Synthesis, Characterization, Aggregation Behavior, Antifungal and Antibacterial Studies of a Novel Tetra-Metronidazole Substituted Zinc (II) Phthalocyanine
Asma Ibrahmi1,2,*, Safa Belaiba1,2, Manel Ben Mansour1, Mohamed Adnen Hadj Ayed1,3 and Jameleddine Khiari1,4
1Laboratory of Didactic Research, Experiment al Sciences and Supramolecular Chemistry, Higher Institute of Education and Continuing Training, Virtual University, Tunis, Tunisia
2Department of Chemistry, Faculty of Sciences of Tunis, University of Tunis El Manar, Tunis, Tunisia
3Department of Materials Engineering, Higher Institute of Applied Sciences and Technology of Mahdia, University of Monastir, Mahdia, Tunisia
4Department of Chemistry, Faculty of Sciences of Bizerte, University of Carthage, Tunisia
Submission: May 22, 2023;Published: August 15, 2023
*Corresponding author: Asma Ibrahmi, Laboratory of Didactic Research, Experiment al Sciences and Supramolecular Chemistry, Higher Institute of Education and Continuing Training, Department of Chemistry, Faculty of Sciences of Tunis, Virtual University, Tunis, Tunisia
How to cite this article: Asma Ibrahmi*, Safa Belaiba, Manel Ben Mansour, Mohamed Adnen Hadj Ayed and Jameleddine Khiari. Synthesis, Characterization, Aggregation Behavior, Antifungal and Antibacterial Studies of a Novel Tetra-Metronidazole Substituted Zinc (II) Phthalocyanine. Organic & Medicinal Chem IJ. 2023; 12(4): 555841. DOI: 10.19080/OMCIJ.2023.12.555841
Abstract
New metronidazole substituted phthalonitrile derivative was prepared by nucleophilic displacement reaction between metronidazole and 4-nitrophthalonitrile. Cyclotetramerization of this phthalonitrile derivative in the presence of zinc acetate gave the new tetrakis (3-(2-(2-methyl-5-nitroimidazol-1(1H)-yl) ethoxy)) phthalocyaninatozinc (II). The new component was purified and characterized via various spectroscopic methods including infrared IR, nuclear magnetic resonance 1H NMR, and UV-visible spectroscopy. The aggregation behavior of the new zinc (II) phthalocyanine was investigated in different solvents such as DMSO and DMF. Antimicrobial activity of this new macrocycle was tested against gram-negative bacteria, gram-positive bacteria, and different varieties of Candida.
Keywords: Peripheral Zinc Phthalocyanine; Metronidazole; Synthesis; Aggregation Behavior; Antimicrobial Activity
(Figure 1)
Abbreviations: DMF: N, N-Dimethylformamide; DMSO: Dimethylsulfoxide; DCM: Dichloromethane; PC: Phthalocyanines; THF: Tetrahydrofuran; MIC: Minimum Inhibitory Concentration
Introduction
One of the important tetrapyrrole derivatives is Phthalocyanines (PC), which are officially known as tetra benzo [5,10,15,20] tetraazaporphyrins (PC). They are the most stable porphyrin analogs, but unlike porphyrins, they do not occur in nature because they are completely synthetic compounds [1]. Phthalocyanines (Pcs) have many considerable physical and chemical features. Thanks to their high electron transfer abilities, phthalocyanines have been utilized in many fields such as dyes and pigments [2],optical data storage [3], photosensitizers [4,5], gas sensors [6,7], liquid crystals [8,9], semiconductors [10], Langmuir-Blodgett films [11], non-linear optical applications [12], materials science, photodynamic therapy [13] and therapeutic medicine [14,15]. Metronidazole is one of the basic drugs for treatment of anaerobic bacterial infections, protozoal infections, and microaerophilic bacterial infections. It is cytotoxic to facultative anaerobic microorganisms. Metronidazole is FDA–approved for the treatment of protozoal infections such as Trichomoniasis vaginalis, Entamoeba histolytica, Giardia lamblia, blastocysts and Balantidium coli.
It is also FDA–approved to treat anaerobic bacterial infections caused by Bacteroides species, Fusobacterium species, Clostridium species, Gardenerella vaginalis, Helicobacter pylori, Prevotella species, Porphyromonas species and Biophilia Wadsworth [16]. In this study, we describe the synthesis and characterization of a new phthalocyanine bearing, 1-(2-(λ1-oxidaneyl) ethyl)-2- methyl-5-nitroimidazole substituent on peripheral positions. The aggregation behaviors of the novel phthalocyanine were investigated in two solvents: dimethylsulfoxide (DMSO) and N, N-dimethylformamide (DMF). We have also reported the results of a comprehensive investigation of the concentration effects on the aggregation properties of phthalocyanine derivatives in DMF. The antifungal and antibacterial activities of the phthalocyanine are investigated by using ELISA plate and disc diffusion method.
Experimental
Materials and Equipment
N, N-dimethylformamide (DMF), dimethylsulfoxide (DMSO), dichloromethane (DCM), 3 n-pentanol, n-hexane, ethyl acetate, ethanol, potassium carbonate, zinc acetate, 1,8-4 diazabicyclo [5.4.0] undec-7-ene (DBU) and metronidazole were used as received from Sigma Aldrich. 4- Nitrophthalonitrile was synthesized starting from the phthalimide according to the literature procedure [17]. The solvents were purified according to standard procedures [18] and stored over molecular sieves (4Å). All reactions were carried out under a dry nitrogen atmosphere. Melting points were measured on an electrothermal apparatus. Electronic spectra were recorded on a Cary 2300 spectrophotometer. Routine IR spectra were recorded on a Perkin- Elmer BX FT-IR system spectrometer by dispersing samples in KBr pellets. 1H NMR and 13C NMR spectral data were obtained by Varian 300 FT-NMR.
Synthesis
4-(2-(2-Methyl-5-Nitroimidazol-1(1h)- Yl) Ethoxy) Phthalonitrile (3)
4-nitrophthalonitrile (200 mg, 1.155 mmol) was mixed with metronidazole (197 mg, 1.155 mmol) in DMF (4 ml). K2CO3 (160 mg, 1.155 mmol) was added in four portions. The mixture was stirring for 72 h at 100 °C, then for 48 h at room temperature. The resulting mixture was transferred to 20 ml of ice-cold water. The formed precipitate was filtered and washed successively with water and diethyl ether. The solid was recrystallized in ethanol to give a brown powder. The product is soluble in acetone, acetonitrile, tetrahydrofuran (THF), dichloromethane, chloroform, DMF and DMSO.
Yield 82 %. Mp 179°C. 1H NMR spectrum (300 MHz, DMSO-d6), δ, ppm (J, Hz): 2.53 (3H, s, CH3); 4.35 (2H, t, J = 4.8, N–CH2); 4.74 (2H, t, J = 4.8, O–CH2); 7.42 (1H, dd, 3J5,6 = 8.7, 4J3,5 = 2.4, H-5 Ar); 7.75 (1H, d, 4J3,5 = 2.4, H-3 Ar); 8.03 (2H, m, H Ar).13C NMR spectrum (75 MHz, DMSO-d6), δ, ppm: 14.2; 44.5; 67.6; 106.4; 115.7; 116.4; 118.5; 119.6; 120.0; 132.9; 136.2; 138.7; 151.6; 162.7. IR spectrum, ν, cm−1: 2938 (C-H aromatic), 2233 (C≡N), 1747 (C=N), 1531 (N=O, asymmetric band), 1368 (N=O, symmetric band), 1435 (C–N).
1(4), 8(11), 15(18), 22(25)-Tetrakis(3-(2-(2-Methyl-5- Nitroimidazol-1(1H)-Yl)Ethoxy)) 8 Phthalocyaninatozinc (II) (4)
A mixture of phthalonitrile (3) (0.05 g, 1.17 mmol), anhydrous Zn (CH3COO)2 (0.015 g, 1.17 mmol) and 1,8-diazabicyclo [5.4.0] undec-7-ene (DBU) (0.1 mL, 0.67 mmol) in dry pentanol (1.5 ml) was stirred and refluxed at 130 °C under an argon atmosphere for 18 hours. Then, 5 ml of hexane was added to the resulting reaction mixture to produce precipitation and the green precipitate was separated by filtering. The crude product was washed several times successively with hexane and acetone to remove impurities. The desired product was purified by basic silica gel column chromatography using a gradient of chloroform/methanol (9/1) as eluents.
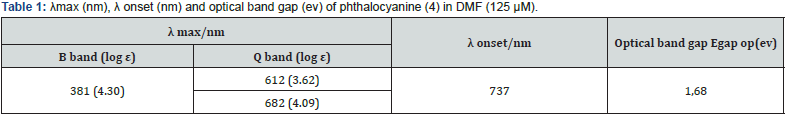
Yield 52 %. Mp > 260°C. 1H NMR spectrum (300 MHz, DMSO-d6), δ, ppm (J, Hz): 2.50-18 2.56 (m, 12H, CH3); 4.28-4.87 (m, 16H, CH2); 7.45-8.12 (m, 16H Ar). 13C NMR spectrum (75 MHz, DMSO-d6), δ, ppm: 13.9, 44.9, 68.2, 102.4, 111.7, 113.2, 115.3, 115.3, 118.6, 121.9, 125.8, 128.0, 128.2, 129.3, 131.1, 131.7, 132.6, 136.8, 138.4, 142.0, 160.3, 168.5, 169.7, 171.4. IR spectrum, ν, cm−1: 3259 (C–H aromatic), 2935 (C–H aliphatic), 1605 (C=N), 1359 (N=O, asymmetric band), 1191 (N=O, symmetric band), 1368 (C–N), 514 (Zn–N). UV spectrum (DMF), λmax (logε): 381 (4.30), 612 (3.62), 682 (4.09).
Antibacterial Activity
T Macrotibacterial activities of Pc complex (4) against three Gram- negative and two Gram-positive bacteria were studied by two different methods; disk diffusion method and minimum inhibitory concentration (macro dilution assay).
Disc Diffusion Assay
Gram-negative bacteria (Escherichia coli (ATCC 10536), Salmonella Entrica (ATCC 35664), Pseudomonas aeruginosa (ATCC 9027)) and gram-positive (Staphylococcus aureus (ATCC 6538), Bacillus subtilis (6051)) were used as the test microorganisms. The disc diffusion method was used for determination of antibacterial activity of phthalocyanine complexe (4) [19].
The synthesized phthalocyanine was solubilized in an anhydrous DMSO. Sterile blank antibacterial test discs were soaked with 15 μL solution of phthalocyanine (4) in DMSO at a concentration of 200 μg/mL, then placed on nutrient agar (Brain Heart Infusion Broth) plates (Petri dishes) and interact with a suspension of the indicator’s microorganisms (0,1 mL of 106 cells per mL). Petri dishes were incubated at 37°C for 24 h. The antibacterial activity was assayed by measuring in millimeters the diameter of the inhibition zone formed around the disk. 50μL of DMSO was used as a negative control to be certain that no bactericid al or bacterio static effects will occur due to the solvent, while spiramycin (200 μg/ mL) was used as positive controls.
Minimum Inhibitory Concentration (Micro Dilution) Assay MIC
According to European Committee for Antimicrobial Susceptibility Testing (EUCAST) criteria [20] the minimum inhibitory concentration (MIC) values of the compound (4) were determined by broth micro-dilution method. Antimicrobial activity testing of the compound (4) was performed using 96-well sterile microplates (with a final volume in each microplate well of 200 μl). The synthesized phthalocyanine (3.6 mg/mL) was solubilized in an anhydrous dimethylsulfoxide (20%). The dilutions of the compounds as previously described were performed between 200 μg /mL and 10 μg /mL. To each test well, 20 μL of the standard bacteria were added to final inoculums of 106 CFU/mL for each microorganism. Negative and positive control wells were added to the microplate. A growth control well contains liquid MH medium and a bacterial suspension while Cells suspension at the same concentration supplemented with gentamicin (40 μg/ ml) was used as negative controls. The plates were then covered with sterile plate covers and incubated at 37°C for 24 h. Next,10 μL of resazurin (MTT) is an indicator of microorganism growth (0.5 mg/mL) dissolved in sterile water was added to the wells and incubated at 37°C for 30 min. The MIC is the lowest concentration of synthesized phthalocyanine at which the microorganism does not show visible growth after incubation.
Antifungal Activities
The antifungal activities of the synthesized phthalocyanine (4) were determined by the minimum inhibitory concentration MIC in accordance with the National Committee for Clinical Laboratory Standards guideline M7-A6 and M38-P [21,22].The aim of this study was to study the photo-dynamic antifungal activities of phthalocyanine derivatives on different types of Candida (C. albicans (ATCC 10231), Candida glabrata (ATCC 90030) and Candida krusei (ATCC 44507)).Candida is the most virulent and common species of fungi and the main cause of invasive fungal infections on humans [23,24]. In this study, microdilution broth method was used to determine the MIC values of the compounds in vitro against different types of Candida.
Single colonies of Candida are picked with a sterile toothpick one day before the screening assay, and used to inoculate YPD medium and then incubated for 24 hours at 30°C. After their growth, colonies are scraped with a loop, then diluted in YPD liquid and their optical densities were fixed. The antifungal activity testing of the compound (4) was performed using 96-well sterile microplates with a final volume in each microplate well of 100 μL. The synthesized compound (100μg/mL) was properly dissolved in dimethyl sulfoxide solution (20%). The dilutions of the compounds as previously described were performed between 100 μg/ml and 10 μg/mL.10 μL of standard bacterial or yeast suspensions (106 CFU/mL for each microorganism) were added to each test well. Positive growth control wells consisted of microorganisms only in their adequate medium. Cells suspension at the same concentration supplemented with fluconazole (50μg/ mL) was used as negative control. Negative and positive control wells were added to the microplate and the microplates were incubated in an incubator set at 37 °C and growth monitored by optical density measurement using a microplate reader at 600 nm. The minimum inhibitory concentration (MIC 50%) which corresponds to the lowest concentration of the tested sample was able to inhibit the growth of Candida by 50%.
Results and Discussion
Synthesis
Our work starts with the synthesis of 4-(2-(2-methyl- 5-nitroimidazol-1(1H)-yl) ethoxy) phthalonitrile (3) by a nucleophilic substitution reaction [25] of 4-nitrophthalonitrile (1) with metronidazole (2) in dry DMF in the presence of dry K2CO3. The desired phthalonitrile derivative (3) was obtained in a yield of 82% after recrystallization in ethanol. The cyclotetramerization of the phthalonitrile derivatives (3) in a high-boiling solvent (n-pentanol) in the presence of a few drops 1,8- diazabicyclo [5.4.0]undec-7-ene DBU as a strong base and anhydrous Zn(CH3COO)2 at 160°C for 18 h was conducted to form 1(4), 8(11), 15(18), 22(25)-tetrakis(3-(2-(2-methyl-4-nitroimidazol-1(1H)- yl)ethoxy)) phthalocyaninatozinc (II) (4) as a mixture of four isomers, in overall yield of 52% after purification. Any separation technique was carried out for resolution of these isomers [26]. The characterization of these compounds was carried out by the combination of several methods, including elemental analysis, IR, 1H NMR, and UV/Vis spectra. The compounds structures were confirmed by the results of these analyses. The synthetic route followed for the synthesis of new dinitrile compound (3) and novel zinc phthalocyanine complex (4) is given in Figure 1.
Gound State Electronic Absorption and Aggregation Properties
The UV/vis spectra are the best indication technique for the phthalocyanine compound which can be used to study and determine the structural properties and aggregation behaviour of phtalocyanines [27]. The ground state electronic absorption spectra of the peripherally tetra-substituted phthalocyanine complex (4) showed monomeric behavior evidenced by a single narrow Q-band, typical of metalated phthalocyanine complexes.
The new phthalocyanine showed typical electronic spectra with two strong absorption regions, one of them showed characteristic Q band absorptions at 600-700 nm which were attributed to the π - π* transition from the highest occupied molecular orbital (HOMO) to the lowest unoccupied molecular orbital (LUMO) of the Pc ring and the other visible region at about 300-400 nm B band called also Soret peak, which represents transition from the deeper π - levels to the LUMO transition [28- 31].The optical band gap was estimated from the absorption onset of the phthalocyanine (4) in DMF, according to the formula E = hc/λ (Table 1). The newly synthesized zinc phthalocyanine (4) was soluble in most of organic solvents, such as dimethylformamide (DMF) and dimethylsulfoxide (DMSO).The spectral profiles of the phthalocyanine compound were recorded in these two organic solvents (DMF and DMSO) because they ensure the best solubility of the product to observe the Pc behaviour variety of solvent parameters such as dielectric constant ε, refractive index n and solvatochromic parameters α, β and π*.
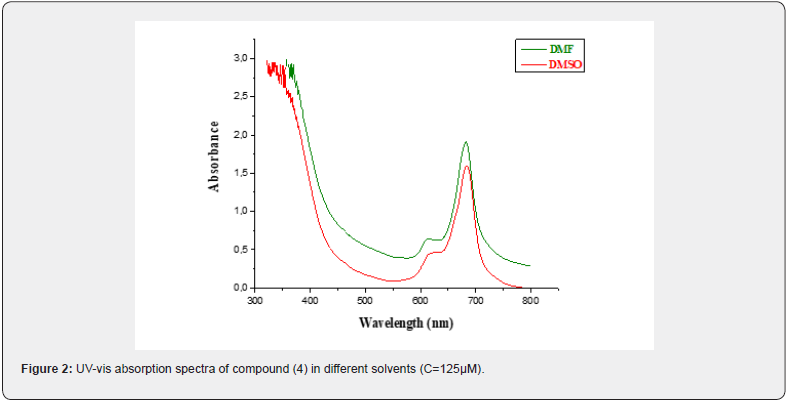
The compound (4) did not aggregate in these solvents at the adopted concentration (125 μM). The spectra of this compound in DMF and DMSO were presented in Figure 2. It is evident that there is no significant change in absorption profile of the synthesized zinc phthalocyanine with variation in polarity of the medium. The synthesized phthalocyanine (4) possesses bathochromic spectral shift (positive solvatochromism) when moving from the least polar (DMF) to the most polar solvent (DMSO). This is due to the fact that a molecule in the ground state and excited state possesses different polarities. Absorption and extinction coefficients are presented in Table 2. The absorption spectra of compound (4) in DMF and DMSO showed two narrow peaks of approximately equal intensity at 677 nm and 679 nm. Sharp and single Q-bands in these solvents are typical of non-aggregated monomeric species.

The UV-visible spectra data presented in Table 2 show that the investigated macrocycle (4) is monomeric in DMF which is a solvent having electron donor atoms binding through the doublebonded oxygen atom to the Zn metal of the ZnPc monomer. Hence, it interacts by coordination with central metal cation of phthalocyanine macrocycle which explains the highest DMF solution compared to other solvents. Besides, DMF has specific solvation ability toward peripheral substituents of phthalocyanine due to its donor- acceptor properties [32,33]. Aggregation is usually depicted as a coplanar association of rings progressing from monomer to dimer and higher order complexes. In general, Pc aggregation is thought to reflect coplanar interactions involving macrocycle ring. These interactions occur because of favorable Van Der Waals forces, π-stacking interactions, and solvent effects [34]. Phthalocyanine compounds have a high aggregation tendency due to the interaction between their 18 π electron systems and the aggregation decreases the solubility of these compounds in solvents. This behavior is dependent on the concentration, nature of the solvent, nature of the substituents, complex metal ion and temperature [35]. Non- aggregated phthalocyanines have received considerable attention. These compounds, normally with bulky substituents, possess good solubility, which can facilitate the purification and characterization processes [36].
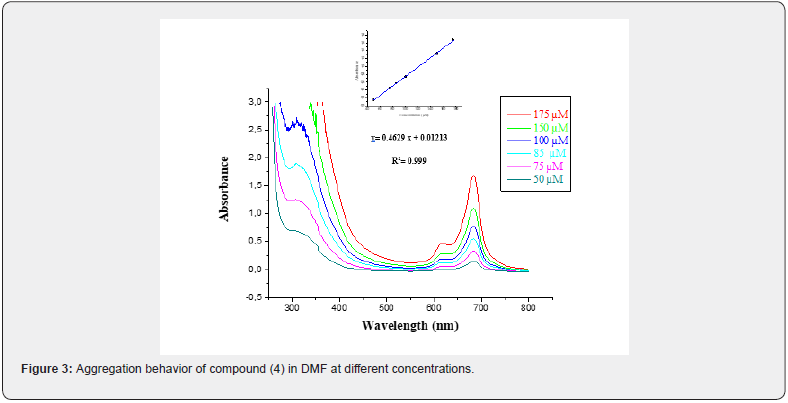
Generally, the increasing concentration of Pcs leads to aggregation, which is observed on the position of Q-bands, which shift to higher energies by a parallel decrease in the molar absorption [37]. The aggregation behavior of the Metallo phthalocyanine complex (4) was investigated at different concentrations in DMF (Figure 3). As the concentration was increased, the intensity of absorption of the Q-band corresponding to monomeric species also increased and there were no new bands due to the aggregated species for the complex. Beer-Lambert law is obeyed for concentrations ranging from 175 μM to 50 μM of compound (4).
Antibacterial Activity
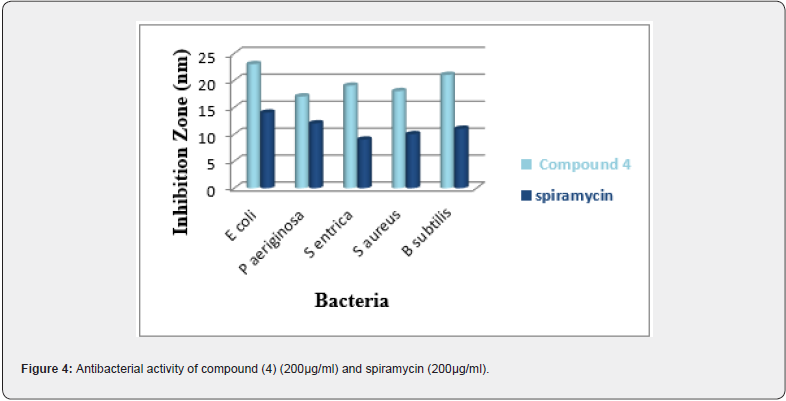
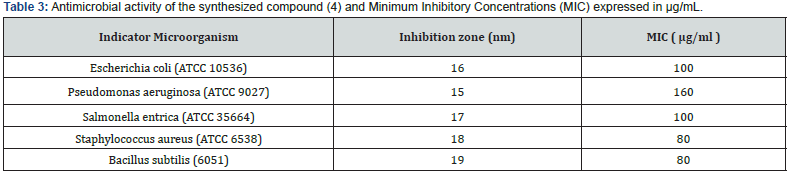
The novel synthesized compound (4) was evaluated for its antibacterial activity in vitro by using zone inhibition technique (disk diffusion method) against gram-negative bacteria (Escherichia coli (ATCC 10536), Salmonella entrica (ATCC 35664), Pseudomonas aeruginosa (ATCC 9027)) and gram-positive (Staphylococcus aureus (ATCC 6538), Bacillus subtilis (6051)). The results obtained were compared with the standard spiramycin drug. The antibacterial activity of the tested compound (4) was demonstrated by the formation of an inhibitory zone, in the order of 15-19 mm, after 24 h of incubation at 37°C (Figure 4).
It was obtained that °C ompound (4) showed antibacterial activities against all studied bacteria. The highest antibacterial activity against Bacillus subtilis [38] with inhibition zone value equal to 17 mm and the minimum inhibitory concentration MIC is equal to 80 μg/ml (Table 3).
Antifungal Activity
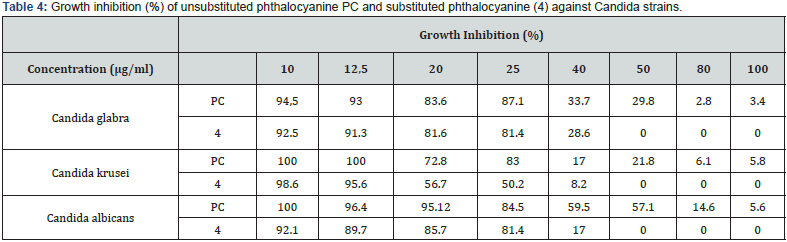
In the present study, antifungal activities of unsubstituted phthalocyanine and substituted phthalocyanine were tested against three strains of Candida (Candida albicans, Candida glabrata and Candida krusei) according to the micro plate method described by Santos and Hamdan [39].Antifungal activity, tested against these microorganisms, was evaluated quantitatively by determining percent growth inhibition and minimal inhibitory concentration (MIC) compared to that of fluconazole which has been used as a reference antibiotic. Results clearly showed variation of the growth inhibition which depends on the concentration of the product and of the tested strain (Table 4).
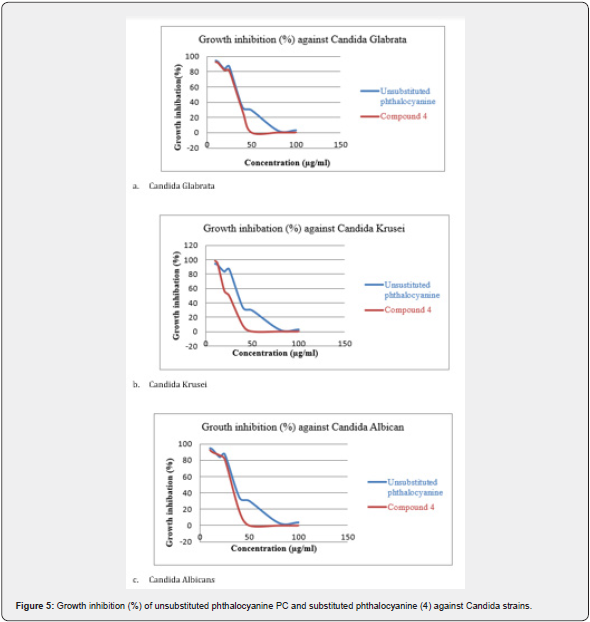
The results presented in Figure 5 indicate the existence of a significant antifungal effect of synthesized zinc phthalocyanines on the growth of Candida strains used at concentrations ranging from 10μM to 100μM. Substituted phthalocyanine compound (4) exhibited more important antifungal activity applied against Candida strains than unsubstituted phthalocyanine. At the concentration of 50μg/ml, inhibitory effect of the tested substituted phthalocyanine on the growth of inhibition was of 100% against all the tested strains. This effect was less important for concentrations ranging from 10 to 40gl/ml.
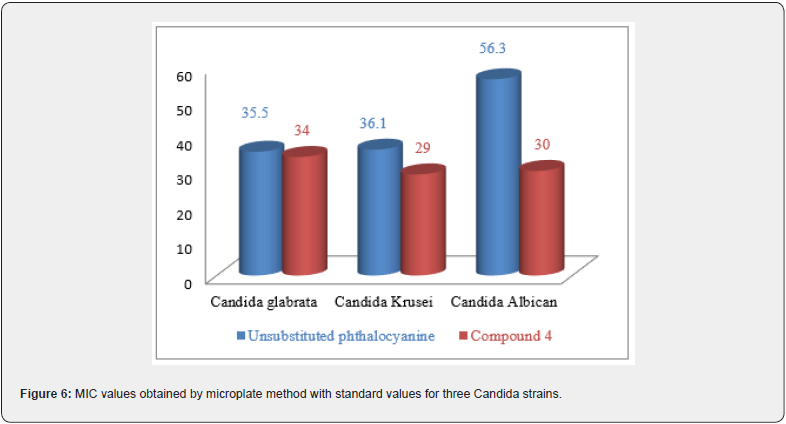
Moreover, the new phthalocyanine (4) showed a greater antifungal activity (MIC is low), against all studied strains, than that of unsubstituted phthalocyanine. The evaluation of antifungal activity of the tested compounds revealed that substituted phthalocyanine (4) was the most potent active compound against Candida strains. MIC values of synthesized phthalocyanine were determined as 34 μg/ml, 29 μg/ml and 30 μg/ml against Candida glabrata, Candida krusei and Candida albicans, respectively (Figure 6).
Conclusion
The present work describes the synthesis and characterization of phthalocyanine bearing 1-(2-(λ1-oxidaneyl) ethyl)-2-methyl-5- nitro-1H-imidazole substituents at the peripheral positions. The synthesized phthalocyanine complex shows excellent solubility in organic solvents such as DMF and DMSO. The aggregation behavior of this compound was investigated. It showed monomeric behaviors in DMF for studied concentrations ranging from 175 μM to 50 μM. In addition, the antibacterial and antifungal activities of this compound were investigated against gram-negative bacteria, gram-positive bacteria and different varieties of Candida. The novel metallophthalocyanine showed antibacterial activity against test microorganisms. It showed maximum antibacterial activity against Bacillus subtilis and showed maximum antifungal activity against Candida krusei.
Acknowledgement
The authors express their grateful thanks to Dr. Sadri ZNAIDI (Pasteur Institute of Tunis) for providing all the necessary equipment to perform the antifungal tests.
References
- McKeown NB (1998) Phthalocyanine Materials: synthesis, structure, and function. Cambridge UK New York 14(3).
- Bıyıklıoğlu Z, Durmus M, Kantekin H (2010) Synthesis, photophysical and photochemical properties of quinoline substituted zinc (II) phthalocyanines and their quaternized derivatives. J Photochem Photobiol A: Chem 211 (1): 32-41.
- Yao CB, Zhang Y, Chen DT, Yin HT, Yu CQ, et al. (2013) Study of All-Optical Switching and Optical Limiting Properties in Phenoxy-Phthalocyanines Liquid. J Opt Laser Technol 47: 228-231.
- Güzel E, Yaşa Atmaca G, Erdoğmuş A, Koçak MB (2017) Novel Sulfonated Hydrophilic Indium (III) and Gallium (III) Phthalocyanine Photosensitizers: Preparation and Investigation of Photophysicochemical Properties. J Coord Chem 70 (15): 2659-2670.
- Güzel E, Günsel A, Bilgiçli AT, Atmaca GY, Erdoğmuş A, et al. (2017) Synthesis and Photophysicochemical Properties of Novel Thiadiazole-Substituted Zinc (II), Gallium (III) and Silicon (IV) Phthalocyanines for Photodynamic Therapy. J Inorganica Chim Acta 467(1): 169-176.
- Çimen Y, Ermiş E, Dumludağ F, Özkaya AR, Salih B, et al. (2014) Synthesis, Characterization, Electrochemistry and VOC Sensing Properties of Novel Ball-Type Dinuclear Metallo phthalocyanines. J Sens Actuators B Chem 202(31): 1137-1147.
- Ağırtaş MS, Altındal A, Salih B, Saydam S, Bekaroğlu Ö (2011) Synthesis, Characterization, and Electrochemical and Electrical Properties of Novel Mono and Ball-Type Metallophthalocyanines with Four 9,9-Bis(4-Hydroxyphenyl) Fluorene. J Chem Soc Dalton trans 40 (13): 3315-3324.
- Jiang Y, Lu Y, Lv X, Han D, Zhang Q, et al. (2013) Enhanced catalytic performance of Pt-Free Iron phthalocyanine by graphene support for efficient oxygen reduction reaction. J ACS Catalysis 3(6): 1263-1271.
- Walter MG, Rudine AB, Wamser CC (2010) Porphyrins and Phthalocyanines in Solar Photovoltaic Cells. J Porphyr Phthalocyanines 14 (9): 759-792.
- Donders CA, Liu SX, Loosli C, Sanguinet L, Neels A, et al. (2006) Synthesis of Tetrathiafulvalene-Annulated Phthalocyanine. J Tetrahedron Lett 62 (15): 3543-3549.
- Shilpa Harish T, Viswanath P (2016) Annealing Assisted Structural and Surface Morphological Changes in Langmuir–Blodgett Films of Nickel Octabutoxy Phthalocyanine. J Thin Solid Films 598(1): 170-176.
- Bilgiçli A. T, Kandaz M, Özkaya A. R, Salih B (2009) Tetracids-Phthalocyanines Bearing Electron-Withdrawing Fluoro Functionality: Synthesis, Spectroscopy, and Electrochemistry. Heteroatom Chemistry 20(5): 262-271.
- Lukyanets EA, Nemykin VN (2010) The Key Role of Peripheral Substituents in the Chemistry of Phthalocyanines and Their Analogs. J Porphyr Phthalocyanines 14(1): 1-40.
- Amaral GP, Puntel GO, Dalla Corte CL, Dobrachinski F, Barcelos RP, et al. (2012) The Antioxidant Properties of Different Phthalocyanines. J Toxicol In Vitro 26 (1): 125-132.
- Ağırtaş MS, Çelebi M, Gümüş S, Özdemir S, Okumuş V (2013) New Water Soluble Phenoxy Phenyl Diazenyl Benzoic Acid Substituted Phthalocyanine Derivatives: Synthesis, Antioxidant Activities, Atypical Aggregation Behavior and Electronic Properties. J Dyes Pigm 99(2): 423-431.
- Pankuch GA, Jacobs MR, Appelbaum PC (1993) New Water Soluble Phenoxy Phenyl Diazenyl Benzoic Acid Substituted Phthalocyanine Derivatives: Synthesis, Antioxidant Activities, Atypical Aggregation Behavior and Electronic Properties. J Antimicrob Agents Chemother 37(8): 1649-1654.
- Young JG, Onyebuagu W (1990) Synthesis and Characterization of Di-Disubstituted Phthalocyanines. J Org Chem Res 55 (7): 2155-2159.
- Perrin BDD, Armarego WLF (1989) Purification of Laboratory Chemicals. Aufl Oxford Pergamon Press J Acta Hydroch Hydrob 17(6): 632-632.
- Kalemba D, Kunicka A (2003) Antibacterial and antifungal properties of essential oils. J Curr Med Chem 10(10): 813-829.
- Arendrup MC, Estrella MC, Florl CL, Hope W, AFST E (2017) Method for the Determination of Broth Dilution Minimum Inhibitory Concentrations of Antifungal Agents for yeasts. EUCAST Defin. Doc E DEF 7. 3.1.
- (1998) National Committee for clinical laboratory standard. Referece method for broth dilution antifungal susceptibility testing of conidium -forming filamentous fungi. Proposed standard M38-P. Wayne, PA: National Committee for clinical laboratory standard.
- Kaspady M, Narayanaswamy VK, Raju M, Gopal (2009) Synthesis, Antibacterial Activity of 2,4-Disubstituted Oxazoles and Thiazoles as Bioisosteres. J Lett Drug Des Discov 6(1): 21-28.
- Lamagni TL, Evans BG, Shigematsu M, Johnson EM (2001) Emerging trends in the epidemiology of invasive mycoses in England and Wales. J Epidemiol Infect 126(3): 397-414.
- Samaranayake LP, Leung WK, Jin L (2009) Oral mucosal fungal infections. J Periodontol 49: 39-59.
- Akçay HT, Bayrak R, Karslioğlu S, Şahin E (2012) Synthesis, Characterization and Spectroscopic Studies of Novel Peripherally Tetra-Imidazole Substituted Phthalocyanine and Its Metal Complexes, the Computational and Experimental Studies of the Novel Phthalonitrile Derivative. Journal of Organometallic Chemistry. J Organomet Chem 713: 1-10.
- Değirmencioğlu İ, Bayrak R, Er M, Serbest K (2009) The Microwave-Assisted Synthesis and Structural Characterization of Novel, Dithia-Bridged Polymeric Phthalocyanines Containing a Substituted Thiophenylamine Schiff Base. J Dyes Pigm 83(1): 51-58.
- Makhseed S, Samuel J (2009) The Synthesis and Characterization of Zincphthalocyanines Bearing Functionalized Bulky Phenoxy Substituents. J Dyes Pigm 82(1): 1-8 5.
- Arslanoglu Y, Mertsevim A, Hamuryudan E, Gul A (2006) Near-IR Absorbing Phthalocyanines. J Dyes Pigm 68(2-3): 129-132.
- Sommerauer M, Rager C, Hanack M (1996) Am Chem Soc 118(42): 10085-10093.
- Koca A, Kalkan A, Bayır ZA (2011) Electrocatalytic Oxygen Reduction and Hydrogen Evolution Reactions on Phthalocyanine Modified Electrodes: Electrochemical, in Situ Spectro electrochemical, and in Situ Electrocolorimetric Monitoring. J Electrochim Acta 56(16): 5513-5525.
- Nyokong T, Isago H (2004) The Renaissance in Optical Spectroscopy of Phthalocyanines and Other Tetraazaporphyrins. J Porphyr Phthalocyanines 8(9): 1083-1090.
- Mayer UA (1979) A semiempirical model for the description of solvent effects on chemical reactions. J Pure & Appl Chem 51: 1697-1712.
- Komarov EV, Komarov VN (1974) Extraction Systems with Associated Extractants. J Russ Chem Rev 43(4) : 263-275.
- Correia RF, Andrade SM, Viseu MI (2012) Aggregation and Disaggregation of Anionic Aluminum Phthalocyanines in Cationic Pre-Micelle and Micelle Media: A Fluorescence Study. J Photochem Photobiol A: Chem 235: 21-28.
- Bıyıklıoğlu Z, Durmuş M, Kantekin H (2010) Synthesis, Photophysical and Photochemical Properties of Quinoline Substituted Zinc (II) Phthalocyanines and Their Quaternized Derivatives. J Photochem Photobiol A: Chem 211(1): 32-41.
- Dominguez DD, Snow AW, Shirk JS, Pong RGS (2001) Polyethylene oxide capped Phthalocyanines: Limiting Phthalocyanine Aggregation to Dimer Formation. J Porphyr Phthalocyanines 5(7): 582-592.
- Akkurt HYY, Okur AI, Gül A (2012) Unsymmetrical Phthalocyanines with Cyclopalladated Azo Functions. J Porphyr Phthalocyanines 16(2): 192-199.
- Salih Ağırtas M, Dede E, Gümüs S, Dündar A, Okumus V (2014) Metallo Phthalocyanines bearing 2-Isopropyl-6-methylpyrimidin-4-yloxy Substituents: Synthesis, Characterization, Aggregation Behavior, Antioxidant and Antibacterial Activity, and Electronic Properties. J Zeitschrift für Anorgan Allgemeine Chem 640(10): 1953-1959.
- Santos DA, Hamdan JS (2005) Evaluation of broth microdilution antifungal susceptibility testing conditions for Trichophyton rubrum. J Clin Microbio 43(4): 1917-1920.






























