Discovery of Original Tricyclic Imidazopyridines during a Search for Nurr1 Nuclear Receptor Agonists
Aline Moulin1, Jean François Peyronel2, Benoit de Carne Carnavalet2 and Dominique Lesuisse2*
1Skyepharma Production SAS, France
2Rare and Neurologic Diseases, Sanofi, France
Submission: January 06, 2020;Published: January 26, 2021
*Corresponding author: Dominique Lesuisse, Rare and Neurologic Diseases, Sanofi, 1 av Pierre Brossolette, Chilly Mazarin, F-91935, France
How to cite this article: Aline M, Jean François P, Benoit de Carne C, Dominique L. Discovery of Original Tricyclic Imidazopyridines during a Search for Nurr1 Nuclear Receptor Agonists. Organic & Medicinal Chem IJ. 2021; 10(3): 555790. DOI: 10.19080/OMCIJ.2021.10.555790
Abstract
This paper describes the exploration of original tricyclic analogues of compound 1, previously identified as a lead compound during a Programme aimed at identifying Nurr1/NOT agonists for potential treatment of Parkinson’s disease. We discovered several tricyclic [5,6,5]-imidazo[1,2-a]pyridines along with efficient ways of preparing them. These new heterocycles are novel starting points for original chemical series.
Keywords: Tricyclic imidazopyridines, Imidazopyridines, Nurr1agonists, NOT agonists, Parkinson’s disease
Introduction
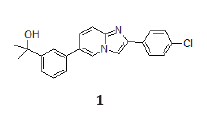
Parkinson’s disease, a devastating neurodegenerative disease, mostly affects dopaminergic neurons resulting in less dopamine and less activation of motor neurons in the central nervous system [1]. The nuclear receptor Nurr1 has been shown to be essential to the development and survival of dopaminergic neurons in substantia nigra and VTA [2]. Nurr1 KO mice display increased vulnerability to MPTP [3] and age-dependent dopaminergic dysfunction [4]. In addition, NOT (human gene) polymorphisms have been reported in Parkinson’s Disease [5]. Nurr1 is an orphan receptor, no endogenous ligand has been identified to-date. We recently reported [6] a potent Nurr1 agonist 1 belonging to 2-Aryl imidazo[1,2-a]pyridines series and its neuroprotective and anti-inflammatory properties. The object of the present paper is to report of an exploration of original tricyclic analogs of 1 to further optimize their overall medicinal chemistry properties.
Materials and Methods
The NMR spectra were recorded on a Bruker Avance II 400 MHz spectrometer using 5 mm PAPPO-BB probe head at 303K temperature in DMSO-d6 solution. The chemical shifts were referred to tetramethylsilane (d, ppm) and the coupling constants in Hertz (s¼ singlet, d¼ doublet, t¼ triplet, q¼ quadruplet, m¼ multiplet). LCMS analyses were performed on a Waters Acquity-SQD UPLC-MS system in positive and negative ESI mode using a short gradient method on an Acquity UPLC Cortecs C18þ, 1.6mm, 2.1_50mm chromatographic column (40_C, flow rate: 1.0 mL/min, 2e98% ACN in 3 min, 0.1% formic acid in both eluents). Compounds representing each class of tricycles are described as an example.
Compound 7 (SAR162770): 2-bromo-4’-chloroacetophenone (1.8g, 1eq) is solubilized in 1,2-dimethoxy-ethane (25 mL). 5-aminopyrrolo[3,2-b]pyridine (1g, 1eq) is then added. The obtained solution is stirred overnight at room temperature. The beige precipitate obtained is dried under vacuum and not isolated. The obtained salt (2.8 g, 1 eq) is introduced in ethanol (40ml), stirred overnight at 110°C, then cooled at room temperature
After evaporation under vacuum, the crude product is purified by preparative chromatography (alumina as stationary phase, dichloromethane / methanol 100/0 to 80/20 v/v as eluant) to obtain the desired product (0.7 g, 33% yield). HPLC=1.07 min. M+H+(C15H10ClN3) = 269.16/270.07. 1H NMR (400MHz, DMSO-d6) δ ppm 6.78 (d, J=2.70 Hz, 1H), 7.23 (d, J=9.50 Hz, 1H), 7.38 (d, J=2.70 Hz, 1H), 7.46 - 7.51 (m, 3H), 8.00 (d, J=8.60 Hz, 2H), 8.65 (s, 1H), 11.66 (br s, 1H).
Compound 8 (SAR197935): 2-bromo-4’-chloroacetophenone (0.4g, 1eq) is solubilized in acetone (3mL). 6-amino-7-azindole (0.2 g, 1 eq) is then added. The obtained solution is stirred under microwave 30 min at 130°C. Water and sodium hydroxide are then added to neutralize the mixture. Ethanol is then added to obtain a brownish solution. Water is then added. The brownish precipitate obtained is cooled with an ice bath, filtered, and dried under vacuum (0.01 g, 2% yield). HPLC= 1.01 min. 1H NMR (400 MHz, DMSO-d6) δ ppm 6.53 (d, J=3.25 Hz, 1H), 7.18 (d, J=3.25 Hz, 1H), 7.20 (d, J=9.20, 1H), 7.51 (d, J=8.50 Hz, 2H), 7.58 (d, J=9.20 Hz, 1H), 7.95 (d, J=8.50 Hz, 2H), 8.44 (s, 1H).
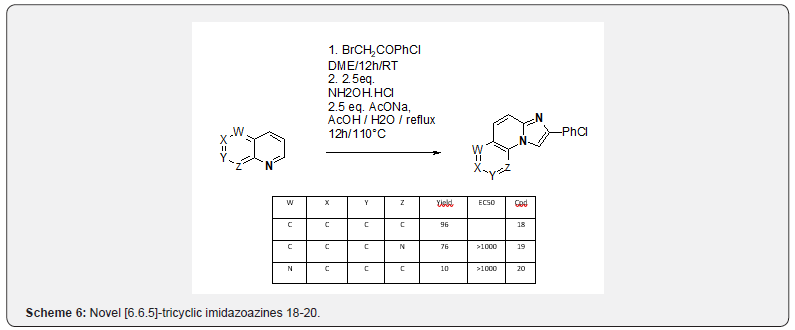
Compound 19 (SAR139598): 2-bromo-4’-chloroacetophenone (2.0g, 1eq) is solubilized in 1,2-dimethoxy-ethane (20 mL). 1.8 naphthyridine (1.1 g, 1eq) is then added. The obtained solution is stirred overnight at room temperature. The red precipitate obtained is filtrated and rinsed with cyclohexane and dried under vacuum (2.4g, 78% yield). The obtained salt (1.7g, 1eq) is introduced in acetic acid / water (10mL/10ml). Sodium acetate (0.9g, 2.5eq) and hydroxylamine hydrochloride (0.8g, 2.5eq) are then added under stirring. The mixture is stirred overnight at 110°C, then cooled at room temperature. Addition of water allows the obtention of an off-white precipitate, which is filtered and dried under vacuum (1.0 g , 76% yield). HPLC= 4.21 min. M+H+ (C16H10ClN3) = 280.15 / 282.00. 1H NMR (400 MHz, DMSO-d6) δppm 7.55 (br d, J=7.55 Hz, 2H), 7.66-7.83 (m, 2H), 7.96 (br d, J=9.30 Hz, 1H), 8.14 (br d, J=7.55 Hz, 2H), 8.56 (br d, J=7.50 Hz, 1H), 8.87 (br s, 1H), 9.20 (br s, 1H).
Compound 20 (SAR140325): 2-bromo-4’-chloroacetophenone (2.0 g, 1 eq) is solubilized in 1,2-dimethoxy-ethane (20 mL). 1.5 naphthyridine (1.1 g, 1 eq) is then added. The obtained solution is stirred overnight at room temparature. The obtained brownish mixture is evaporated and dried under vacuum to obtain a brownish oil (2.8g, 89% yield). The obtained salt (2.8g, 1eq) is introduced in acetic acid / water (15mL/15 ml). Sodium acetate (1.6g, 2.5eq) and hydroxylamine hydrochloride (1.3g, 2.5eq) are then added under stirring. The mixture is stirred overnight at 110°C, then cooled at room temperature. After evaporation under vaccum, the crude product is purified by preparative chromatography (alumina as stationary phase, dichloromethane / methanol 100/0 to 95/5 v/v as eluant). The desired product is obtained with a very low yield (0.1 g, 6% yield). M+H+ (C16H10ClN3)=280.09/282.09. 1H NMR (400 MHz, DMSO-d6) δppm 7.55 (d, J=8.40 Hz, 2H), 7.73 - 7.87 (m, 3H), 8.03 (d, J=8.40 Hz, 2H), 8.78 (d, J=8.05 Hz, 1H), 8.84 (dd, J=4.40, 1.40 Hz, 1H), 9.30 (s, 1H).
Results and Discussion
Characterization of the first leads
Full optimization leading to 1 had not permitted to get rid of one liability, i.e., hErg channel inhibition. Since the discovery of a link between drug-induced hERG inhibition and cardiac safety related to Torsade de Pointes (TdP) [7], this parameter has been intensely scrutinized and optimized before entering development. This hErg inhibition of 1 (IC50=1.5μM) has been since derisked [6] and shown not to give rise to cardiac related safety issues, but before this was the case, we actively searched how to design Nurr1 agonists with comparable or better potency and devoid of this (and any other) liability.
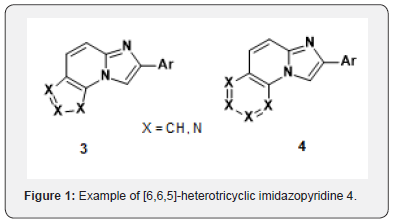
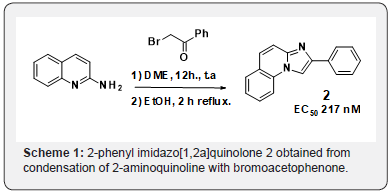
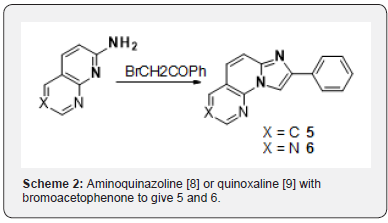
Amongst a very narrow structure-activity relationship in these series, initial exploratory work had pointed to tricyclic imidazo[1,2-a]pyridines as potentially promising scaffods. 2-phenyl imidazo[1,2a]quinolone 2 obtained from condensation of 2-aminoquinoline with bromoacetophenone (Scheme 1) gave comparable potency as 1 in our in vitro assay [6]. We the aim of discovering novel scaffolds with better solubility, we embarked on the search for new tricyclic analogs. At the time, no examples of [5,6,5]-heterotricyclic imidazopyridine 3 and very few examples of [6,6,5]-heterotricyclic imidazopyridine 4 existed in the literature (Figure 1). For the latter, the only reported method was analogous to the one we had used for 2 namely condensation of the aminoquinazoline [8] or quinoxaline [9] with bromoacetophenone to give 5 and 6 (Scheme 2).
This methodology was applied to several commercially available bicyclic amines and bromo-4-chlorophenylacetophenone to afford the corresponding tricyclic imidazo[1,2-a] pyridines 7-11 (Scheme 3). The 4-chloroaryl substitution allowed direct comparison with the optimized imidazopyridine 1 showing that aside from the triazolo scaffold 11, all scaffolds were well tolerated by the nuclear receptor. Starting from there, around 100 analogs of these tricyclic imidazopyridines differing by their aryl motif were prepared and evaluated for their activity on the receptor. A few were extremely potent but none of them satisfied the full target product profile. The most often issue encountered was metabolic stability and CYP inhibition or induction (data not shown).
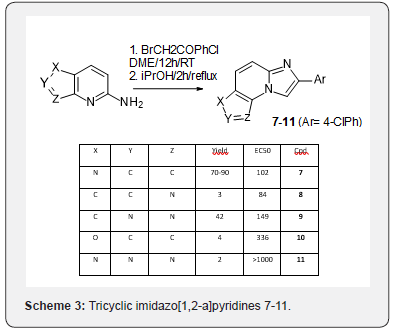
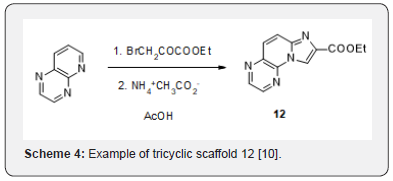
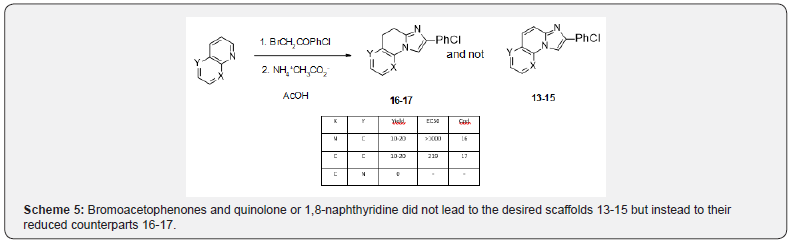
Our access to these series was limited by the very few commercially available starting bicyclic amines. Another potential access to imidazo [1,2-a]pyridines has been long reported starting from quinolones in place of aminoquinolines. An example is given in scheme 4 for the access to the tricyclic scaffold 12 [10] (Scheme 4). Applying these conditions to bromoacetophenones and quinolone or 1,8-naphthyridine did not lead to the desired scaffolds [13-15] but instead to their reduced counterparts [16,17]. In the case of 1,5-naphthyridine, no identified compound could be isolated (Scheme 5).
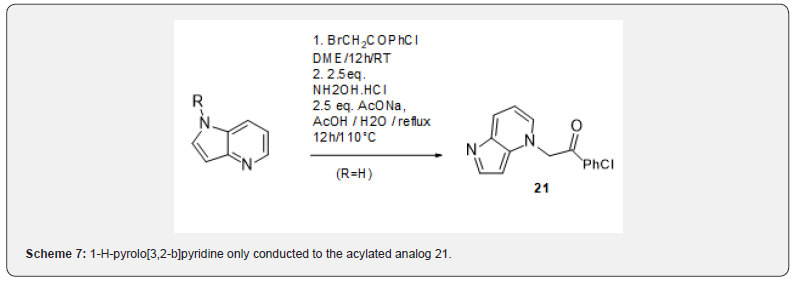
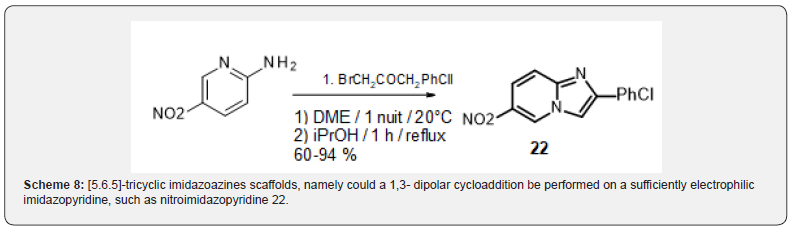
Taking a closer look at the literature, three precedents of synthesis of imidazo[1,2-a]pyridines from quinolines indicated that an oxidative step is required to obtain the desired products, either hydroxylamine11, ferric chloride [12] or manganese dioxide [13]. The oxidative methodology reported by Krohnke could be successfully reproduced and applied to the synthesis of the novel [6.6.5]-tricyclic imidazoazines 18-20 (scheme 6). However, the scope of this reaction seemed limited as application to 1.4.5-triazanaphthalin (W=Z=N) or quinazoline only led to tars, while 1-H-pyrolo[3,2-b]pyridine only conducted to the acylated analog 21 (Scheme 7). Various protecting groups on the pyrrole moiety (mesyl, acetyl, tosyl, FMOC) did not stand the condtions. These observations along with the very few sources of commercially available bicyclic precursors, led us to devise a new strategy to access the [5.6.5]-tricyclic imidazoazines scaffolds, namely could a 1,3- dipolar cycloaddition be performed on a sufficiently electrophilic imidazopyridine, such as nitroimidazopyridine 22 ? Several aryls substituted nitroimidazopyridines 22 were synthesized from 4-nitro-2-aminopyridine using a reported protocol [14] (Scheme 8).
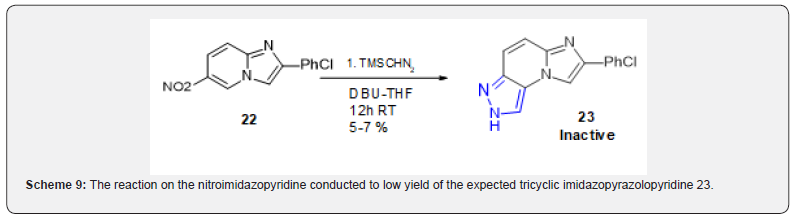
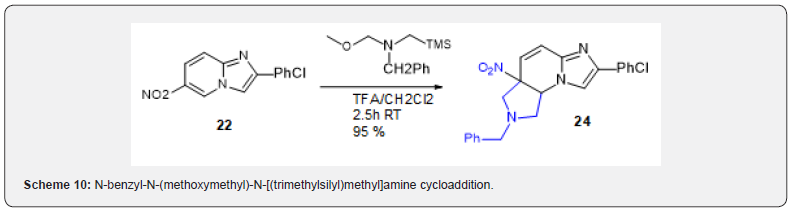
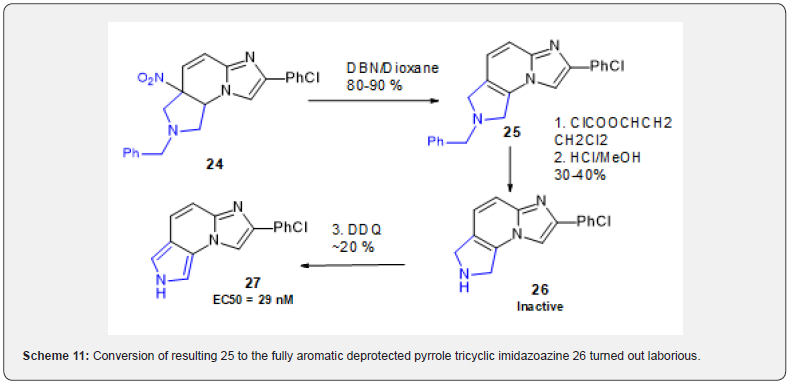
The only precedents of trimethyl silyl diazomethane cycloaddition on aromatic compounds are on arynes [15]. The reaction on the nitroimidazopyridine conducted to low yield of the expected tricyclic imidazopyrazolopyridine 23 (Scheme 9). This compound turned out inactive on Nurr1 receptor. There are two recent reports for cycloaddition of azomethine ylids on nitroarenes [16,17] however there is no precedent on imidazoazine. It could be anticipated that this bond would be more reactive than a fully aromatic nitroarene. This was in fact the case and when 22 was engaged with N-benzyl-N-(methoxymethyl)-N-[(trimethylsilyl) methyl]amine cycloaddition indeed proceeded smoothly and with very high yield (Scheme 10). However, in this case as opposed to the two above-cited precedents, cycloaddition only occurred on one bond of the aryle to afford 24.
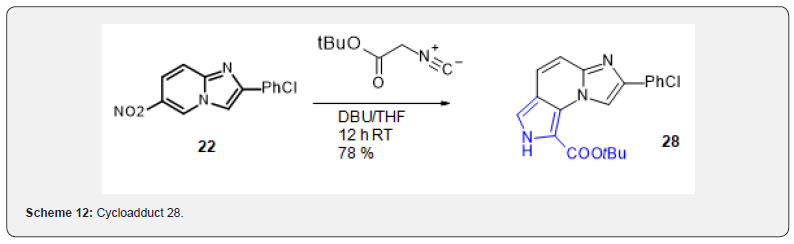
The nitro group could be smoothly eliminated using DBN, however, conversion of resulting 25 to the fully aromatic deprotected pyrrole tricyclic imidazoazine 26 turned out laborious. Low yied deprotection could be realized using vinyl chloroformate and aromatization could be performed using DDQ albeit the process turned out whimsical and poorly reproducible (Scheme 11). Strikingly while 26 was totally devoid of activity, 27 turned out one of the most active compounds in the series. Therefore, the need for better chemical way to the series became stringent and we reasoned that a potential access could be through isocyanide cycloaddition. Synthesis of polycyclic pyroles by cycloaddition on nitroarenes has been reported [18,19] but no example on imidazopyridine. In this case also the reaction of tButyl isocyanoacetate with 22 proceeded very smoothly to afford a good yield of the expected cycloadduct 28 (Scheme 12).
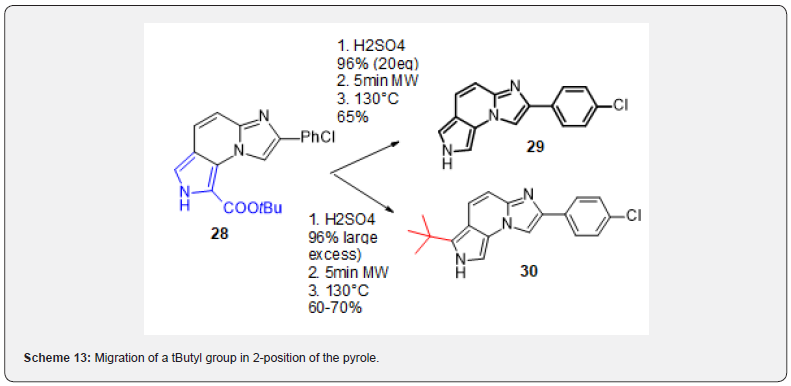
The conditions for deprotection of the tButyl group turned out exquisitely sharp: upon addition of a controlled amount of 96% sulfuric acid, the fully deprotected compound 27 was obtained with good yield. On the other hand, when the same conditions were used with an excess of sulfuric acid, compound 30 resulting from migration of a tButyl group in 2-position of the pyrole was obtained with the same yield (Scheme 13). Given the promising activity of compound 29 on the Nurr1 receptor, we further explored this chemical series.
Optimization of the potency
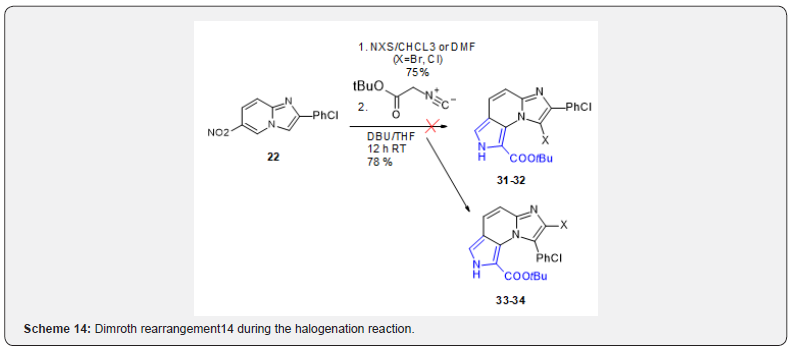
One way of expanding the chemical space around a promising lead is to introduce a halogen on the scaffold that will them allow substitution by diverse nucleophilic partners. Compound 22 was treated with N-halogen succinimides to halogenate position 3 before the isocyanide cycloaddition. However, in place of obtaining the expected 31 and 32, compounds 33 and 34 were obtained (Scheme 14). It was hypothesized that these compounds could arise from a Dimroth rearrangement14 during the halogenation reaction.
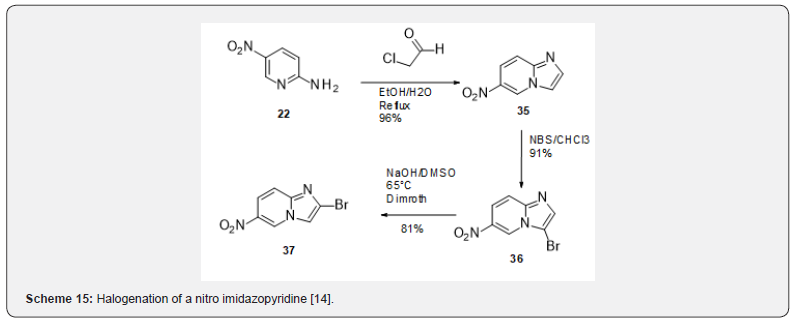
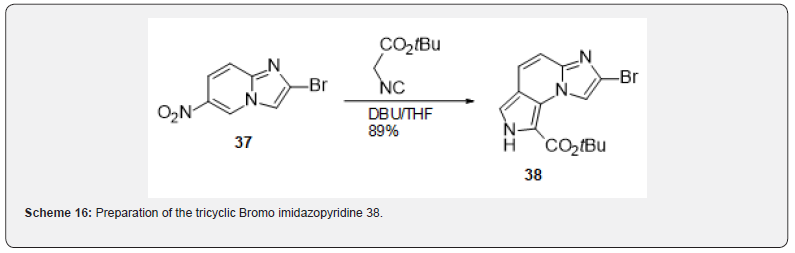
In fact, a Dimroth rearrangement had been reported in the literature during the halogenation of a nitro imidazopyridine [14] (Scheme 15) and was reproduced for the synthesis of compound 37. The nitro imidazopyridine 35 was obtained from 5-Nitro-2- aminopyridine after chloroacetaldehyde treatment. Bromination led to the expected 3-bromoimidazopyridine 36 which rearranged to 37 upon basic treatment. Compound 37 allowed the preparation of the tricyclic Bromo imidazopyridine 38 (Scheme 16) which was the starting point of a wide exploration of the substitution in several positions of the tricyclic skeleton. This series turned out very difficult to optimize from a medicinal chemistry point of view. It is probable that the flat aromatic scaffold might be responsible for promiscuous interactions with other targets giving rise to safety liabilities [20].
Conclusion
During the search for new and original scaffolds as Nurr1 agonists, we discovered several tricyclic [5,6,5]-imidazo [1,2-a] pyridines along with efficient ways of preparing them. These new heterocycles have not been described and are novel starting points for original chemical series.
Acknowledgement
We are grateful to Laszlo Balazs and the analytical department for compound analyses..
References
- Tansey M G, Goldberg M S (2010)Neuroinflammation in Parkinson's disease: its role in neuronal death and implications for therapeutic intervention. Neurobiol Dis 37: 510-518.
- Zetterstrom R H, L Solomin, L Jansson, B J Hoffer, L Olson, T Perlmann(1997) Dopamine neuron agenesis in Nurr1-deficient mice. Science 276: 248-250.
- Le W, Conneely O M, He Y, Jankovic J, Appel S H (1999) Reduced Nurr1 expression increases the vulnerability of mesencephalic dopamine neurons to MPTP-induced injury. J Neurochem 73: 2218-2221.
- Jiang, C, Xinhua Wan, Yi He, Tianhong Pan, Joseph Jankovic, et al. (2005) Age-dependent dopaminergic dysfunction in Nurr1 knockout mice. Exp Neurol 191: 154-162.
- Zheng K, Heydari B, Simon D K (2003) A common NURR1 polymorphism associated with Parkinson disease and diffuse Lewy body disease. Arch Neurol 60: 722-725.
- Lesuisse D, et al. (2019) Development of a novel NURR1/NOT agonist from hit to lead and candidate for the potential treatment of Parkinson's disease. Bioorg Med Chem Lett 29: 929-932.
- Wallis R M (2010) Integrated risk assessment and predictive value to humans of non-clinical repolarization assays. Br J Pharmacol 159: 115-121.
- Dauphin, A (1997) Synthesis, structure and reactivity of imidazo[1,2‐a][1,8]naphthyridines. J. Heterocyclic Chemistry 34: 765-771.
- H W (2016)Chinese patent CN105481858 A.
- Blache Y G Alain, Chavignon Olivier, Elhakmaoui Ahmed, Viols Henry, Teulade Jean Claude, et al. (1994) Reaction of 1-ethoxyalylmethylpyrido[2,3-b]pyrazinium bromide with ammonium acetate: synthesis of imidazo[1',2':1,2]pyrido[5,6-b]pyrazines. Heterocycles 38: 283-289.
- ZeCher F K u W (1962) ZurKonstitution der Betaine aus Phenacyl-cyclimmoniumsalzen, I . Phenacyl-isochinolinium- und -chinoliniumbetaine. Chem Ber 95: 1128-1135.
- Toja E O S A, Favara D, Cattaneo C, Gallico L, Galliani G (1983) Synthesis and pregnancy terminating activity of 2-arylimidazo[2,1-a]isoquinolines and isoindoles. ArzneimittelForschung 33: 1222-1226.
- Parenty A D, Song Y F, Richmond C J, Cronin L (2007) A general and efficient five-step one-pot procedure leading to nitrogen-bridgehead heterocycles containing an imidazole ring. Org Lett 9: 2253-2256.
- Jacquier R L, Helene, Maury Georges (1973) Intermediates in the Dimroth rearrangement of imidazo[1,2-a]pyridines. J Heterocyclic Chemistry 10: 755-762.
- Shoji Y, Hari Y, Aoyama T (2004) Facile synthesis of 3-trimethylsilylindazoles by [3+2]cycloaddition reaction of lithium trimethylsilyldiazomethane with benzynes. Tetrah Let 45: 1769-1771.
- Bastrakov M A, et al. (2010) Double 1,3-dipolar cycloaddition of N-methyl Azomethine ylide to meta-Dinitrobenzene annelated with nitrogen aromatic heterocycles. Journal of Heterocyclic Chemistry 47: 893-896.
- Lee S, Chataigner I, Piettre S R (2011) Facile dearomatization of nitrobenzene derivatives and other nitroarenes with N-benzyl azomethine ylide. Angew Chem Int Ed Engl 50: 472-476.
- Murashima T, Ryuji Tamai, Keiji Nishi, Kentaroh Nomura, KenichiFujit, et al. (2000) Synthesis and X-ray structure of stable 2H-isoindoles. J Chem Soc Perk Trans 1: 995-998.
- Timothy D Lash, C W Augustine T Osuma, Jyoti R Patel (1997) Synthesis of Novel Porphyrin Chromophores from Nitroarenes: Further Applications of the Barton-Zard Pyrrole Condensation. Tetrah Let 38: 2031-2034.
- Lovering F, Bikker J, Humblet C (2009) Escape from flatland: increasing saturation as an approach to improving clinical success. J Med Chem 52: 6752-6756.






























