Freezing the Rotation around Carbon-Carbon Bond in Two Inseparable Conformers by Cyclisation and Isolation of their Products
Neetu and Arif Ali Khan*
University School of Basic & Applied Sciences, India
Submission: October 21, 2021;Published: November 12, 2021
*Corresponding author: Arif Ali Khan, University School of Basic & Applied Sciences, Guru Gobind Singh Indraprastha University, Dwarka, Sector 16-C, New Delhi-110078 (INDIA)
How to cite this article: Neetu, Arif Ali Khan. Freezing the Rotation around Carbon-Carbon Bond in Two Inseparable Conformers by Cyclisation and Isolation of their Products. Organic & Medicinal Chem IJ. 2021; 11(2): 555810. DOI: 10.19080/OMCIJ.2021.11.555810
Abstract
A simple method of cyclisation of two conformers of 2-(2-Hydroxy-phenyl)-2,3-dihydro-1H-quinazolin-4-one with PhPCl2 in presence of a suitable base is described. And the isolation of cyclisation products of two conformers and the synthesis of their chalcogenides is reported here. Each of the cyclized products contained oxazaphosphinine ring in it.
Keywords: Benzoxazaphosphinine; Conformers; P-heteerocycle; Chalcogenides
Introduction
The chemistry of six-member phosphorus heterocycles and their derivatives is well known for their biological activities [1,2]. Cyclophosphamide containing an oxazaphosphinine is proven alkylating antitumor agent against a wide spectrum of human cancer including slow-growing solid tumour [3]. The organophosphorus heterocycles containing O and N in a six-member ring possess high antitumor and antimicrobial activity [4]. The clinical significance, unique conformation and stereo chemical aspects of oxazaphosphinine have attracted much interest in drug design and synthesis [5]. The synthesis of oxazaphosphinine and similar compounds is described in the literature [6-8]. Organo-Phosphorus compounds also constitute a family of flame retardants due to their unique combustion inhibition properties [9,10].
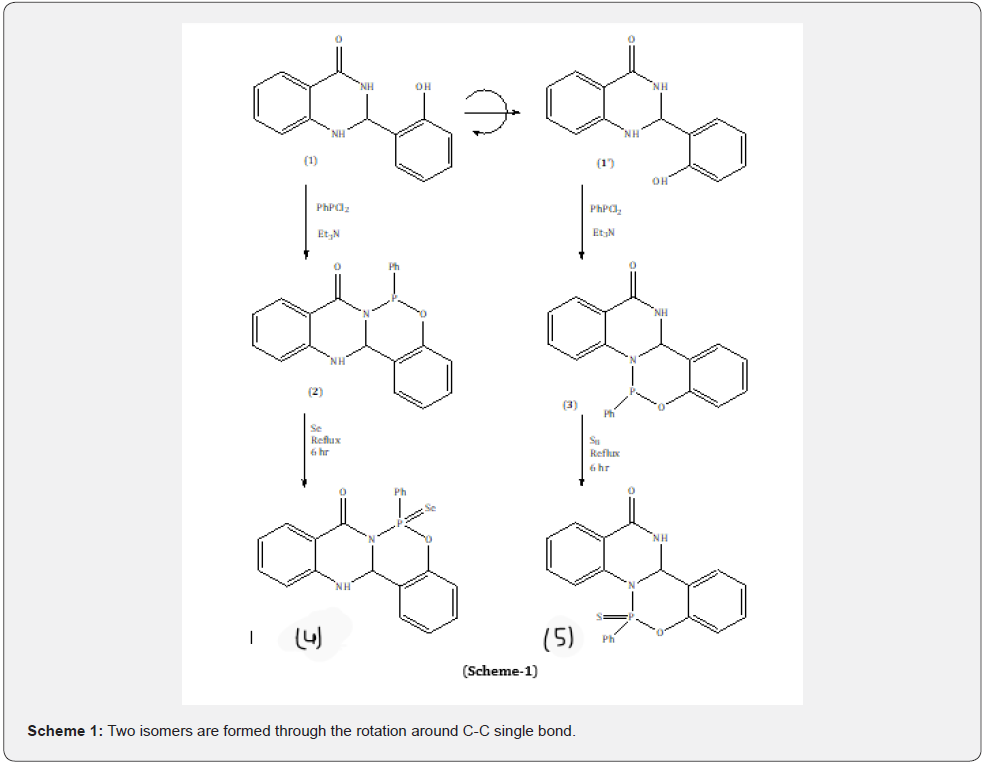
Recently, an article on eco-friendly synthesis of quinazolinones appeared in the literature [11], where the condensation product of salicylaldehyde with 2-anthranilamide provides a unique opportunity to give two inseparable conformational isomers. The two isomers are formed through the rotation around C-C single bond as shown in (Scheme-1). So far such isomers have not been isolated. However, in this case the rotation around C-C single bond of these isomers could be easily frozen by simple cyclization with PhPCl2 in presence of suitable base. We have reported [12-16] a number of such compounds containing benzodioxophosphinone,benzoxazaphosphinine and their chalcogens. However, to the best of our knowledge the synthesis of such benzoxazaphosphinines by freezing the rotation around C-C single bond of two inseparable rotomers is unknown in the literature. The one conformer afforded the product as P-heterocycle derivative of Benzo[a]anthracen-7-one (2) and the other as P-heterocycle derivative of chrysene-12-one (3).
Materials and Methods
Phosphorus (III) compounds are air/ moisture sensitive and their synthesis & manipulation require inert atmosphere of inert/dry gases like helium, argon and nitrogen. And therefore the synthesis of these compounds was carried under inert atmosphere of dry nitrogen with the help of Schlenk Line/Apparatus.
Synthesis of 6-Phenyl-12,12a-dihydro-5-oxa-6a,12-diaza-6-phospha-benzo[a]anthracen-7-one (2) and Synthesis of 5-Phenyl-10b,11-dihydro-6-oxa-4b,11-diaza-5-phospha-chrysen-12-one (3)
The Compound 2-(2-Hydroxy-phenyl)-2, 3-dihydro-1H-quinazolin-4-one (1) was prepared according to literature [11]. To a toluene (40mL) solution of compound (1) (0.240gm; 1mmol), phenyldichlorophosphine (0.179gm; 1mmol) and triethylamine (0.202gm; 2mmol) were added under dry nitrogen atmosphere. Then reaction mixture was refluxed for 3h at 110oC. The progress of reaction was monitored by 31P-nmr. Two compounds were obtained in a typical ratio but could not be separated in pure form even with the low temperature column chromatography. In GC-MS both compounds have same molecular mass but different retention time (R. time- 29.17sec, R.time-27.03sec). A mixture of two compounds was isolated (0.150 gm) and which yielded following partial analysis data. (31P-NMR (161.5MHz, CDCl3): δ106/111; 13C-NMR(100MHz, CDCl3): δ 167, 155, 147, 138, 130, 129, 128, 123, 116, 115, 65, 45; 1H-NMR(400MHz, CDCl3): δ 6-8(m,13H), δ 4(broad, NH), GC-MS: 346 ( base peak)/R. time- 29.17sec, 346 (base peak)/R.time-27.03sec. Melting point-140- 150oC (As Mixture)
Synthesis of compound (4)/Selenide of compound (2)
To a reaction mixture in toluene with compound (2) as major, an access of selenium powder (0.77g, 10mmol) was added and refluxed for 5h. The resulting dark red color solution was filtered hot under nitrogen and concentrated under reduced pressure. The crude product thus obtained was separated by crystallization. (Melting point- 160-170oC); 31P-NMR (161.5MHz, CDCl3): δ 73.4(t) (1JP=Se: 895.66Hz); 31C-NMR (100MHz, CDCl3: δ 165, 148, 146, 137, 135, 133, 132, 131, 129, 128, 125, 121, 117, 67, 45; 1H-NMR (400MHz, CDCl3): δ 6-8(m,13H), δ 4(broad, NH); MS (EI, 30eV)m/ z=424 (62%, M+), 267 (100%, base peak); IR Data: 578.64(P=Se), 3282.84 (20 NH Stretching), 1670.35 (C=O Stretching).
modelling
To a reaction mixture in toluene with compound (3) as major, an access of elemental sulfur (0.320g, 10mmol) was added and refluxed for 5h. The resulting dark red color solution was filtered hot under nitrogen and concentrated under reduced pressure. The crude product thus obtained was separated by crystallization. (Melting point- 200-205oC); 31P-NMR (161.5MHz, CDCl3): δ71.4; 31C-NMR(100MHz, CDCl3): δ 167, 149, 133, 132, 131, 128, 127, 125, 120, 68, 31, 25; 1H-NMR(400 MHz, CDCl3): δ 6-8(m,13H), δ 6(broad, CONH); MS (EI, 30eV)m/z= 378 (40%, M+), , 377 (100%, base peak); IR Data: 677.01(P=S), 3153.61(2o NH Stretching), 1664.57(C=O Stretching).
Results and Discussion
The molecule of 2-(2-Hydroxy-phenyl)-2,3-dihydro-1Hquinazolin- 4-one is a unique example of conformational isomerism where the two rotomers are separated by negligible amount of energy and therefore cannot be separated from each other in pure form. However, the cyclisation of this compound with PhPCl2 in presence of a suitable base eventually afforded the products of two rotomers. It is a remarkable consequence of rotation around C-C single bond in parent compound. 2-(2-Hydroxy-phenyl)-2,3- dihydro-1H-quinazolin-4-one has two amino groups and a C-C single bond between quinazolinone ring and hydroxyl phenyl ring. The hydroxyl phenyl ring can freely rotate around C-C single bond axis and the hydroxyl group on phenyl ring may cyclize with any of two amino groups present on quinazolin-4-one ring at a time in presence of PhPCl2.
The progress of reaction was monitored by 31P-nmr. Two compounds (31P-nmr: 106ppm (2) and 111ppm (3)) were obtained in a typical ratio. In GC-MS both compounds have same molecular mass but different retention time (GC-MS: 346 ( base peak), R. time- 29.17sec, R.time-27.03sec). Two products obtained are time dependent, in first 2.30-3hrs of reaction one product (31P-nmr: 106ppm) will be major and after 3hrs the other one (31P-nmr: 111ppm) will be major. Both are present in reaction mixture in either 1:3 or 3:1 ratio, but could not be separated in pure form even with the help of low temperature column chromatography. However, when the reaction mixture with compound (2) as major was refluxed for 6hr with Se powder in toluene a new compound (31P-nmr: 73.4 ppm (t) with a coupling constant of 1JP=Se: 895.66Hz) (4) was obtained in pure form and good yield. 31P NMR chemical shifts for benzoxazaphosphinine, P=S and P=Se were observed at 106 and 111, 71.4, and 73.4 respectively. Coupling constant for 1JP=Se: 895.66Hz is within the reported range [16].
Conclusion
The cyclisation products of two conformers are very interesting as these molecules contain the oxazaphosphinine ring and may have potential as antibacterial, diuretic and anticancer reagents. The chalcogens of the two products are easily formed. The selective protections of either N-H bonds could be carried out by PhPCl2 and the substitution of other N-H bond with a variety of groups could provide a number of novel compounds.
Acknowledgement
Authors are thankful to the GGSIP University for providing necessary facilities and chemicals in the form of FRGS grant to carry out this research work.
References
- D L Quin (1981) the Heterocyclic Chemistry of Phosphorus, Wiley, New York p 21.
- A A Prishchenko, M V Livantsov (1998) Abstracts of XIVTH International Conference on Phosphorus Chemistry, Cincinnati, Ohio, pp 213.
- S M Ludeman, V L Boyd, J B Regan, L N Gallo, G Zon et al. (1986) Synthesis and antitumor activity of cyclophosphamide analogs. Preparation, kinetic studies, and anticancer screening of phenylketophosphamide and similar compounds related to the cyclophosphamide metabolite aldophosphamide. J Med Chem 29: 716.
- B F Schmidt, W C Tang, G Eisenbrand (1962) Synthesis 701.
- H Kivela, Z Zalam, P Tahtinen, R Sillanpaa, F Fulop et al. (2005) Synthesis and Conformational Analysis of Saturated3,1,2-Benzoxazaphosphinine 2-Oxides. Eur J Org Chem 6: 1189.
- B S Kumar, A U R Sankar, G C S Reddy, M V N Reddy, C D Reddy, et al. (2008) Synthesis and antimicrobial activity of some oxazaphosphinine oxides. ARKIVOC 109.
- KRKK Reddy, CB Reddy, KS Kumar, CN Raju, CS Reddy (2009) Iodine catalyzed and tertiary butyl ammonium bromide promoted p reparation of b enzoxazaphosphininyl phenylboronates. Org Commun 2: 28.
- KRKK Reddy, GS Prasad, MA Kumar, CS Reddy, CN Raju (2008) EJ Chem 5: 1025.
- ED Weil, R Engel (1992) Phosphorus-based Flame Retardants in Hand book of Organophosphorus Chemistry. Marcel Dekker Inc, New York, USA P 14.
- SV Levchik, ED Weil (2005) Overview of recent developments in the flame retardancy of polycarbonates. Polym Int 54(7): 981-998.
- M Wang, TT Zhang, ZG Song (2011) Eco-friendly synthesis of 2-substituted-2,3-dihydro-4(1H)-quinazolinones in water. Chin Chem Let 22: 427-430.
- Manju Rani, Davender Kumar Shukla, Arif Ali Khan (2014) Synthesis of Chalcogenides and Metal Complex of 2-Phenyl-benzo[ 1,3,2]dioxophosphinin-4-one. Asian J Chem 26 (7): 1998.
- Manju Rani, Davender Kumar Shukla, Arif Ali Khan (2013) One Pot Synthesis of 3,9-Bis-(2,4-di-tert-butyl phenoxy)-2,4,8,10-tetraoxa-3,9-diphospha-spiro[5.5]undecane, their Chalcogenides and Metal Complexes. Asian J Chem 25: 4556.
- Davender Kumar Shukla, Manju Rani, Arif Ali Khan (2013) Mercury-Free Catalyst Preparation for Acetylene Hydrochlorination and it’s Catalytic Performance on Single-Tube Pilot Unit Compared with HgCl2 Catalyst. Asian J Chem 25: 4537.
- Manju Rani, Davender Kumar Shukla and Arif Ali Khan (2013) Synthesis and Application of Bisazo Acid Dyes for Water Repellent Polyamides. Asian J Chem 25: 3449.
- Davender Kumar Shukla, Manju Rani, Arif Ali Khan (2013) An Efficient Route for the Synthesis of 3-(4-Bromophenyl)-2-Phenyl-3,4-Dihydro-2H-Benzo[E][,32,]Oxazaphosphinine, its P-Chalcogenides and Metal Complexes. Phosphorus, Sulfur, Silicon Rel Elem 188(8): 1088.






























