Pharmacognostic Standardization of Megacarpaea Polyandra Benth Leaf
Shabir Ijaz1*, Anjum Perveen1, Ahmed Shahid Mirza2, Saima Ashraf3 and Imtiaz Bashir4
1Center for Plant Conservation, University of Karachi, Pakistan
2Department of Pharmacognosy, Faculty of Pharmacy, Ziauddin University, Karachi
3Department of Botany, University of Azad Jammu and Kashmir Muzaffarabad, Pakistan
4Department of Horticulture, University of Haripur, Pakistan
Submission: May 22, 2023;Published: August 15, 2023
*Corresponding author: Shabir Ijaz, Center for Plant Conservation, University of Karachi, Pakistan
How to cite this article: Shabir Ijaz*, Anjum Perveen, Ahmed Shahid Mirza, Saima Ashraf and Imtiaz Bashir. Pharmacognostic Standardization of Megacarpaea Polyandra Benth Leaf. Organic & Medicinal Chem IJ. 2023; 12(3): 555840. DOI: 10.19080/OMCIJ.2023.12.555840
Abstract
M. polyandra Benth. (Locally known as Chach) is a perennial medicinal herb confined to the Himalayas from Kashmir to C. Nepal. Traditionally, young leaves are cooked as vegetables; the root is used to cure fever, gastric problems, and pneumonia. Owing to the presence of analogous species in its habitat the plant often suffers from quality disputes for collectors. To maintain the plant’s uniqueness and avoid adulteration, pharmacognostic approaches, and microscopic investigations were used to report its features. Tests such as physicochemical study and chemical analysis were applied. The anatomical features of the plant parts were studied and identified through light microscopy. Pharmacognostic parameters viz ash%, moisture%, extractive values, and pH showed different values; various classes of plant metabolites have been detected. Fluorescence analysis, FTIR Spectroscopy, and elemental analysis revealed unique features of this plant. Different types of tissues of stomata, midrib, and powdered leaf were illustrated and identified. The parameters indicated above are being reported for the first time in M. polyandra and are important in setting microscopic and pharmacopoeias criteria for future identification and authentication of adulterantion in this plant species.
Keywords: Megacarpaea polyandra; Medicinal plants; Himalaya; Brassicaceae; pharmacognosy
(Figure 1)
Introduction
In recent centuries, researchers have paid close attention to complementary and alternative medicine, with a particular focus on plant-derived medicines. Alternative remedies and the use of common objects to treat, avoid, and lighten the infection have progressed for a variety of reasons, including the fact that standard pharmaceuticals have several side effects [1]. Natural substances are chosen for screening based on their traditional use, compound organization, toxicity, randomized determination, or a combination of characteristics [2]. The institutionalization of natural medications depends on thorough investigations of the plant species, phytochemical examinations assurance of following components spectroscopy, determination of functional groups and structural components found in organic compounds by Fourier transform infrared FTIR, physicochemical analysis to evaluate the quality of the herbal drugs by determining ash values, moisture content, extractive values in different solvent systems, pharmacological activities, and so forth, to ascertain its importance in medicinal and culinary purposes [3].
The quality control of herbal medicine is currently a source of concern. The quality of these medications is dependent on the correct verification of the plant parts used and the use of established techniques to standardize herbal remedies. Due to the close likeness of several species belonging to the same genus or even the family, there is always the potential of unintentional adulteration while collecting [4]. M. polyandra is a high-valued wild medicinal perennial crucifer herb (Figure 1) belong to Brassicaceae family and endemic to the Himalayas in Kashmir, Pakistan, India, and Nepal [5,6] found at 3000 to 3400m in alpine and subalpine environments [7].
In several ethnobotanical reports M. polyandra is recognized by its use among pastoralists and local farmers, young leaves of this plant cooked as vegetable and root is effective against pneumonia [8]. In others studies the root is reported as coolant, antidote for scorpion sting and snake bite and to cure fever [5,6]. A report regarding the growth stages of M. polyandra was published by Singh et al [9] they argued that the complete maturity (germination to flower/seed production) depends on a three years’ time scale. The first two years are important in which vegetative growth arises. On the other hand, at the vegetative stages collection of leaves is high because of the young leaves and tender petiole. In vegetative stage this species phenotypically resembles with its neighboring species- Angelica archangelica var. himalaica (Clarke) E. Nasir, comb., and Heracleum candicans Wall. ex-DC. Occasionally, collectors are confused to identify their desired species. As there are no known standards for this species with no identifying characters. Therefore, the present study was designed to standardize M. polyandra pharmacognostically, microscopically, and chemically to decrease the risks of adulteration of this valuable herbal drug with other species.

Methodology
Plant Collection, Identification and Submission of Specimen
Fresh and completely mature plant specimens were collected during August 2021 from Lawat, District Neelum Azad Kashmir. Plant was identified through available literature ‘’Brassicaceae. vol. No 55, page 81-82, Flora of Pakistan’’ [10] and taxonomy followed ‘’World Flora Online’’ [11] the plant was air dried, mounted on Herbarium sheet and assigned Herbarium number (KUH735) and deposited in the Prof. Dr. S. I. Ali Herbarium, Center for Plant Conservation, University of Karachi.
Physicochemical Analysis
Physicochemical analysis of leaf, which includes ash values (total ash, acid insoluble, and water-soluble ash), moisture content, extractive value, and pH value was carried out following the methods of [12-14].
Morphological and Macroscopic Observations
Macromorphology of plant was carried out with the help of “Flora of Pakistan, no 55 family Brassicaceae’’ [10] and followed the method of Wallis [15].
Light Microscopy
Dried leaves were soaked in distilled water for 24 hours to maximum swelling and forming the shape of tissues. After 24 hours the section of the midrib (near to the leaf apex) was then cut into thin pieces and fine sections were treated with glycerin and chloral hydrate separately. For the study of stomata wet leaf was slightly crushed then a pinch of crushed material was placed on slides. One slide was treated with chloroform and the second with glycerin. For powdered leaf study a pinch of dry powder was treated with glycerin. The sections and powder were observed under the digital microscope (B- 290TB) [16].
Phytochemical Screening
Preparation of Extracts
5g of powdered leaf material was accurately weighed. Following that, 100 mL of each solvent-acetone, chloroform, methanol, and water was filled in conical flask. The flask with leaf powder was placed on a stirrer for 48 hours. After that, the substance was filtered. The filtrate was placed in Petri dishes for complete drying. The dried extract was used to calculate the extractive value [17].
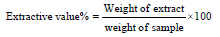
Phytochemical Analysis
Extracts of four different solvents were checked to know the presence or absence of metabolites. For that purpose, 21 different standard protocols were applied. These standard protocols for preliminary phytochemical analyses have been discussed in the study of [16].
Fluorescence Study
The powdered leaf material was subjected to fluorescence analysis. Powder was treated with a number of chemicals (ether, hydrochloric acid, methanol, methyl trichloride, acetic acid, sulfuric acid, nitric acid, phosphoric acid, benzene, ammonia, water, and potassium hydroxide) and observed in ordinary daylight, under UV light- short wavelength (254 nm) and long wavelength (366 nm) [17].
FTIR Spectroscopy
Dried powdered leaf (10 mg) was subjected to FTIR analysis. After that, the sample disc was stacked in a spectrometer with an output range of 400 to 4,000 cm1 and 4 cm goals [18].
Scanning Electron Microscopy-Energy Dispersive Spectroscopy (Sem-Eds)
SEM-EDS (JSM-IT100 InTouchScopeTM) was used to analyze the leaf’s microscopic features and element distribution. The surface areas of leaf (10 μm) (i) lamina with stomata (ii) lamina without stomata were selected for the field view and to capture elements distribution, and the microscope was operated at 20 KV accelerating voltage with the pixel size 1280 x 960μm.
Results
Morphology
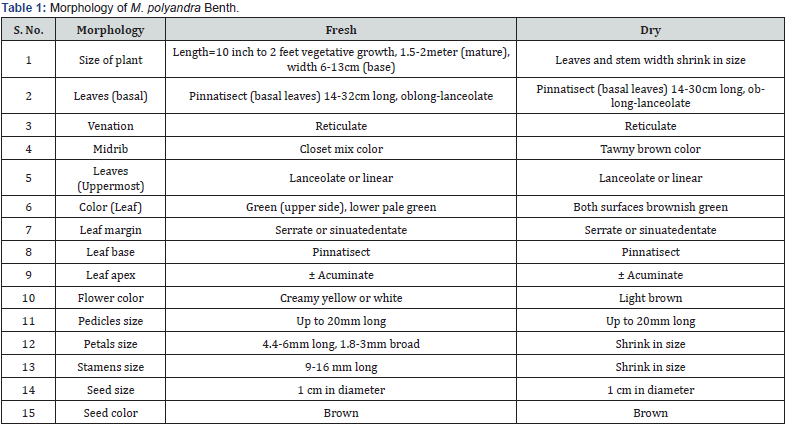
Macromorphological properties of above ground parts of the plants are often employed for description and explanation of species in floras. Macromorphology is only useful for experts (plant taxonomists) when all morphological aspects of the plant (vegetative, flower, and seed) are available at the same time. On the other hand, Shinwari and Qaiser [19] says that there are few plant taxonomists/field botanists in Pakistan. Morphology of fresh and dried parts of this plant reported in this study (Table 1). Reported morphological characters are supportive for researchers during characterization of crud drug in future. Otherwise searching for plants within the flora is a difficult task for nonexperts. The macromorphology of aboveground parts (stem, leaves, flower, and seed) is summarized in Table 1. Change in morphology of fresh and dried parts was seen in size and color of leaves and flower parts other features were same in both forms.
Physicochemical Analysis
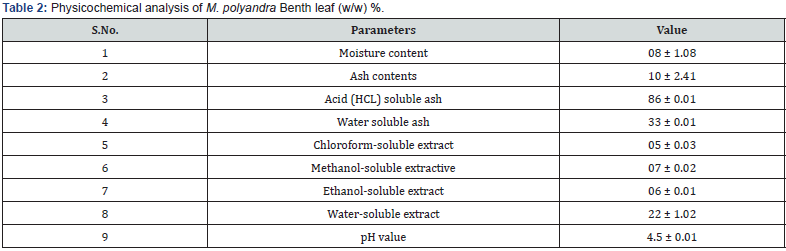
Physicochemical/proximate analysis (Table 2) of the leaf was carried out which includes moisture content, ash values (total ash, water soluble ash, acid soluble ash), extractive values and pH. Physicochemical analyses are the most important tests for estimating the purity and quality of the collected crude drug material when compared to referenced values, as well as assisting in the establishment of standardization parameters and pharmacopoeial standards. The results for moisture percentage, total ash, acid soluble ash, water soluble ash and pH value were 8%, 10%, 86%, 33% and 4.5 respectively. All of the results were within the WHO’s guidelines for herbal medications [13]. The existence of extraneous matter adhering to the plant part is determined by ash values, and their collection also gives a clear indication of the presence of low-grade mineral matter or inorganic contaminants in the crude medicines [16]. Moisture content measurement is an important parameter which is linked to the creation of microbial contaminants and high moisture can induce deterioration of the crude medication due to fungal/ bacterial growth. Our sample’s moisture content was within the specified range of 8 to 14 percent. Lower moisture content, on the other hand, ensures plant material’s long-term storability and stability [13]. The pH value of the crude medication is also included in the proximate analysis. The pH value of the drug determines its acidic and basic nature. The pH value of the sample was 4.5± 0.01 which was lower than 7, which falls in the acidic range that clearly shows that the constituents are slightly acidic. Extractive values of leaf were estimated in four solvents, which was high in water (33%), followed by methanol (7%), ethanol (6%) and chloroform (5%), these values are to Figure out the best suitable solvent for the extractive yield respectively. A solvent that produces a high extract yield is suitable and will aid in obtaining the maximum amount of components. With water as the solvent, a good yield was obtained (Table 2). Based on the nature of constituents and the solvent used, extractive values indicate distinct types of active phytoconstituents and their amounts in medicinal plants. It produces various amounts and types of phytoconstituents in response to a specific solvent and is used to detect adulterants and exhausted materials in crude drugs [20]. The active portion of a drug is separated from crude drugs and powder from various parts of the plant using different solvents, which is a significant marker of the nature of chemical ingredients in drugs [21]. Several researchers have published reports about extractive values of crude powder in which they were used variety of organic and inorganic solvents e.g Aslam et al [17]; Khan et al, [22]; Wang, et al, (2021); Nafees et al; Uza and Dastagir [23].
Microscopic Investigation of Midrib, Stomata and Powdered Leaf
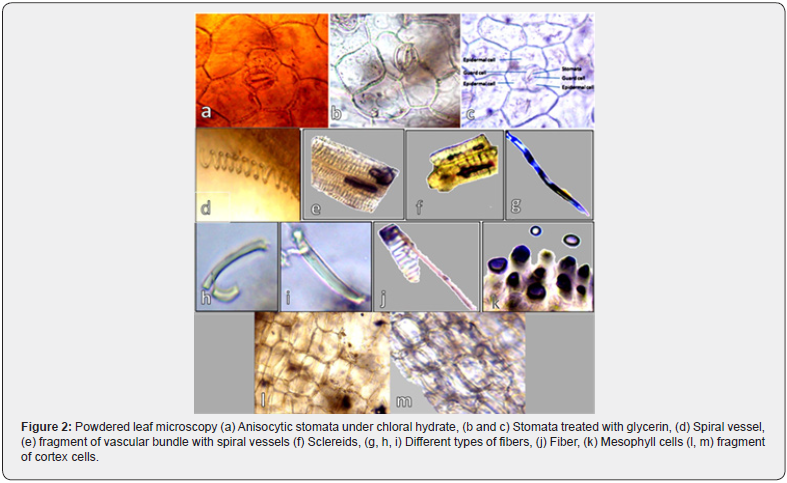
Stomatal study revealed that the leaf of the plant bear anisocytic stomata via three companion cells that surrounds the stomata (Figure 2a, b, c). Leaf powdered showed different fragments of cells/tissues including spiral vessel (Figure 2d), fiber and vessels (Figure 2e), sclereids (Figure 2f), fibers with different colors and shapes (Figure 2g, h, i) fiber (Figure 2j), mesophyll cells (Figure 2k) and fragment of cortex cells (Figure 2, l-m).
The transverse section of the midrib showed various types of cells, midrib was covered by both the upper and lower epidermis from abaxial as well as adaxial surfaces. The shape of midrib is more or less likely to the turtle. Vascular bundles are surrounded by inner and outer phloem and cambium (dark brown in color) is present between the outer phloem and xylem. On the abaxial side, (lower) midrib vascular bundles were arranged in oval shape and no vascular bundle was found in upper side of midrib. The adaxial side of midrib consists of two types of tissue viz epidermis and endodermis. Epidermis of the lower side of midrib consists of two layers of cells. Phloem towards the pith side was prominent with creamy white color and thick at the center, size of phloem towards cortex side was large than inner side phloem. The cortex region was brown in color with an irregular shape of tissue. The shape and size of pith tissues were not prominent; area of each xylem vessel was milky white in color (Figure 3).
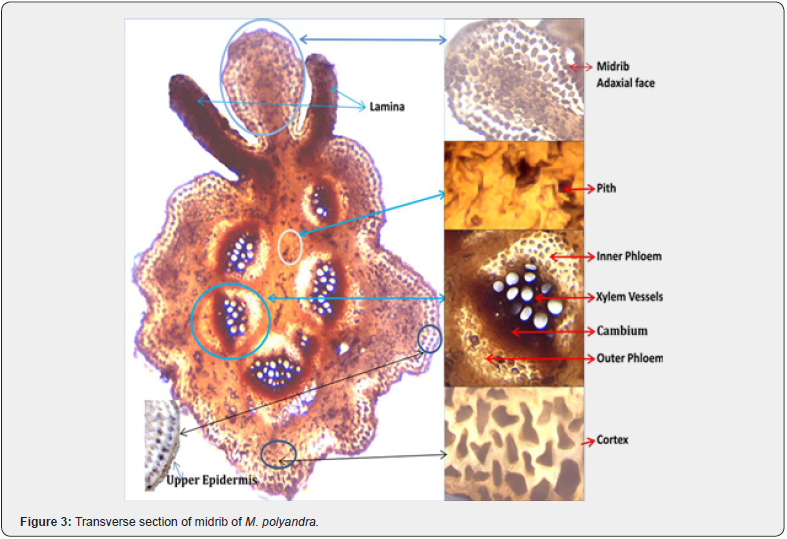
Midrib is an important part of studying tissues and have been reported in many studies such as Nafees, Ullah, & Ikram, [23] carried out the midrib studies of Buddleja asiatica. Mahmud et al, [24] studied Holoptelea integrifolia leaves, and they explained that the leaf anatomy is the foundation for standardizing and correctly identifying plants, as well as distinguishing them from closely related species.
Fluorescence and Phytochemical Analysis
Fluorescence analysis of the powdered leaf of M. polyandra showed vast variation in color, when was treating with 13 different types of reagents under low (254nm) Visible light and high (366 nm). The most popular approaches identifying chemical substances include labeling with fluorescent chemicals and assessing color changes for [13]. The powder was treated with different reagents and different colors were observed under both visible and ultraviolet light according to the different contents of the powder (Table 3). This method can quickly verify the validity of medicinal materials and determine their quality in a tentative manner.

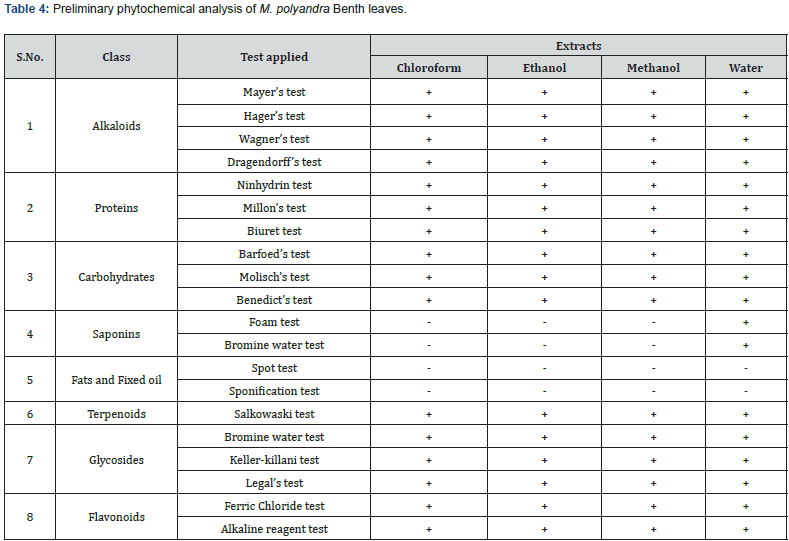
The leaves of the aforementioned plant contained a variety of bioactive chemicals. The existence of active secondary phytoconstituents such as flavonoids, saponins, alkaloids, terpenoids, and phenols determines the medicinal potential of the plant (Table 4). Qualitative phytochemical screening must be responsible for the detection of secondary metabolites in plant crude materials, pharmacological amplification of crude pharmaceuticals, and the provision of genuine drugs for businesses [17]. Flavonoids have been shown to have anti-anaphylactic, anti-inflammatory, antioxidant, anti-allergic, antimicrobial, and anticancer properties, as well as anti-inflammatory, antioxidant, anti-allergic, anti-allergic, antimicrobial, and anticancer properties [25]. The presence of high amounts of steroids, terpenoids, and saponins in plants is thought to be responsible for their astringent characteristics. Tannins are used to treat bacterial, viral, and fungal infections, as well as burns, inflammation, and wound healing [27]. Saponins and glycosides must be utilized as immune regulators, anti-cancer agents, and in the treatment of the majority of heart illnesses. Phenolic constituents have been shown to have cytotoxic, anti-mutagenic, anti-oxidative, and astringent actions against pathogens such as bacteria [27]. Metabolites like anthraquinones are employed as laxatives, antimalarials, and anticancer drugs [28].
To determine the presence of different novel compounds in the plant 20 different tests were performed. We identified seven different classes of bioactive compounds (except fixed oil) in the leaves of M. polyandra, which show that leaves are also a rich source of therapeutic compounds. Similar work was carried out by various workers; for example, Aslam et al. 2019 carried out the phytochemical’s investigation of Caralluma edulis; Uza and Dastagir (2022) Astragalus scorpiurus; Nafees et al. [23] authenticated of Buddleja asiatica for correct identification, authentication and quality assurance of the preliminary resources is an important requirement to make sure the reproducible quality of phytomedicine which will show the safety and effectiveness of herbal products. Pharmacognosy is an easy step to discriminate adulterated drug from an unadulterated one.
SEM-EDS and FTIR Analysis
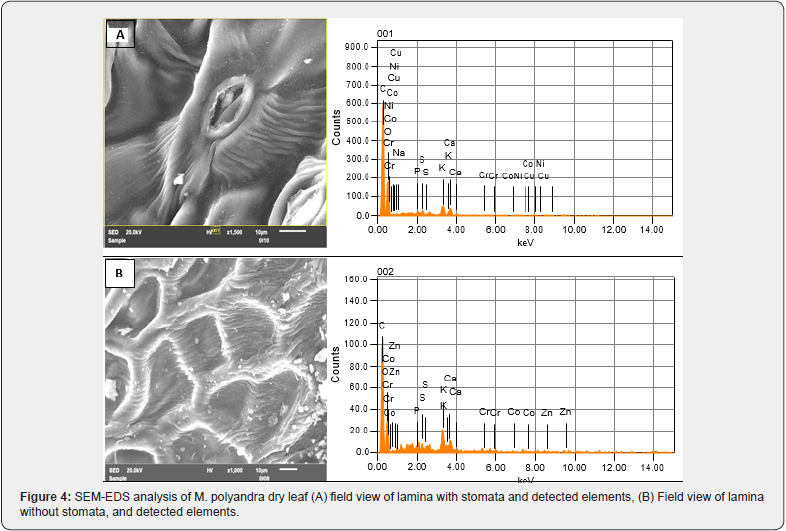
SEM-EDS (scanning electron microscopy with energy dispersive X-ray spectrometry) is an elemental microanalysis technique used in a variety of fields including physical and biological sciences, engineering, technology, and forensic investigations [29]. X-ray peaks give identification and quantification for all elements of the periodic. To gather, analyze, and measure X-ray spectra, modern SEM-EDS X-ray microanalysis systems are often equipped with a robust, flexible, and extremely helpful collection of software tools [30]. This technique was used by Boi et al, [31] for element detection in the root part of Helichrysum microphyllum subsp Tyrrhenicum. By rastering a focused electron beam across the surface and detecting secondary or back scattered electron signals, SEM offers detailed high-resolution pictures of the material. The elemental identification and quantitative compositional information are also provided by an Energy Dispersive X-Ray analyzer. The elements detected by SEM-EDS from M. polyandera dry leaf (lamina with stomata) (Figure 4a) are C, O, K, Ca, S, Cu, P, Co, Ni, Na, and Cr with the mass percentage of 52.01, 37.59, 4.34, 3.03, 1.17, 0.71, 0.42, 0.34, 0.23, 0.14, and 0.02 respectively. The detected elements with percentages from the lamina (10 μm) without stomata were C (49.70%), followed by O (35.31%), K (8.81,%), Ca (3.84%), S (1.30%), Zn (0.74%), P (0.14%), Co (0.10%) and Cr (0.06%) (Figure 4b).
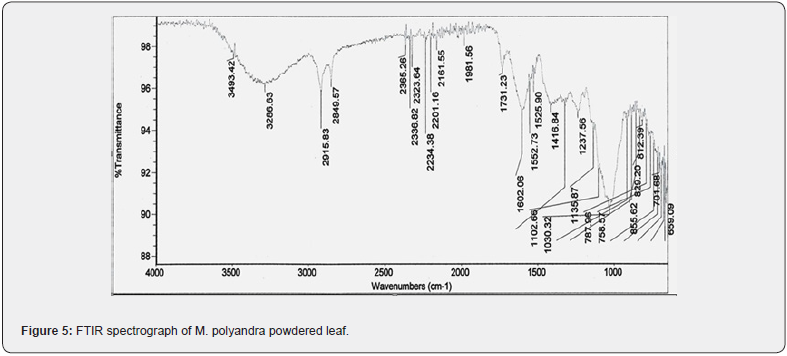
The FTIR analysis was performed for the powdered leaf. The transmittance scan was run over 4,000- 1000 cm-1 (Figure 5). There were 27 sharp bends observed in the IR spectrum. First peak was at 3493.42 cm-1 which indicates the presence of O-H bonds. Other values such as 3286.63 indicates OH alcohol stretch, 2915.83 indicates O-H aliphatic stretch, 2849.57 show CH₃ group, 2365.26 C-H aromatic bond, 2336.82 O=C=O stretching, 2323.64 O=C=O, 2234.38 C=C Alkyne, 2201.16 C≡C medial alkyne, 2161.55 terminal alkyne, 1981.56 C=C=C alkane, 1731.23 Six-membered ring lactone, 1602.06 N=N, 1552.73 aliphatic nitro compounds, 1525.90 N-O starching , 1416.84, peaks such as 1237.56, 1135.87, 1102.66, 1030.32 represents -C-O stretching sulphate ion, 855.62 P-O-C stretch, 829.20, 812.39, three peaks 787.96, 758.57 and 701.68 shows C-Cl stretch, 659.09 represents thioether and CH2-S (Figure 5) [32]. The FTIR analysis has great importance which indicates the functional groups/ compound variety of the raw material during authentication of medicinal plants [33-36].
Conclusion
Raw material collected from most therapeutic plants loses its morphological characteristics. Sometimes only a single part of the plant is collected and used, or occasionally one common name is given to different species. That is why it is difficult to find out the correct scientific name of raw material by searching flora. Pharmacognostic study is a tool that can be used for the correct identification of raw materials of medicinal plants and also useful for specific differentiation among closely related species. It can be used to gain complete information about a crude drug and to promote traditional herbal medicine knowledge in a meaningful way. Accurate identification of the plants paves the way for further research such as pharmacological activities, isolation of therapeutic bioactive compounds and drug preparation. The parameters that have been studied in this study provides monograph for M. polyandra plant. These characteristics will aid in recognizing the species from its co-species and preventing the plant from becoming adulterated.
References
- Ekor M (2014) The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Frontiers in Pharmacol 4: 177.
- Lahlou M (2013) The success of natural products in drug discovery. Pharmacology and Pharmacy 4(3A): 17-31.
- Manek RV, Builders PF, Kolling WM, Emeje M, Kunle OO (2012) Physicochemical and binder properties of starch obtained from Cyperus esculentus. AAPS Pharm Sci Tech 13(2): 379-388.
- Ijaz S, Perveen A, Ashraf S, Bibi A, Dogan Y (2021) Indigenous wild plants and fungi traditionally used in folk medicine and functional food in district Neelum Azad Kashmir. Environment Development and Sustainability 23(6): 8307-8330.
- Misra S, Maikhuri RK, Kala CP, Rao KS, Saxena KG (2008) Wild leafy vegetables: A study of their subsistence dietetic support to the inhabitants of Nanda Devi Biosphere Reserve. India Journal of Ethnobiology Ethnomedicine 4(1): 1-9.
- Singh A, Nautiyal MC, Kunwar RM, Bussmann RW (2017) Ethnomedicinal plants used by local inhabitants of Jakholi block, Rudraprayag district, western Himalaya, India. J Ehnobio Ethnomed 13(1): 1-29.
- Korner C (2003) Alpine plant life. Springer Berlin Heidelberg Berlin Heidelberg.
- Ijaz S, Perveen A, Ashraf S, Abid R, Kousar S, et al. (2022) Traditional Knowledge of Wild Edible Plants Used by the People of Lawat, District Neelum, Azad Jammu & Kashmir, Pakistan. Ethnobotany Research and Applications 23: 1-16.
- Singh A, Nautiyal MC, Curti RN, Fenu G (2022) The phenological growth stages of Megacarpaea polyandra Benth. ex-Madden: a high valued traditional medicinal plant of the Himalaya. Genetic Resources and Crop Evolution 69(1): 431-441.
- Jafri SMH, Ali SI, M Qaiser (Eds.) (1973) Flora of Pakistan 55: 81-82.
- Bot HJ. Gard K (2023) WFO Megacarpaea polyandra Benth.
- WHO (1998) Quality control methods for medicinal plant materials. World Health Organization Geneva.
- WHO (2007) WHO guidelines for assessing quality of herbal medicines with reference to contaminants and residues. World Health Organization.
- Perveen A, Ijaz S, Ghaffar N (2020) Comparative phytochemical and physicochemical study of seeds of the genus Angelica L. from Neelum valley Azad Kashmir, Pakistan. Pakistan Journal of Botany 52(1): 257-260.
- Wallis TE (2009) Textbook of pharmacognosy New Delhi, India: CBS Publisher and Distributors (5th ed pp. 572-575).
- Aslam I, Afridi MSK (2018) Pharmacognostic characterization of Beaumontia grandiflora (Roxb) Wall. Leaf for taxonomic identification for quality control of a drug. Journal of Applied Research on Medicinal and Aromatic Plants 8: 53-59.
- Aslam I, Iqbal J, Peerzada S, Afridi MS., Ishtiaq S (2019) Microscopic investigations and pharmacognostic techniques for the standardization of Caralluma edulis (Edgew.) Benth. ex-Hook. f. Microsc Res Tech 82(11): 1891-1902.
- Theng PA, Korpenwar AN (2015) Analysis of bioactive compounds in Geodorum densiflorum (lam.) Schltr Pseudobulb using UV-VIS, FTIR and GC-MS techniques. Journal of Chemical, Biological and Physical Sciences 5(2): 2151.
- Shinwari ZK, Qaiser M (2011) Efforts on conservation and sustainable use of medicinal plants of Pakistan. Pak J of Bot 43(1): 5-10.
- Atasie VN, Akinhanmi TF, Ojiodu CC (2016) Proximate analysis and physicochemical properties of groundnut (Arachis hypogaea L.). Pakistan Journal of Nutrition 8(2): 194-197.
- Khan S, Khan GM (2013) In vitro antifungal activity of Rhazya stricta. Pakistan Journal of Pharmaceutical Sciences 20(4): 279-284.
- Khan SA, Khan B (2020) Anatomy, micromorphology, and physiochemical analysis of Rhus succedanea var. himalaica root. Microsc Res Tech 83(4): 424-435.
- Nafees M, Ullah S Ikram N (2022) Phytochemical and pharmacognostic studies of Buddleja asiatica leaves. Microscopy Research and Technique 85(2): 510-520.
- Mahmud S, Shareef H, Ahmad M, Gouhar S, Rizwani GH (2010) Pharmacognostic studies on fresh mature leaves of Holoptelea integrifolia (Roxb) planch. Pak J of Bot 42(6): 3705-3708.
- Yun TK, Lee YS, Kwon HY, Choi KJ (1996) Saponin contents and anticarcinogenic effects of ginseng depending on types and ages in mice. Zhongguo yao li xue bao 17(4): 293-298.
- Savithramma N, Rao ML, Suhrulatha D (2011) Screening of medicinal plants for secondary metabolites. Middle East Journal of Scientific Research 8(3): 579-584.
- Edeoga HO, Eriata DO (2009) Alkaloid tannin and saponin contents of some Nigerian medicinal plants. Journal of Medicinal and Aromatic Plant Science 23(6): 344-349.
- Deore SL, Jajoo NB, Chittam KP, Deshmukh TA 2015. Comparative pharmacognostic, phytochemical and biological evaluation between five Chlorophytum species. Pharmacognosy Journal 7(5): 316-325.
- Goldstein JI, Newbury DE, Michael JR, Ritchie NW, Scott JH, et al. (2003) Scanning electron microscopy and X- ray microanalysis. 3rd Ed New York: Springer.
- Newbury DE, Ritchie NW (2013) Is scanning electron microscopy/energy dispersive X‐ray spectrometry (SEM/EDS) quantitative. Scanning 35(3): 141-168.
- Boi ME, Medas D, Aquilanti G, Bacchetta G, Birarda G, et al. (2020) Mineralogy and Zn chemical speciation in a soil-plant system from a metal-extreme environment: a study on Helichrysum microphyllum subsp. tyrrhenicum (Campo Pisano Mine, SW Sardinia, Italy). Minerals 10(3): 259.
- He Z, Liu S, Nam S, Klasson KT, Cheng HN (2023) Molecular level characterization of the effect of roasting on the extractable components of glandless cottonseed by Fourier transform ion cyclotron resonance mass spectrometry. Food Chem 403: 134404.
- Coates J (2000) Interpretation of infrared spectra, a practical approach. Encyclopedia of analytical chemistry 12: 10815- 10837.
- Schulze ED, Schweingruber FH, Borner A (2011) Atlas of stem anatomy in herbs, shrubs and trees Springer 1.
- Singh A, Nautiyal MC, Kunwar RM, Bussmann RW (2017) Ethnomedicinal plants used by local inhabitants of Jakholi block, Rudraprayag district, western Himalaya, India. J Ethnobi Ethnomed 13(1): 1-29.
- Uza NU, Dastagir G (2022) Microscopic and pharmacognostic standardization of stragalus scorpiurus bunge. Microsc Res Tech 85(1): 324-338.






























