USA-Kenya Collaboration in Natural Products Research Involving Isolation of Medicinal Compounds from Zanthoxylum Usambarense
Joyce J Kiplimo1, Eric Towett1 and Joseph Kipronoh Rugutt2*
1Department of Physical Sciences, School of Science and Technology University of Kabianga, Kenya
2Department of Chemistry, Missouri State University-West Plains, USA
Submission: September 25, 2022;Published: November 10, 2022
*Corresponding author: Joseph Kipronoh Rugutt, Department of Chemistry, Missouri State University-West Plains, 128 Garfield, MO 65775, USA
How to cite this article: Joyce J K, Eric T, Joseph K R. USA-Kenya Collaboration in Natural Products Research Involving Isolation of Medicinal Compounds from Zanthoxylum Usambarense. Organic & Medicinal Chem IJ. 2022; 12(1): 555828. DOI: 10.19080/OMCIJ.2022.12.555828
Abstract
The present research is part of an ongoing USA-Kenya collaboration in medicinal chemistry. The overarching goal is to enhance multicultural and professional development opportunities for faculty members, undergraduates, and graduate students. We unanimously chose a common topic involving medicinal plants endemic to Kenya. The use of medicinal plants dates back to the 15th century when hundreds of plants were used in traditional medicine. To date, medicinal plants are still used in some developing countries primarily because they are readily available and cheaper than pharmaceutical drugs. The earliest historical records of herbs are found from the Sumerian civilization, where different medicinal plants including opium were used. In Kenya, Zanthoxylum usambarense (Engl.) Kokwaro is used in herbal medicine to treat different diseases. The present study is aimed at assessing the antimicrobial activity of extracts from Z. usambarense plant materials that were collected from Elgeyo Marakwet County, Kenya. The plant materials were taken to the chemistry laboratory at the University of Kabianga. Crude extracts and pure compounds were isolated from the plant materials and biotested for antimicrobial activity. Staphylococcus aureus was used as the biological test organism. Undergraduate and graduate students played an integral part in implementing the research project. The bioassay data provided lead compounds that could be further developed as natural antibiotics for the treatment of bacterial and fungal infections.
Keywords: Natural products; Zanthoxylum usambarense; Antimicrobial activity; Structural elucidation; Nuclear magnetic resonance (NMR) spectroscopy; Mass spectrometry; Mentoring undergraduate and graduate students
Introduction
Zanthoxylum usambarense (Engl.) Kokwaro [1,2] also known as Fagara usambarensis (Engl. 1905), Fagara becquetii and Zanthoxylum becquetii, belong to the family Rutaceae [3]. It is a much-branched tree up to 15 meters tall with conical woody protuberances 2-3cm long ending in sharp straight thorns 5-9mm long. It has a bark rough with longitudinal ridges and furrows as deep as 5 m, greyish brown, peeling yellow underneath and branches with sharp straight to slightly up curved dark red prickles 6-12mm long [1]. Leaves are usually alternating, and flowers are unisexual. The fruits are usually a pair of almost globose follicles.
usambarense is highly valued as an important medicinal plant. It is found in Ethiopia, Kenya, Tanzania, and Rwanda [1]. In Ethiopia, a dried stem infusion is taken as a remedy for kidney infections while fresh stem bark is crushed and used to poultice swollen joints [4]. A stem bark and root bark decoction are commonly taken by the Maasai people of Kenya as an emetic and purgative as well as for the treatment of malaria, backache, joint pain, and rheumatism. The fruits and the leaves are chewed to treat mouth infections, intestinal worms, diarrhea, dysentery, cough, vomiting, and stomachache. An infusion of the fruit is mixed with milk to treat fever, sore throat, tonsillitis, and chest pains. A hot decoction of the seeds is taken to treat malignant catarrhal fever and respiratory tract infections.
In Kenya, Z. usambarense plant is used for dying clothes. The root and stem barks yield yellow and beige dyes, respectively [4]. Wood is used in house construction and to make furniture and bows. The young twigs are used as chew sticks for dental hygiene. Z. usambarense is also used as a “life” fence.
Previous studies have indicated that the extracts of Z. usambarense show antimalarial activity in an animal model [1]. Different extracts of the stem bark and the root bark exhibited significant antibacterial activities against Bacillus subtilis, Micrococcus luteus, and Staphylococcus aureus. The hexane, methanol, and water extracts of the leaves, root bark, and stem bark showed significant anti-inflammatory activity [5] in the cyclooxygenase (COX-1) assay.
Experimental Section
Zanthoxylum usambarense roots were collected from Soy location, in Elgeyo Marakwet County (Kenya). It was identified by Mr. Tuei at the University of Kabianga (UoK). A voucher specimen number ZU 01 was deposited at UoK’s herbarium. The roots were washed using tap water to remove dirt, chopped into small pieces, and then dried under the shade. The dried samples were ground to powder using mortar and pestle.
Organic Extraction
About 500g of the powdered plant material was extracted successively with dichloromethane (DCM) and methanol (MeOH) at a ratio of 1:1 (v/v) and MeOH, respectively. Extraction with MeOH took 48 hours while that of DCM took 24 hours. The extracts obtained were decanted and filtered through Whatman No.1 filter paper. The resulting filtrates were concentrated using a rotary evaporator yielding solid crude organic extracts.
Column Chromatography
The crude extracts from the root bark of Z. usambarense were separated on silica gel column chromatography [6]. The extracts were dissolved in just enough distilled solvents to form a slurrylike paste before loading on an evenly packed chromatographic column using clean pipettes. Thin-layer Chromatography (TLC) [6] was performed to determine the solvent system to be used for separation on column chromatography; [6] a 30:70 (v/v) mixture of hexane:ethyl acetate (EtOAc) gave the best separation. The column was then eluded with the identified solvent system yielding four fractions based on TLC analysis: F1 (23.20 mg), F2 (40.3 mg), F3 (56.78 mg), and F4 (100.05 mg). On further purification through chromatography, F2 yielded two white crystals (compounds 1 and 2). Fraction 4 was further purified using EtOAc in hexane in the ratio 20:80 (v/v) to yield compound 3.
Phytochemical analysis

Structural elucidation using NMR and mass spectrometry
Characterization and structure determination was done using spectroscopic methods. Structure determination and mass analysis were done using Nuclear Magnetic Resonance (NMR [9,10] spectroscopy and Mass Spectrometry (MS), [11] respectively.
All 1D and 2D NMR spectra [10,12] were recorded on a Bruker Avance 500 MHz NMR spectrometer. [13] Samples were dissolved in deuterated chloroform before loading onto an NMR probe and acquiring data. Tetramethylsilane (TMS) was used as an internal standard and the chemical shift was reported as δ (ppm). The offdiagonal elements were used to identify the spin-spin coupling interactions in the 1H-1H Correlation spectroscopy (COSY) [14]. The proton-carbon connectivity, up to three bonds away, was determined using 1H-13C Heteronuclear Multiple Bonds Coherence (HMBC), [6] whereas Heteronuclear Single Quantum Coherence (HSQC) [15] was used to determine the connectivity of hydrogen to their respective carbon atoms.
Mass spectrometry
The mass spectra [11] for all pure compounds were recorded on Finnigan Tipple Stage Quadrupol Spectrometer (TSQ-70) with electron spray ionization (ESI) method in the analysis. Thermo Xcalibur Qual computer software [16] was used in the analysis of the mass chromatograms.
Bioassays
Pure standard isolates of Staphylococcus aureus [17] were cultured at UoK’s microbiology laboratory and used for evaluating the antimicrobial activity of extracts and pure compounds isolated from Z. usambarense.
Antimicrobial bioassays
Antimicrobial activity of the extract from Z. usambarense was carried out using disc diffusion method [18]. Sterile filter paper discs (5 mm in diameter, Whatman No.1) were individually loaded with 30 microlitres of tested extract at a concentration of 0.1 gm/ ml, 0.01 gm/ml, 0.001 gm/ml and were dried. After incubation, the antimicrobial activity was evaluated by measuring the inhibition zones (including the diameter of the discs).
Results And Discussion
Compound 1 was obtained as a yellow solid. Its mass was established to be 388 amu, based on HREIMS data, [7] corresponding to a molecular formula of C21H24O7, which indicates a double bond equivalence [19] of 3, one being due to the furanyl ring, one being due to the carbonyl group and one to exo methylenic double bond [7] attached to furanyl skeleton. The Infrared (IR) [20] spectrum showed a carbonyl stretching band at 1705 cm-1 and a hydroxyl absorption band at 3364 cm-1. This compound was identified as a furanyl group based on its characteristic 1H NMR spectral pattern.
1H NMR spectrum showed resonance at δH 3.55 attributed to a methine proton in close proximity to a hydroxyl group H-4. Another methine resonance in the furan ring was observed at δH 2.66 H-3; this position was confirmed by HMBC correlation of H-3 to C-7 and C-4. Also present were the exo methylenic protons resonating at δH 4.66, 4.91 (H-6), these protons correlated with C-4 and C-5 in the HMBC spectrum. The long aliphatic chain of methylene protons was observed as overlapping peaks between δH 1.30- 2.07 (H-7 to H-21). Methyl peak was observed at δH 0.91 attributed to H-22 at the tail end of the methylene chain.
The 13C spectrum exhibited the presence of 21 carbon resonances, one methyl (CH3) peak, 16 methylene (CH2), 2 methine (CH) carbons, and two fully substituted (quaternary) carbons.
Structure determination
Fraction 2 obtained from DCM:MeOH (1:1 v/v) solvent in column chromatography yielded two compounds after series of purification on 30:70 (v/v) of hexane:ethyl acetate (EtOAc) and later on isocratic solvent containing a mixture of EtOAc and hexane.
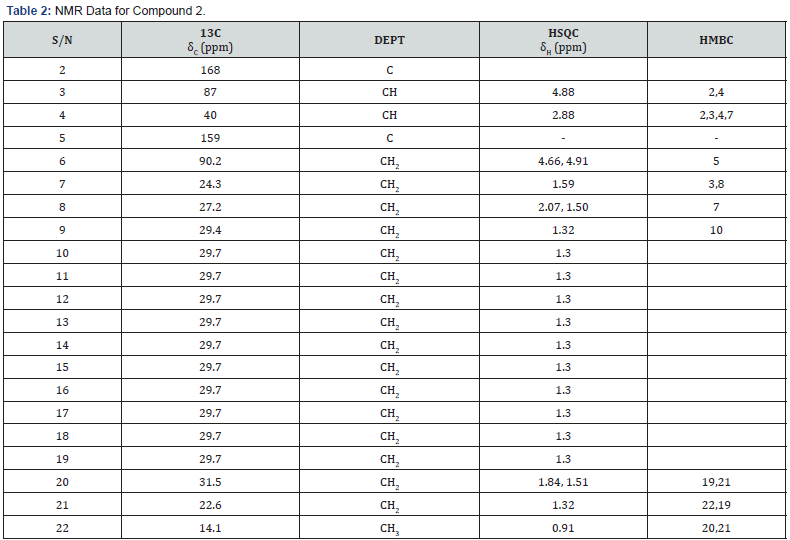
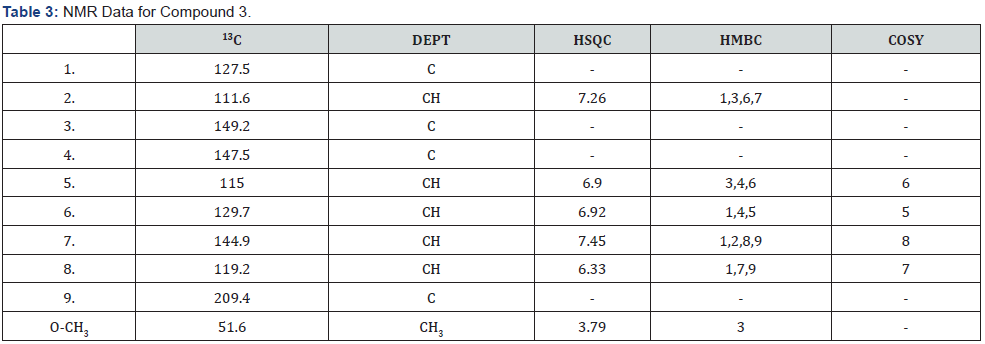
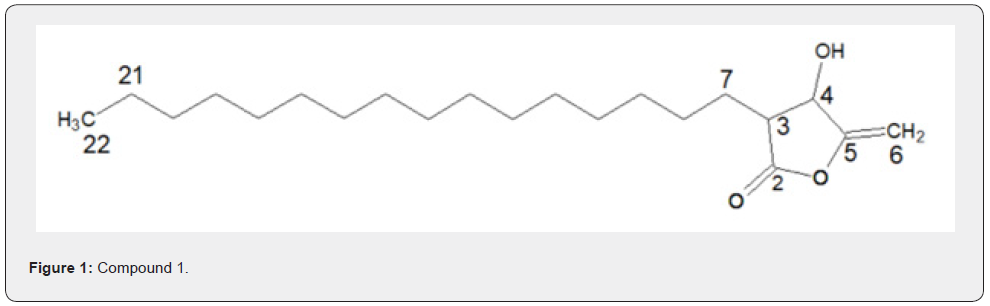
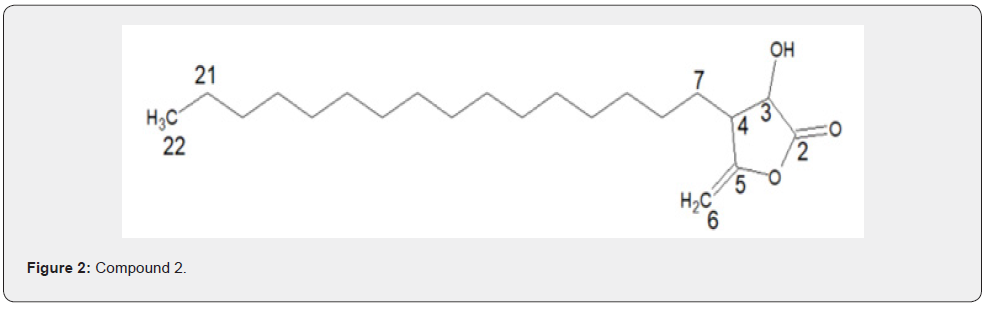
Compounds 1 and 2 were isolated both as white solids at room temperature. They were identified as structural isomers having the same molecular weight but a different order of attachments (Figure 1 & 2). The NMR data for compounds 1 and 2 are similar with slight difference of the absorption of C-2 (δC 174.0), C-5 (δC 158.0) for compound 1, and C-2 (δC 168.0), C-5 (δC 159.0), for compound 2. The slight variation in 13C of the compounds is attributed to the resonance effects which in compound 1, a deshielded C-2 is created while a shielded electron rich C-2 is created due to resonance; these two effects then made carbons in furan ring of compound 1 absorbs up field while those of compound 2 absorbing downfield.
The connection of the two compounds to the hexadecyl group was determined using the HMBC correlations of H-3 (δH 2.65) to C-7 and C-8 for compound 1 and H-4 (δH 2.88) to C-7 in compound 2, respectively.
Additional NMR data for compounds 1 and 2 are shown in Figure 1 & 2 and Table 1 & 2, respectively. Based on these data, compound 1 was assigned the IUPAC name 3-hexadecyl-dihydro- 4-hydroxy-5-methylenefuran-2(3H)-one while compound 2 was assigned the IUPAC name 4-hexadecyl-dihydro-3-hydroxy-5- methylenefuran-2(3H)-one. The two compounds are structural isomers with differences in the attachment of the hexadecyl site chain and the positions of the ketone functional group as well as that of the exocyclic double bonds. The two compounds are reported for the first time and hence considered to be new.
The two compounds (1&2) are furan derivatives having site chain attachment of saturated alkane as well as hydroxy, ketone, and exocyclic double bounds within the furan system. Formerly, spiculisporic acid B, spiculisporic acid C, spiculisporic acid, and secospiculisporic acid were isolated from the fungus Aspergillus fumigatu. These furan derivatives possess anti-inflammatory, anti-microbial as well as anti-cancer activities [21]. On the other hand, Chan et al (2017) [22] isolated six new furan derivatives from the fungal fermentation of Coriolopsis sp on solid rice media, the compounds were assigned the following IUPAC [23,24] names: 5-(3-methoxy-3-oxopropyl)-furan-2-carboxylic acid, 1-(5-(2-hydroxypropanoyl) furan-2-yl)-pentan-3-one, 2-hydroxy-1-(5-(1-hydroxypentyl)-furan-2-yl)-propan-1-one, 1-(5-(1,2-dihydroxypropyl)-furan-2-yl)-pentan-1-one, 5-(1-hydroxypent- 4-en-1-yl)-furan-2-carboxylic acid, and 5-(3-hydroxypentyl)- furan-2-carboxylic acid. The six isolated compounds showed no antimicrobial activities against Staphylococcus aureus (ATCC51650), Ralstonia solanacearum, Fusarium oxysporum f. sp. cubense race 4, Fusarium oxysporum f. sp. niveum, Fusarium oxysporum f. sp. vasinfectum, and Candida albicans (ATCC10231).
The diversity of the furan-based [25] natural products could be attributed to the flexibility of the 5-membered ring to accept site attachments as well as its reactive nature [26]. In the present study, the isolated compounds are new and hence provide useful information. This study then shows diverse sources of furan-based natural products [26] (Figure 3).
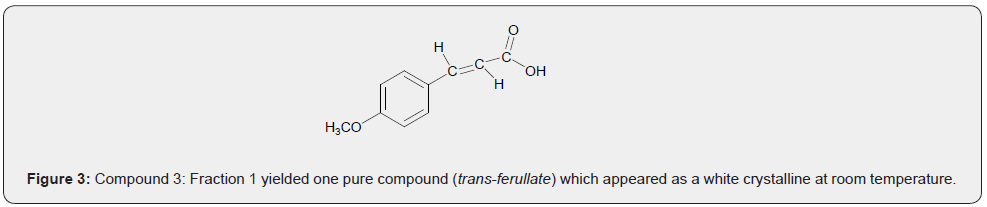
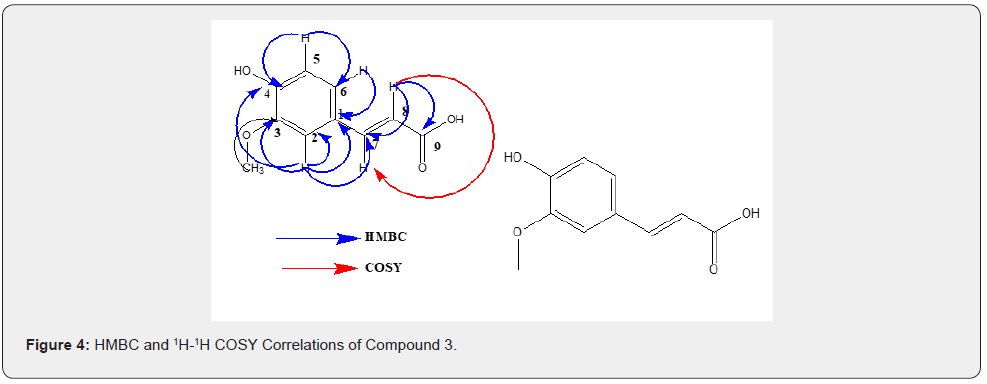
The crude extract obtained in MeOH:DCM (1:1 v/v) solvent system was purified in column chromatography using DCM:EtOAc (20:80 v/v) which resulted in Fraction 1 (F1). A white solid that crystalized at room temperature was analyzed using NMR and Mass spectrometers. It was found to be compound 3 (transferullate). The molecular mass of 3 was established to be 194.18 amu, with molecular formulae of C10H10O4. This indicates a hydrogen deficiency index (HDI) [19] of 5, attributed to the three (3) conjugated double bonds in the benzene ring, a trans double bond on the side chain and carbonyl carbon of the carboxylic acid moiety (C-9). Compound 3 was identified as a trans-ferulate [27] bearing the methoxy and hydroxyl groups on the phenyl ring at positions 3 and 4, respectively. The 1H NMR spectrum indicates the presence of benzylic protons resonating at δH 7.26 (H-2), δH 6.90 (H-5), and δH 6.92 (H-6) corresponding to carbon signals at δC 111.6 (C-2), δC 115.0 (C-5) and δC 129.7 (C-6) in the HSQC spectrum. Also present are the methylene protons with varied multiplicities at δH 7.45 (H-7) and δH 6.33 (H-8) corresponding to carbon signals at δC 144.9 (C-7) and δC 119.2 in HSQC spectrum. Methoxy protons were observed at δH 3.79, corresponding to the carbon signal at δC 51.6 in the HSQC spectrum. Additionally, a hydroxyl proton resonating at δH 5.30 was observed.
The 13C NMR and DEPT [6,28] spectra show that compound 3 has a total of 10 carbons, 5 methine carbons resonating at δC 111.6 (C-2), δC 115.0 (C-5), δC 129.7 (C-6), δC 144.9 (C-7) and δC 119.2 (C- 8). Three quaternary carbons at δC 129.5 (C-1), δC 149.2 (C-3), and δC 147.5 (C-4). One carbonyl carbon resonating at δC 209.4 (C-9) and a methoxy carbon at δC 51.6.
The HMBC [12] spectrum showed that proton resonating at δH 7.26 (H-2) correlates with carbon signals; C-1, C-3, and C-6. Consequently, proton resonating at δH 6.90 (H-5) correlates with carbon signals; C-3, C-4, and C-6. Also observed in the HMBC spectrum was a strong correlation between methoxy protons resonating at δH 3.79 and carbon signal at δC 149.2 (C-3). In the 1H- 1H COSY [6] spectrum correlations between proton at δH 6.90 (H- 5) with that at δH 6.92 (H-6) were observed. Also, olefinic protons at δH 7.45 (H-7) and δH 6.33(H-8) correlated. Other HMBC and COSY spectra are shown in Figure 4. Compound 3 was assigned IUPAC and common names, (E)-3-(4-hydroxy-3-methoxyphenyl) acrylic acid and trans-ferulate, respectively.
Compound 3 is a cinnamic acid derivative having both methoxy and hydroxy substituents on the phenyl and is generally referred to as ferulic acids. The insecticidal activity of compound 3 has been attributed to the conjugation in the benzene rings as well as the activation caused by the ring activators (-OH and -OCHδ3 groups) on the benzene ring [29]. Consequently, the trans double bond on the side chain of the carboxylic acid moiety offers greater reactive stability to the compound which can be used as an insecticide or a precursor in synthesizing bioactive compounds (Table 3).
As part of an ongoing NSF-funded “Undergraduate Student Achievers in Research (USTAR) Program at Missouri State University-West Plains (MSU-WP), one of the end-of-semester exit survey questions posed by faculty to students in STEMeligible disciplines [30] is, “How can the STEM faculty motivate undergraduate/graduate students to participate in research?” While the responses from undergraduates are usually varied, the two most common responses emphasize: 1) the need for scholarships, academic support services, and mentoring programs that promote student participation in on-campus faculty-led research instead of working at local “stores” (e.g., flipping burgers at Burger King), and 2) the need for interesting and cutting-edge research projects that are not only inspiring but also full of the “Aha” moments. Some students go even further and indicate that “inspiring” research should involve beautiful “colored chemical reactions” or spectacular “crystalline compounds.”
The collaborating faculty members from MSU-WP and the University of Kabianga (UoK) unanimously selected natural products research to inspire undergraduate and graduate students. [6,31] Natural products cut across many scientific disciplines such as organic chemistry, analytical chemistry, biochemistry, botany, microbiology, and other chemical sciences. Moreover, the isolation, chemical modifications, structural elucidation, and biotesting of natural products promote high-impact practices [32] (i.e., first-year seminars, engaged student learning communities, undergraduate/graduate research, service-learning, and capstone experiences). To inspire students and create many WOW! Moments in faculty-led natural products research, we use an industry [33] model. For fun and to enhance personal ownership of the projects, students in each research group are told to imagine as if they are part of a new startup company called Plants Pharmaceutical Company. Students would be the employees while the faculty members would be the presidents. The goal of the new pharmaceutical company is to make money by developing new and legal plant-based drugs. The employees need to work as a team and must understand how drugs work, how targets and lead compounds are identified, how they are optimized, how clinical trials are conducted, intellectual property rights, and issues surrounding patents.
To meet the goals of the new pharmaceutical company, the employees write five-page capstone natural products research proposals. [34] Each team develops a grant proposal requesting a “theoretical” two million dollars in funds to support their research project. The proposal will contain the following components: (1) An outline of the research plan stating: a hypothesis, research question(s), and weekly schedules for the entire semester; (2) Annotated bibliography of literature search using ACS citation format [35] and the Japanese KENSHU Method; [36] (3) A list of special glassware, equipment, MSDS of chemicals, reaction schemes, budget and justification, purification schemes, and product identification schemes; (4) How barrier banding protocols will be implemented to prevent lab-related exposure to COVID-19; (5) Experimental procedures for bio-testing of natural products against various biological targets; (6) A five-PowerPoint slide oral presentation [37] summarizing the proposal; and (7) faculty member’s approval signature.
Acknowledgment
Professor Joseph Kipronoh Rugutt gratefully acknowledges the National Science Foundation for a grant (# 1920137) that supports an ongoing (2019-2022) project entitled “Undergraduate Student Achievers in Research (USTAR) Program” at Missouri State University-West Plains (MSU-WP) and collaborating institutions in Kenya. Both professors ( Joseph Kipronoh Rugutt and Joyce J. Kiplimo) acknowledge two grants from the Carnegie African Diaspora Fellowship Program (CADFP) that supported the natural products research at the University of Kabianga (Kenya) and Missouri State University-West Plains (USA).
References
- Ozkan M, Patrick BC Mutiso, Lutfun Nahar, Peng Liu, Sarah Brown, et al. (2013) Zanthoxylum usambarense (Engl.) Kokwaro (Rutaceae) extracts inhibit the growth of the breast cancer cell lines MDA-MB-231 and MCF-7, but not the brain tumour cell line U251 in vitro. Phytother Res 27(5): 787-790.
- Were PS, P Kinyanjui MM Gicheru, E Mwangi, HS Ozwara (2010) Prophylactic and curative activities of extracts from Warburgia ugandensis Sprague (Canellaceae) and Zanthoxylum usambarense (Engl.) Kokwaro (Rutaceae) against Plasmodium knowlesi and Plasmodium berghei. J Ethnopharmacol 130(1): 158-62.
- Tan MA, N Sharma, SSA An (2022) Phyto-Carbazole Alkaloids from the Rutaceae Family as Potential Protective Agents against Neurodegenerative Diseases. Antioxidants (Basel) 11(3): 493.
- Kato A (1996) Isolation of Alkaloidal Constituents of Zanthoxylum usambarense and Zanthoxylum chalybeum Using Ion-Pair HPLC. Journal of Natural Products 59(3): 316-318.
- Agyare C (2013) 19 - Anti-Inflammatory and Analgesic Activities of African Medicinal Plants, in Medicinal Plant Research in Africa, V Kuete, Editor, Elsevier: Oxford, pp. 725-752.
- Rugutt JK, KJ Rugutt, DK Berner (2001) Limonoids from Nigerian Harrisonia abyssinica and their stimulatory activity against Striga hermonthica seeds. J Nat Prod 64(11): 1434-1438.
- Hooper GJ, MT Davies-Coleman, M Schleyer (1997) New Diterpenes from the South African Soft Coral Eleutherobia aurea. Journal of Natural Products 60(9): 889-893.
- Khuda F, Nida Alam, Atif Ali Khan Khalil, Asif Jan, Faiza Naureen, et al. (2022) Screening of Rhamnus Purpurea (Edgew.) Leaves for Antimicrobial, Antioxidant, and Cytotoxic Potential. ACS Omega 7(26): 22977-22985.
- Palazzo TA, Tiana T Truong, Shirley MT Wong, Emma T Mack, Michael W Lodewyk, et al. (2015) Reassigning the Structures of Natural Products Using NMR Chemical Shifts Computed with Quantum Mechanics: A Laboratory Exercise. Journal of Chemical Education 92(3): 561-566.
- Soulsby D (2013) Modern NMR Experiments: Applications in the Undergraduate Curriculum, in NMR Spectroscopy in the Undergraduate Curriculum. American Chemical Society, pp. 7-41.
- Zeeb DJ (2000) Separation and Identification of Twelve Catechins in Tea Using Liquid Chromatography/Atmospheric Pressure Chemical Ionization-Mass Spectrometry. Analytical Chemistry 72(20): 5020-5026.
- Rugutt JK (2000) GR 24 enantiomers: synthesis, NMR spectroscopy, X-ray crystallography, and separation by chiral electrokinetic capillary chromatography. Anal Chem 72(16): 3887-95.
- Serber Z (2001) High-Resolution Macromolecular NMR Spectroscopy Inside Living Cells. Journal of the American Chemical Society 123(10): 2446-2447.
- Rugutt JK (1999) NMR and molecular mechanics study of pyrethrins I and II. J Agric Food Chem 47(8): 3402-3410.
- Qu C (2011) Heteronuclear Single-Quantum Coherence Nuclear Magnetic Resonance (HSQC NMR) Characterization of Acetylated Fir (Abies sachallnensis MAST) Wood Regenerated from Ionic Liquid. Journal of Agricultural and Food Chemistry 59(10): 5382-5389.
- Goecker ZC (2021) Alternative LC–MS/MS Platforms and Data Acquisition Strategies for Proteomic Genotyping of Human Hair Shafts. Journal of Proteome Research 20(10): 4655-4666.
- Kariba RM, PJ Houghton, A Yenesew (2002) Antimicrobial Activities of a New Schizozygane Indoline Alkaloid from Schizozygia coffaeoides and the Revised Structure of Isoschizogaline. Journal of Natural Products 65(4): 566-569.
- Johnson JW (2022) Antibacterial Activity of Metergoline Analogues: Revisiting the Ergot Alkaloid Scaffold for Antibiotic Discovery. ACS Medicinal Chemistry Letters 13(2): 284-291.
- Klemm LH (1995) Use of Degrees of Unsaturation in Solving Organic Structural Problems. Journal of Chemical Education 72(5): 425.
- Koenig JL (1981) Fourier transform infrared spectroscopy of chemical systems. Accounts of Chemical Research 14(6): 171-178.
- Guo Z, Gai C, Cai C, Chen L, Liu S, (2017) Metabolites with Insecticidal Activity from Aspergillus fumigatus JRJ111048 Isolated from Mangrove Plant Acrostichum specioum Endemic to Hainan Island. Marine drugs 15(12): 381.
- Chen LL, WP, Chen HQ, Guo ZK, Wang H, Dai HF, et al. (2017) New Furan Derivatives from a Mangrove-Derived Endophytic Fungus Coriolopsis sp. J5. Molecules 22(2): 261.
- Cook WB (1962) The Chemistry of Natural Products (IUPAC Symposium). Journal of Chemical Education 39(5): A416.
- Natural Products, in Nomenclature of Organic Compounds. 1974, AMERICAN CHEMICAL SOCIETY, pp. 234-240.
- Montagnon T, M Tofi, G Vassilikogiannakis (2008) Using Singlet Oxygen to Synthesize Polyoxygenated Natural Products from Furans. Accounts of Chemical Research 41(8): 1001-1011.
- Lou Y (2020) Oxidation of Furfural and Furan Derivatives to Maleic Acid in the Presence of a Simple Catalyst System Based on Acetic Acid and TS-1 and Hydrogen Peroxide. ACS Omega 5(6): 2561-2568.
- Luo H-F (2005) Cytotoxic Hydroxylated Triterpene Alcohol Ferulates from Rice Bran. Journal of Natural Products 68(1): 94-97.
- Soulsby D (2012) Using Cloud Storage for NMR Data Distribution. Journal of Chemical Education 89(8): 1007-1011.
- Xu H (2009) Semisynthesis and Quantitative Structure−Activity Relationship (QSAR) Study of Novel Aromatic Esters of 4′-Demethyl-4-deoxypodophyllotoxin as Insecticidal Agents. Journal of Agricultural and Food Chemistry 57(17): 7919-7923.
- Vestal SS, M Miller, L Browning (2017) Metacognition across the STEM Disciplines, in Metacognition in Chemistry Education: Connecting Research and Practice American Chemical Society: 17-30.
- Rugutt JK, KJ Rugutt (2012) Antimycobacterial activity of steroids, long-chain alcohols and lytic peptides. Nat Prod Res 26(11): 1004-1011.
- Barr CA (2022) Integrating Authentic Research, Peer Learning, and High-Impact Project Work into the General Chemistry Laboratory. Journal of Chemical Education.
- Beichl GJ, WA Kriner (1986) Why not prepare chemistry majors to work in industry? Journal of Chemical Education 63(8): 699.
- Pirinelli AL (2021) Integrating A Structured Research Proposal into An Undergraduate Glycobiology Class. Journal of Chemical Education 98(11): 3542-3548.
- Schildcrout SM (2002) Learning Chemistry Research outside the Laboratory: New Graduate and Undergraduate Courses in Research Methodology. Journal of Chemical Education 79(11): 1340.
- Drake BD, GM Acosta, RL Smith (1997) An Effective Technique for Reading Research Articles - The Japanese KENSHU Method. Journal of Chemical Education 74(2): 186.
- Hites RA (2014) How to Give a Scientific Talk, Present a Poster, and Write a Research Paper or Proposal. Environmental Science & Technology 48(17): 9960-9964.






























