Physicochemical Studies of Homoeopathic Formulations of Ammonium Aceticum by using Volumetric, Acoustic and Viscometric Measurements at Different Temperatures
Anil Kumar Nain1*, Neha Chaudhary1, Anil Khurana2, Raj Kumar Manchanda2 and Debadatta Nayak2
1Department of Chemistry, Dyal Singh College, University of Delhi, India
2Central Council for Research in Homeopathy, Ministry of AYUSH, India
Submission: February 13, 2023;Published: March 01, 2023
*Corresponding author: Anil Kumar Nain, Department of Chemistry, Dyal Singh College, University of Delhi, New Delhi – 110 003, India
How to cite this article: Anil Kumar Nain, Neha Chaudhary, Anil Khurana, Raj Kumar Manchanda and Debadatta Nayak. Physicochemical Studies of Homoeopathic Formulations of Ammonium Aceticum by using Volumetric, Acoustic and Viscometric Measurements at Different Temperatures. Organic & Medicinal Chem IJ. 2023; 12(1): 555830. DOI: 10.19080/OMCIJ.2023.12.555830
Abstract
Ocean covers about one-third of the earth and is considered as a vital natural resource. Seaweeds accompany a large areas of the ocean coastal area, which are considered as among the primary producers. Many seaweeds have food values and so many indigenous populations relay on them as their nutrient source. Besides, seaweeds are also rich in many bioactive compounds having pharmacological significance. Seaweeds contain higher number of proteins, vitamins, minerals, essential fatty acids, polysaccharides and so on. Gracilariopsis lemaniformis is a red sea alga and is also rich in bioactive compounds having medicinal values. In this review article, we focused on the pharmacological importance of this species based on recent updates of literature survey and we discussed here about its antimicrobial, antitumor, antidiabetic and antioxidant properties, which would demand its therapeutic relevance for future drug development.
Keywords: Covid-19; Sars-Cov-2; Copper Metal; Cancer; Viral Infections
Introduction
Homoeopathy is one of the traditional and most widespread alternative systems of medicine after conventional therapy. The efficacy of homoeopathic medicines is well supported by research evidence; however, there are controversies regarding implausibility in biological activity of homoeopathic medicines in which the source drug is diluted beyond Avagadro’s limit, i.e., the highly diluted medicine formulation must be identical to the solvent. There have been a few research studies pertaining to explore the presence of drug in extremely diluted formulations [1-7] and its mechanism of action, but the question still remains answered acceptably. The measurements of physicochemical properties and derived parameters of aqueous and mixed-aqueous solutions of amino acids, carbohydrates, drugs, etc. have been supportive in characterizing the solute-solute and solute-solvent interactions, which are subsequently useful in understanding of solute-solvation/hydration behavior of solute and preferential solvation of solute by the solvent [8-15]. Since homoeopathic formulations are extremely dilute solutions, their physicochemical properties, like density, ultrasonic speed, viscosity, refractive index, etc. can be measured easily as a function of concentration/potency and temperature.
The physicochemical properties calculated from these experimental data can deliver valuable information on the understanding of physicochemical behavior and mechanism of drug action. Recently, there have been few physicochemical studies on extremely diluted solutions of inorganic salts [16-21] and homoeopathic medicines [2,3,22-28] by using physicochemical methods. These studies provided interesting and convincing information on the behavior of these extremely diluted solutions.
To the best of our information, very few physicochemical studies on homoeopathic medicines using volumetric, acoustic, viscometric and optical methods have been reported in the literature [2,3,22]. These considerations led us to undertake the present study on the physicochemical behavior of extremely diluted homoeopathic formulations of ammonium aceticum. Ammonium aceticum dilution is helpful in the treatment of many types of conditions, viz., heavy feeling of the head and scraping sensation of the throat, stomach and reducing sweat. It also helps in reducing urination urge.
The homoeopathic medicine formulations are generally obtained through the combination of two processes: a dilution of 1:100 in mass followed by succussion. In the present study, the densities, ρ, ultrasonic speeds, u and viscosities, η of pure ethanol control (91% ethanol in water) and 33 formulations of ammonium aceticum with potencies ranging from 1C to 200C (with intervals of 2C till 30C, and then with intervals of 10C till 200C) at 293.15, 298.15, 303.15, 308.15, 313.15 and 318.15 K and atmospheric pressure. From these experimental data, the isentropic compressibilities κs, intermolecular free length, Lf , acoustic impedance, Z, relative association, RA, deviations in isentropic compressibility, deviations in intermolecular free length, deviations in acoustic impedance, and deviations in viscosity, have been calculated. The results have been qualitatively discussed in terms of interactions/physicochemical behavior of ammonium aceticum in these homoeopathic formulations.
Experimental
Ethanol control and homoeopathic formulations of various potencies of ammonium aceticum used in the study were procured from Dr. Wilmer Schwabe India Pvt. Limited, India. The densities and ultrasonic speeds of the samples were measured by using high precision digital vibrating tube Density and Sound Analyzer (DSA 5000M, Anton Paar, Austria). This two-in-one instrument is equipped with both density and ultrasonic cells, with reproducibility of ±1 × 10−3 kg·m−3 and ±1 × 10−2 m·s−1 for density and ultrasonic speed, respectively. The temperature for both cells was kept constant by using built in peltier thermostat within ±0.01 K. The calibration of instrument was done by using triply-distilled, degassed water and with dry air at atmospheric pressure and 293.15 K [11]. The operating working frequency used for ultrasonic speed measurements is 3 MHz. The principle used in density measurement is based upon oscillating U-tube principle while the speed of sound is measured using a propagation time technique. The standard uncertainties related to the measurements of density, ultrasonic speed and temperature were found within ±0.05 kg·m−3, ±0.5 m·s−1 and ±0.01 K, respectively.
The viscosity measurements were done by using microviscometer (Lovis 2000M, Anton Paar, Austria) at temperatures, (293.15 − 318.15) K, and atmospheric pressure p = 101 kPa. The temperature was controlled to ±0.02 K by an automatic build in Peltier technique . The rolling ball principle was used in the measurement of viscosity, having a calibrated glass capillary with a steel ball as supplied by manufacturer. The calibration of capillary was accomplished by using viscosity standard fluids. The relative standard uncertainty in viscosity measurements was found to be within ±0.5 %.
Results
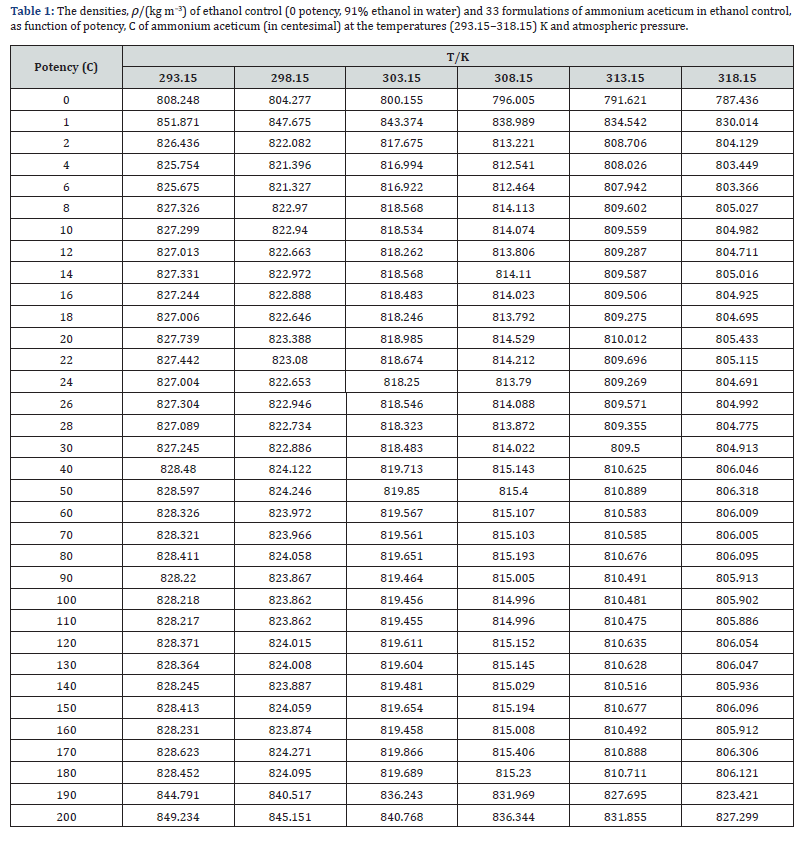
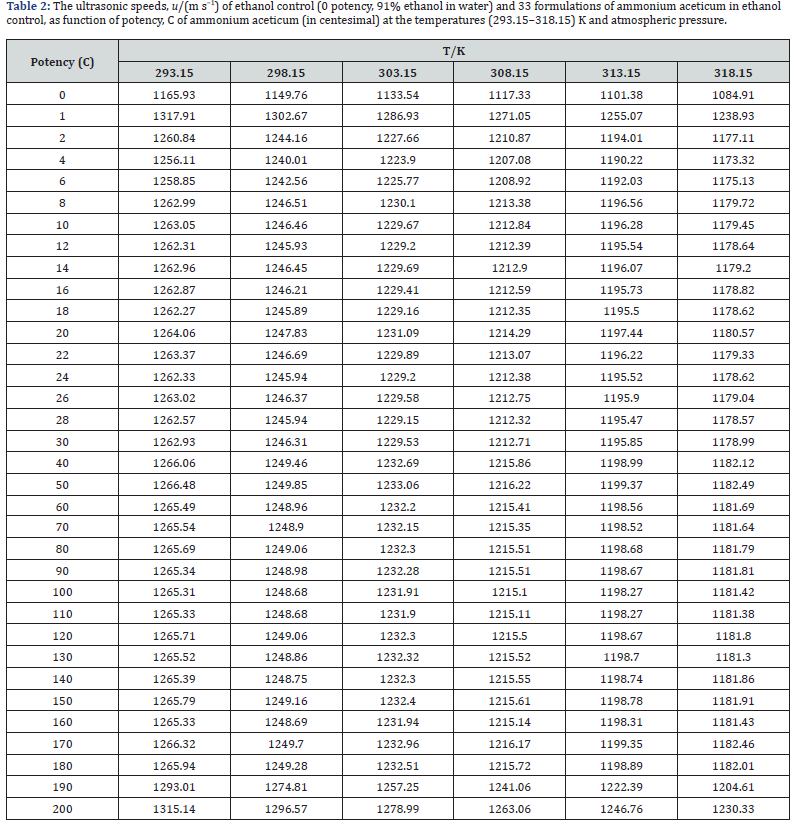
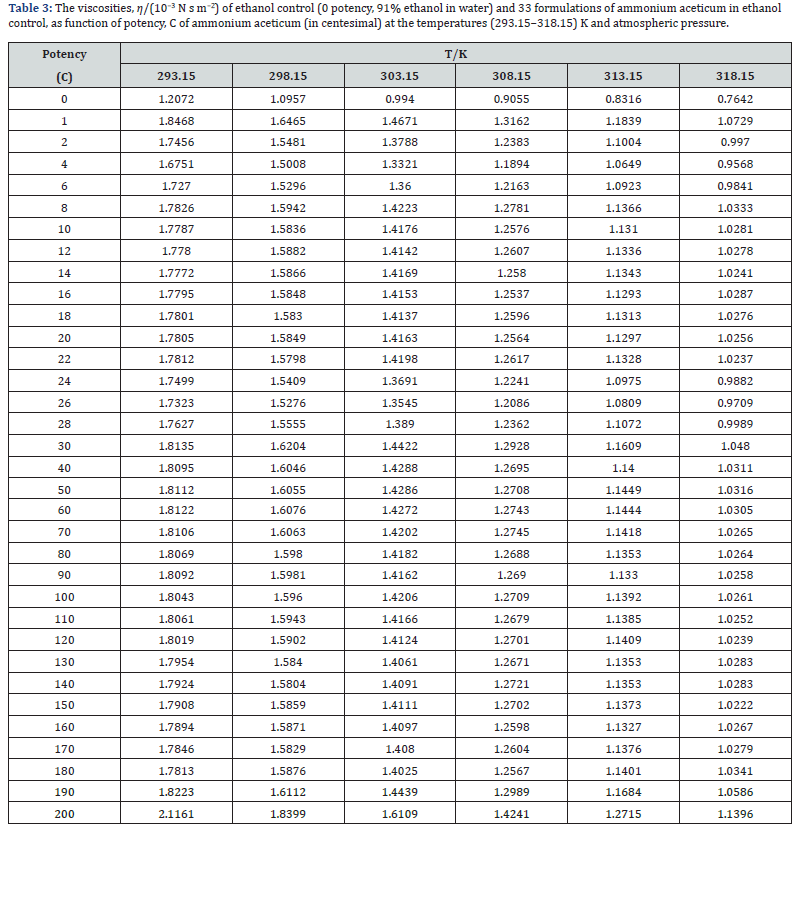
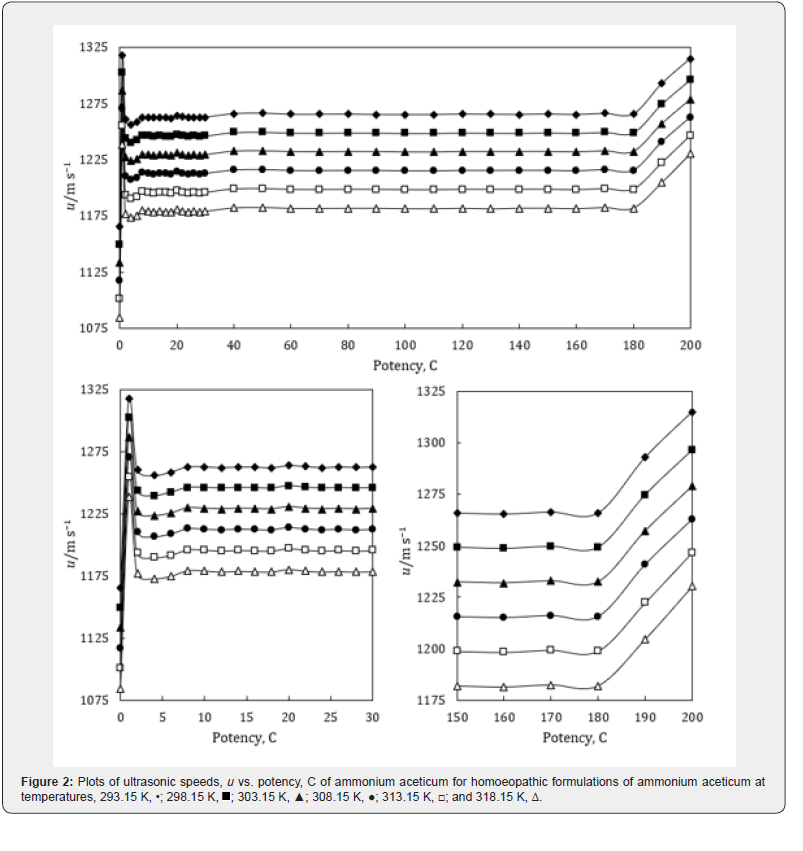
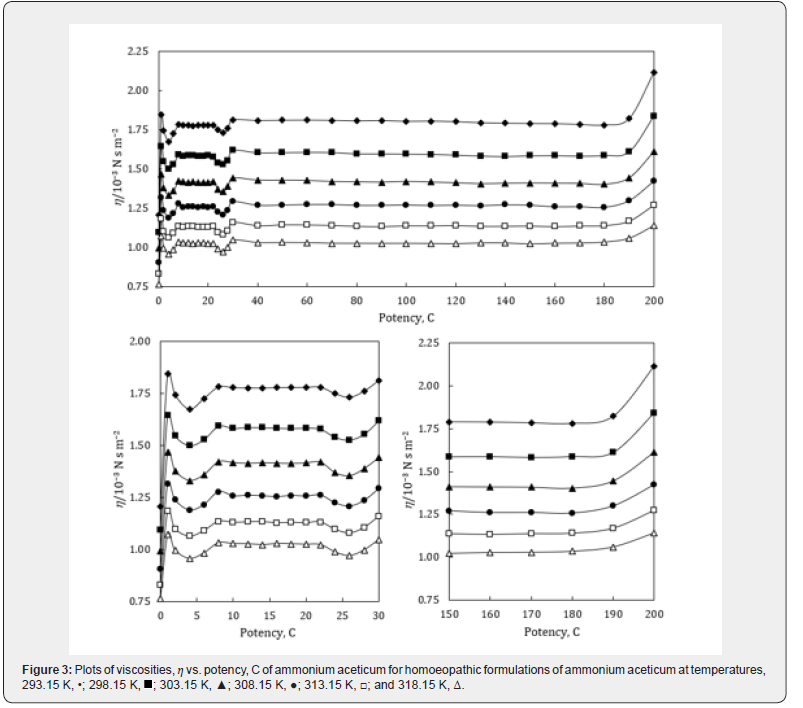
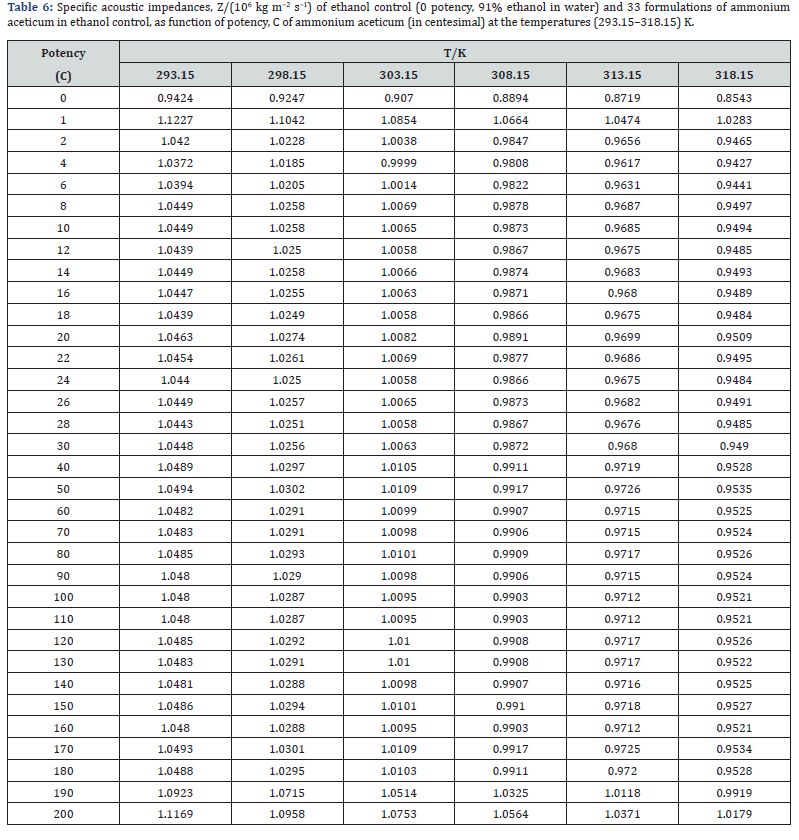
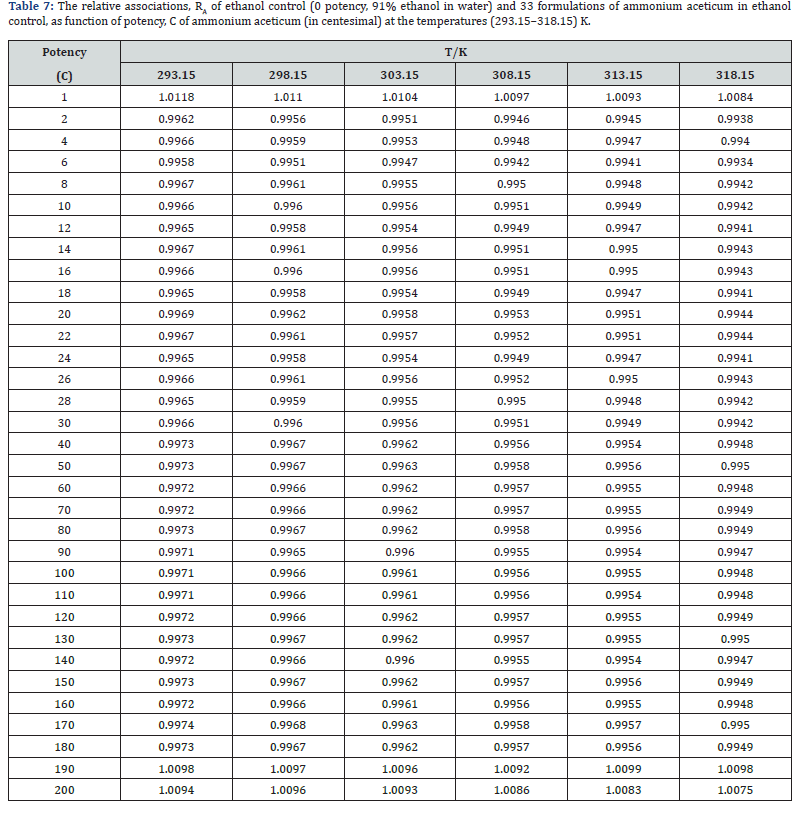
The experimental values of densities, ρ, ultrasonic speeds, u and viscosities, η of ammonium aceticum formulations as function of potency (in centesimal) at different temperatures are listed in Tables 1-3, and are presented graphically in Figures 1-3, respectively. The values of the isentropic compressibility, intermolecular free length, acoustic impedance, Z and relative association, RA have been calculated by using the following relations
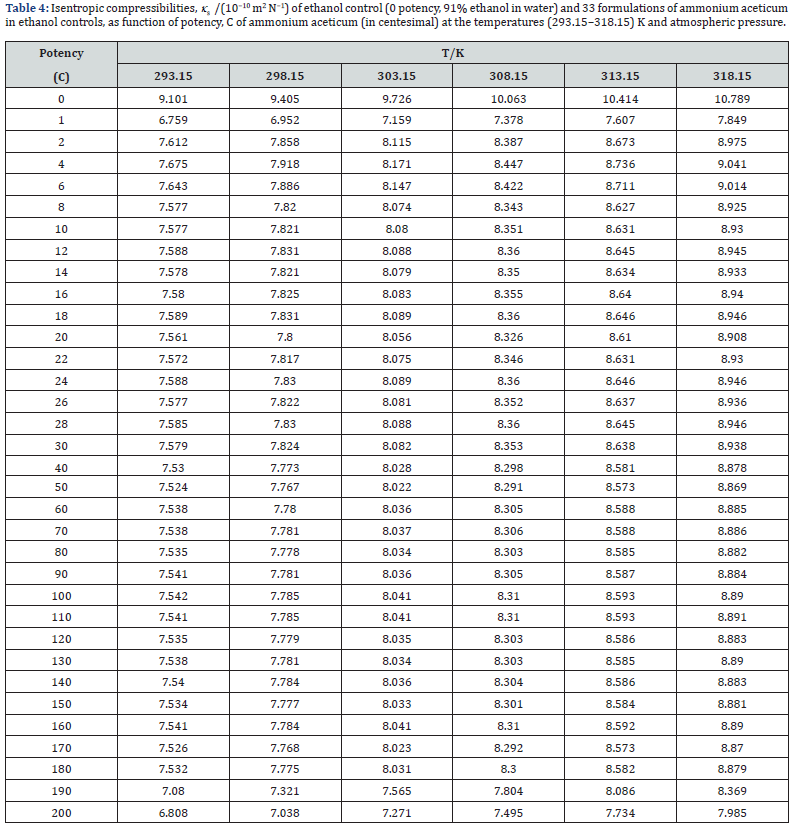
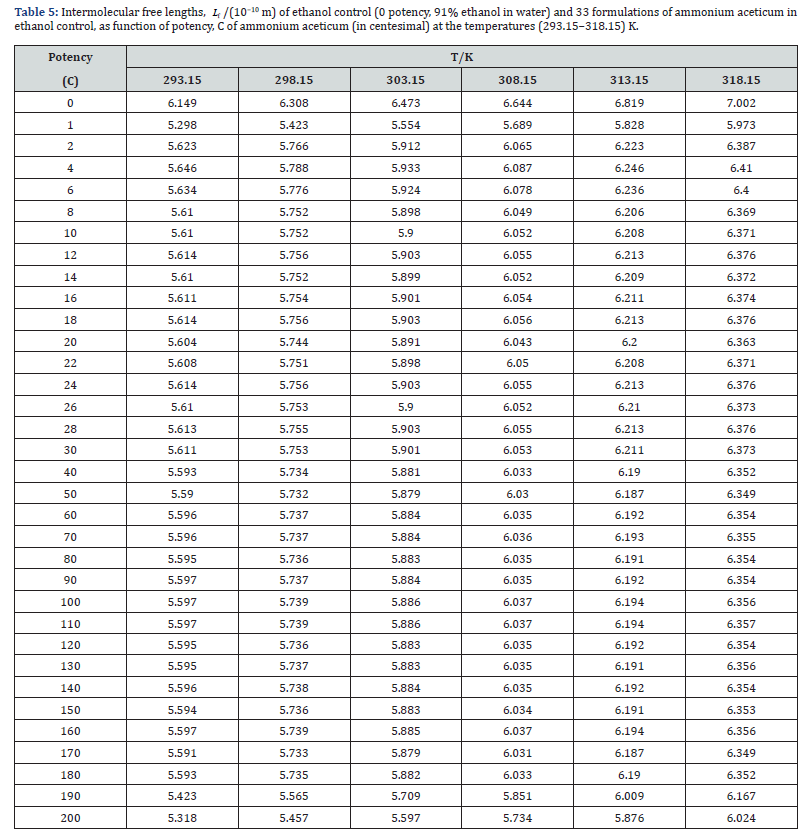
where K’ is temperature dependent constant [= (93.875 + 0.375T) × 10−8]; T is the absolute temperature, ρo, ρ, uo and u are the densities and ultrasonic speeds of the solvent and the solution, respectively. The values of κs, Lf , Z and RA are given in Tables 4-7. The deviations in κs, Lf , Z, η and nD of ethanol due to addition of ammonium aceticum with dilution and succussion are represented by deviation values of these properties. The deviations in isentropic compressibility, Δκs , deviations in intermolecular free length, ΔLf , deviations in acoustic impedance, ΔZ and deviations in viscosity, Δη have been calculated by using the following standard relations

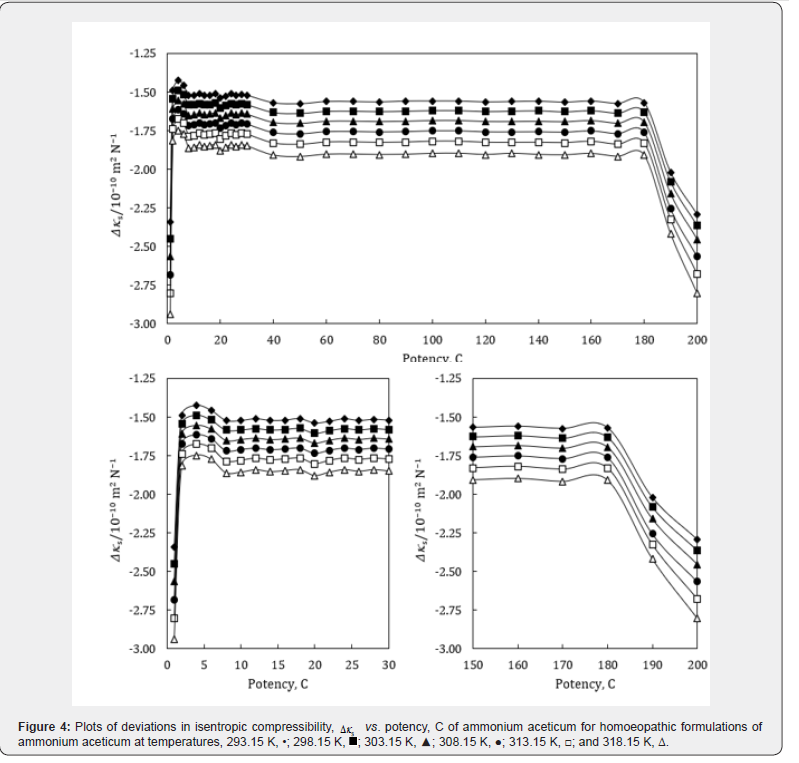
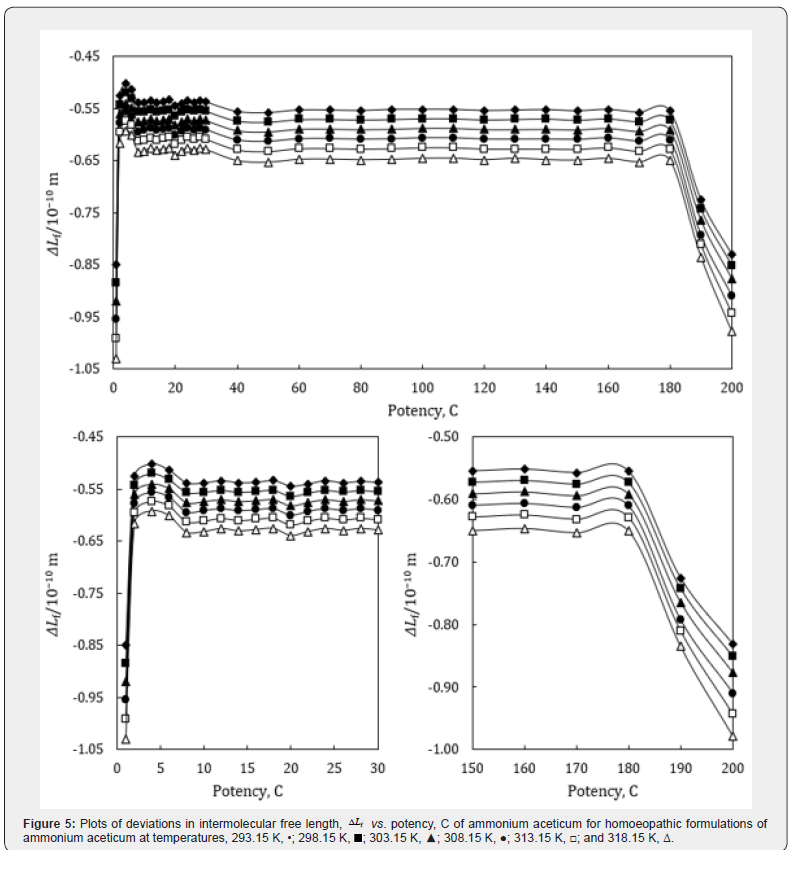
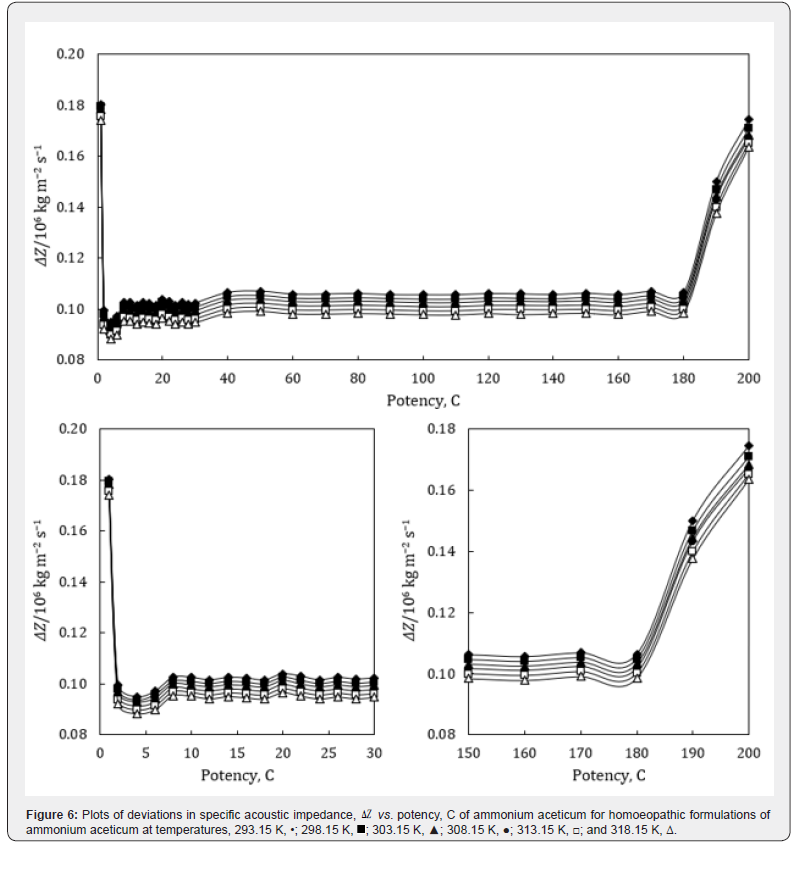
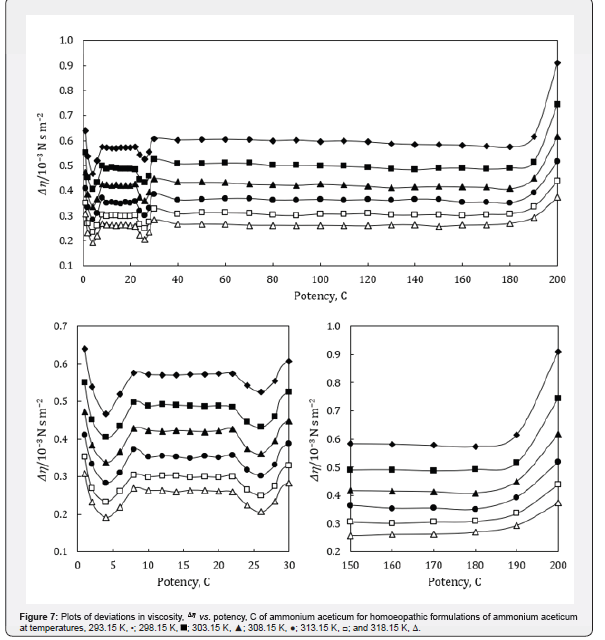
where the superscript ‘o’ represents the values for pure ethanol control (91% ethanol in water). The variations of Δκs , ΔLf , ΔZ and Δη with potency, C of ammonium aceticum and temperature are presented graphically in Figures 4-7, respectively.
Discussion
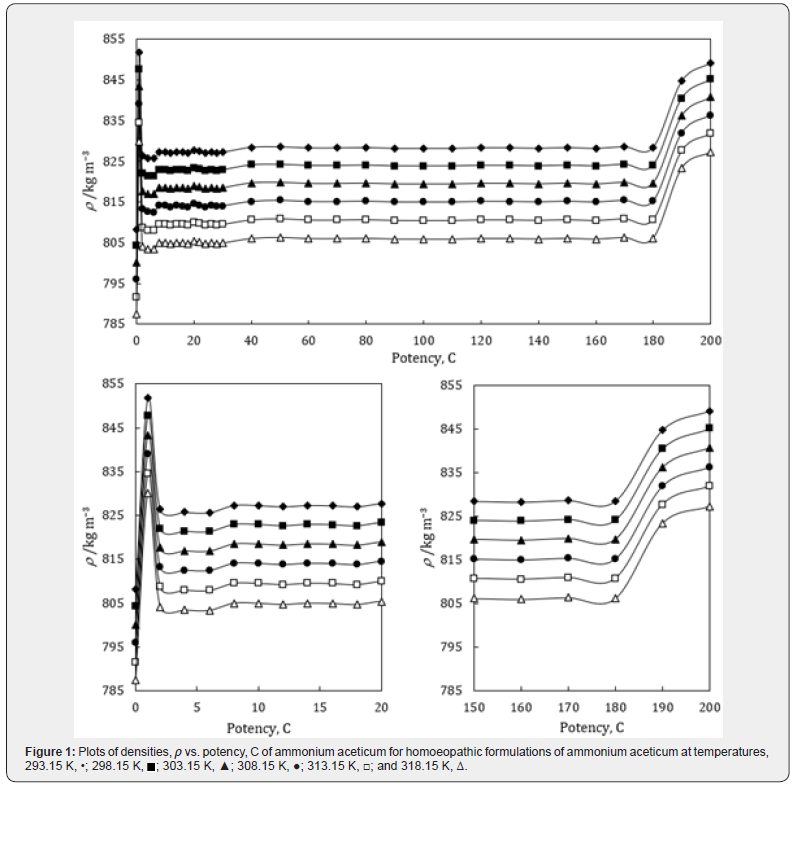
A close perusal of Tables 1-3 and Figures 1-3 indicates that the values of ρ and u and η of ammonium aceticum in ethanol are more than those of ethanol control for all the potencies (1C to 200C) at each investigated temperature and the values decrease with increase in temperature. The values of ρ and u are maximum at 1C and then decrease significantly in presence of ammonium aceticum for simple successive dilution to the potency 4C and after that these values increase slightly till 8C and remain nearly constant up to potency 22C and then decrease and exhibit a minimum at 26C and thereafter 30C the values remain nearly constant up to 180C, and thereafter, again increase significantly after potency 180C up to 200C (Figures 1 & 2). The values of values of η are maximum at 1C decrease significantly in presence of ammonium aceticum for simple successive dilutions from potency 1C to 4C and then increase up to 8C and thereafter remain nearly constant up to potency 22C and then decrease and exhibit a minimum at 26C and then after 30C the values remain nearly constant up to 180C. Thereafter, values of η again increase after potency 180C up to 200C (Figure 3). The observed anomalous trends in ρ, u and η at certain potencies, viz., 1C to 4C, 22C to 30C and 180C to 200C indicate that these potencies exhibit different solution structure as compared to other potencies and ethanol control.
A close perusal of Tables 4 & 5 indicate that the values of κs and Lf for ammonium aceticum potencies are less than those of ethanol controls for all the potencies at each investigated temperature and these values increase with increase in temperature, which indicates significant interaction between ammonium aceticum and ethanol molecules. The values of κs and Lf decrease significantly in presence of ammonium aceticum for simple successive dilutions up to potency 4C and after that value pass through maxima/minima at some potencies with dilution and these values again decrease after potency 180C up to 200C (Tables 4 & 5). These variations in κs and Lf are expressed in terms of deviations in isentropic compressibility, Δκs and deviations in intermolecular free length, ΔLf and are shown graphically in Figures 4 & 5.
Figures 4 & 5 indicate that the values of Δκs and ΔLf are negative and these values are minimum for potency 1C and then increase till potency 4C and after that values decrease till potency 8C and then remain nearly constant on more dilution till 22C, thereafter these values increase and exhibit a maximum at 26C and then after 30C the values remain nearly constant up to 180C, finally decrease again after potency 180C up to 200C. This indicates that at potencies 2C to 6C, 24C to 28C are more compressible and 1C, 190C to 200C are less compressible than the other potencies, indicating that the potencies 2C to 6C, 24C to 28C exhibit less compact solution structure and the potencies 1C, 190C to 200C exhibit less compact solution structure as compared to other potencies and ethanol control. The minimum in Δκs and ΔLf values at potencies 1C, 190C and 200C indicate that these have most compact solution structure as compared to other potencies, hence, these potencies may have different behavior in terms of efficacy when used in practice.
A close perusal of Table 6 and Figure 6 indicate that the acoustic impedances, Z of potencies of ammonium aceticum are more than those of ethanol control for all the potencies at each investigated temperature and the values decrease with increase in temperature, which indicates significant interaction between ammonium aceticum and ethanol molecules. These variations in Z are expressed in terms of deviations in acoustic impedance, ΔZ and are shown in Figure 6. it indicates that ΔZ values are positive, i.e., Z values for ammonium aceticum are more than those of ethanol control. These ΔZ values are maximum for potency 1C and then decrease till potency 4C and after that values increase till potency 8C and then remain nearly constant on more dilution till 22C, thereafter these values decrease and exhibit a minimum at 26C and then after from 30C the values remain nearly constant up to 180C, finally increase again after potency 180C up to 200C.
This indicates that at potencies 2C to 6C, 24C to 28C offer less impedance to sound waves due to less compact structure and 1C, 190C to 200C offer more impedance to sound waves due to more compact structure than the other potencies and ethanol control. The variations in values of Z and ΔZ of these potencies may be due interaction between ammonium aceticum and ethanol molecules.
A close perusal of Tables 1-3 and Figures 1-3 indicates that the values of ρ and u and η of ammonium aceticum in ethanol are more than those of ethanol control for all the potencies (1C to 200C) at each investigated temperature and the values decrease with increase in temperature. The values of ρ and u are maximum at 1C and then decrease significantly in presence of ammonium aceticum for simple successive dilution to the potency 4C and after that these values increase slightly till 8C and remain nearly constant up to potency 22C and then decrease and exhibit a minimum at 26C and thereafter 30C the values remain nearly constant up to 180C, and thereafter, again increase significantly after potency 180C up to 200C (Figures 1 & 2). The values of values of η are maximum at 1C decrease significantly in presence of ammonium aceticum for simple successive dilutions from potency 1C to 4C and then increase up to 8C and thereafter remain nearly constant up to potency 22C and then decrease and exhibit a minimum at 26C and then after 30C the values remain nearly constant up to 180C. Thereafter, values of η again increase after potency 180C up to 200C (Figure 3). The observed anomalous trends in ρ, u and η at certain potencies, viz., 1C to 4C, 22C to 30C and 180C to 200C indicate that these potencies exhibit different solution structure as compared to other potencies and ethanol control.
A close perusal of Table 7 and Figure 7 indicate that the viscosities, η of potencies of ammonium aceticum are more than those of ethanol control for all the potencies at each investigated temperature and the values decrease with increase in temperature, which indicates substantial interaction between ammonium aceticum and ethanol molecules. These variations in η are expressed in terms of deviations in acoustic impedance, Δη and are shown in Figure 7. it indicates that Δη values are positive, i.e., η values for ammonium aceticum are more than those of ethanol control. These Δη values are maximum for potency 1C and then decrease till potency 4C and after that values increase till potency 8C and then remain nearly constant on more dilution till 22C, thereafter these values decrease and exhibit a minimum at 26C and thereafter from 30C the values remain nearly constant up to 180C, finally increase again after potency 180C up to 200C. The variations in values of η and Δη of these potencies may be due interaction between ammonium aceticum and ethanol molecules. It is observed that the variations observed in the values of measured properties, ρ and u and η ; and calculated parameters, κs, Lf , Δκs , ΔLf , ΔZ and Δη support each other.
A close perusal of Table 7 indicates that the values of RA for 1C, 190C and 200C potencies of ammonium aceticum are more than 1 while for all other potencies the values are less than 1, and other potencies show varying values with slight variations at certain potencies. The changes in values of RA of solution in presence of ammonium aceticum are due to different extents of breaking/ formation of hydrogen-bonded associates in ethanol controls and their interaction with ammonium aceticum with successive dilutions and succussion.
Conclusion
The densities, ultrasonic speeds and viscosities of ethanol control, 33 formulations of ammonium aceticum in ethanol control are measured for potencies from 1C to 200C (with an interval of 2C up to 30C and then of 10C up to 200C) at six different temperatures and atmospheric pressure. From these experimental data, various physicochemical parameters, viz., κs ,Lf , Z , Δκs , ΔLf , ΔZ , Δη and RA have been calculated. The results have been qualitatively discussed in terms of interactions/ physicochemical behavior of these extremely dilute homoeopathic formulations of ammonium aceticum in ethanol. The potencies 2C to 6C, 24C to 28C exhibit less compact solution structure and the potencies 1C, 190C to 200C exhibit less compact solution structure as compared to other potencies and ethanol control. Hence, these potencies may have different behavior in terms of efficacy when used in practice. It can be qualitatively concluded that even in extreme dilutions the molecules of ammonium aceticum may be present in these homoeopathic formulations, however it needs to be confirmed from other techniques.
Acknowledgement
The authors are thankful to Central Council for Research in Homeopathy (CCRH), Ministry of AYUSH, Govt. of India for providing financial assistance to carry out this collaborative study.
References
- Ciavatta L, Elia V, Napoli E, Niccoli M (2008) New physico-chemical properties of extremely diluted solutions. Electromotive Force Measurements of Galvanic Cells Sensible to the Activity of NaCl at 25 °C. J Solution Chem 37: 1037-1049.
- Elia V, Elia L, Marchettini N, Napoli E , Niccoli M, et al. (2008) Physico-chemical properties of aqueous extremely diluted solutions in relation to aging. J Therm Anal Cal 93: 1003-1011.
- Maity T, Ghosh D & Mahata CR (2008) Theory and instrumentation related to potentised homoeopathic medicines. Indian J Res Homeopathy 2(3): 1-5.
- Maity T , Ghosh D, Mahata CR (2010) Effect of dielectric dispersion on potentised homeopathic medicines. Homeopathy 99(2): 99-103.
- Mahata CR (2013) Dielectric dispersion studies indicate change in structure of water by potentised homeopathic medicines. J Inst Eng India Ser B 93: 231-235.
- Nain AK , Chand D (2009) Volumetric ultrasonic and viscometric behaviour of glycine DL-alanine and L-valine in aqueous 1,4-butanediol solutions at different temperatures. J Chem Thermodyn 41(2): 243-249.
- Zhao C, Ma P, Li J (2005) Partial molar volumes and viscosity B-coefficients of arginine in aqueous glucose sucrose and L-ascorbic acid solutions at T = 298.15 K. J Chem Thermodyn 37(1): 37-42.
- Zhuo K, Liu Q, Yang Y, Ren Q & Wang J (2006) Volumetric and viscosity properties of monosaccharides in aqueous amino acid solutions at 298.15 K. J Chem Eng Data 51(3): 919-927.
- Nain AK, Pal R, Neetu (2014) Physicochemical study of solute-solute and solute-solvent interactions of l-phenylalanine in water + arabinose/glucose/sucrose solutions at different temperatures. J Chem Thermodyn 68: 169-182.
- Nain A K, Lather M, Neetu (2013) Probing solute-solute and solute-solvent interactions in l-arginine + d-xylose/l-arabinose + water solutions at different temperatures by using volumetric and viscometric methods. J Chem Thermodyn 63: 67-73.
- Nain AZ, Lather M, Sharma RK (2011) Volumetric ultrasonic and viscometric behaviour of l-methionine in aqueous-glucose solutions at different temperatures. J Mol Liq 159(3): 180-188.
- Nain A K, Pal R, Sharma R K (2012) Physicochemical study of solute-solute and solute-solvent interactions of l-histidine in water + sucrose solutions at different temperatures. J Mol Liq 165: 154-160.
- Pal A, Chauhan N (2012) Interactions of amino acids and peptides with the drug pentxifylline in aqueous solution at various temperatures: A volumetric approach. J Chem Thermodyn 54: 288-292.
- Kumar H, Kaur K (2012) Investigation on molecular interaction of amino acids in antibacterial drug ampicillin solutions with reference to volumetric and compressibility measurements. J Mol Liq 173: 130-136.
- Iqbal MJ, Chaudhary MA (2010) Effect of temperature on volumetric and viscometric properties of some non-steroidal anti-inflammatory drugs in aprotic solvents. J Chem Thermody 42(8): 951-956.
- Elia V, Marchese M, Montanino M, Napoli E, Niccoli M, et al. (2005) Hydrohystretic phenomana of extremely diluted solutions induced by mechanical treatments: A calorimetric and conductometric study at 25°C. J Solution Chem 34: 947-960.
- Elia V, Napoli E, Niccoli M (2009) A molecular model of interaction between extremely diluted solutions and NaOH solutions used as titrants. Conductometric and pHmetric titrations. J Mol Liq 148: 45-50.
- Cacace C M, Elia L, Elia V, Napoli E, Niccoli M (2009) Conductometric and pHmetric titrations of extremely diluted solutions using HCl solutions as titrant. A molecular model. J Mol Liq 146: 122-126.
- Elia V, Niccoli M (2000) New physico-chemical properties of water induced by mechanical treatments: A calorimetric study at 25°C. J Therm Anal Cal 61: 527-537.
- Elia V, Napoli E, Niccoli M (2008) On the stability of extremely diluted aqueous solutions at high ionic strength: A calorimetric study at 298.15 K. J Therm Anal Cal 92: 643-648.
- Elia V, Napoli E, Niccoli M (2010) Thermodynamic parameters for the binding process of the OH- ion with the dissipative structures. Calorimetric and conductometic titrations. J Therm Anal Cal 102 : 1111-1118.
- Elia V, Elia L, Cacace P, Napoli E, Niccoli M, et al. (2006) Extremely diluted solutions as multi-variable systems: A study of calorimetric and conductometric behaviour as a function of the parameter time. J Therm Anal Cal 84: 317-323.
- Elia V, Napoli E, Niccoli M, Marchettini N(2008) New physico-chemical properties of extremely dilute solutions. A conductivity study at 25 °C in relation to ageing. J Solution Chem 37: 85-96.
- Belon P, Elia V, Elia L, Montanino M, Napoli E, et al. (2008) Conductometric and calorimetric studies of the serially diluted and agitated solutions. On the combined anomalous effect of time and volume parameters. J Therm Anal Cal 93: 459-469.
- Elia V, Napoli E, Niccoli M (2013) On the stability of extremely diluted solutions to temperature. J Therm Anal Cal 113: 963-970.
- Elia V, Marchettini N, Napoli E & Niccoli M (2014) The role of ethanol in extremely diluted solutions. Calorimetric and conductometric measurements. J Therm Anal Cal 116: 477-483.
- Wang J, Zhao F, Chen B, Li Y, Na P, et al. (2013) Small water clusters stimulate microcystin biosynthesis in cyanobacterial Microcystis aeruginosa. J Appl Phycol 25:329-336.
- Singh PP, Chhabra HL (1993) Topological investigation of the ethanol/water system and its implication for the homoeopathic medicines. Br Homeopath J 82(3): 164-171.
- Homoeopathic Pharmacopoia of India (1971) Controller of Publications, Ministry of Health & Family Welfare. Volume I, Govt. of India, New Delhi.
- Riddick JA, Bunger WB, Sakano T (1986) Organic Solvents: Physical Properties and Methods of Purification, 4th ed., Wiley-Interscience, New York.
- Ortega J (1982) Densities and refractive indices of pure alcohols as a function of temperature. J Chem Eng Data 27: 312-317.
- Nain AK (2008) Ultrasonic and viscometric studies of molecular interactions in binary mixtures of formamide with ethanol. 1-propanol, 1,2-ethanediol and 1,2-propanediol at different temperatures. J Mol Liq 140(1-3): 108-116.
- Ali A, Nain AK, Kamil M (1996) Physico-chemical studies of non-aqueous binary liquid mixtures at various temperatures, Thermochim Acta. 274: 209-221.
- Nain A K, Chand D (2009) Refractive indices of formamide + alkanol/alkanediol binary mixtures at different temperatures, J Indian Chem Soc. 86: 92-96.
- Nain AK (2007) Densities and volumetric properties of (formamide + ethanol, or 1-propanol, or 1,2-ethanediol, or 1,2-propanediol) mixtures at temperatures between 293.15 and 318.15 K. J Chem Thermodyn. 39(3): 462-473.
- Aminabhavi TM, Banerjee T (1998) Density, viscosity, refractive index, and speed of sound in binary mixtures of 2-chloroethanol with alkanols (C1-C6) at 298.15, 303.15, and 308.15 K. J Chem Eng Data 43:509-513.
- Aminabhavi TM, Patil VB (1998) Density, viscosity, and speed of sound in binary mixtures of 1-chloronaphthalene with methanol, ethanol, propan-1-ol, butan-1-ol, pentan-1-ol, and hexan-1-ol in the temperature range (298.15-308.15) K. J Chem Eng Data 43(4): 504-508.
- Krestov GA (1991) Thermodynamics of Solvation. Ellis-Horwood Ltd, England, UK.
- Hildebrand JH, Scott RL (1950) Solubility of Nonelectrolytes. 3rd edition, Reinhold Publishing Co, New York.
- Eyring H, Hirshfelder JO (1937) The theory of the liquid state. J Phys Chem 41: 49-257.































