous bone structure. J Clin Endocrinol Metab. 70(4): 930-938.
- Mosekilde L, Eriksen EF, Charles P (1990) Effects of thyroid hormone on bone and mineral metabolism. Endocrinol Metab Clin North Am 19(1): 35-63.
- Rosen CJ, Alder RA (1992) Longitudinal changes in lumbar bone density among thyrotoxic patients after attainment of euthyroidism. J Clin Endocrinol Metab 75(6): 1531-1534.
- Obermayer-Pietsch B, Dobnig H, Warnkross H, Dimai HP, Weber K, et al. (2000) Variable bone mass recovery in hyperthyroid bone disease after radioiodine therapy in postmenopausal patients. Maturitas 35(2): 159-166.
- Faber J, Galloe AM (1994) Changes in bone mass during prolonged subclinical hyperthyroidism due to L-thyroxine treatment: a meta-analysis. Eur J Endocrinol 130(4): 350-356.
- Cummings SR, Nevitt MC, Browner WS, Stone K, Fox KM, et al. (1995) Risk factors for hip fracture in white women: study of Osteoporotic Fractures Research Group. N Engl J Med 332(12): 767-773.
- Biller BM, Saxe V, Herzog DB, Rosenthal DI, Holzman S, et al. (1989) Mechanisms of osteoporosis in adult and adolescent women with anorexia nervosa. J Clin Endocrinol Metab 68(3): 548-554.
- Bianchi ML, Bardella MT (2008) Bone in celiac disease. Osteoporos Int 19(12): 1705-1716.
- Mora S, Weber, G, Barera, G, Bellini, A, Pasolini D, et al. (1993) Effect of gluten-free diet on bone mineral content in growing patients with celiac disease. Am J Clin Nutr 57(2): 224-228.
- Mora S, Barera G, Ricotti A, Weber G, Bianchi C, et al. (1998) Reversal of low bone density with a gluten-free diet in children and adolescents with celiac disease. Am J Clin Nutr 67(3): 477–448.
- Capriles VD, Martini LA, Arêas JAG (2009) Metabolic osteopathy in celiac disease: importance of a gluten-free diet. Nutr Rev 67(10): 599–606.
- Larussa, T, Suraci E, Nazionale I, Leone I, Montalcini T, et al. (2012) No evidence of circulating autoantibodies against osteoprotegerin in patients with celiac disease. World J Gastroenterol 18(14): 1622–1627.






























