Docking Study of Novel Thiophene Derivatives Synthesized by Stille Cross Coupling with Potent Anti-Cancer Activity
Ibrahim F Nassar1* and Nasser SM Ismail2
1Faculty of Specific Education, Ain Shams University, Abassia, Cairo, Egypt
2Pharmaceutical Chemistry Department, Faculty of Pharmaceutical Sciences and Pharmaceutical Industries, Future University, Cairo 12311, Egypt
Submission: August 28, 2023;Published: September 08, 2023
*Corresponding author: Ibrahim F Nassar, 1Faculty of Specific Education, Ain Shams University, 365 Ramsis street, Abassia, Cairo, Egypt
How to cite this article: Ibrahim F N, Nasser SM I. Docking Study of Novel Thiophene Derivatives Synthesized by Stille Cross Coupling with Potent Anti-Cancer Activity. Organic & Medicinal Chem IJ. 2023; 12(5): 555847. DOI: 10.19080/OMCIJ.2023.12.555847
Abstract
Background: Novel diaryldithiophene and hetero conditions. Isophane Derivatives (7-11) were designed and synthesized under stille cross coupling reaction conditions.
Materials and Methods: The new compounds were evaluated for their antitumor activities in 3 different cell lines; MCF7, HEPG2 and Lung Cancer.
Results: Compounds 7, 8, 9, 10 and 11 exhibited highly potent cytotoxic activity with IC50 values in 0.042 - 4.09 μM ranges. A docking study of the synthesized compounds was carried out using complex (H10V).
Conclusion: Molecular docking simulation was performed to position compounds 7-11 into the EGFR active site to determine the probable binding mode.
Keywords: Carbon-Carbon Bond Formation; Stannane; Cytotoxic Activity; Docking Study; CDK2
Abbreviations: SRB: Sulforhodamine B; HBV: Hepatitis B Virus; CDK2: Cyclin-Dependent Kinase 2; DMEM: Dulbecco’s Modified Eagle’s Medium; FBS: Heat Inactivated Fetal Bovine Serum
Introduction
The development of new anticancer therapeutic agents is one of the fundamental goals in medicinal chemistry as cancer causes about 13 % of all the death [1]. Uracil and other pyrimidine derivatives are effective anticancer agents especially for prostate cancer with cathepsin B inhibition [2], mouse tumor (EAC), human liver (HepG2), breast (MCF-7) and colon (HCT-29) cancer cell lines [3-10]. Also, Pyrimidine derivatives have moderate inhibition activity against HBV with mild cytotoxicity activity [11]. While some pyridine derivatives exhibit considerable vasodilation properties on thoracic aortic rings of Wister rats [12]. On the other hand, Palladium metal was found to be the most effective transition-metal for catalyzing C-C coupling reactions of the last decades [13-17]. This reaction has been used in the syntheses of various biaryl compounds, which are important intermediates in the syntheses of natural products, functionalized polymers, and pharmaceuticals [18]. Pd-complex catalysts are generally used in cross-coupling reactions, not only in Migitae Kosugie Stille cross coupling reactions, but also in other relevant cross-couplings such as Suzukie Miyaura [19], Mizorokie Heck [20], and Hiyamae Hatanaka [21] cross-coupling reactions. In continuation of the previous work with Carbon-Carbon coupling reactions of thiophene with various aryl and heterocyclic rings as, tetrahydroisoquinoline bromide, and other allyl halides [22-25], and searching for new effective anticancer agents, this study was performed with the stille cross coupling reaction between thiophene and dihalobiphenyldiol, its benzyloxy derivative, dihalopyridine as well as with monohalopyrimidine-2,4-dione and with monohaloquinoline-3-carbaldehyde to explore the effect of the [Pd]0 catalyst on this reactions. The yielded compounds were tested for their antitumor activity in 3 different cell lines; MCF7, HEPG2 and Lung Cancer which show good activity against the tumor cells with docking study.
Materials and Methods
Experimental
Chemistry
All melting points were measured using a kofler block apparatus and are uncorrected. Yields listed are of isolated compounds. The IR spectra were recorded on a perkin-Elmer model 1720 FTIR spectrometer for KBr disc. 1H NMR and 13C NMR spectra were recorded on a varian Gemini 300 BB NMR Spectrometer at 400 MHz. Chemical shifts were reported in δ scale (ppm) relative to TMS as a reference standard and the coupling constants J values are given in Hz. Mass spectra were recorded on GC/MS Finnegan SSQ 7000 spectrophotometer & GC Ms-QP 1000 EX mass spectrometer at 70 ev. The progress of the reactions was monitored by TLC using aluminum silica gel plates 60 F245. Elemental analyses were performed at the Microanalytical centre at Faculty of science, Cairo University, Egypt. The Antitumor Evaluation of the synthesized compounds was carried out at the faculty of pharmacy, Ain Shams University, Cairo, Egypt.
Synthesis of Bis(Dibenzylidineacetone)Palladium(0) Catalyst
A mixture of PdCl2 (1.77 g, 0.01 mol) and NaCl (0.58 g, 0.01 mol) was stirred in methanol (50 ml) at room temperature for 16 hrs. The resulting solution was filtered through a cotton plug and the filtrate was diluted to 300 ml with methanol. The diluted filtrate was then heated to 60oC and 7.28 g of (dibenzylideneacetone) were added to warm, stirred Na2PdCl6 filtrate. Stirring was continued for further 15 minutes followed by addition of anhydrous sodium acetate (15g) where the reaction commences at once. The solution was stirred for 1hour till cooling to room temperature; the resulting dark brown precipitate was filtered, washed with (5 x 25 ml) methanol, with (5 x 25 ml) water and with (5 x 25 ml) acetone affording pure [Pd(dba)2] (4.59 g, 80 %).
General Procedure for the Preparation of the Stille Coupling Compounds
In a capped flask were placed a solution of the mono or dibromoaryl derivative (0.01 mol), Pd(dba)2 (10 mol %) and one or two equivalents of tributyl(thiophen-2-yl)stannane 1 in toluene (20 ml), the flask was sealed under nitrogen and the reaction mixture was heated at 110oC for 24 hrs., then cooled to room temperature. The reaction mixture was quenched with water (25 ml) and KF (250 mg) was added to the mixture with stirring for 5 hrs., then neutralized with aqueous ammonium chloride solution. The resultant mixture was filtered to remove the unwanted solid (Bu3SnF) and the filtrate was evaporated in vacuum to give an oily residue, which was isolated by extraction with ethyl acetate. The organic layer was washed with brine, dried over MgSO4, and evaporated under vacuum to produce the pure products.
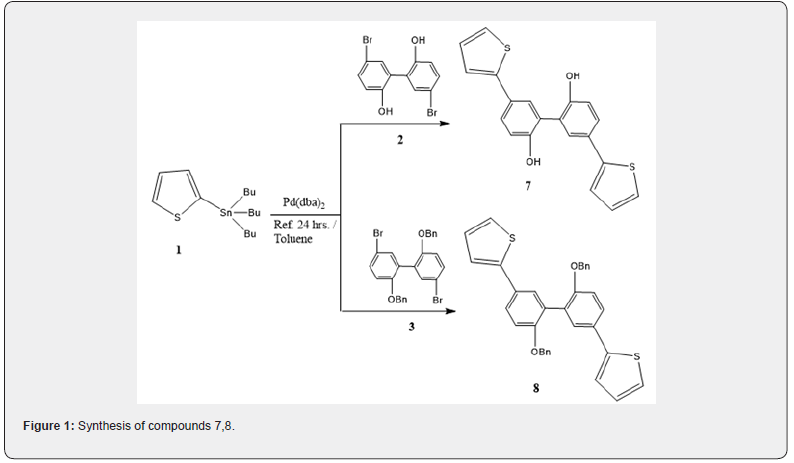
5,5`-Bi(Thiophen-2- yl)Biphenyl-2,2`-Diol (7)
Yellowish solid, recrystallized from methanol; yield (45%); mp 165-166 oC; IR (KBr) νmax: 3340, 3248 (OH), 1612 (C=C) cm- 1; 1H NMR (DMSO-d6, 400 MHz): δ 7.83- 7.12 (m, 12H, Ar-H), 9.0 (b s, 2H, 2 OH) ppm; 13C NMR (DMSO-d6,100 MHz): δ = 117.0 - 140.5 (Ar-C + thiophene-C), 155.5 (C-O) ppm; GC-MS: (M+, %) m/z 350 (25.95), 333 (9.31), 291 (8.65), 233 (8.91), 151 (7.21), 79.95 (100), 72 (8.91); Analysis Calcd. for C20H14O2S2 (350.45) C, 68.55; H, 4.03; S, 18.30; Found C, 68.60; H, 4.15; S, 18.55.
2,2`-Bis-Benzyloxy-5,5`-Bi(Thiophen-2-yl)-Biphenyl (8)
Green yellowish crystals; recrystallized from ethyl acetate; yield (65%), mp 170-171 oC; IR (KBr) νmax: 3062, 3032 (C-H) cm- 1; 1H NMR (DMSO-d6, 400 MHz): δ 7.90 - 7.10 (m, 22H, Ar-CH), 5.11, 5.07 (2s, 4H, 2 CH2) ppm; 13C NMR (DMSO-d6,100 MHz): δ = 71.1 (CH2), 115.0 - 140.5 (Ar-C + thiophene-C), 148.5 (C-O) ppm; GC-MS: (M+, %) m/z 530 (59.32), 447 (44.07), 340 (69.49), 256 (60.17), 152 (91.53), 106 (100), 82 (27.12); Analysis Calcd. for C34H26O2S2 (530.70) C, 76.95; H, 4.94; S, 12.08; Found C, 76.75; H, 4.95; S, 12.15.
2,4-Bi(Thiophen-2-yl)Pyridine (9)
Brown yellowish crystals, recrystallized from ethyl alcohol, yield (40%); mp 140-141oC, IR (KBr) νmax: 3062, 3032 (C-H), 1603 (C=N) cm-1; 1H NMR (DMSO-d6, 400 MHz): δ 8.70 (s, 1H, pyridine- CH), 8.72, 8.65 (2 d, J1 = 2.1, J2 = 2.4 Hz, 2H, pyridine-CH), 7.90- 7.17 (m, 6H, thiophene-CH) ppm; 13C NMR (DMSO-d6,100 MHz): δ = 112.5 - 152.3 (pyridine-C + Thiophene-C) ppm; GC-MS: (M+, %): m/z 243 (33.67), 159 (29.59, 77 (100), 61 (27.55); Analysis Calcd. for C13H9NS2 (243.34) C, 64.17; H, 3.73; N, 5.76; S, 26.35; Found C, 64.05; H, 3.65; N, 5.55; S, 26.50.
5-(Thiophen-2-yl)Uracil (10)
Black crystals; recrystallized from toluene; yield (50%); mp 155-156 oC; IR (KBr) νmax: 3162 (NH), 3061, 3029 (C-H), 1698, 1650 (2 C=O), 1603 (C=C) cm-1; 1H NMR (DMSO-d6, 400 MHz): δ 11.30, 11.10 (bs, 2H, 2 NH exchangeable), 7.8 (s, 1H, pyrimidine-CH), 7.60 - 7.10 (m, 3H, thiophene-CH) ppm; 13C NMR (DMSO-d6,100 MHz): δ = 112.3 - 137.5 (Pyrimidine-C + thiophene-C), 151.0, 166.0 (2C=O) ppm; GC-MS: (M+, %): m/z 194 (3.5) 178 (26.82), 146.95 (100), 121.95 (68.60), 80 (17.27); Analysis Calcd. for C8H6N2O2S (194.21) C, 49.48; H, 3.11; N, 14.42; S, 16.51; Found C, 50.0; H, 3.35; N, 14.60; S, 16.32.
2-(Thiophen-2-yl)Quinoline-3-Carbaldehyde (11)
Green crystals, recrystallized from ethyl acetate; yield (60%); mp 135-136 oC; IR (KBr) νmax: 3061, 3029 (Ar-CH), 2957, 2851 (Ald-CH), 1655 (C=O), 1616 (C=C) cm-1; 1H NMR (DMSO-d6, 400 MHz) ppm: δ 10.40 (s, 1H, aldehydes-CH), 9.0 (s, 1H, quinoline- CH), 8.30 (m, 4H, quinoline-CH), 7.46-7.2 (m, 3H, thiophene- CH); 13C NMR (DMSO-d6,100 MHz): δ = 120.7 - 154.0 (quinoline-C + thiophene-C), 192.0 (C=O) ppm; GC-MS: (M+, %): m/z 239 (13.7), 210 (1.55), 178 (100), 127 (5.29), 76 (2.56); Analysis Calcd. for C14H9NOS (239.29) C, 70.27; H, 3.79; N, 5.85; S, 13.40; Found C, 70.10; H, 3.95; N, 5.60; S, 13.30.
Biological Evaluation
Cytotoxicity Assessment
Methodology
Each of MCF-7 and HepG2, were grown in RPMI-1640 medium, A549 human lung cancer cells were grown in DMEM, supplemented with 10% heat inactivated FBS, 50 units/mL of streptomycin and maintained at 37oC in a humidified atmosphere containing 5% CO2. The cells were maintained as “monolayer culture” by serial sub culturing.
SRB Cytotoxicity Assay
Cytotoxicity was determined using SRB method as described by Skehan et al. Exponentially growing cells were collected using 0.25 % Trypsin-EDTA and seeded in 96-well plates at 1000-2000 cells/well in RPMI-1640 supplemented medium. After 24 h, cells were incubated for 72h with various concentrations of the tested compounds. Following 72h treatment, the cells were fixed with 10 % trichloroacetic acid for 1h at 4oC. Wells were stained for 10 min at room temperature with 0.4 % SRB dissolved in 1% acetic acid. The plates were air dried for 24h and the dye was solubilized with Tris-HCl for 5 min on a shaker at 1600 rpm. The optical density (OD) of each well was measured spectrophotometrically at 564 nm with an ELISA microplate reader (ChroMate-4300, FL, USA). The IC50 values were calculated according to the equation of Boltzman sigmoidal concentration - response curve using the nonlinear regression fitting models (Graph Pad, Prism Version 5).
Results and Discussion
Chemistry
Tributyl(thiophen-2-yl)stannane 1 was prepared according to a previous procedure and was separated by chromatography using neutral Alumina [26]. Coupling of compound 1 with 5,5`-dibromobiphenyl-2,2`-diol (2) in 10% mol of Pd(dba)2 under reflux in toluene in a nitrogen atmosphere afforded 5,5`-di(thiophen-2-yl)biphenyl-2,2`-diol (7) with 45% yield. The reaction mechanism includes coupling of two thiophene moieties at the 5 and 5` positions of the 5,5`-dibromobiphenyl-2,2`-diol reagent via the common stille coupling mechanism which suggests that when an aryl halide was coupled with a thiophenestannane derivative in the presence of [Pd] salt as a catalyst, the product is the arylthiophene beside tributylstanylbromide as a by-product as shown in [Equation 1] [27].

Equation 1: Reaction of alkyl halide with alkyltributylstannane in the presence of Pd [0] catalyst [28].
The toxic tin by-product was precipitated by stirring the product with KF and removed by filtration. The IR spectrum of compound 7 revealed a broad band at νmax = 3340 and 3248 cm-1 attributed to two OH groups. While it’s 1H NMR spectrum showed signals of the aromatic C-H and thiophene groups from δ 7.83 - 7.12 ppm while the OH protons were appeared as singlet at δ 9.0 ppm and the 13C NMR spectrum showed signals of the aromatic-C and thiophene-C groups at δ = 117.0 - 140.5 in addition to C-O group at 155.5 ppm. The GC-MS of compound 7 showed the [M+] with m/z = 350 by 26 % rel. Int. Reaction of the stannane 1 with the benzyloxy derivative of the compound 2 named, 2,2`-(benzyloxy)-5,5`-dibromobiphenyl (3) afforded 2,2`-bis-benzyloxy-5,5`-di(thiophen-2-yl)-biphenyl (8). The reaction product was found to have more yield product (65% yield) than the one with the hydroxyl derivative and so the antitumor activity is much better than the one with the hydroxyl derivative 7, which indicated that the reactivity of the coupling reaction was increased with increasing the aromaticity of the aryl bromide because of increasing the electron withdrawing effect of the benzyloxy moiety of the halide reagent. The IR spectrum of 8 revealed strong bands for aromatic C-H at νmax = 3062, 3032 cm-1. While its 1H NMR spectrum showed the signals of the aromatic and thiophene moiety in addition to two singlet signals at δ = 5.1 and 5.07 ppm attributed to two methylene groups of the benzyl moiety and the 13C NMR spectrum showed signals of two CH2 groups at δ = 71.1 ppm. The GC-MS of 8 showed the [M+] with m/z = 530 by 59 % rel. Int. (Figure 1).
Scheme-1
On the other hand, the reaction of stannane 1 with 2,4-dibromopyridine (4) under the same reaction conditions afforded 2,4-di(thiophen-2-yl)pyridine (9) with 40 % yield. The reaction proceeds with the same stille coupling reaction mechanism. The IR spectrum of 9 showed bands at νmax = 3061, 3032 and 1603 cm-1 attributed to aromatic C-H and C=N groups respectively. Its 1H NMR spectrum revealed signals at δ = 8.70- 8.65 ppm for the pyridine moiety and at δ = 7.90 - 7.17 ppm for the thiophene moiety and the 13C NMR spectrum showed signals of the pyridine and thiophene moieties. The GC-MS of 9 showed the [M+] with m/z = 243 by 34 % rel. Int (Figure 2).
Reaction of the stannane 1 with 5-bromouracil (5) under the same conditions afforded 5-(thiophen-2-yl)uracil (10) in 50 % yield. The reaction mechanism includes coupling of one thiophene moiety with 5-bromopyrimidindione moiety at the 5 positions. The IR spectrum of 10 revealed a band at νmax = 3162 cm-1 for the NH group in addition to two bands at νmax = 1698 and 1650 cm-1 for the two C=O groups of uracil moiety. Its 1H NMR spectrum revealed two singlet signals at δ = 11.3, 11.1 ppm attributed to two NH groups and the 13C NMR spectrum of 10 showed signals of pyrimidine and thiophene carbons in addition to two signals of two C=O groups at δ = 151.0, 166.0 ppm. The GC-MS of the same compound showed the [M+] with m/z = 194 by 3.5 % rel. Int.
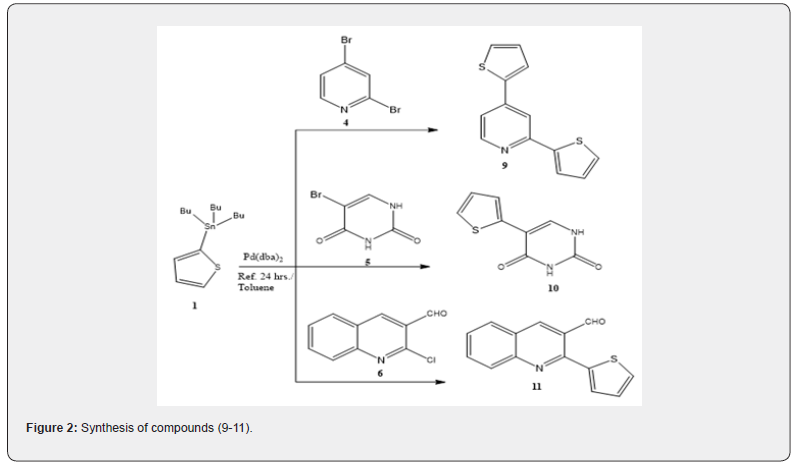
Coupling of the stannane 1 with 2-chloroquinoline-3- carbaldehyde (6) under the same reaction conditions afforded 2-(thiophen-2-yl)quinoline-3-carbaldehyde (11) in 60 % yield. The reaction mechanism includes coupling of one thiophene moiety at the position 2 of the 2-chloroquinoline-3-carbaldehyde moiety. The IR spectrum of compound 11 revealed bands at νmax = 2957, 2852, 1655 and 1616 cm-1 attributed to the aldehyde C-H, C=O and C=C respectively, the 1H NMR of the same compound revealed signals at δ = 10.39, 9.0, 8.30, 8.0, 7.82, 7.46, 7.28, 7.25 and 7.20 ppm due to C-H group of the aldehyde and 8 aromatic CH groups of the quinoline and thiophene moieties and the 13C NMR spectrum of 11 showed signals of quinoline and thiophene carbons in addition to a signal of C=O group of the aldehyde at δ = 192.0 ppm. The GC-MS of the same compound showed the [M+] with m/z = 239 by 13.7 % rel. Int (Figure 2).
Scheme 2
Molecular Docking Study of CDK2 Inhibitors and Binding Conformation
The Molecular Docking study for the new synthesized compounds into CDK2 was carried out by using Discovery Studio 2.5 software to investigate the detailed intermolecular interactions between the ligand and the target protein. The structure of the enzyme and lead compound 1 (H10V) having resolution of 1.95 was used in the determination of the important amino acids in the predicted binding pocket. [24] Re-docking lead 1 with the same binding site showed docking energy = - 37.45 kcal/mol with small RMSD (0.045) deviation in comparison to its crystal structure. The small RMSD values proved the validity of the used docking processes [29]. Interactive docking using C-DOCKER protocol for all compounds 7-11 was carried out relative to the selected active site and each docked compound was assigned according to binding mode. The score according to its binding mode onto the binding site was calculated [27]. The predicted binding energies and the corresponding experimental values were outlined in (Figure 3).
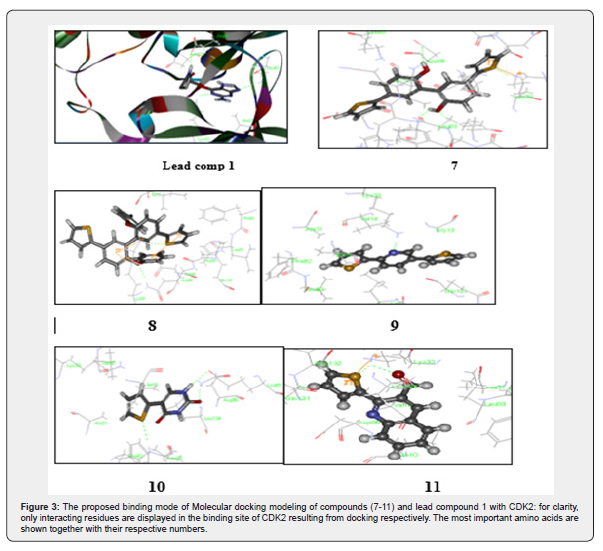
Figure 3
Alignment Study of Docked Compound 8 And 9 With the Lead Compound 1 Inside the Binding Pocket Of CDK2, As Shown In Figure 2 Revealed That
For compound 8, the Biphenyl moiety of 8 was perfectly aligned with purine nucleus of ligand 1, Benzyloxy group of 8 superimposed with 4-anino substituent of lead compound 1, the 2-thiophene part of 8 was aligned with o-pyrrolidin-2-one methyl substituent of ligand 1, also both compound 8 and ligand 1 make the same hydrogen bonding interaction with Asp86 and Lys33. Additionally, compound 8 makes two hydrophobic interactions with Lys33. Also, compound 9 revealed good alignment with lead compound 1 (Figure 4 A&B).
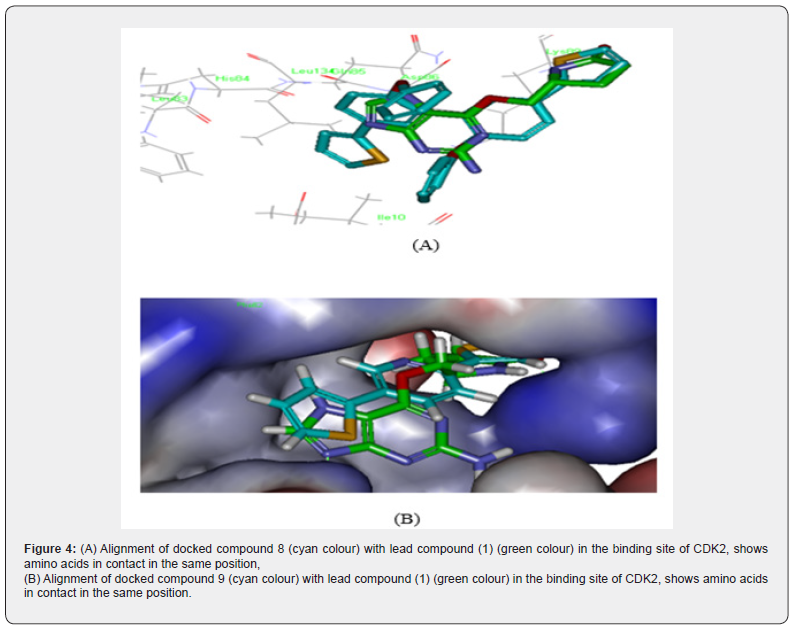
Figure 4
Cytotoxicity Evaluation
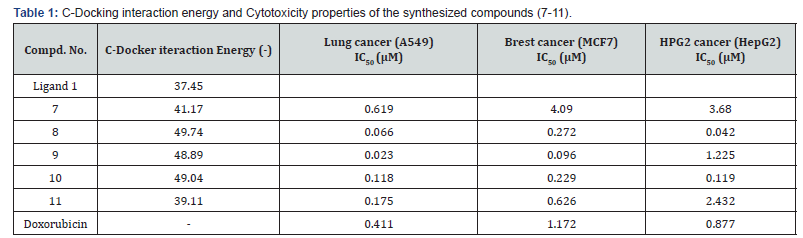
The synthesized compounds 7-11 were screened for their cytotoxicity against A549 (Lung Cancer), MCF7 (breast) and HEPG2 (liver) human tumor cell lines utilizing the Sulfo-Rhodamine-B (SRB) standard method [28-33]. From the observed results (Table 1), it has been concluded that compounds 8, 9, 10 and 11 exhibited highly potent anti-tumor activity against MCF7 (breast) and A549 (Lung Cancer) cell lines more than that of Doxorubicin that was used as a reference standard during this study (Table 1), while, compounds 8, 9 and 10 revealed highly potent cytotoxicity against HEPG2 (liver cancer) and A549 (Lung Cancer) cell lines more than that of Doxorubicin.
Table 1
The high docking results and biological activities can be explained by; first, the thiophene ring in all the new compounds increases the hydrophobic binding interaction with the deep hydrophobic pocket created by His84, Phe82, Glu81 and Ala31. Second, hydrogen bonding interactions were found to happen between the compounds (7-11) with the high docking scores and N-H group of Lys33, N-H of Lys89 and Asp86. H-bond is displayed as line. Compounds 8 and 9 are nicely bound to the CDK2. The oxygen atom of 8 projects toward the side chain NH group of Asp86, forming a more optimal H-bond interaction. Also, the sulfur atom of the thiophene ring of compound 8 forms hydrogen bond with Lys33. For compound 9 its N atom of pyridine ring make hydrogen bonding interaction with the N-H of Lys33. The docking results had provided us with useful information to understand the binding mode and the structural features of the target and chemical structure of the ligand. This was extended to the successful designing of highly active analogs of CDK2 inhibitor, (Table 1 and Figure 3,4).
Conclusion
We have successfully synthesized a series of new mono and bithiophene compounds (7-11) by using stille cross coupling technique of one or two moles of Tributyl(thiophen-2-yl) stannane with aryl halides and/or mono or dihaloheterocyclic compounds. The new synthesized compounds were evaluated for their antitumor activities in 3 different cell lines; MCF7, HEPG2 and A549 where compounds 8, 9 and 10 exhibited highly potent cytotoxicity activity comparable to that of Doxorubicin. A Molecular Docking study for the new synthesized compounds into CDK2 was carried out by using Discovery Studio 2.5 software.
References
- (2006) WHO, Cancer World Health Organization.
- Sarg MTM, El-Shaer SS (2014) Efficient Utilization of 6-Aminouracil to Synthesize Fused and Related Heterocyclic Compounds and Their Evaluation as Prostate Cytotoxic Agents with Cathepsin B Inhibition Open Journal of Medicinal Chemistry 4: 39.
- Nassar IF, El-Assaly SA (2011) Synthesis, reactivity, and antitumor activity of some new pyrazolo[3,4-d] pyrimidine and their triazole derivatives. Der Pharma Chemica 3: 229.
- Nassar IF, Atta-Allah SR, Elgazwy ASSH (2015) A convenient synthesis and molecular modeling study of Novel Pyrazolo[3,4-d]pyrimidine and Pyrazole derivatives as antitumor agents J Enzyme Inhib Med Chem 30: 396-405.
- Elgazwy ASSH, Ismail NSM, Elzahabi HAS (2010) Molecular modeling study bioactive natural product of khellin analogues as a novel potential pharmacophore of EGFR inhibitors. J Enzyme Inhib Med Chem 28(6): 1171-1181.
- Megally Abdo NY (2015) Synthesis, and antitumor evaluation of novel dihydropyrimidine, thiazolo [3,2-a] pyrimidine and pyrano [2, 3-d] pyrimidine derivatives. Acta Chim Slov 62(1): 168-180.
- Fares M, Eldehna WM, Abou-Seri SM, Abdel-Aziz HA, Aly MH, et al. (2015) Design, synthesis and in vitro antiproliferative activity of novel isatin-quinazoline hybrids. Archiv der Pharmazie 348: 144-154.
- Ashry ES, Rashed N, Abdel-Rahman A, Awad LF, Rasheed HA (2006) Synthesis of 2-Bromomethyl-3-Hydroxy-2-Hydroxymethyl-Propyl Pyrimidine and Theophylline Nucleosides Under Microwave Irradiation. Evaluation of Their Activity Against Hepatitis B Virus. Nucleosides Nucleotides Nucleic Acids 25(8): 925-939.
- Girgis AS, Ismail NS, Farag H (2011) Synthesis, hypnotic properties, and molecular modeling studies of 1, 2, 7, 9-tetraaza-spiro [4.5] dec-2-ene-6, 8, 10-triones Eur J Med Chem 46(10): 4964-4969.
- Beletskaya IP, Cheprakov AV (2000) The Heck Reaction as a Sharpening Stone of Palladium Catalysis. Chem Rev 100(8): 3009-3066.
- Liu Y, Wang SS, Liu W, Wan QX, Wu HH (2009) Transition-Metal Catalyzed Carbon-Carbon Couplings Mediated with Functionalized Ionic Liquids, Supported-Ionic Liquid Phase, or Ionic Liquid Media. Curr Org Chem 13: 1322-1346.
- Moreno-Manas M, Pleixats R (2003) Formation of carbon− carbon bonds under catalysis by transition-metal nanoparticles. Acc Chem Res 36(8): 638-643.
- Phan NTS, Van Der Sluys M, Jones CW (2006) On the Nature of the Active Species in Palladium Catalyzed Mizoroki-Heck and Suzuki-Miyaura Couplings-Homogeneous or Heterogeneous Catalysis, A Critical Review. Adv Synth Catal 348(6): 609-679.
- Yin LX, Liebscher J (2007) Carbon- carbon coupling reactions catalyzed by heterogeneous palladium catalysts. Chem Rev 107(1): 133-173.
- (A)Bringmann G, Menche D (2001) Stereoselective total synthesis of axially chiral natural products via biaryl lactones. Acc Chem Res 34(8): 615-624.
- (b) Bringmann G, Reichert Y, Kane VV (2004) The total synthesis of streptonigrin and related antitumor antibiotic natural products. Tetrahedron 60(16): 3539-3574.
- (c) Roberts TC, Smith P, Cirz RT, Romesberg FE (2007) Structural and initial biological analysis of synthetic albomycin A2 J Am Chem Soc 129(51): 15830-15838.
- (d) Wallace TW (2006) Biaryl synthesis with control of axial chirality. Org Biomol Chem 4: 3197.
- Miyaura N, Suzuki A (1995) Palladium-catalyzed cross-coupling reactions of organoboron compounds Chem. Rev pp: 2457.
- b) Martin R, Buchwald SL (2008) Palladium-catalyzed Suzuki-Miyaura cross-coupling reactions employing di alkyl aryl phosphine ligands. Acc Chem Res 41(11): 1461-1473.
- Oestreich M (2009) The Mizorokie Heck Reaction. Wiley.
- (b) Firouzabadi H, Iranpoor N, Ghaderi A (2011) Palladium nanoparticles supported on aminopropyl-functionalized clay as efficient catalysts for phosphine-free C-C bond formation via Mizoroki-Heck and Suzuki-Miyaura Reaction. J Mol Catal A Chem 42(1): 38.
- Hiyama T, Hatanaka Y (194) Palladium-catalyzed cross-coupling reaction of organ metalloids through activation with fluoride ion. Pure Appl Chem 25(41): 1471.
- (b) Denmark SE, Sweis RF (2002) Design and implementation of new, silicon-based, cross-coupling reactions: importance of silicon- oxygen bonds. Acc Chem Res 35: 835.
- Sattar A, Elgazwy HS (2006) Synthetic analogs of dioncophylline and michellamines by stille coupling. J Heterocyclic Chem 43(5): 1151.
- Sattar A, Elgazwy HS (2004) Improved studies of cross‐coupling reactions of 5‐(tri‐n‐butystannyl)‐ and 5,5′‐bis(tri‐n‐butylstannyl)‐2,2′‐bithiophene with aryl halides. J Heterocyclic Chem 41: 755.
- Sattar A, Elgazwy HS (2006) Stille coupling of thiophene with a tetrahydroisoquinoline alkaloid. Mendeleev commun 16: 92.
- Sattar A, Elgazwy HS (2001) Separation of triorganostannyl-and bis (triorganostannyl) bithiophene. Phosphorous Sulfur and Silicon 175(1): 237.
- Espinet P, Echavarren AM (2004) The mechanisms of the Stille reaction. Angew Chem Int Ed 43(36): 4704-4734.
- Gibson AE, Arris CE, Bentley J, Boyle FT, Curtin NJ, et al. (2002) Probing the ATP Ribose-Binding Domain of Cyclin-Dependent Kinases 1 and 2 with O(6)-Substituted Guanine Derivatives. J Med Chem 45(16): 3381-3393.
- Sattar A, Elgazwy HS, Nasser SMI (2010) Bioorg Med Chem 18: 763.
- Sattar A, Elgazwy HS, Mastoura ME, Nasser SMI (2013) Molecular modeling study bioactive natural product of khellin analogues as a novel potential pharmacophore of EGFR inhibitors. J Enzyme Inhib Med Chem 28(6): 1171-1181.
- Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, et al. (1990) New Colorimetric Cytotoxicity Assay for Anticancer-Drug Screening J Natl Cancer Inst 82(13): 1107-1112.






























