Michael Addition Reaction Acrylamide and Malonic Ether in PTC System
GH Torosyan* and NR Hovhannisyan
Doctor of Chemical sciences, Yerevan Gagik Torosyan, National polytechnic university of Armenia, Armenia
Submission: August 2, 2023;Published: August 24, 2023
*Corresponding author: GH Torosyan, National polytechnic university of Armenia, Armenia, Doctor of Chemical sciences Yerevan Gagik Torosyan, Armenia
How to cite this article: GH Torosyan, NR Hovhannisyan. Michael Addition Reaction Acrylamide and Malonic Ether in PTC System. Organic & Medicinal Chem IJ. 2023; 12(4): 555842. DOI: 10.19080/OMCIJ.2023.12.555842
Abstract
Here was discussed also the Michael addition reaction for acrylamide as α, β-unsaturated compound with Malonic acid ethyl ester as classical CH-acid. It has been shown the catalytic activity of quaternary ammonium salts as phase transfer catalyst in this interaction. The PTC made the possibility to avoid the use of toxic and flammable organic solvents. The rate of the additional reaction was also calculated.
Keywords: Acrylamide; Malonic Acid Ethyl Ester; Quaternary Ammonium Salt; Michael Addition Reaction; Phase Transfer Catalysis
Abbreviations: AC: Acrylamide; MEA Malonic Acid Ethyl Ester; MAR: Michael Addition Reaction; PTC: Phase Transfer Catalysis; QUAT: Quaternary Ammonium Salt; TMBACH: Trimethylbenzylammoium Chloride
Introduction
Acrylamide belongs to large-tonnage products of the main organic synthesis, and its production and consumption in industrialized countries is steadily increasing [1]. Acrylamide (CH2CHCONH2), an industrially produced α,β-unsaturated reactive molecule, is used worldwide to synthesize polyacrylamide. Polyacrylamide has found numerous applications as a soil conditioner, in wastewater treatment, in the cosmetic, paper, and textile industries, and in the laboratory as a solid support for the separation of proteins by electrophoresis. Acrylamide has two reactive sites, the conjugated double bond, and the amide group. The electrophilic double bond can participate in nucleophilic reactions with active hydrogen bearing functional groups both in vitro and in vivo. A better understanding of the chemistry and biology of pure acrylamide in general and its impact in a food matrix in particular can lead to the development of improved food processes to decrease the acrylamide content of the diet.
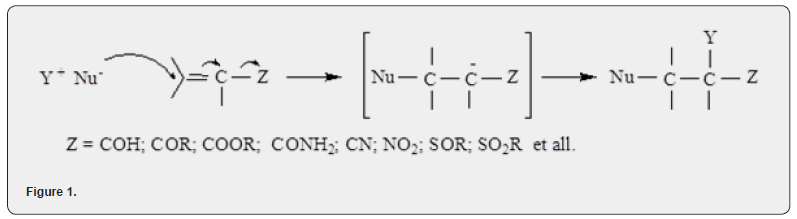
The interest in research in the field of acrylamide chemistry is also growing, because, on the one hand, the easy availability of acrylamide makes it an attractive starting material for the synthesis of new compounds, on the other hand, the ever-increasing prevalence of acrylamide requires expanding research aimed at improving environmental safety. its use. Michael addition, also known as the Michael reaction, is one of the most well-known reactions in organic synthesis [2-4]. The conjugated, carbonyl, double bond in the acrylamide molecule creates very favorable conditions (strong bond polarization, no steric hindrance) for a nucleophilic attack to the β-position, with respect to the carbonyl (Nu- is the nucleophilic part of the attacking molecule) (Figure 1).
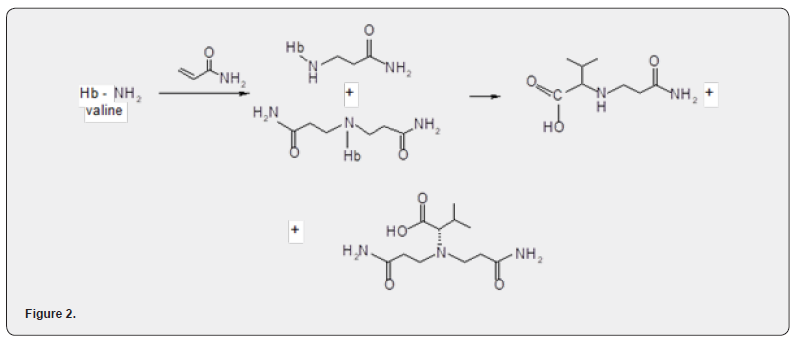
Previously, it was found that acrylamide is able to enter into nucleophilic addition reactions with biogenic molecules containing active hydrogen atoms both in vitro and in vivo. So, with the sulfhydryl group of cysteine contained in the protein, acrylamide forms, as a final result, S - (2 - carbamoyl ethyl) cysteine. The addition of acrylamide to the terminal amino groups of valines contained in hemoglobin can lead to the formation of both mono- and di-substituted adducts. (Figure 2)
The product of enzymatic hydrolysis of the monosubstituted adduct of acrylamide with the valine fragment of hemoglobi - N-(2-carbamoylethyl)valine, was isolated from in vivo interaction products and identified. Acrylamide has all the necessary properties for entering into nucleophilic addition reactions, incl. and the Michael reaction [3-5] - activated, conjugation with the amide group, double bond, no steric hindrance. This group of reactions, in comparison with the reactions of the amide group, has not been studied enough, which is apparently explained by the fact that acrylamide was considered mainly as a monomer and the reactions leading to its modification precisely as a monomer, i.e., without affecting the double bond. Studies of the reactions of acrylamide at the double bond were mainly carried out with the aim of modeling its behavior in biological objects also.
Results and Discussion
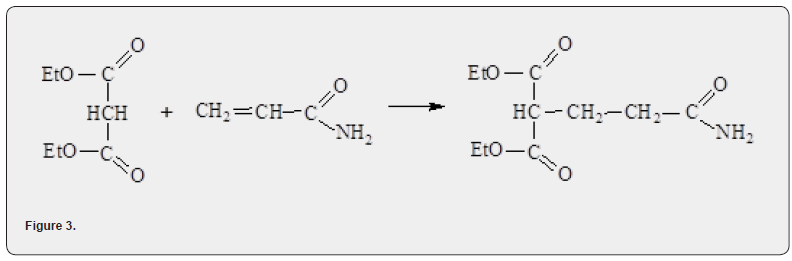
It had been studied the interactions of acrylamide with classical CH-acid, i.e., compounds containing a mobile “acidic” proton as MEA. Here were expected to react with acrylamide in a 1,4-type addition -Michael addition. The addition was studied under conditions of PTC with QUAT, which made it possible to avoid the use of toxic and flammable organic solvents. The interaction of malonic acid diethyl ester with acrylamide leads to 1,1-dicarbethoxy-3-carbamoylpropane (Figure 3).
Comparison of the results of experiments with various reagents and catalysts was carried out by comparing the reaction rate constants, assuming that at sufficiently high catalyst concentrations (0,1 mol/l), when a change in the catalyst concentration ceases to noticeably affect the reaction rate, the course of the process can be approximately described the kinetic equation of the second-order reaction, i.e., equation of the form:

where [Z] is the current concentration of the reaction product; [X ] and [Y ] - concentrations of initial reagents; k is the reaction rate constant.
The equation 1 can have the following form (2), when the initial concentrations of reagents are equal,[X0] = [Y0] :

The solution to this equation for k will be:

[X0] and [Xτ] are the concentrations of the reagent X, respectively, at the initial and after the time τ has elapsed. The time τ is taken for the initial period of the reaction when the reaction proceeds at a constant rate. In all cases not specifically specified, the reaction rate constant was calculated for the time τ = 10 min from the start of the reaction; the period of constant reaction rate was at least 20 min. To determine the yield of the target product, the reaction was continued for a time corresponding to the calculated time for 95% conversion (τ ) . In the case when the calculated duration of the reaction significantly exceeded the duration of the working day, a sample was taken to determine the yield of the product on the next day; in all cases when the calculated reaction time exceeded 3 hours, ~ 0,1 g of hydroquinone was introduced into the reaction mixture to suppress the polymerization of acrylamide. The results of the study of the effect of the catalyst on the reaction rate and the yield of the final product are given in Table 1.
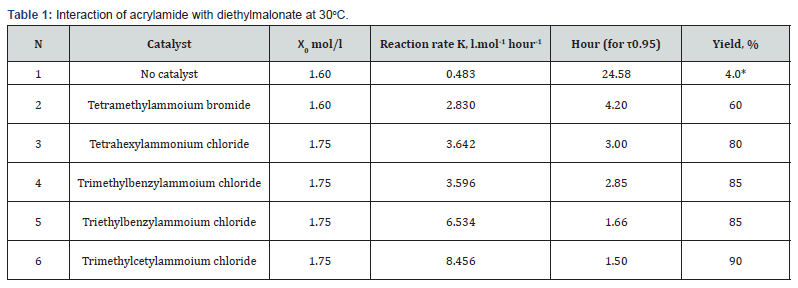
Note: * - next day; response time approximate
Experimental Part
1,4 ml of diethyl malonate (0,075 mol) with 12,5 ml of a saturated solution of potash (0,075 mol of K2CO3) stirred for 5 min.; then 0,93 g (0,005 mol) of TMBACH was added and solution of 5,33 g of acrylamide in 35 ml of water added dropwise over 5 minutes. The reaction mixture stirred at 30oC for 3 hours. After the reaction mixture was neutralized with concentrated hydrochloric acid, it was extracted three times with benzene (30 ml each), the benzene extracts were combined, and benzene was distilled off under vacuum (20 mm Hg).
14,7 g (0,064 mol) of product were obtained. Yield 85% of theory.
Calculated (% mass): C - 51,94, Н - 7,41, N - 6,06, О - 34,59 (С10Н17NО5)
Found (% масс): С - 52,3, Н - 7,02 N - 5,95;Rf = 0,72 IR spectra , ν, sm-1 : 3420, 3300 { vas ,vs (NH2)}, 2965, 2870 { v (CH3) }, 2895{v (CH)}, 1735 {v (CO) C-C(CO)-O}, 1680{v (CO) C-C(O)-NH2 }, 1640{δ (NH)}, 1465, 1380 {δ (CH3)}, 1140, 1030 {δ NMR, 1Н, (СDСl3), δ, м.д.: 1,25 m (6H, -OCH2CH3 J 5,2 Гц), 2,15 -2,35 m (4H, -CH-CH2CH2C(O)NH2), 3,15 m (1H, C(O)-CH-C(O) J 6,4 Гц), 4,1 m (4H, CH3CH2CH-O-), 7,04 s (2H, NH2 ).
References
- Gokmen V (2015) Acrylamide in Food, Analysis, Content and Potential Health Effects,1st Edition eBook.
- Michael A (2017) Ueber die Addition von Natriumacetessig- und Natriummalonsäureäthern zu den Aethern ungesättigter Säuren. J Prakt Chem 35: 349-356.
- Gangliang H, Xue Li (2017) Applications of Michael Addition Reaction in Organic Synthesis. Current organic synthesis 14 (4): 568 -571.
- Rekala Sh MJ, Mydhili PS, Ponnaboina T (2022), Recent Advances in Base-assisted Michael Addition Reactions, Currrent Organic chemistry 26(13): 1264 -1293.
- Friedman M (2001) Application of the S-pyridylethylation reaction to the elucidation of the structures and functions of proteins. J Protein Chem 20(6): 431-455.






























