Synthesis of Pyrimidinones with (E/Z)-2-Chlorobutene-2-Enyl Group
Hakobyan MR, Sumbatyan AS and Harutyunyan AA*
Scientific and Technological Center of Organic and Pharmaceutical Chemistry of the National Academy of Sciences of the Republic of Armenia, Armenia
Submission: April 8, 2023;Published: May 10, 2023
*Corresponding author: Harutyunyan AA, Scientific and Technological Center of Organic and Pharmaceutical Chemistry of the National Academy of Sciences of the Republic of Armenia, 0014 Armenia, 0014 Azatutyan Ave 26, Armenia
How to cite this article: Hakobyan MR, Sumbatyan AS, Harutyunyan AA. Synthesis of Pyrimidinones with (E/Z)-2-Chlorobutene-2-Enyl Group. Organic & Medicinal Chem IJ. 2023; 12(2): 555833. DOI: 10.19080/OMCIJ.2023.12.555833
Abstract
Substituted pyrimidines are considered as one of the important building blocks for designing new drugs. The functionalization of the pyrimidine ring with reactive groups creates opportunities for inhibition of intracellular metabolism by ligand-receptor interaction, including the type of irreversible inhibition.
Keywords: Pyrimidines; 5-(2-Chlorobutene-2-enyl) group; Doking; DNA-methylation inhibitors
Introduction
It is known that the affinity of natural pyrimidines for biochemical processes in all living systems creates a rational concept for creating drugs based on molecular target-ligand interactions of substituted pyrimidines. In light of this, it seems important that a reactive chemical group be present in the structure of a biologically active compound, due to which an effective interaction between the receptor and the ligand is possible. In this regard, our attention was drawn to the (E/Z)-2-chlorobutene-2-enyl group, information on the use of which in works on the synthesis of biologically active compounds is absent.
Obviously, the presence of a reactive 2-chlorobutene-2-enyl group in the pyrimidine ring creates opportunities for the interaction of the ligand molecule with various nucleophilic groups of cellular target macromolecules. In view of the foregoing, in continuation of the research on the search for biologically active pyrimidines [1,2] (Figure 1 & 2).

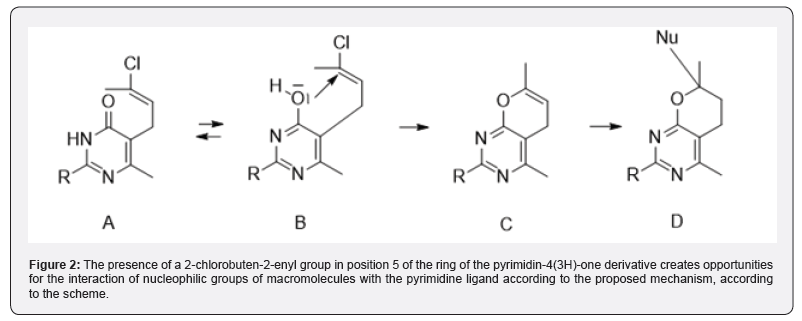
Pyrimidin-4-one A, in the form of pyrimidin-4-ol tautomer B, attacks the C2 atom of the vinyl group to eliminate the chlorine atom and form the 6-methyl-,4-dihydro-2H-pyran derivative C. The latter reacts with suitable nucleophilic groups, for example, with hydroxyl groups of macromolecules according to the wellknown reaction of protection of hydroxyl groups by interaction with 2,3-dihydropyran, with the formation of compound D. The synthesized compounds were docked and their effect on the level of tumor DNA methylation was studied under in vitro conditions in a 180-mouse sarcoma model.
References
- Harutyunyan AA, Gukasyan GT, Grigoryan AG, Stepanyan HM (2021) Antibacterial and mao-inhibiting properties of new styryl derivatives of pyrimidines, quinazolines and bis- quinazolines. Biol J Armenia 73(2): 26-30.
- Safaryan MS, Sumbatyan AS, Israelyan SG, Harutyunyan AA (2022) N1- and N1, N3- dialkylated uracils and 5-alogenuracils. Collected Papers XLIX International Scientific- Practical conference. Moscow, Russia, p. 88-89.






























