Phytochemical Analysis of Tsuga Dumosa
Pramod Kumar Ojha1 and Bipin Chandra Joshi2*
1Department of Chemistry, L.S.M. Govt Post Graduate College, Pithoragarh, Uttarakhand, India
2Department of Chemistry, L.B.S. Govt Post Graduate College, Halduchaur, Nainital, Uttarakhand, India
Submission: July 13, 2023;Published: July 24, 2023
*Corresponding author: Bipin Chandra Joshi, Department of Chemistry, L.B.S. Govt Post Graduate College, Halduchaur, Nainital, Uttarakhand, India
How to cite this article: Pramod Kumar Ojha and Bipin Chandra Joshi*. Phytochemical Analysis of Tsuga Dumosa. Organic & Medicinal Chem IJ. 2023; 12(3): 555838. DOI: 10.19080/OMCIJ.2023.12.555838
Abstract
There is increasing evidence showing the potential of plant constituents as antioxidant agents. Plants contain some organic compounds which provide definite physiological action on the human body and these bioactive substances include tannins, alkaloids, carbohydrates, terpenoids, steroids and flavonoids. These compounds are synthesized by primary or rather secondary metabolism of living organisms. Essential oils (EO) from Tsuga dumosa, have various medicinal properties, including antibacterial and antioxidant activity. The plant Tsuga dumosa leaves extracted by hydro distillation method for 6 hours using Clevenger apparatus. The oil was analyzed by Gas Chromatography-Mass spectrophotometry (GC-MS). A total of 35 compounds were identified constituting 98.93% of the total oil. The main compounds were Bornyl acetate (18.87%), α-pinene (17.67%), Limonene (17.05%), Intermedeol (14.13%) and minor compounds was trans-Piperitol acetate (0.09 %), Thunbergol (0.13%), Cryptone (0.14%).
Keywords: Tsuga Dumosa; Phytochemical Analysis; Essential Oil; GC-MS
Abbreviations: E0: Essential Oils; GC-MS: Gas Chromatography-Mass Spectrophotometry; RI: Retention Indices
Introduction
Medicinal plants have the ability to inhibit the growth of a wide range of pathogenic microorganisms due to the presence of essential oils. The antimicrobial impact of essential oils and its various components extracted from medicinal plants has been well documented. Many plant species and herbs exert antioxidant activity due to their essential oil fractions. Some scientists reported the antioxidant activity of essential oils from oregano, thyme, sage, rosemary, clove, coriander, garlic, and onion against both bacteria and molds. The composition, structure, as well as functional groups of the oils play an important role in determining their antioxidant activity. The aromatic oils from plant leaves are used as pharmaceutical raw material in the formulation of many drugs. [1]
The genus Tsuga (Pinaceae) is comprised of nine species. T. dumosa D. Don is an economically as well as medicinally important conifer. It is commonly known as “Hemlock Spruce” and locally called as “Dhupi” or “Thingre Salla”. The plant has been extensively used for timbering and lumber products because of its resistance to decay. The bark of this plant is a rich source of tannin, hence can be used for dying. Himalaya, the youngest mountain system of the world, constitutes an important link between the vegetation of the southern peninsular India on the one hand, the eastern Malaysian, the north-eastern Sino-Japanese and the northern Tibetan areas on the other. Biodiversity is essential for human survival and economic well-being and for the ecosystem function and stability [2-6]. The present paper deals with the estimation of essential oil present in the leaves of plants.
Materials and Methods
Plant Material
The leaves of T. dumosa was collected in the month of January 2020 from Thalkedar near Pithoragarh, Uttarakhand, India in the Kumaon Himalayas. The plant was first identified in the Department of Botany, Kumaun University, Nainital. The collected plant material was first washed with cold water to remove the soil particles and then shade dried.
Chemicals
Isolation of Essential Oil
The leaves of T. dumosa were extracted by hydro-distillation method for 6 hours using Clevenger apparatus. The oil was dried over anhydrous sodium sulphate and stored at room temperature in a sealed vial until analysis was performed. The percentage oil yield was calculated based on the dry weight of the plant. The oil yield was (0.09%).
GC and GC/MS Analyses and Identification
Essential oil analyses were performed by GC-MS and GC-FID on a Shimadzu QP-2010 instrument, equipped with FID, in the same conditions. The percentage composition of the oil sample was computed from the GC peak areas without using correction for response factors. The oil was analyzed using a Shimadzu GC/ MS Model QP 2010 Plus, equipped with Rtx-5MS (30 m × 0.25 mm; 0.25 mm film thickness) fused silica capillary column. Helium (99.99%) was used as a carrier gas adjusted to 1.21 ml/min at 69.0 K Pa, spitless injection of 1 mL, of a hexane solution injector and interface temperature was 2700C, oven temperature programmed was 50-2800C at 3 0C/min. Mass spectra was recorded at 70 eV. Ion source temperature was 2300C.
The identification of the chemical constituents was assigned on the basis of comparison of their retention indices and mass spectra with those given in the literature.[7] Retention indices (RI) were determined with reference to a homologous series of normal alkanes, by using the following Kovats formula. [8]
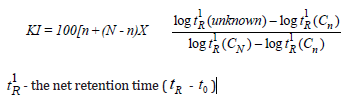
tO - the retention time of solvent (dead time) tR - the retention time of the compound.
CN - number of carbons in longer chain of alkane Cn - number of carbons in shorter chain of alkane
n - is the number of carbon atoms in the smaller alkane N - is the number of carbon atoms in the larger alkane
Results and Discussion
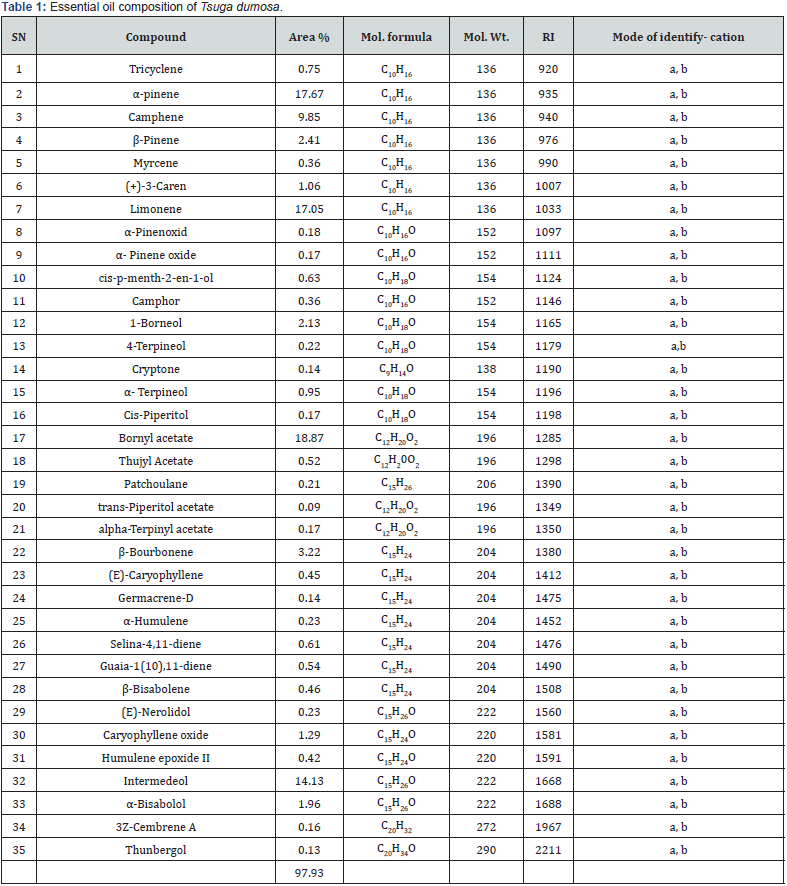
The GC and GC-MS analyses of essential oil of T. dumosa resulted in the identification of 35 compounds (Table 1). The oil yield was (0.09%) by raw material weight. Both, the major as well as minor constituents were identified by their retention indices and comparison of their mass spectra. A total of 35 compounds were identified constituting 98.93 % of the total oil. The main compounds were Bornyl acetate (18.87%), α-pinene (17.67%), Limonene (17.05%), Intermedeol (14.13%), Camphene (9.85%), β-Courbonne (3.22%), β-Pinene (2.41%) and 1- Borneol (2.13%). The main minor compounds were trans-Piperitol acetate (0.09 %), Thunbergol (0.13%), Cryptone (0.14%), Germacrene-D (0.14%), 3Z-Cembrene A (0.16%), α- Pinene oxide (0.17%), Cis-Piperitol (0.17%), alpha-Terpinyl acetate (0.17%) and α-Pinenoxid (0.18%). The presence of 18.87% Bornyl acetate, 17.67% α-pinene and 17.05% Limonene show good source of these natural compound.
Conclusions
Our study concludes that the oil extract has a good number of essential oils. The essential oil from Tsuga dumosa showed a qualitative and quantitative make-up of constituents. A total of 35 compounds were identified constituting 98.93% of the total oil. Clinically, these plant leaves can be a good source of herbal medicine for the treatment of diseases indigenously.
Acknowledgement
The authors are thankful to the Head, Department of Botany, Kumaun University, Nainital for the identification of Plant. The authors are grateful to Prof. Veena Pande, Department of Biotechnology, Sir J. C. Bose Technical Campus, Kumaun University, Nainital (Bhimtal) for the evaluation of antioxidant property of EO and AIRF, Jawaharlal Nehru University, New Delhi for GC-MS. The authors are grateful to Departments of Chemistry, of L.S.M. Govt Post Graduate College, Pithoragarh, and L.B.S. Govt Post Graduate College, Halduchaur (Nainital), Uttarakhand for providing the necessary facilities.
References
- Celiktas OY, Kocabas EEH, Bedir E., Sukan FV, Ozek T, et al. (2007) Antimicrobial activities of methanol extracts and essential oils of Rosmarinus officinalis, depending on location and seasonal variations. Food Chemistry 100(2): 553-559.
- Mabberley DJ (2008) Mabberley’s Plant-Book, 3rd Cambridge University Press, Cambridge, UK.
- Fang JM, Wu SK, Cheng YS (1985) Journal of the Chinese Chemical Society 32(4): 477-480.
- Craft JD, Setzer WN (2017) Leaf essential oil composition of Tsuga canadensis growing wild in North Alabama and Northwest Georgia. American Journal of Essential Oils and Natural Products 5(4): 26-29.
- Puri GS, Meher-Homji VM, Gupta RK, Puri S (1983) Phytogeographical ecology. In: Forest Ecology. 2nd edn Oxford & IBH Publishing Company, India, pp. 115-210.
- Singh JS (2002) The biodiversity crisis: a multifaceted review. Curr Sci 82(25): 638-647.
- Sarikurkcu C (2011) Antioxidant activities of solvent extracts from endemic Cyclamen mirabile Hildebr tubers and leaves. African Journal of Biotechnology 10(5): 831-839.
- Kovats E (1958) Gas-chromatographische Charakterisierung organischer Verbindungen Teil 1: Retentionsindices aliphatischer Halogenide, Alkohole, Aldehyde und Ketone. Helv Chim Acta 41 (7): 1915-1932.






























