Delivery of FITC-Dextran Loaded Calcium Phosphate Nanoparticles Across the Blood-Brain Barrier
Sheetal Budhiraja1*, Rakesh Kumar Sharma2 and Rakesh Kumar Sharma3
1Ramjas College, University of Delhi, India
2SGT University, Gurugram, India
3Department of Chemistry, University of Delhi, India
Submission: July 03, 2023;Published: July 10,2023
*Corresponding author: Sheetal Budhiraja, Department of Chemistry, Ramjas college, University of Delhi, India
*Co -corresponding author: Rakesh Kumar Sharma, Department of Chemistry, University of Delhi, India
How to cite this article: Sheetal Budhiraja*, Rakesh Kumar Sharma and Rakesh Kumar Sharma. Delivery of FITC-Dextran Loaded Calcium Phosphate Nanoparticles Across the Blood-Brain Barrier. Organic & Medicinal Chem IJ. 2023; 12(3): 555836. DOI: 10.19080/OMCIJ.2023.12.555836
Abstract
While the blood-brain barrier’s (BBB) function in the development of treatments for neurodegenerative disorders is becoming more widely acknowledged, there are currently few methods for delivering non-BBB crossing molecules directly to the brain, where they can have an impact. However, the new development of nanomedicines may offer an effective method to apply tailored delivery of active substances to the central nervous system (CNS). Here, polysorbate 80 coated calcium phosphate nanoparticles encapsulating FITC dextran were prepared and characterized for their size and morphology by techniques like Dynamic light scattering (DLS) and transmission electron microscopy (TEM). The synthesized particles were then further examined for their delivery across the blood-brain barrier in-vivo. The organ distribution studies were also done using fluorescence microscopy. This study can further be extended for the delivery of other drugs across the blood brain barrier for various brain disorders.
Keywords: Nanoparticles; Reverse Micelles; FITC Dextran; Calcium Phosphate; Brain Delivery; Brain Disorders; Blood Brain Barrier
Abbreviations: BBB: Blood-Brain Barrier’s; CNS: Central Nervous System; DLS: Dynamic Light Scattering; TEM: Transmission Electron Microscopy; EGFR: Epidermal Growth Factor Receptor; RES: Reticuloendothelial System; PMMA: Poly Methyl Methacrylate; LDL: Low-Density Lipoprotein; PBS: Phosphate Buffer Saline
Introduction
Nanotechnology is one of the most effective and novel areas of research in modern material science. This field of science is developing day by day and is making a valuable impact on the life science, especially biotechnology and biomedical science. Nanoparticles, because of their certain unique properties, exhibit characteristic shape, size, and distribution. Nowadays nanosized delivery vehicles are being used for delivering drugs and genes to the target areas. Among them metal nanoparticles, because of their high surface area, low toxicity, and stability are fast emerging as new drug delivery vehicles. It is reported that metallic nanoparticles can be used as strong antiviral agents, because they inhibit the entry of viruses into the host system. Targeting HSC [1-5] cancer cells frequently used citrate-stabilized gold nanoparticles (human oral squamous cell carcinoma).
To specifically target cancer cells, these gold nanoparticles were coated with anti-EGFR (Epidermal Growth Factor Receptor). Also, gold and platinum nanoparticles are used for preventing inflammation naturally. These divalent cations form constituents of body fluids and are highly biocompatible in the trace amounts required for gene delivery applications. Also, for efficient and secure gene delivery, both viral and non-viral vectors have recently been utilized as gene carriers. Viral vectors provide the benefit of high levels of gene transfection because of the intracellular gene delivery mechanism. Non-viral vectors can also be produced in huge quantities and chemically modified with relative ease. The non-viral vector system, most crucially, is not constrained by the molecular size of the gene to be inserted, as a result of these benefits, non-viral vectors, including polymers, lipids, and functional vectors, have been created and used in gene therapy [6-15]. There are numerous reviews that provide in-depth descriptions of these vectors. DNA delivery vectors have been made from a variety of synthetic and organic polymers. Additionally, naturally occurring polysaccharides like chitosan and proteins like albumin and histones have been employed as nonviral vectors for DNA transport [16,17].
Drug delivery to the central nervous system (CNS) poses significant challenges for both clinical and basic neuroscientists. The blood-brain barrier (BBB) is a crucial protective mechanism in the CNS that hinders the transfer of numerous drugs from the blood to the brain. This barrier is formed by the brain capillary endothelial wall, which exhibits minimal permeability in vivo. A major obstacle in targeted drug delivery is the rapid recognition and uptake of injected carriers by the reticuloendothelial system (RES), particularly by macrophages in the liver and spleen.
In the case of nanoparticles, an in vivo study demonstrated that modifying the surface properties of poly (methyl methacrylate) (PMMA) nanoparticles, a model carrier, through coating with nonionic surfactants like poloxamers, poloxamines, and polysorbates [18-20] could alter their distribution in the body. Increasing the hydrophilicity of the particles prolonged their circulation time in the bloodstream, resulting in lower concentrations in RES organs and tissues such as the liver, spleen, lungs, bone marrow, and lymph nodes, while showing significantly higher levels in non- RES organs like the heart, brain, and intestine. Notably, coating the PMMA particles with polysorbate 80, the most effective substance for brain targeting in vivo, enhanced nanoparticle uptake by a factor of 5 compared to the control group of uncoated nanoparticles, as demonstrated by Borchard et al. [21-24].
Apart from polymeric and biological nanoparticles, inorganic nanoparticles such as iron oxides, silica, gold, and calcium phosphate have gained considerable attention due to their mechanical stability, ease of preparation, tunable size, and versatile surface chemistry. Among these, calcium phosphate nanoparticles offer distinct advantages, particularly their high biocompatibility and biodegradability [25-28]. Calcium phosphate, known as a biomaterial in bulk form or as a coating, has found numerous applications in vitro and in vivo. It is naturally present in biomineralized hard tissues as nanoplatelets embedded in a collagen matrix, constituting the mineral component of bones and teeth in the form of calcium-deficient hydroxyapatite with various ionic substitutions. Unlike many artificial nanoparticle materials such as iron oxide, polymers, silica, or nanodiamonds, calcium phosphate is ubiquitous in the body, present in bone, teeth, saliva, and blood, which contributes to its high biocompatibility and nontoxic nature.
In contrast to other inorganic nanoparticles like gold, magnetite, silica [29,30] and certain polymers, calcium phosphate nanoparticles readily dissolve in the acidic environment within endo lysosomes or phagosomes after cellular uptake while remaining stable at neutral pH, such as in the blood. This characteristic renders them highly biodegradable compared to bio persistent materials like gold, nanodiamonds, magnetite, or carbon nanotubes. Furthermore, calcium phosphate nanoparticles [31] fulfill crucial requirements for an effective delivery system, including the ability to incorporate drugs or biomolecules both internally and on the surface through physical or covalent binding. They can retain these biomolecules until reaching the target site, where they dissolve, and subsequently undergo biodegradation into harmless compounds, namely calcium and phosphate ions. In this study, calcium phosphate nanoparticles encapsulating FITC dextran were prepared and coated with polysorbate 80. The size and morphology of the particles were characterized using techniques such as Dynamic light scattering (DLS) and transmission electron microscopy (TEM). Subsequently, the synthesized particles were evaluated for their ability to cross the blood-brain barrier in vivo.
Materials and Methods
Experimental
Preparation of Calcium Phosphate Nanoparticles Encapsulating FITC Dextran
According to a previously described method (19), the calcium phosphate nanoparticles were created in the aqueous core of AOT/Hexane reverse micellar droplets. In a nutshell, 0.1 solutions in hexane were made. By continuously stirring for 12 hours, 70 μl of 1.36 M calcium chloride aqueous solution and 125 μl of FITC dextran (10 mg/ml) aqueous solution were added to 25 ml of AOT in hexane to create microemulsion A. Another 25ml of AOT solution was used to dissolve 50 ml of 0.2 M Tris-HCl buffer (pH 7.4), 70 ml of 0.35 M NaH2PO4 in water, and 125 ml of FITC Dextran (10 mg/ml) in water to create microemulsion B. This process took place over the course of 12 hours with constant stirring. To adjust WO, or the molar ratio of water to AOT, at 10, further water was added before swirling both microemulsions, bringing the total amount of added water to 450 μl. They were both optically clear microemulsions. Then, at 4°C in a cold chamber, microemulsion B was continuously stirred in while being introduced to microemulsion A at an exceedingly slow rate (20 drops per minute). The precipitation was permitted to occur at 4°C to have the smallest possible particle size because temperature has a significant impact on particle size. The resulting mixture was then churned once more for a further 12 hours in a cold environment. Translucency growth suggested the development of nanoparticles in the aqueous core of the microemulsion droplets. The resultant solution was completely free of hexane, and by vortexing, the nanoparticles holding the solid mass of AOT were dissolved in 10 ml of 99.9% pure ethanol. The solutions were then centrifuged in a cold centrifuge for 30 minutes at 8000 rpm and 4°C (Sigma 3K18). Three times pure ethanol was used to wash the pelleted nanoparticles. In order to achieve a clean dispersion, the pelleted nanoparticles were lastly vortexed in double-distilled water at 4°C. Using a 12kD dialysis membrane bag, the dispersed nanoparticles were dialyzed overnight in a cool room. After that, the fluid was lyophilized to create a dry powder for later usage. Figure 1 shows a schematic illustration of the full preparation process for nanoparticles (Figure 1).
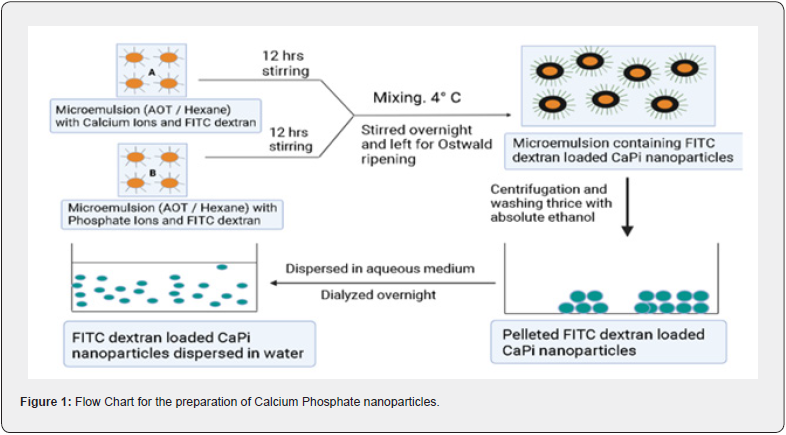
Surface Modification of Calcium Phosphate Nanoparticles Using Polysorbate 80
Polysorbate 80 is the trade name for Sorbiton-mono-9- octadecenolatepoly(oxy-1,2-ethanediyl) derivative. Surfactants such as polysorbate 80 were previously shown to significantly alter the body’s distribution of nanoparticles, upon intravenous injection of the coated nanoparticles in rats and mice. Among the substances that led to a significant prolonged blood circulation time were poloxamer 338, polyoxamine 908 and polysorbate 80. These substances significantly increased the whole brain concentration after intravenous injection of nanoparticles. Therefore polysorbate 80 was chosen as the lead substance for the coating of nanoparticles in our studies. The lyophilized powder of calcium phosphate nanoparticles was dispersed in buffer and 100 μl of aqueous solution of polysorbate 80 was added to have 1%w/w coating. The formulation was then used for further experiments.
Biodistribution Studies of Polysorbate 80 Coated/Uncoated Calcium Phosphate Nanoparticles
Biodistribution of polysorbate 80 coated/uncoated calcium phosphate nanoparticles was studied in Swiss Albino Strain ‘A’ mice (2-3monthsold) weighing ~25-30g. The lyophilized powder of FITC Dwereran loaded calcium phosphate nanoparticles was dispersed in double distilled water and coated with polysorbate 80 (1%w/w). The solutions were thoroughly vortexed and sonicated. An aliquot of 100 μl of coated/uncoated calcium phosphate nanoparticles containing ~18 μg if FITC dextran was administered through the tail vein of each mouse. The animals were sacrificed by cervical dislocation at different time intervals and various organs were removed, washed in cold sterile phosphate buffer saline (PBS), and dried in paper folds. The tissues were stored in reweighed vials. The tissues under observation were the brain (the target organ), heart and other organs belonging to RES i.e., lungs and liver. The tissues were homogenized in phosphate buffer saline under ice cooled conditions. 10% homogenate was prepared for each organ. The homogenates obtained were then centrifuged for 10 minutes at a speed of 10,000 rpm and the supernatant was separated for further evaluation. 200 μl of each homogenate was taken and added to the titer plate. The fluorescence was read at an emission wavelength of 490nm and excitation wavelength at 520nm using spectrofluorometer (Varian, USA). In the control experiments 18 μg of naked FITC dextran was administered to the mice and the tissues were taken out and processed as described above.
Characterization
Quasi Elastic Light Scattering Studies
The synthesized particles were formed in the aqueous core of the AOT microemulsion in hexane. The size of the particles obtained in reverse micelles was determined by using dynamic light scattering experiment using a Brookhaven instrument with BI200SM goniometer and an air-cooled argon ion laser as a light source.
Transmission Electron Microscopic Studies
The TEM photographs of the particles were taken using an electron microscope (JEOL JEM 2000 Ex200 model). In order to prepare the sample; 1mg of sample was dispersed in 5 ml of distilled water by sonication and a drop of clear dispersion was put on the formvar coated grid and dried in a desiccator. The dried grid was then examined under electron microscope.
Results and Discussion
Determination of Size and Morphology of Nanoparticles
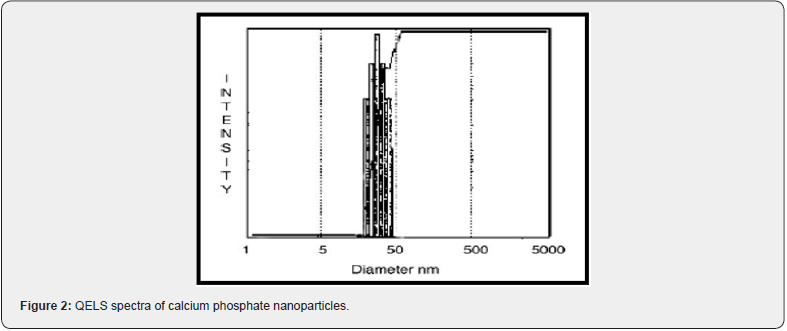
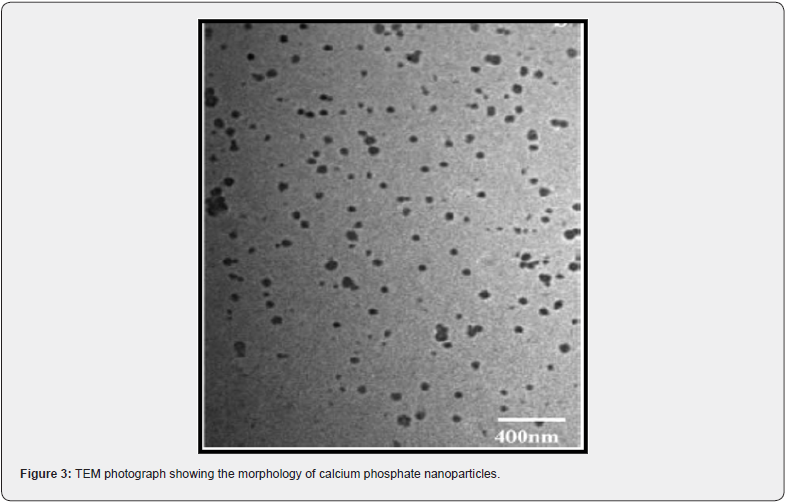
The mean size distribution (as seen from Figure 2) of the calcium phosphate nanoparticles was found to be in the range of 30-40 nm. The particles show narrow size distribution and unimodal pattern. The transmission electron micrographs of calcium phosphate nanoparticles (Figure 3) revealed the formation of dense particles with spherical morphology as well as their narrow size distribution. The size and size distribution of these nanogels were found to be the same before and after lyophilization. It thus confirms that aggregation of these particles is not occurring (Figure 3,4).
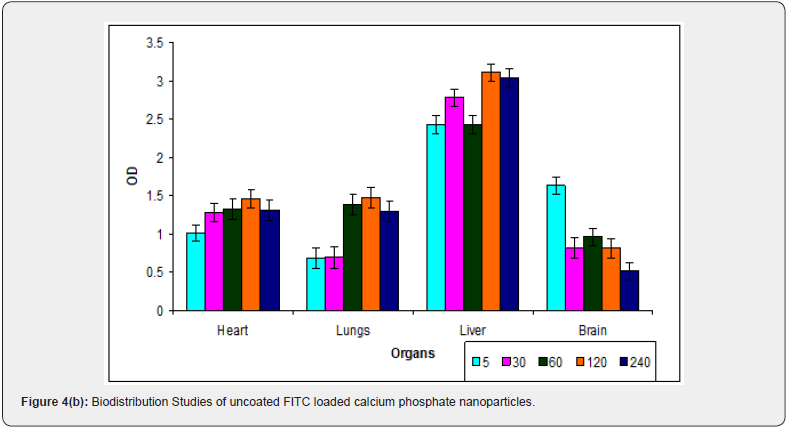
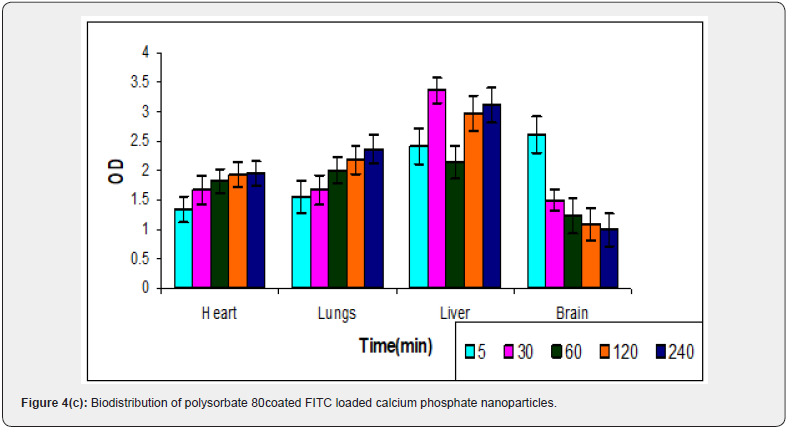
Biodistribution of Calcium Phosphate Nanoparticles Coated/Uncoated with Polysorbate 80
Fluorescence spectroscopy works on the principle of light emission. The emission of light can reveal properties of biological molecules quite different from the properties revealed by light absorption. Fluorescence generally is much more sensitive to the environment of the chromophore than light absorption. Fluorescence technique was used in the experiments to assess the uptake of nanoparticles by brain tissue. Fluorescein Isothiocyanato Dextran was encapsulated inside the calcium phosphate nanoparticles. It is a fluorescent molecule with absorbance at 490 nm and emission at 520nm. The fluorescence intensity was measured in each organ.
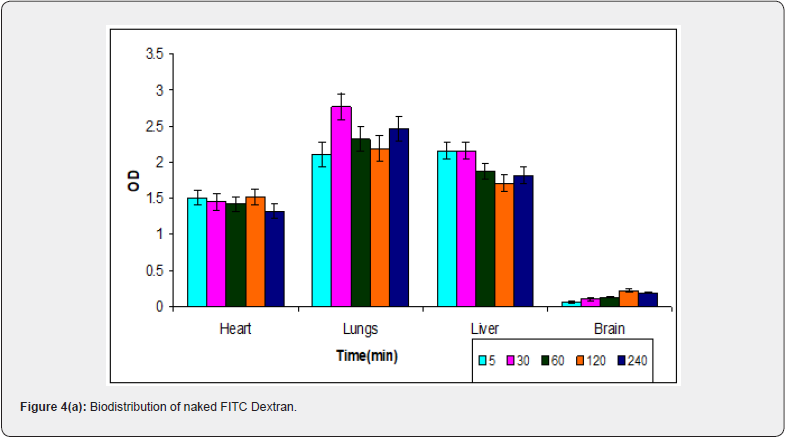
Figure 4(a-c) shows the fluorescence intensity with time in various organs such as brain, lungs, liver, and heart in naked FITC Dextran/uncoated calcium phosphate nanoparticles/ poysorbate 80 coated calcium phosphate nanoparticles loaded with FITC dextran. When solution of naked FITC dextran was administered to mice biodistribution pattern was different from that obtained when it is encapsulated in the nanoparticles. Higher concentrations of naked FITC dextran were found in heart as compared to what was observed in uncoated calcium phosphate nanoparticles. The reason may be that FITC dextran, being larger molecule is interacting strongly with the blood and serum proteins whereas when it is entrapped in the nanoparticles, it is well protected from the outer environment and hence shows lesser interaction. Lesser fluorescence intensity was seen in lungs in uncoated nanoparticles as compared to FITC dextran alone. This may be attributed to the particle size of calcium phosphate nanoparticles. Since the particles are smaller than 100nm in diameter, they are easily filtered out of the lungs whereas FITC dextran being a larger sized molecule gets trapped by the lungs. The overall uptake of uncoated nanoparticles by liver was relatively higher than that of naked FITC dextran. The uptake of naked FITC dextran by liver was high initially followed by a decrease afterwards.
f4
Slightly higher concentrations were observed in the organs belonging to RES i.e., lungs and liver in the case of nanoparticles coated with polysorbate 80 as compared to uncoated calcium phosphate nanoparticles. Perhaps the surface hydrophobicity of coated nanoparticles is responsible for greater accumulation in organs belonging to RES. When these particles are coated with polysorbate 80, the hydrophobic tail of the surfactant protrudes towards the outer surface of the particles. As a result, the nanoparticles acquire partial surface hydrophobicity and, thus, are expected to become susceptible to enhanced RES uptake. Additionally, because of partial surface hydrophobicity, these particles may have higher uptake by brain endothelial cells also, thus, enabling them to cross BBB. This has been clearly manifested in figure 5 in which polysorbate 80 coated FITC dextran loaded nanoparticles have shown more uptake by brain compared to uncoated nanoparticles. The highest level of uptake has been observed within 5 minutes post injection of mice. At five minutes post injection the fluorescence intensity in case of coated nanoparticles was found to be 2.61 as compared to 1.63 in uncoated nanoparticles. Significantly lower concentrations of naked FITC dextran were observed in brain as compared to both coated/uncoated nanoparticle preparations. However, the concentration of the particles was found to be decreasing with time but there was relatively higher retention of the particles at four hours post injection.
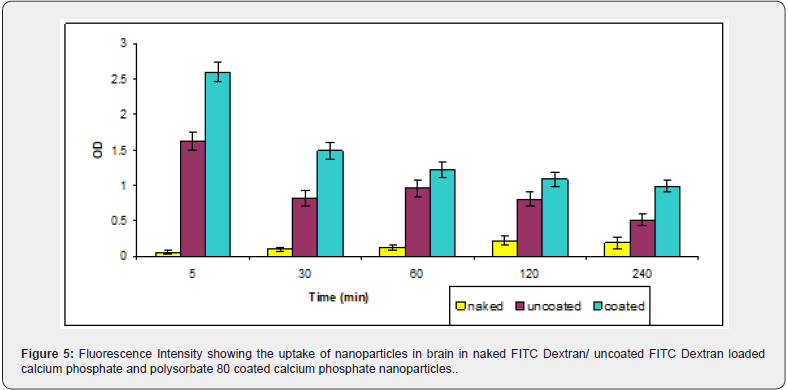
This can be well correlated to the data obtained by Schroder et al where they concluded that a peptide Dalargin could be transported across the BBB following intravenous injection after binding of the two nanoparticles and coating these particles with polysorbate 80. The preparation was able to induce a significant analgesic effect about 30 to 45 minutes after intravenous injection into mice at a dose of 7.5mg/kg. The reason for greater uptake of polysorbate 80 coated calcium phosphate nanoparticles by coating with poysorbate80 may lead to the adsorption of apolipoprotein E from blood plasma onto the nanoparticles surface. The particles then seem to mimic low-density lipoprotein (LDL) particles and could interact with the LDL receptor leading to their uptake by the brain endothelial cells. After this FITC dextran may be released in these cells and diffuse into the brain interior or the particles may be transcytosed. Other processes such as tight junction modulation or P-glycoprotein inhibition may also occur. Moreover, these mechanisms may run in parallel or may be cooperative thus enabling a drug delivery to the brain.
Conclusion
The interest in developing targeted systems, able to reach the CNS, represented one of major sections of neuroscience, over the last 20 years. The pathologies affecting the “Brain” are surely the most difficult to be treated and the least favorable in terms of rescue of the integrity of the neuronal structures. When nanoparticles coated with polysorbate 80 were compared to uncoated calcium phosphate nanoparticles, larger quantities were accumulated in RES organs, namely the lungs and liver. The reason for uptake by the RES organs is due to the hydrophobic surface of calcium phosphate nanoparticles. These particles may also be more readily absorbed by brain endothelial cells, allowing them to pass the blood-brain barrier (BBB). Phagocytosis could be one of the prominent reasons for efficient uptake in the brain. Furthermore, the coating of calcium phosphate by surfactants like polysorbate 80 plays an important role for body distribution and the time of circulation of the particles. Also, as opposed to both coated and uncoated nanoparticle preparations, smaller amounts of naked FITC dextran were found in brain and the result defines two important requirements to take into account in the design of adequate brain delivery systems, long circulating properties of the carrier and appropriate surface characteristics to permit interactions with endothelial cells. Thus, the application of nanotechnology to CNS diseases (namely Nano-Neuroscience) is surely representing one of the most innovative strategies to improve the current treatments and, possibly, to find newer and, most of all, more efficacious therapies.
References
- Khandel P, Yadaw RK, Soni DK, Kanwar L, Shahi SK (2018) Biogenesis of metal nanoparticles and their pharmacological applications: present status and application prospects. Journal of Nanostructure in Chemistry 8(3): 217-254.
- Huang X, Qian W, El‐Sayed IH, El‐Sayed MA (2007) The potential use of the enhanced nonlinear properties of gold nanospheres in photothermal cancer therapy. Lasers in Surg Med 39(9): 747-753.
- Wang X, Shao N, Zhang Q, Cheng Y (2014) Mitochondrial targeting dendrimer allows efficient and safe gene delivery. Journal of Materials Chemistry B 2(17): 2546-2553.
- Guan L, Huang S, Chen Z, Li Y, Liu K, et al. (2015) Low cytotoxicity fluorescent PAMAM dendrimer as gene carriers for monitoring the delivery of siRNA. Journal of Nanoparticle Research 17(9):1-9.
- Liang X, Li X, Chang J, Duan Y, Li Z (2013) Properties, and evaluation of quaternized chitosan/lipid cation polymeric liposomes for cancer-targeted gene delivery. Langmuir 29(27): 8683-8693.
- Ding X, Wang W, Wang Y, Bao X, Wang Y, et al. (2014) Versatile reticular polyethyleneimine derivative-mediated targeted drug and gene co-delivery for tumor therapy. Mol pharm 11(10): 3307-3321.
- Moran LA, Horton RA, Scrimgeour KG, Perry MD (2014) Principles of Biochemistry. Murray, RK, Granner, DK, Mayes, PA & Rodwell, VW 2014. Harper’s Illustrated Biochemistry. McGraw-Hill, New York.
- Wu P, Chen H, Jin R, Weng T, Ho JK, et al. (2018) Non-viral gene delivery systems for tissue repair and regeneration. J Transl Med 16(1): 1-20.
- Qadir A, Gao Y, Suryaji P, Tian Y, Lin X, et al. (2019) Non-viral delivery system and targeted bone disease therapy. Int J Mol Sci 29 20(3): 565.
- Gao YG, Alam U, Ding AX, Tang Q, Tan ZL, et al. (2018) [12] ane N3-based lipid with naphthylamide moiety for enhanced gene transfection efficiency. Bioorg chem 79: 334-340.
- Bates K, Kostarelos K (2013) Carbon nanotubes as vectors for gene therapy: past achievements, present challenges, and future goals. Adv Drug Deliv Rev 65(15): 2023-2033.
- Liu L, Zhang H, Meng X, Yin J, Li D, et al. (201) Di nuclear metal (II) complexes of polybenzimidazole ligands as carriers for DNA delivery. Biomaterials 31(6): 1380-1391.
- Li H, Yang Y, Hong W, Huang M, Wu M, et al. (2020) Applications of genome editing technology in the targeted therapy of human diseases: mechanisms, advances, and prospects. Signal transduc Targeted Ther 5(1): 1-23.
- Chirila TV, Rakoczy PE, Garrett KL, Lou X, Constable IJ (2002) The use of synthetic polymers for delivery of therapeutic antisense oligodeoxynucleotides. Biomaterials 23(2): 321-342.
- Gebhart CL, Kabanov AV (2001) Evaluation of polyplexes as gene transfer agents. J Control Release 73(2-3): 401-416.
- Niidome T, Huang L (2002) Gene therapy progress and prospects: nonviral vectors. Gene Ther 9(24): 1647-1652.
- Mao HQ, Roy K, Troung-Le VL, Janes KA, Lin KY, et al. (2001) Chitosan-DNA nanoparticles as gene carriers: synthesis, characterization, and transfection efficiency. J control release 70(3): 399-421.
- Balicki D, Reisfeld RA, Pertl U, Beutler E, Lode HN (2000) Histone H2A-mediated transient cytokine gene delivery induces efficient antitumor responses in murine neuroblastoma. Proc Natl Acad Sci 97(21): 11500-11504.
- Bisht S, Bhakta G, Mitra S, Maitra A (2005) pDNA loaded calcium phosphate nanoparticles: highly efficient non-viral vector for gene delivery Int J Pharm 288(1): 157-168.
- Zhou X, Huang L (1992) Targeted delivery of DNA by liposomes and polymers. Journal of controlled release 19(1-3): 269-274.
- Banerjee T, Mitra S, Singh AK, Sharma RK, Maitra A (2002) Preparation, characterization and biodistribution of ultrafine chitosan nanoparticles. Int J pharm 243(1-2): 93-105.
- Banerjee T, Mitra S, Singh AK, Sharma RK, Maitra A. Preparation, characterization and biodistribution of ultrafine chitosan nanoparticles. Inter J pharm 243(1-2): 93-105.
- Schröder U, Sabel BA (1996) Nanoparticles, a drug carrier system to pass the blood-brain barrier, permit central analgesic effects of iv dalargin injections. Brain Res 710(1-2): 121-124.
- Ramge P, Unger RE, Oltrogge JB, Zenker D, Begley D, et al. (2000) Polysorbate‐80 coating enhances uptake of polybutylcyanoacrylate (PBCA)‐nanoparticles by human and bovine primary brain capillary endothelial cells. Eur J Neurosci 12(6): 1931-1940.
- Chong Qiu, Yanyan Wu, Qiuyan Guo, Qiaoli Shi, Junzhe Zhang, et al. (2022) Preparation and application of calcium phosphate nanocarriers in drug delivery. Mater Today Bio 17: 10051.
- Ginebra MP, Canal C, Espanol M, Pastorino D, Montufar EB (2012) Calcium phosphate cement as drug delivery materials. Adv Drug Deliv Rev 64 (12): 1090-1110.
- Huang D, He B, Mi P (2019) Calcium phosphate nanocarriers for drug delivery to tumors: imaging, therapy and theranostics. Biomater Sci 7(10): 3942-3960.
- Sokolova V, Epple M (2021) Biological and medical applications of calcium phosphate nanoparticles. Chemistry A European Journal 27(27): 7471-7488.
- Albinali KE, Zagho MM, Deng Y, Elzatahry A (2019) A perspective on magnetic core–shell carriers for responsive and targeted drug delivery systems. Int J Nanomedicine 14: 1707-1723.
- Roggers R, Kanvinde S, Boonsith S, Oupický D (2014) The Practicality of Mesoporous Silica Nanoparticles as Drug Delivery Devices and Progress Toward This Goal. AAPS Pharm SciTech 15(5): 1163-1171.
- Ramachandran R, Paul W, Sharma CP (2009) Synthesis, and characterization of PEGylated calcium phosphate nanoparticles for oral insulin delivery. J Biomed Mater Res Part B: Appl Biomater Sect B 88(1): 41-48.






























